Antilipidemic drugs
Antilipidemic Drugs
Overview of Drug Classes
| Class | Primary Target | Key Agents |
|---|---|---|
| HMG-CoA reductase inhibitors | ↓ LDL-C | Statins |
| Cholesterol absorption inhibitor | ↓ LDL-C | Ezetimibe |
| Bile acid sequestrants | ↓ LDL-C | Cholestyramine, Colesevelam |
| PCSK9 inhibitors | ↓↓ LDL-C | Alirocumab, Evolocumab |
| ATP-citrate lyase inhibitor | ↓ LDL-C | Bempedoic acid |
| MTP inhibitor | ↓ LDL-C (HoFH only) | Lomitapide |
| Fibrates | ↓ Triglycerides, ↑ HDL | Fenofibrate, Gemfibrozil |
| Niacin | ↓ TG, ↓ LDL, ↑ HDL | Nicotinic acid |
| Omega-3 fatty acids | ↓ Triglycerides | EPA/DHA, Icosapent ethyl |
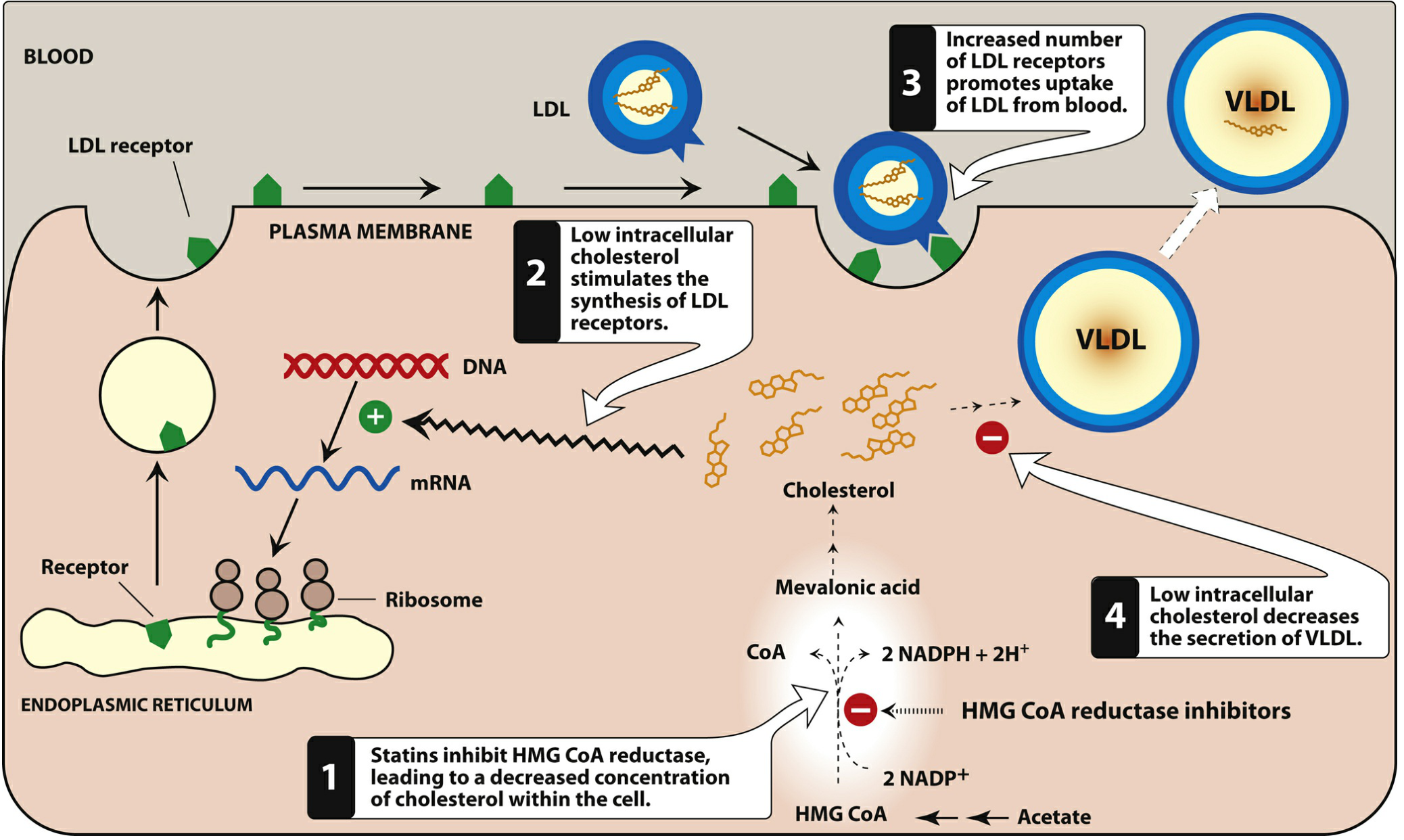
1. Statins (HMG-CoA Reductase Inhibitors)
- Inhibit HMG-CoA reductase → ↓ intracellular cholesterol
- Low intracellular cholesterol → upregulation of LDL receptors on hepatocyte surface
- More LDL receptors → increased clearance of LDL-C, VLDL, and IDL from blood
- Also ↓ VLDL secretion from liver
- Improved endothelial function
- Increased nitric oxide bioavailability
- Anti-inflammatory and antioxidant properties
- Atherosclerotic plaque stabilization and partial resorption
- Short-acting (t½ 1–4 h): Lovastatin, simvastatin, pravastatin, fluvastatin → taken with evening meal (peak HMG-CoA reductase activity at midnight)
- Long-acting (t½ ~14–19 h): Atorvastatin, rosuvastatin → can be taken at any time
- Atorvastatin, simvastatin, and lovastatin are metabolized by CYP3A4 — avoid with macrolides, azole antifungals, cyclosporine, HIV protease inhibitors, and >1 qt/day grapefruit juice
- Clinical ASCVD
- LDL-C ≥ 190 mg/dL
- Diabetes aged 40–75 years
- Elevated 10-year ASCVD risk (≥7.5%), aged 40–75 years
- Myopathy/Rhabdomyolysis (most serious): Risk <0.1%; creatine kinase >10× ULN = myopathy diagnosis; presents with proximal weakness, myalgia, brown/red urine
- Hepatotoxicity: ALT elevation ≥3× ULN on two occasions; routine LFT monitoring no longer universally recommended by FDA due to low yield
- New-onset diabetes mellitus: ~1/1000 patients per year; higher risk with metabolic syndrome; benefits still outweigh risks
2. Ezetimibe
3. Bile Acid Sequestrants (Resins)
