Teach me beta oxidation
Reading File
Finding Sources
Finding Sources
Searching Images
beta oxidation fatty acid spiral pathway diagram
Reading File
Reading File
Reading File
Reading File
Searching Images
beta oxidation spiral four steps acyl-CoA dehydrogenase enoyl hydratase thiolase mitochondria
Reading File
Loading Image
Loading Image
I now have everything needed. Here is a comprehensive, textbook-sourced lesson on beta oxidation.
Beta Oxidation of Fatty Acids
What Is It and Why Does It Matter?
Beta (β) oxidation is the primary catabolic pathway for fatty acids — it systematically dismantles fatty acyl chains into 2-carbon acetyl-CoA units, generating large amounts of ATP in the process. It is a major energy source for heart muscle, skeletal muscle, and liver, especially during fasting, exercise, and prolonged starvation.
- Basic Medical Biochemistry - A Clinical Approach - 6e, p. 1081
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease
Step 0: Getting the Fatty Acid into the Mitochondria
Before any oxidation can occur, the fatty acid must enter the mitochondrial matrix. This is the carnitine shuttle — one of the most clinically important regulatory points.
Activation (Cytoplasm → Outer Mitochondrial Membrane)
A fatty acid is first activated to fatty acyl-CoA by an acyl-CoA synthetase on the outer mitochondrial membrane. This consumes 2 ATP equivalents (ATP → AMP + PPi):
Fatty acid + CoASH + ATP → Fatty acyl-CoA + AMP + PPi
Chain-length specificity of acyl-CoA synthetases:
| Enzyme | Chain Length |
|---|---|
| Very-long-chain acyl-CoA synthetase | 14–26 C (peroxisomes only) |
