Hepatopulmonaru syndrome full details for md exam answer 15 marks introduction etiology pathogenesis clinical features and diagnostic and treatment and complications and drugs dosage
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive data from the authoritative textbooks. I also have both key diagrams. Let me compose the complete MD exam answer.
Hepatopulmonary Syndrome (HPS) — Complete MD Exam Answer (15 Marks)
1. INTRODUCTION & DEFINITION
Hepatopulmonary syndrome (HPS) is a pulmonary vascular complication of chronic liver disease characterized by the clinical triad of:
Liver disease + Intrapulmonary vascular dilatation (IPVD) + Arterial hypoxemia
It is formally defined as a widened alveolar-arterial oxygen gradient (AaPO₂) on room air — exceeding 15 mmHg (or 20 mmHg in patients ≥64 years of age) — due to intrapulmonary vasodilation, with or without frank hypoxemia, occurring in the setting of advanced liver disease or portal hypertension.
- Prevalence: 5–35% of patients with cirrhosis being evaluated for liver transplantation (LT)
- HPS significantly increases mortality in patients with cirrhosis
- It can also occur in non-cirrhotic portal hypertension, ischemic hepatitis, and chronic hepatitis without confirmed cirrhosis
Severity grading (based on resting PaO₂ on room air):
| Grade | PaO₂ |
|---|---|
| Mild | ≥ 80 mmHg |
| Moderate | 60–80 mmHg |
| Severe | 50–60 mmHg |
| Very Severe | < 50 mmHg |
2. ETIOLOGY
HPS arises in the context of conditions that cause portal hypertension or hepatic dysfunction:
Primary hepatic causes:
- Cirrhosis (most common) — alcoholic, viral (HBV, HCV), cryptogenic, NASH, autoimmune, primary biliary cirrhosis, primary sclerosing cholangitis
- Wilson's disease, hemochromatosis
Non-cirrhotic causes:
- Extrahepatic portal hypertension (e.g., portal vein thrombosis)
- Congenital absence of portal vein (abernethy malformation)