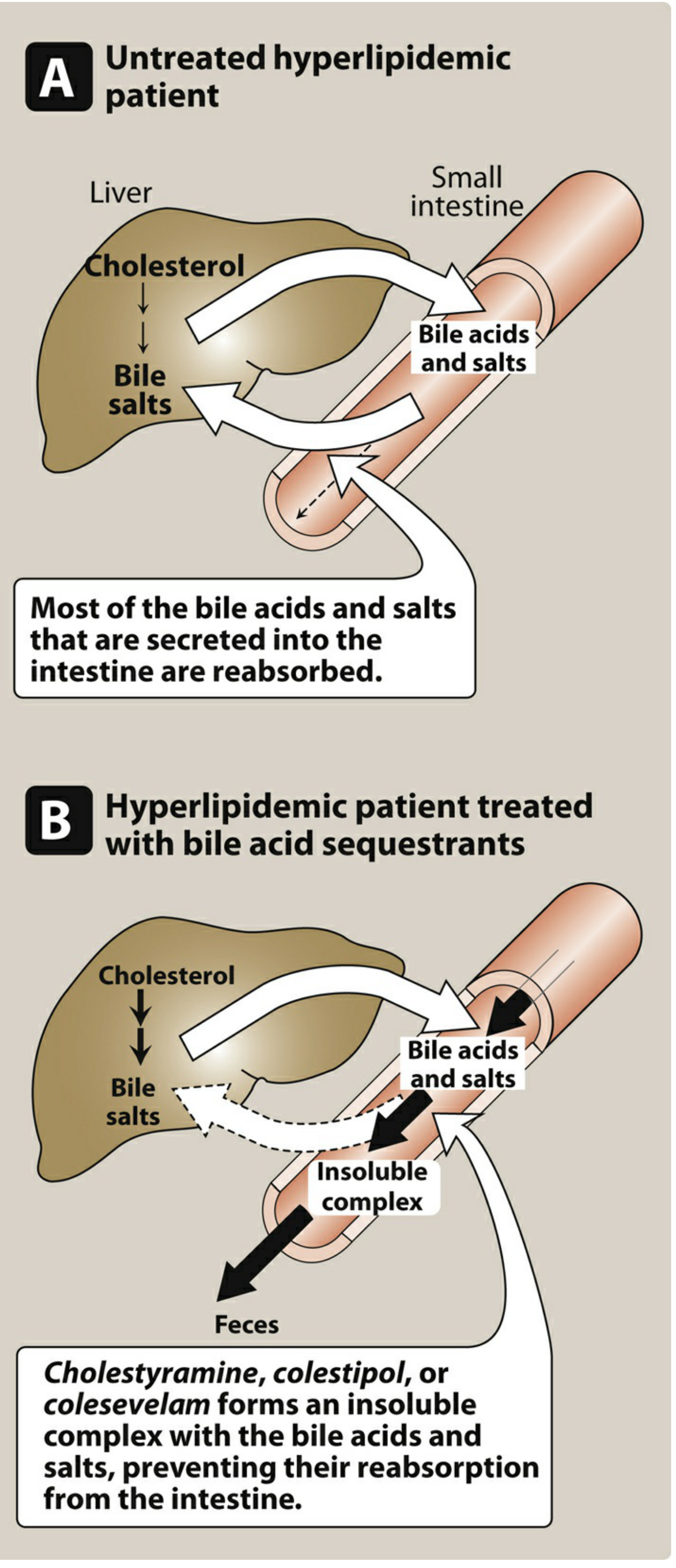
- GI: Constipation, nausea, flatulence (most common)
- Impair absorption of fat-soluble vitamins (A, D, E, K)
- Interfere with absorption of many drugs (digoxin, warfarin, thyroid hormone) — other drugs should be taken 1–2 h before or 4–6 h after the resin
- Contraindicated in significant hypertriglyceridemia (TG >400 mg/dL) — may raise TG further
4. PCSK9 Inhibitors
- Heterozygous or homozygous familial hypercholesterolemia
- Clinical ASCVD requiring additional LDL-C lowering beyond maximally tolerated statins
- Statin-intolerant patients with high ASCVD risk
5. Bempedoic Acid (ATP-Citrate Lyase Inhibitor)
6. Lomitapide (MTP Inhibitor)
7. Fibrates
- ↑ Lipoprotein lipase (LPL) expression → enhanced VLDL triglyceride hydrolysis
- ↓ Apolipoprotein CIII (an LPL inhibitor)
- ↑ ApoAI and ApoAII expression → ↑ HDL-C by 15–25%
- Net effect: ↓ TG by 40–60%, modest ↓ LDL-C
- Hypertriglyceridemia (especially severe cases to prevent pancreatitis)
- Type III hyperlipidemia (dysbetalipoproteinemia)
- Not indicated primarily for ASCVD event reduction
- Mild GI disturbances (most common)
- Gallstone formation (↑ biliary cholesterol excretion)
- Myositis/rhabdomyolysis — significant risk when gemfibrozil + any statin is combined; gemfibrozil is contraindicated with simvastatin
- Potentiate warfarin anticoagulation — monitor INR
- Contraindicated in severe hepatic or renal dysfunction, preexisting gallbladder disease
8. Niacin (Nicotinic Acid)
- Strongly inhibits lipolysis in adipose tissue (via GPR109A receptor) → ↓ free fatty acid flux to liver → ↓ hepatic VLDL/TG synthesis → ↓ LDL-C
- Blocks HDL catabolism by hepatocytes → ↑ circulating HDL
- Reduces apoB100 catabolism → ↓ LDL
- ↓ TG: 20–50%
- ↓ LDL-C: 10–20%
- ↑ HDL-C: 10–30% (most effective agent for raising HDL)
- Cutaneous flushing (most common, prostaglandin-mediated) — reduced by taking aspirin 30 min prior, slow dose titration, or sustained-release formulation
- Nausea, abdominal pain, pruritus
- Hyperuricemia/gout (inhibits tubular urate secretion)
- Impaired glucose tolerance
- Hepatotoxicity
- Contraindicated in active hepatic disease or active peptic ulcer
9. Omega-3 Fatty Acids