| Long-chain acyl-CoA synthetase | 12–20 C |
| Medium-chain acyl-CoA synthetase | 6–12 C |
| Acetyl CoA synthetase | 2–4 C |
The Carnitine Shuttle (Crossing the Inner Membrane)
Long-chain fatty acyl-CoA cannot cross the inner mitochondrial membrane directly. It is transferred to carnitine by carnitine palmitoyltransferase I (CPT-I) on the outer face of the inner membrane, forming fatty acylcarnitine. This crosses via the carnitine:acylcarnitine translocase. Inside, CPT-II regenerates fatty acyl-CoA, releasing free carnitine (which exits in exchange).
Key regulation: Malonyl-CoA (the first committed intermediate of fatty acid synthesis) is a potent inhibitor of CPT-I. This ensures that fatty acid synthesis and oxidation do not run simultaneously — when you are building fat, you don't burn it.
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease
The β-Oxidation Spiral: Four Repeating Reactions
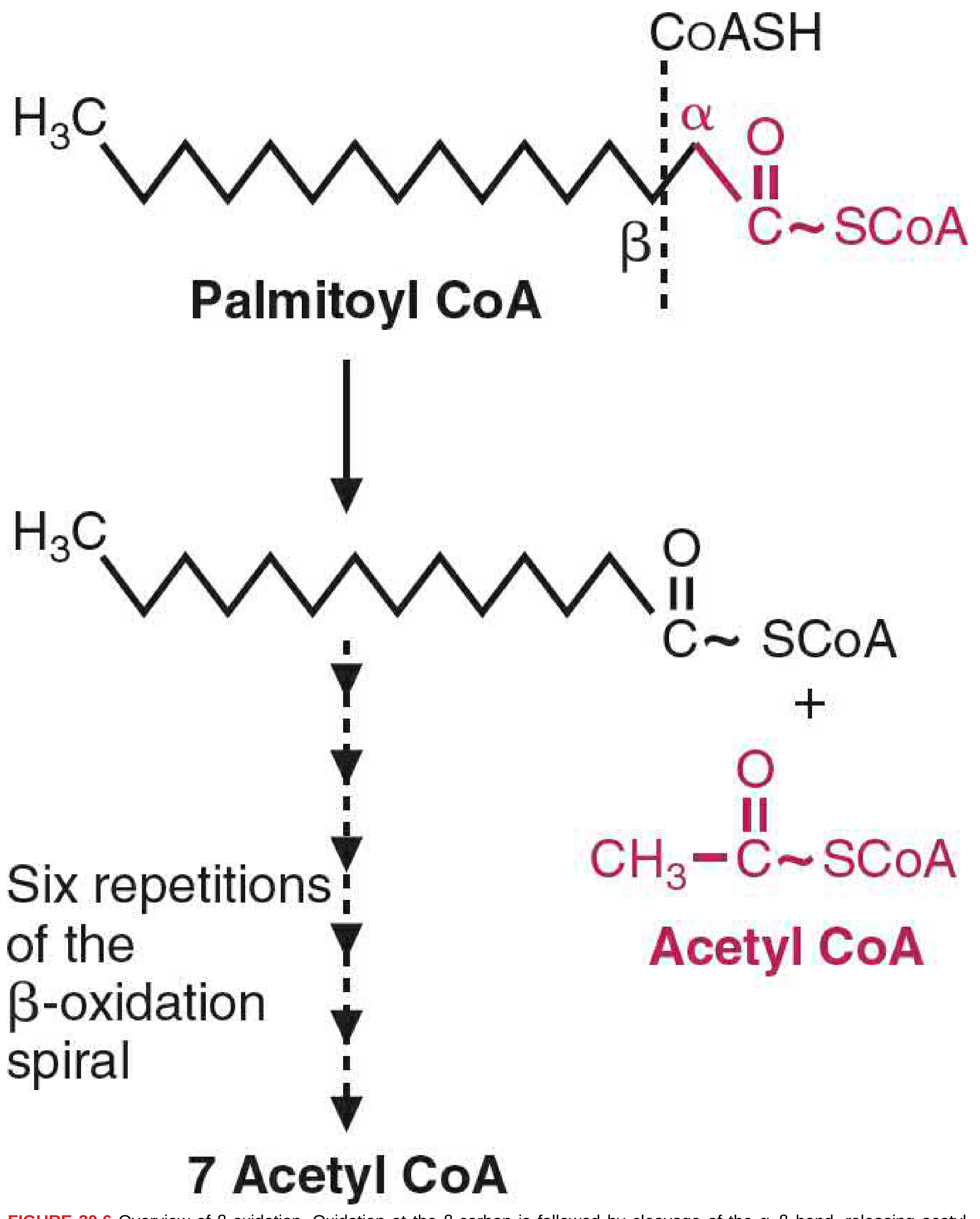
Once inside the mitochondrial matrix, fatty acyl-CoA undergoes the four-step spiral. Each turn clips off one acetyl-CoA and shortens the chain by 2 carbons.
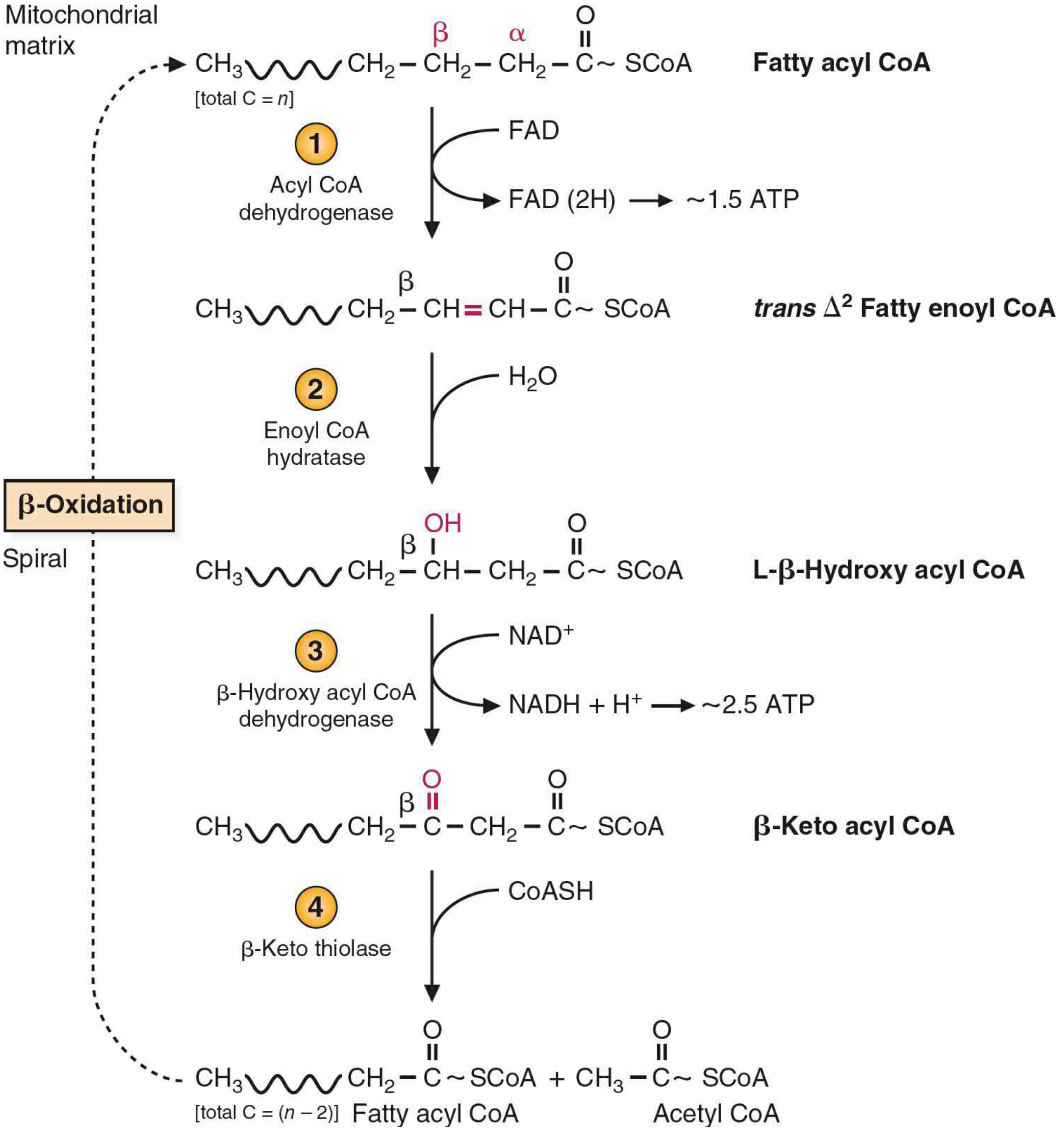
Step 1 — Oxidation: Acyl-CoA → trans-Δ²-Enoyl-CoA
Enzyme: Acyl-CoA dehydrogenase (FAD-linked; VLCAD, LCAD, MCAD, SCAD — each chain-length specific)
A double bond is inserted between the α- and β-carbons in the trans configuration. Two electrons are transferred to FAD, producing FADH₂ (~1.5 ATP via oxidative phosphorylation).
Step 2 — Hydration: trans-Enoyl-CoA → L-β-Hydroxyacyl-CoA
Enzyme: Enoyl-CoA hydratase (crotonase)