- Budd-Chiari syndrome
- Hereditary hemorrhagic telangiectasia
Key point: HPS has no correlation with the severity of hepatic dysfunction (MELD score), meaning mild liver disease can sometimes be associated with severe HPS.
3. PATHOGENESIS
The fundamental mechanism is intrapulmonary vascular dilation leading to ventilation-perfusion mismatch (V/Q mismatch), diffusion-perfusion impairment, and shunting.
Pathophysiologic cascade:
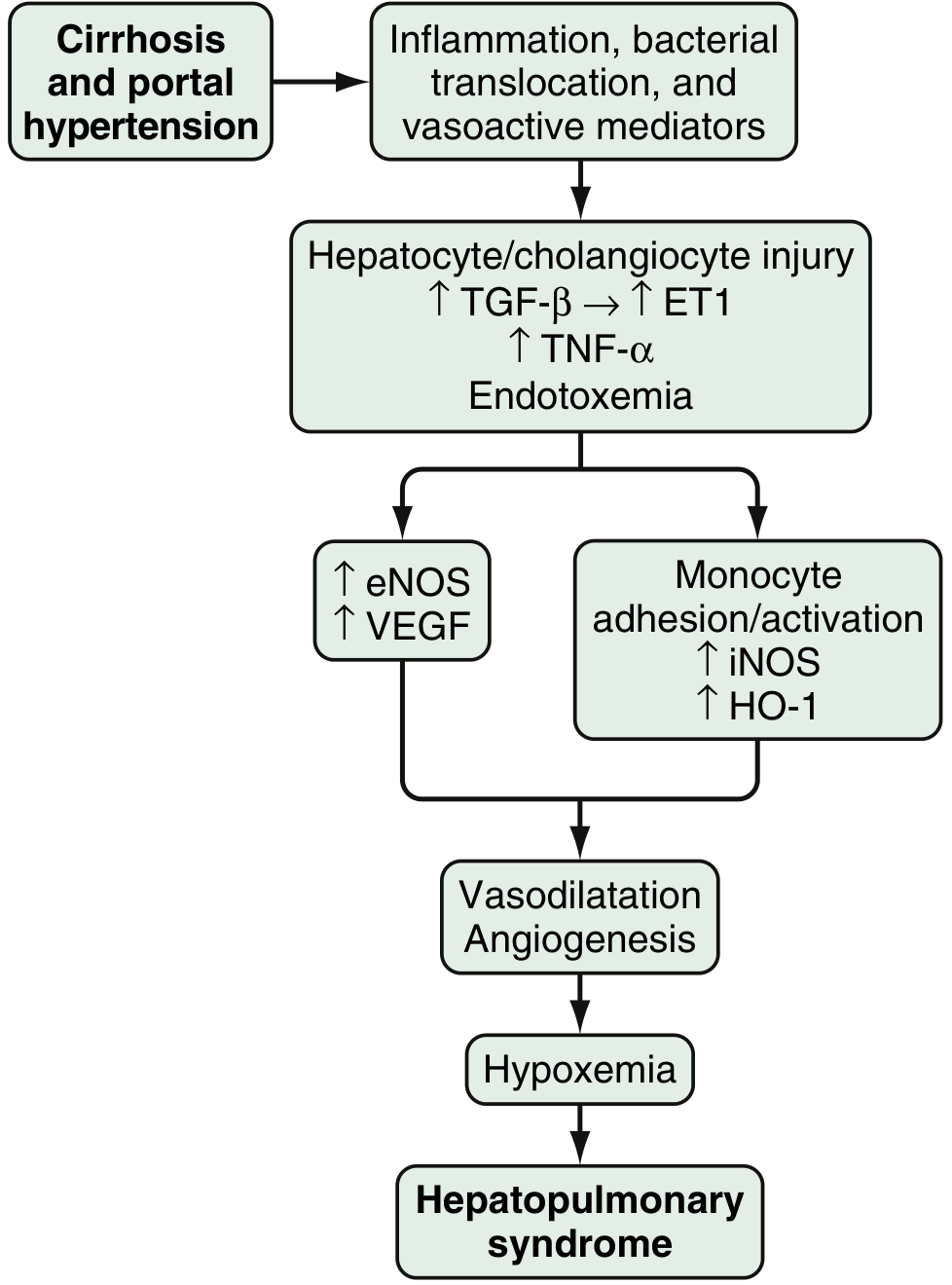
Step-by-step mechanism:
- Cirrhosis → Portal hypertension → inflammation, bacterial translocation, and release of vasoactive mediators
- Hepatocyte/cholangiocyte injury → ↑ TGF-β → ↑ Endothelin-1 (ET-1), ↑ TNF-α, endotoxemia
- Two parallel pathways:
- ET-1 pathway: Shear stress in pulmonary microvasculature → overexpression of endothelin-B (ETB) receptors → stimulates eNOS → ↑ Nitric Oxide (NO) → pulmonary vasodilation
- Monocyte adhesion pathway: Driven by ETB receptor activation, fractalkine receptor overexpression, and TNF-α → monocyte adherence in pulmonary vasculature → activates iNOS (more NO), heme oxygenase-1 (HO-1) (↑ carbon monoxide), and VEGF (angiogenesis)
- Result: Vasodilatation + angiogenesis in pulmonary microvasculature → hypoxemia → HPS
Mechanisms of hypoxemia (three components):
| Mechanism | Explanation |
|---|---|
| V/Q mismatch (major) | Dilated vessels at lung bases get excess perfusion relative to ventilation; worsens in upright posture (explains orthodeoxia) |
| Diffusion limitation | Dilated capillaries → ↑ distance from alveolar gas to central erythrocytes; ↑ cardiac output → reduced RBC transit time |
| Intrapulmonary shunting | Large arteriovenous dilations allow blood to bypass alveoli; predominates in severe HPS (PaO₂ 35–67 mmHg) |
Note: Even in areas of "shunt," many patients partially respond to 100% O₂, suggesting low V/Q or diffusion limitation behavior.