Summary: Drug Selection by Lipid Target
| Goal | First-line Agent | Add-on / Alternative |
|---|---|---|
| ↓ LDL-C | Statin | Ezetimibe → PCSK9 inhibitor → Bempedoic acid |
| ↓ TG (severe, >500 mg/dL) | Fibrate or Omega-3 FA | Niacin |
| ↑ HDL-C | Niacin (limited ASCVD benefit) | Fibrate |
| ↓ LDL in HoFH | Lomitapide + statin | PCSK9 inhibitor |
| Mixed dyslipidemia | Statin ± fibrate (avoid gemfibrozil) | Niacin (adjuvant) |
- CYP3A4 inhibitors + atorvastatin/simvastatin/lovastatin → ↑ myopathy risk
- Gemfibrozil + any statin → ↑ rhabdomyolysis (contraindicated with simvastatin)
- Bile acid sequestrants → ↓ absorption of many drugs; give other drugs 1–2 h before or 4–6 h after
- Fibrates + warfarin → ↑ INR; monitor closely
New guidelines
2025 2026 dyslipidemia guidelines lipid lowering ACC AHA ESC updated
"dyslipidemia" AND "guidelines" AND "lipid lowering"
2026 ACC AHA dyslipidemia guideline key recommendations LDL targets PCSK9 Lp(a) risk categories
ESC 2025 dyslipidemia guidelines new recommendations LDL targets treatment
2026 ACC AHA dyslipidemia guideline full details risk score Lp(a) evinacumab inclisiran children statin benefit groups
site:jacc.org OR site:ahajournals.org "2026 dyslipidemia guideline" LDL targets primary secondary prevention non-HDL apoB
2025 ESC EAS dyslipidemia focused update new drugs ACS bempedoic acid evinacumab inclisiran Lp(a) targets
2026 ACC AHA dyslipidemia guideline PREVENT score non-HDL apoB targets hypertriglyceridemia children familial hypercholesterolemia
2025 ESC EAS dyslipidemia update LDL targets risk categories inclisiran siRNA SCORE2 HIV cancer statin
New Dyslipidemia Guidelines (2025–2026)
1. 2026 ACC/AHA Multisociety Dyslipidemia Guideline
🔵 New Risk Assessment Tool: PREVENT™
- Estimates 10-year and 30-year risk of fatal and non-fatal ASCVD events
- Incorporates kidney function (eGFR) and metabolic factors
- Better calibrated across sex, race, and age groups
- Aligns with the concept of cardiovascular-kidney-metabolic (CKM) syndrome
🔵 Restored LDL-C Treatment Goals
| Prevention Setting | Risk Level | LDL-C Goal |
|---|---|---|
| Primary prevention | Borderline/Intermediate risk | < 100 mg/dL |
| Primary prevention | High risk | < 70 mg/dL |
