Endometriosis
Reading File
Finding Sources
Searching PubMed
endometriosis
Reading File
Finding Sources
Searching Images
endometriosis pathology histology
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive content from multiple authoritative sources. Let me compile the full answer.
Endometriosis
Definition
Endometriosis is the presence of endometrial glands and stroma outside the uterine cavity. It affects approximately 10% of women in their reproductive years and up to 25–40% of infertile women. Prevalence is higher (>30%) in women with chronic pelvic pain (CPP). — Berek & Novak's Gynecology; Robbins & Kumar Basic Pathology
Classification (Three Types)
| Type | Description |
|---|---|
| Superficial peritoneal | Implants on pelvic peritoneum |
| Ovarian endometriosis | Endometriomas ("chocolate cysts") |
| Deep infiltrating endometriosis (DIE) | Penetrates >5 mm beneath the peritoneal surface; highest risk of malignant transformation |
Common sites: ovaries, pouch of Douglas, uterosacral ligaments, fallopian tubes, bladder, rectum/sigmoid colon. Rare distant sites include diaphragm, pleura (thoracic endometriosis), umbilicus, and laparotomy scars. — Robbins & Kumar
Pathogenesis
The pathogenesis remains incompletely understood. Four leading theories:
- Retrograde menstruation (Sampson's theory) — Menstrual endometrium refluxes through the fallopian tubes and implants at ectopic sites. Most widely accepted.
- Benign metastasis theory — Endometrial tissue spreads to distant sites (bone, lung, brain) via blood vessels and lymphatics.
- Coelomic metaplasia theory — Coelomic epithelium (mesothelium) undergoes metaplastic differentiation into endometrial tissue, driven by its shared Müllerian origin.
- Extrauterine stem/progenitor cell theory — Bone marrow–derived stem cells differentiate into ectopic endometrial tissue.
Molecular mechanisms: Endometriotic implants express elevated aromatase (→ local estrogen production), COX-2/PGE₂, VEGF, and MMPs. Activated macrophages sustain a proinflammatory milieu that promotes implant survival and angiogenesis.
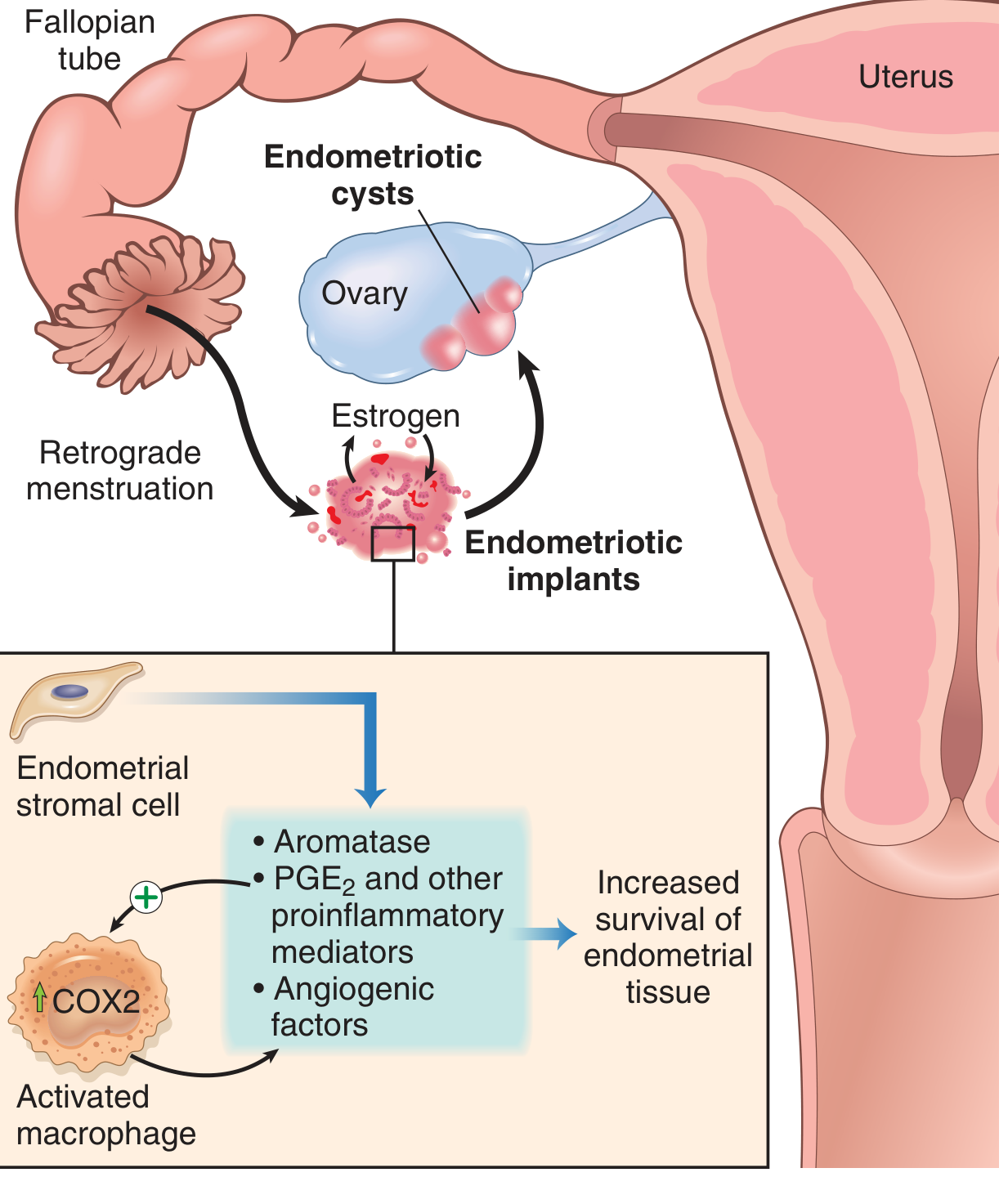
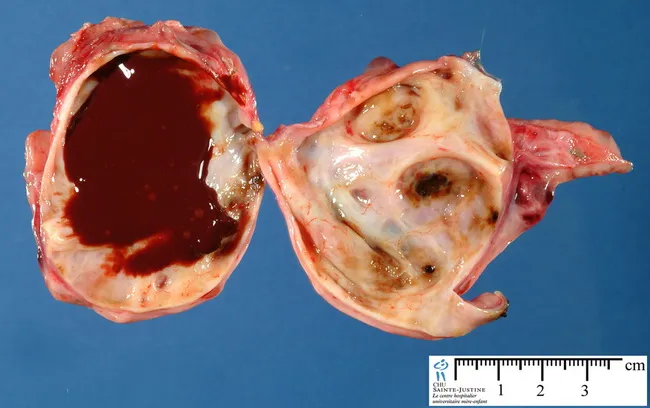