4. CLINICAL FEATURES
Symptoms:
- Dyspnea — insidious onset, most common; often progressive
- Platypnea — dyspnea worsening in upright posture, relieved by lying supine (pathognomonic but uncommon)
- Orthodeoxia — O₂ desaturation worsening in upright posture (PaO₂ falls ≥4 mmHg or SpO₂ falls ≥5% on standing) — highly characteristic
- Cough
- Up to 70% of patients have significant nocturnal hypoxemia (often ahead of daytime symptoms)
Signs:
- Digital clubbing — one of the most reliable signs; highly suggestive of HPS when present with hypoxemia and liver disease
- Central cyanosis (distal cyanosis)
- Signs of underlying liver disease: jaundice, spider angiomata, splenomegaly, ascites, palmar erythema, leukonychia
- Spider nevi (cutaneous manifestation of the same vascular dilatation process)
Important clinical correlations:
- Many patients with early HPS are asymptomatic or symptomatic only on exertion
- HPS does not correlate with severity of hepatic dysfunction (MELD score)
- Clubbing + hypoxemia (PaO₂ <60 mmHg) in a liver disease patient without intrinsic cardiopulmonary disease = highly suggestive of HPS
5. DIAGNOSIS
The diagnosis requires:
- Clinical suspicion
- Measurement of arterial blood gases
- Detection of intrapulmonary vascular dilation (IPVD)
- Exclusion of intrinsic cardiopulmonary disease
Diagnostic Criteria (ERS Task Force):
- Liver disease or portal hypertension (any etiology)
- Widened AaPO₂ ≥ 15 mmHg (≥20 mmHg if age ≥64) on room air
- Evidence of IPVD on contrast echocardiography or macroaggregated albumin (MAA) lung perfusion scan
Screening Algorithm:
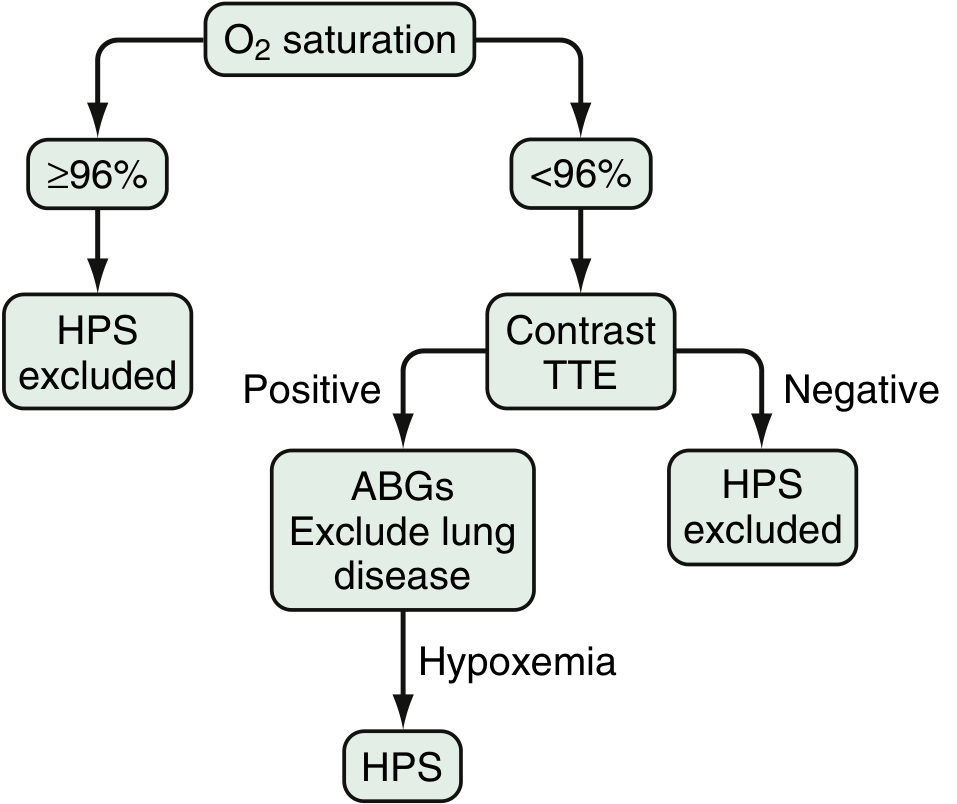
Diagnostic Tests:
1. Pulse Oximetry (Screening):
- SpO₂ <96% on room air → proceed to further evaluation
- Insensitive if used alone; must be confirmed with ABG
2. Arterial Blood Gas (ABG) — Gold standard for hypoxemia:
- Room air ABG: widened AaPO₂ ± reduced PaO₂
- Perform both sitting and standing to detect orthodeoxia
- AaPO₂ = PAO₂ – PaO₂ (normal <15 mmHg)
3. Contrast-Enhanced Transthoracic Echocardiography (CE-TTE) — Gold standard for IPVD:
- Peripheral IV injection of agitated saline creates microbubbles
- Normally absorbed in the pulmonary capillaries — do NOT reach left heart
- In intracardiac shunt: microbubbles appear in left heart within 1–2 cardiac cycles
- In IPVD/HPS: microbubbles appear in left heart after 3–6 cardiac cycles (delayed appearance = passage through dilated pulmonary vessels)
- Most sensitive test for intrapulmonary shunting
4. ⁹⁹mTc-Macroaggregated Albumin (MAA) Lung Perfusion Scan:
- MAA particles (20–90 μm) normally trapped in pulmonary capillaries