| Secondary prevention | Very high risk (ASCVD + risk enhancers) | < 55 mg/dL |
- Non-HDL-C = LDL-C target + 30 mg/dL
- ApoB target for very high risk: < 65 mg/dL
🔵 Updated Primary Prevention Strategy
- LDL-lowering therapy may be considered for primary prevention in adults with 10-year PREVENT-ASCVD risk of 3–<5% (borderline risk), with shared decision-making
- Statin therapy recommended at ≥5–<7.5% risk with risk-enhancing factors
- Statin therapy recommended at ≥7.5% 10-year ASCVD risk
🔵 Expanded Risk-Enhancing Factors
- Lipoprotein(a) [Lp(a)] — should be measured at least once in all adults; elevated Lp(a) is a risk-enhancing factor and an indicator for more aggressive LDL-C lowering
- Coronary artery calcium (CAC) scoring — recommended for men ≥40 years and women ≥45 years to refine risk assessment; absolute CAC value and sex/age/race-standardized percentile are both used
- Remnant particles and TG-rich lipoproteins — elevated TG and remnant cholesterol now recognized as independent ASCVD risk factors
- CKM syndrome — kidney disease and metabolic factors formally integrated into risk
🔵 New and Expanded Drug Recommendations
| Drug | Update |
|---|---|
| Statins | Remain first-line; high-intensity preferred for high/very high risk |
| Ezetimibe | Second-line add-on; use earlier in treatment algorithm |
| PCSK9 inhibitors (alirocumab, evolocumab) | Recommended for very high-risk ASCVD or FH not at target on statin + ezetimibe |
| Inclisiran (siRNA) | Formally recommended; 2 doses/year provides sustained ≥50% LDL-C reduction; novel mechanism (RNA interference of PCSK9 synthesis) |
| Bempedoic acid | Recommended for statin-intolerant patients; add-on therapy |
| Omega-3 (icosapent ethyl) | Recommended for high TG (≥150 mg/dL) in ASCVD patients already on statin (based on REDUCE-IT) |
| Evinacumab | Recommended for homozygous FH (HoFH) — ANGPTL3 inhibitor |
🔵 Hypertriglyceridemia
- TG ≥ 150 mg/dL: lifestyle modification first
- TG ≥ 500 mg/dL: fibrate or omega-3 to prevent pancreatitis
- Icosapent ethyl preferred over mixed omega-3 for ASCVD risk reduction
🔵 Children and Adolescents
- Lipid screening recommended starting at age 9–11 (and again 17–21)