Morphology
Grossly, endometriotic lesions appear as:
- Early/active lesions: Red "flame-like" lesions, colorless vesicles, petechial spots
- Established lesions: "Powder-burn" black/brown nodules (1–2 cm) on serosal surfaces
- Ovarian endometriomas: Large cysts (3–5 cm) filled with dark brown, tarry fluid from repeated hemorrhage — the classic "chocolate cysts"
Organizing hemorrhage creates fibrous adhesions that can obliterate the pouch of Douglas and fuse pelvic organs. Histologically, both endometrial glands AND stroma must be identified for definitive diagnosis. — Robbins & Kumar
Clinical Features
Symptoms (often cyclic, worsening with menses):
- Dysmenorrhea — severe, often beginning up to 2 weeks before menses
- Chronic pelvic pain — diffuse, pressure-like, or sharp; referred to lower back, rectum
- Dyspareunia — deep thrust type
- Subfertility/infertility — 30–40% of patients present with infertility
- Dyschezia, urinary urgency/frequency, bloating (depending on site)
- Cyclic hemoptysis, pneumothorax, hemothorax (thoracic endometriosis)
- Menstrual irregularities
Signs on examination:
- Uterosacral nodularity and focal tenderness on bimanual/rectovaginal exam
- Fixed retroverted uterus or laterally deviated cervix (due to adhesions)
- Adnexal fullness (endometrioma)
The degree of visible disease often does not correlate with symptom severity. — Berek & Novak's Gynecology
Diagnosis
| Method | Notes |
|---|---|
| Clinical (nonsurgical) | ~50% accuracy; based on history and examination |
| Ultrasound | Can identify endometriomas (homogeneous ground-glass cysts that persist >1–2 menstrual cycles); cannot diagnose peritoneal implants |
| MRI | Best for mapping DIE |
| CA-125 | Elevated in some cases; low specificity and sensitivity — not diagnostic |
| Laparoscopy ± biopsy | Gold standard — direct visualization with histologic confirmation |
Suspicious laparoscopic findings (red/flame, powder-burn, peritoneal windows) should be biopsied. — Berek & Novak's Gynecology
Management
Medical (Hormonal Suppression)
The goal is to create a hypoestrogenic state that causes decidualization or atrophy of ectopic tissue.
