Atrial fibrilation
Reading File
Finding Sources
Searching PubMed
"atrial fibrillation"[MeSH Terms] AND "management"[MeSH Terms]
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Searching Images
atrial fibrillation ECG rhythm strip
Reading File
Reading File
I now have comprehensive content to provide a thorough overview of atrial fibrillation. Let me compose the answer.
Atrial Fibrillation (AF)
Definition & ECG Features
AF is a supraventricular arrhythmia characterized by:
- Low-amplitude baseline oscillations (fibrillatory / f-waves) from chaotically firing atria at 300–600 beats/min, variable in amplitude, shape, and timing
- Irregularly irregular ventricular rhythm — the hallmark clinical sign
- Absent discrete P waves
- Ventricular rate during untreated AF is typically 100–160 beats/min
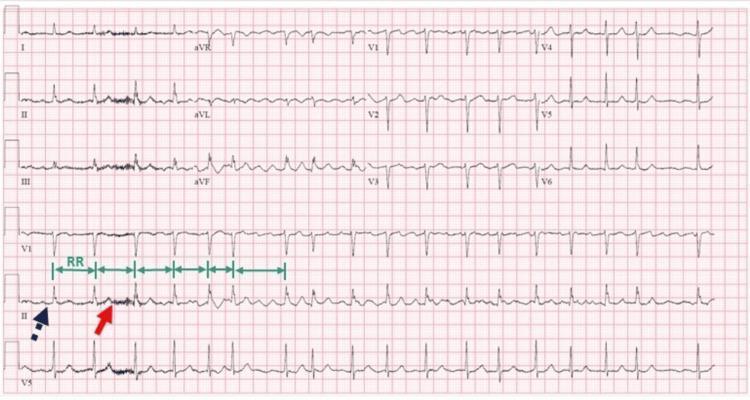
Classic 12-lead ECG of AF: absent P waves (red arrow), fibrillatory baseline, and irregularly irregular R-R intervals (green arrows).
Classification
| Type | Definition |
|---|---|
| Paroxysmal | Terminates spontaneously within 7 days |
| Persistent | Continuously present >7 days |
| Long-standing persistent | Present >1 year |
| Permanent | Patient and clinician decide to accept AF and abandon rhythm control; this is a therapeutic attitude, not a pathophysiological category |
| Vagotonic (paroxysmal subtype) | ~25% of paroxysmal AF; triggered by high vagal tone (evening, sleep); worsened by digoxin |
| Adrenergic (paroxysmal subtype) | Triggered by exercise or emotional stress; β-blockers preferred |
— Braunwald's Heart Disease, Chapter 66
Epidemiology
- Affects ~12% of adults ≥75 years and 18% of those ≥85 years
- About one-third of all AF patients are ≥80 years (2019 AHA/ACC/HRS guideline estimate)
- Common comorbidities: hypertension, coronary heart disease, obesity, sleep apnea, hyperlipidemia, heart failure
Mechanisms
AF is maintained by multiple wavelet re-entry and/or focal triggers (most commonly from pulmonary vein ostia). The key elements are:
- Trigger: Ectopic impulses, often from the pulmonary veins, initiate AF
- Substrate: Atrial fibrosis, conduction abnormalities, and stretch provide a favorable re-entrant environment
- Remodeling: AF itself causes electrical and structural remodeling ("AF begets AF") — progressive shortening of the atrial refractory period and loss of rate adaptation
- Autonomic influences: Vagal or adrenergic tone can both precipitate and maintain AF depending on the subtype
Causes / Risk Factors
- Hypertension (most common)
- Ischemic / structural heart disease
- Valvular disease (especially mitral stenosis — MS + AF carries near-prosthetic-valve stroke risk; warfarin, not DOACs, required in rheumatic MS)
- Thyrotoxicosis
- Heart failure (both cause and consequence)
- Obesity, sleep apnea
- Alcohol ("holiday heart")
- Post-cardiac surgery
- Genetic factors (familial AF, mutations in ion channel and gap junction genes)
Clinical Features
Symptoms:
- Palpitations, fatigue, reduced exercise tolerance
- Dyspnea, lightheadedness, chest discomfort
- Acute pulmonary edema (loss of atrial kick in a stiff LV)
- Syncope, fall, or stroke as initial presentation (especially in elderly)
- Frequently asymptomatic — subclinical AF detected on implanted devices in up to 50% of pacemaker/ICD patients
Complications:
- Stroke / thromboembolism: Nonvalvular AF → 5-fold increase in stroke risk; strokes tend to be severe
- Heart failure (rate-related cardiomyopathy)
- Cognitive impairment, reduced physical performance, increased mortality
Diagnostic Evaluation
- ECG — gold standard; 12-lead or rhythm strip
- Holter / event monitor — for paroxysmal AF
- Implantable loop recorder — high yield for cryptogenic stroke workup
- Echocardiography — assess structural disease, LA size, LV function, thrombus (especially transesophageal echo before cardioversion)
- TFTs, CMP, CBC — exclude reversible causes
- CHA₂DS₂-VASc score — stroke risk stratification
- HAS-BLED score — bleeding risk assessment
Stroke Prevention (Anticoagulation)
CHA₂DS₂-VASc Score
| Factor | Points |
|---|---|
| Congestive heart failure | 1 |
| Hypertension | 1 |
| Age ≥75 years | 2 |
| Diabetes mellitus | 1 |
| Stroke / TIA / thromboembolism (prior) | 2 |
| Vascular disease (prior MI, PAD, aortic plaque) | 1 |
| Age 65–74 years | 1 |
| Sex category (female) | 1 |
2019 AHA/ACC/HRS Guidelines (Class I):
- Men with CHA₂DS₂-VASc ≥ 2 → anticoagulate
- Women with CHA₂DS₂-VASc ≥ 3 → anticoagulate
- All patients ≥75 years (score ≥ 2 by definition) → anticoagulate regardless of AF type
Key points:
- DOACs (dabigatran, rivaroxaban, apixaban, edoxaban) preferred over warfarin for nonvalvular AF — no dietary restrictions, no INR monitoring, similar or better efficacy with less bleeding in elderly