- Earlier statin initiation considered for familial hypercholesterolemia in children ≥8–10 years
- Expanded guidance for pediatric dyslipidemia management
🔵 Referral to Lipid Specialist
- HoFH or suspected FH with inadequate response
- Very high Lp(a) (≥ 100 mg/dL or ≥250 nmol/L)
- Statin intolerance with persistent LDL-C above goal
- Complex mixed dyslipidemias
2. 2025 ESC/EAS Focused Update on Dyslipidaemias
🔴 Risk Assessment: SCORE → SCORE2 / SCORE2-OP
- SCORE2 — estimates both fatal and non-fatal CVD events in adults 40–89 years without known CVD or diabetes
- SCORE2-OP (Older Persons) — better calibrated for elderly patients
- Additional risk modifiers: family history, ethnicity, hs-CRP, Lp(a), subclinical atherosclerosis (imaging or CAC score by CT)
| Risk Category | LDL-C Target | % Reduction |
|---|---|---|
| Low risk | < 3.0 mmol/L (116 mg/dL) | — |
| Moderate risk | < 2.6 mmol/L (100 mg/dL) | ≥30% |
| High risk | < 1.8 mmol/L (70 mg/dL) | ≥50% |
| Very high risk | < 1.4 mmol/L (55 mg/dL) | ≥50% |
🔴 New Drug Recommendations
- Class IB — recommended for patients unable to take statins (statin intolerance)
- Class IIaC — may be used as add-on to maximally tolerated statin ± ezetimibe in high-risk patients not at LDL-C target
- Recommended specifically for homozygous familial hypercholesterolemia (HoFH) as adjunct to other LDL-lowering therapy (based on Phase 3 ELIPSE HoFH trial)
- Can reduce LDL-C by ~47% in HoFH where LDL receptors are non-functional
- Formally endorsed; given twice yearly subcutaneously; reduces LDL-C by ~50%
🔴 ACS: Start Lipid Lowering at Index Hospitalization
- High-intensity statin + ezetimibe should be started during the index ACS hospitalization
- LDL-C should be rechecked at 4–6 weeks; if target not met, add PCSK9 inhibitor or inclisiran
- Goal: LDL-C < 1.4 mmol/L (55 mg/dL) and ≥50% reduction within 4–6 weeks of ACS
🔴 Lipoprotein(a) [Lp(a)]
- Lp(a) is formally recognized as an independent cardiovascular risk factor
- Measurement recommended at least once in all adults; elevated Lp(a) (≥50 mg/dL or ≥125 nmol/L) should trigger more intensive LDL-C lowering
- No specific Lp(a)-lowering drug is yet approved (pelacarsen, olpasiran in trials); current management is to treat overall ASCVD risk more aggressively