Water is added across the double bond. The –OH goes to the β-carbon (stereospecifically producing the L isomer).
Step 3 — Second Oxidation: L-β-Hydroxyacyl-CoA → β-Ketoacyl-CoA
Enzyme: L-β-Hydroxyacyl-CoA dehydrogenase (NAD⁺-linked)
The β-hydroxyl group is oxidized to a ketone. NAD⁺ accepts the electrons → NADH (~2.5 ATP).
Step 4 — Thiolysis: β-Ketoacyl-CoA → Acetyl-CoA + Shortened Acyl-CoA
Enzyme: β-Keto thiolase (β-ketoacyl-CoA thiolase)
CoASH attacks the β-keto group, cleaving the α–β bond. Products: one acetyl-CoA (2 carbons) + a fatty acyl-CoA shortened by 2 carbons. The shortened chain re-enters Step 1.
- Basic Medical Biochemistry - A Clinical Approach - 6e, pp. 1081–1082
Energy Accounting: Palmitate (C16:0) as the Example
Palmitate (16 carbons, fully saturated) requires 7 turns of the spiral to yield 8 acetyl-CoA.
| Source | Yield |
|---|---|
| 7 × FADH₂ | 7 × 1.5 = 10.5 ATP |
| 7 × NADH | 7 × 2.5 = 17.5 ATP |
| 8 × Acetyl-CoA → TCA cycle | 8 × 10 = 80 ATP |
| Subtotal | 108 ATP |
| Activation cost (ATP → AMP + PPi) | −2 ATP equivalents |
| Net yield | ~106 ATP |
This is far more than glucose oxidation (~30–32 ATP per molecule), which is why fats are such a dense energy store.
Fates of Acetyl-CoA
In most tissues, acetyl-CoA enters the TCA cycle → CO₂ + ATP + NADH + FADH₂.
In the liver (and only the liver), when acetyl-CoA accumulates faster than the TCA cycle can handle it (e.g., fasting, diabetic ketoacidosis), it is diverted into ketone body synthesis (acetoacetate, β-hydroxybutyrate, acetone) via the HMG-CoA pathway. Ketone bodies are exported to peripheral tissues (brain, heart, muscle) as alternative fuel.