- In HPS: particles pass through dilated vessels → detected in brain, kidneys
- Cerebral uptake >6% = positive (quantifies shunt fraction)
- Shunt fraction >20% = poor prognosis post-LT
5. High-Resolution CT Chest:
- Can identify peripheral pulmonary arteriovenous malformations (AVMs) amenable to embolization
- Also excludes intrinsic lung disease
6. Pulmonary Angiography:
- Reserved for patients with severe HPS considering embolization
- Pattern A: diffuse fine "spongy" vascular dilatation (most common) — responds to O₂
- Pattern B: discrete arteriovenous communications — may not respond to O₂; candidate for embolization
7. Liver Biopsy:
- Not required for diagnosis but may identify underlying liver pathology
6. TREATMENT
A. Medical Therapy
No medical treatment has been proven definitively effective in large RCTs. Options:
| Drug | Mechanism/Evidence | Dose |
|---|---|---|
| Oxygen supplementation | Symptomatic; recommended when resting/exercise PaO₂ <60 mmHg; may slow progression | 2–4 L/min via nasal cannula; titrate to SpO₂ ≥88% |
| Garlic (Allium sativum) | Inhibits NO production; 2 small uncontrolled studies + 1 RCT showed improvement in oxygenation | 250 mg three times daily (pilot trials used) |
| Pentoxifylline | TNF-α inhibitor; anti-inflammatory; conflicting case series results | 400 mg three times daily |
| Norfloxacin | Reduces bacterial translocation → reduces endotoxemia → reduces NO | 400 mg twice daily (used in experimental models) |
| N-acetylcysteine (NAC) | Antioxidant; limited case reports | Variable |
| Methylene blue | NO scavenger; acute improvement in oxygenation reported in case reports | 3 mg/kg IV (acute, bridge to LT) |
| Sorafenib | Anti-VEGF/angiogenesis; animal model data; not established in humans | Under investigation |
| Octreotide | Reduces splanchnic vasodilation; limited evidence | 100–200 μg SC three times daily (case reports) |
Drugs NOT recommended:
- Indomethacin, almitrine — used in early case reports but not validated
- β-blockers (propranolol) — may worsen hypoxemia in HPS
B. Radiologic/Interventional Therapy
- TIPS (Transjugular Intrahepatic Portosystemic Shunt): Case reports of improvement by reducing portal pressure; invasive, not routinely recommended for HPS
- Pulmonary Angiography + Embolization: For Pattern B (discrete AVMs/fistulae on CT); may improve severe refractory hypoxemia as bridge to LT; not standard of care
C. Liver Transplantation (LT) — Definitive Treatment
- LT is the ONLY proven curative therapy for HPS
- Reverses HPS in up to 80% of patients
- Resolution of hypoxemia may take >12 months post-transplant, especially in severe cases