| Agent | Mechanism | Notes |
|---|---|---|
| Combined oral contraceptives (COCPs) | Suppress gonadotropins → ↓ estrogen | First-line for symptomatic relief |
| Progestins (medroxyprogesterone, dienogest) | Decidualization of ectopic tissue | Oral or injectable; dienogest is highly effective |
| Levonorgestrel IUS | Local progestin action | Off-label for endometriosis/menorrhagia |
| GnRH agonists (leuprolide, goserelin, nafarelin) | Medical castration | Indicated if COCPs fail; "add-back" therapy (CEE or norethindrone) required after 6 months due to bone loss |
| GnRH antagonist — elagolix | Oral; blocks GnRH receptor | Effective for dysmenorrhea and pelvic pain; hypoestrogenic side effects (hot flashes, ↓ BMD) |
| Aromatase inhibitors | Block local estrogen synthesis | Used in refractory cases |
| Danazol | Synthetic androgen; inhibits gonadotropins | FDA-approved but rarely used (hirsutism, hepatotoxicity) |
| NSAIDs / COX-2 inhibitors | Reduce PGE₂-mediated inflammation and pain | Useful for symptom control |
Goodman & Gilman's Pharmacological Basis of Therapeutics; Harrison's Principles of Internal Medicine
Surgical
- Laparoscopy is preferred over laparotomy
- Excision > ablation — Cochrane review found excisional surgery reduces recurrence of endometriomas, return of pain, and improves spontaneous pregnancy rates
- Endometrioma: stripping/excision preferred; avoid compromising remaining ovarian tissue
- DIE: requires expertise in retroperitoneal dissection; multidisciplinary involvement (urology, colorectal surgery) often needed
- Hysterectomy ± bilateral salpingo-oophorectomy: definitive treatment for women who have completed childbearing — Berek & Novak's Gynecology
Complications
- Infertility (tubal involvement, impaired oocyte maturation, adhesions)
- Malignant transformation: mainly in DIE; associated with clear cell and endometrioid ovarian carcinoma
- Bowel/urinary tract obstruction from extensive adhesions
- Fistula formation (post-surgical)
Key Associations
- Endometrioid and clear cell ovarian carcinomas arise in a background of endometriosis
- Risk of malignancy is primarily confined to deep infiltrating endometriosis
- Patients with endometriosis have nerve fibers in endometrial tissue, contributing to pain mechanisms
Sources: Robbins & Kumar Basic Pathology (p. 689); Berek & Novak's Gynecology (pp. 577, 1299–1300); Goodman & Gilman's Pharmacological Basis of Therapeutics; Harrison's Principles of Internal Medicine 22E
Recent literature: A 2024 systematic review (PMID 39067455, Human Reproduction Update) highlights the role of fibrosis in endometriosis pathobiology — fibrotic remodeling of implants is increasingly recognized as a driver of pain and organ dysfunction beyond hormonal factors alone.
This is a shared conversation. Sign in to Orris to start your own chat.