🔴 Special Populations
| Population | New Recommendation |
|---|---|
| HIV | Statin therapy recommended from age ≥40 years, regardless of LDL-C (due to elevated CVD risk) |
| Cancer (anthracyclines) | Statin therapy recommended in high-risk patients on anthracycline chemotherapy |
| Diabetes (T2DM) | SCORE2-Diabetes algorithm used; PCSK9 inhibitors considered earlier |
| Hypertriglyceridemia | New treatment algorithm; fibrates, omega-3s, pemafibrate considered |
| Supplements/vitamins | Explicitly advised against for CVD prevention (no benefit) |
Key Comparisons: 2026 ACC/AHA vs. 2025 ESC/EAS
| Feature | 2026 ACC/AHA | 2025 ESC/EAS Focused Update |
|---|---|---|
| Risk score | PREVENT™ | SCORE2 / SCORE2-OP |
| LDL-C targets | Restored (<55 / <70 / <100 mg/dL) | Unchanged from 2019 (<55 / <70 / <100 / <116 mg/dL) |
| Lp(a) screening | Once in all adults | Recommended; ≥50 mg/dL = risk modifier |
| CAC scoring | Recommended (men ≥40, women ≥45) | Recommended for moderate-risk patients |
| ACS: early therapy | Emphasized | High-intensity statin + ezetimibe at index hospitalization |
| Bempedoic acid | Recommended | Class IB (statin intolerance); IIaC (add-on) |
| Evinacumab | Recommended for HoFH | Recommended for HoFH |
| Inclisiran | Formally recommended | Endorsed |
| Children | Expanded guidance (screening from age 9) | Less detailed |
| HIV / Cancer | Not specifically featured | New recommendations |
| Supplements | Not recommended | Explicitly advised against |
Bottom Line
- Lower is better — LDL-C targets are now more aggressive, especially for very high-risk patients (< 55 mg/dL / 1.4 mmol/L)
- Lp(a) matters — universal screening is now recommended; it refines risk stratification
- New drugs have a formal role — bempedoic acid, evinacumab, and inclisiran are now guideline-endorsed
- Treat earlier and more intensively — especially in ACS, start combination therapy at hospitalization
- Personalized risk assessment — PREVENT™ and SCORE2 replace older tools; CAC and Lp(a) refine the picture
- Supplements do not work — dietary supplements and vitamins are not recommended for CVD prevention