- MELD exception points are allocated for HPS patients with PaO₂ <60 mmHg on room air, increasing transplant priority
Post-LT complications specific to HPS:
- Transient worsening of hypoxemia post-operatively
- Development of pulmonary hypertension post-LT
- Embolic cerebrovascular events
Predictors of poor outcome after LT:
- PaO₂ <50 mmHg (very severe)
- Shunt fraction >20% on MAA scan
- Failure to improve with 100% O₂ (suggests true anatomical shunt)
7. COMPLICATIONS
| Complication | Details |
|---|---|
| Progressive hypoxemia | Natural course — severity worsens over time without LT |
| Nocturnal desaturation | Up to 70% of patients; occurs before daytime hypoxemia |
| Right heart failure (Cor pulmonale) | Chronic hypoxemia → pulmonary vasoconstriction → cor pulmonale |
| Polycythemia | Compensatory response to chronic hypoxemia |
| Cerebrovascular events (paradoxical emboli) | Intrapulmonary vascular channels act as conduit; may lead to cerebral abscess or stroke |
| Pulmonary hypertension (post-LT) | Paradoxical development after LT in some HPS patients |
| Increased surgical mortality | LT carries higher perioperative mortality in HPS vs. non-HPS patients |
| Transition to POPH | HPS and portopulmonary hypertension (POPH) can coexist in the same patient |
8. DIFFERENTIAL DIAGNOSIS
| Condition | Key Differentiator |
|---|---|
| Portopulmonary Hypertension (POPH) | Pulmonary arterial hypertension; no IPVD; responds to pulmonary vasodilators, not O₂ |
| Primary pulmonary disease (COPD, ILD) | Abnormal PFTs; no IPVD on echo; different CT pattern |
| Intracardiac shunt (ASD, PFO) | Microbubbles appear in left heart within 1–2 beats on echo |
| Hereditary Hemorrhagic Telangiectasia (HHT) | Skin/mucosal telangiectasias; positive family history; pulmonary AVMs on CT |
9. SUMMARY TABLE
| Feature | Details |
|---|---|
| Definition | Liver disease + IPVD + ↑AaPO₂ (≥15 mmHg) |
| Key mechanism | NO-mediated pulmonary vasodilatation |
| Pathognomonic features | Platypnea + Orthodeoxia |
| Best screening test | SpO₂ <96% → proceed further |
| Gold standard for IPVD | Contrast TTE (CE-TTE) — microbubbles in LA after ≥3 beats |
| Quantification of shunt | ⁹⁹mTc-MAA lung scan (cerebral uptake >6%) |
| No proven medical therapy | O₂ symptomatic; garlic + pentoxifylline investigational |
| Definitive treatment | Liver transplantation (reversal in ~80%) |
| MELD exception | PaO₂ <60 mmHg on room air qualifies |
Sources:
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease, Chapter 94
- Murray & Nadel's Textbook of Respiratory Medicine, Chapter 126
- Fishman's Pulmonary Diseases and Disorders
This is a shared conversation. Sign in to Orris to start your own chat.