- Warfarin (target INR 2–3) remains first choice in rheumatic mitral stenosis and mechanical valves (DOACs are contraindicated in these)
- Aspirin is NOT effective for stroke prevention in AF; no longer recommended for this purpose
- In elderly, typical warfarin maintenance dose is 2–5 mg/day, often started without a loading dose
Acute Management
Hemodynamically Unstable AF
- Immediate direct-current cardioversion (DCCV) — biphasic shock, typically starting at 200 J
Hemodynamically Stable AF
Rate control (first-line in most):
- IV β-blocker (metoprolol, esmolol) or non-dihydropyridine CCB (diltiazem, verapamil)
- Digoxin — slower onset, useful in HF with reduced EF
- Amiodarone — when other agents fail or in hemodynamically compromised patients
Rhythm control / Cardioversion:
- If AF onset ≤48 hours, cardioversion can proceed after anticoagulation with heparin
- If AF onset >48 hours or unknown duration: anticoagulate for ≥3 weeks before cardioversion OR perform transesophageal echo (TEE) to rule out LA thrombus, then cardiovert with adequate anticoagulation
- Anticoagulation must continue for ≥4 weeks after successful cardioversion regardless of CHA₂DS₂-VASc score (due to atrial stunning)
Long-Term Management: Rate vs. Rhythm Control
| Strategy | Approach | Agents / Methods |
|---|---|---|
| Rate control | Control ventricular rate; accept AF | β-blockers, CCBs (diltiazem, verapamil), digoxin |
| Rhythm control | Restore and maintain sinus rhythm | Antiarrhythmics (flecainide, propafenone, sotalol, amiodarone, dofetilide); cardioversion; catheter ablation |
Key principle: Rate control is recommended as first-line for asymptomatic or mildly symptomatic patients — it is safer and as effective as pharmacologic rhythm control (AFFIRM, RACE trials). Rhythm control is preferred for:
- Symptomatic patients despite rate control
- First-episode or young patients
- AF contributing to HF or tachycardia-induced cardiomyopathy
Target heart rate: Generally ≤110 bpm at rest ("lenient" rate control per RACE II trial) is acceptable; stricter control (<80 bpm rest, <110 bpm with exertion) for symptomatic patients.
Antiarrhythmic Drug (AAD) Selection
| Drug | Best for | Cautions |
|---|---|---|
| Flecainide / Propafenone | No structural heart disease | Contraindicated in CAD, HF, significant LVH |
| Sotalol | CAD, mild LVH | QTc monitoring; avoid in CrCl <40 |
| Amiodarone | HF, complex structural disease | Thyroid, pulmonary, hepatic toxicity; multiple drug interactions |
| Dofetilide | HF, CAD | In-hospital initiation; QTc monitoring |
| Dronedarone | Paroxysmal AF, no HF | Contraindicated in permanent AF with HF (increased mortality) |
Nonpharmacologic Management
Catheter Ablation (Pulmonary Vein Isolation, PVI)
- First-line rhythm control option in symptomatic paroxysmal AF refractory to or intolerant of AADs (Class I)
- Superior to AADs for maintaining sinus rhythm in paroxysmal AF
- Growing evidence supports early ablation, even as first-line, in selected patients
- In HF with reduced EF + AF, ablation significantly reduces mortality and hospitalization vs. rate control (CASTLE-AF trial)
Left Atrial Appendage (LAA) Occlusion
- Watchman device (percutaneous LAA closure) — for patients with nonvalvular AF who have contraindications to long-term anticoagulation
- Surgical LAA excision/ligation during concomitant cardiac surgery
Upstream Risk Factor Modification
- Weight loss, treatment of sleep apnea, blood pressure control, and exercise can reduce AF burden
- Addresses the AF substrate directly
Special Situations
| Situation | Key Consideration |
|---|---|
| WPW + AF | Risk of ventricular fibrillation via accessory pathway conduction (rates >250 bpm); avoid AV nodal blockers (digoxin, CCBs, adenosine); treat with DCCV or procainamide/ibutilide; refer for ablation |
| AF + Mitral Stenosis | Use warfarin (not DOACs); oral anticoagulants regardless of CHA₂DS₂-VASc; restore sinus rhythm after balloon mitral valvuloplasty (BMV) for better durability |
| AF + HFrEF | Catheter ablation preferred; amiodarone or dofetilide for AAD; avoid flecainide/propafenone/sotalol; DOACs preferred |
| AF in Pregnancy | β-blockers (metoprolol) preferred for rate control; electrical cardioversion safe at any trimester; heparin preferred over warfarin in first trimester |
| Elderly (≥75 years) | All have CHA₂DS₂-VASc ≥2; anticoagulate unless bleeding risk overwhelming; DOACs preferred; lower warfarin doses needed |
| Subclinical AF (device-detected) | Anticoagulation advised for episodes ≥5 hours with elevated stroke risk profile |
Sources: Braunwald's Heart Disease: A Textbook of Cardiovascular Medicine, Chapter 66 (Atrial Fibrillation); Chapter 54 (Heart Failure with Preserved/Mid-range EF); Chapter 77 (Cardiovascular Disease in the Elderly); Chapter 75 (Valvular Heart Disease).
This is a shared conversation. Sign in to Orris to start your own chat.