Oxidation of Unsaturated Fatty Acids
Unsaturated fatty acids (e.g., oleate C18:1, linoleate C18:2) require extra enzymes because their natural cis double bonds don't match the normal β-oxidation intermediate (which needs a trans Δ² bond):
- Enoyl-CoA isomerase — converts a cis-Δ³ double bond to a trans-Δ² double bond, bypassing Step 1 (no FADH₂ produced for that turn).
- 2,4-Dienoyl-CoA reductase — for polyunsaturated fatty acids; uses NADPH to reduce a conjugated pair of double bonds to one trans-Δ³ bond, which is then isomerized. This costs one NADPH.
Net result: unsaturated fatty acids yield slightly less ATP than their saturated counterparts due to fewer FADH₂ produced and NADPH consumed.
- Basic Medical Biochemistry - A Clinical Approach - 6e, pp. 1481–1490
Odd-Chain Fatty Acids
The final spiral of an odd-chain fatty acid (rare in humans; found in ruminant products) yields propionyl-CoA (3 carbons) instead of acetyl-CoA. This is converted:
Propionyl-CoA → (propionyl-CoA carboxylase, biotin) → Methylmalonyl-CoA → (methylmalonyl-CoA mutase, vitamin B₁₂) → Succinyl-CoA → TCA cycle
This is the only route by which fatty acid carbons can contribute to gluconeogenesis (succinyl-CoA → OAA).
Peroxisomal β-Oxidation
Peroxisomes handle very-long-chain fatty acids (>C22) and branched-chain fatty acids that mitochondria cannot process directly:
| Feature | Mitochondria | Peroxisomes |
|---|---|---|
| Chain length handled | Short to long (C4–C20) | Very long (C10–C24) |
| Carnitine needed for entry? | Yes (CPT-I) | No |
| Step 1 electron acceptor | FAD → ETC → ATP | FAD → O₂ → H₂O₂ (no ATP) |
| H₂O₂ disposal | — | Catalase |
| End products | Acetyl-CoA → TCA | Acetyl-CoA + octanoyl-CoA (exits to mitochondria) |
Because the first step donates electrons directly to O₂ (not the electron transport chain), peroxisomal β-oxidation is less ATP-efficient and generates oxidative stress if catalase is insufficient.
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease
Regulation
| Regulator | Effect |
|---|---|
| Malonyl-CoA (high = fed state) | Inhibits CPT-I → blocks entry of fatty acids into mitochondria |
| Insulin (fed state) | Activates ACC → raises malonyl-CoA → inhibits oxidation |
| Glucagon / epinephrine (fasting) | Inactivates ACC → lowers malonyl-CoA → activates CPT-I → promotes oxidation |
| NADH/NAD⁺ ratio (high) | Inhibits β-hydroxyacyl-CoA dehydrogenase (Step 3) |
| Acetyl-CoA accumulation | Inhibits thiolase (Step 4); signals ketogenesis in liver |
Clinically Relevant Enzyme Deficiencies
| Deficiency | Key Feature | Presentation |
|---|---|---|
| MCAD (most common) | Medium-chain acyl-CoA dehydrogenase | Hypoketotic hypoglycemia, elevated C8 acylcarnitine in urine; can cause sudden death in infants |
| LCAD / VLCAD | Long/very-long-chain dehydrogenase | Cardiomyopathy, muscle weakness, hypoglycemia |
| CPT-I / CPT-II | Carnitine palmitoyltransferase | Myoglobinuria after exercise (CPT-II); hepatic failure (CPT-I) |
| Carnitine deficiency | Primary or secondary | Muscle weakness, cardiomyopathy, hypoglycemia |
MCAD deficiency is diagnosed by newborn screening — elevated octanoylcarnitine (C8) in dried blood spot is pathognomonic.
- Basic Medical Biochemistry - A Clinical Approach - 6e, pp. 1492–1494
- Goldman-Cecil Medicine
Summary Flowchart (Text)
Fatty acid (cytoplasm)
↓ acyl-CoA synthetase (−2 ATP)
Fatty acyl-CoA
↓ CPT-I [inhibited by malonyl-CoA]
Fatty acylcarnitine → crosses inner membrane
↓ CPT-II
Fatty acyl-CoA (matrix)
↓ β-oxidation spiral (×n turns):
Step 1: −FAD → FADH₂ (1.5 ATP)
Step 2: +H₂O
Step 3: −NAD⁺ → NADH (2.5 ATP)
Step 4: +CoASH → Acetyl-CoA released
↓
Acetyl-CoA → TCA cycle (10 ATP each)
→ Ketone bodies (liver only, when acetyl-CoA excess)
This is a shared conversation. Sign in to Orris to start your own chat.