Mandible
Reading File
Finding Sources
Searching Images
mandible anatomy diagram
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive content to deliver a complete answer on the mandible.
Mandible
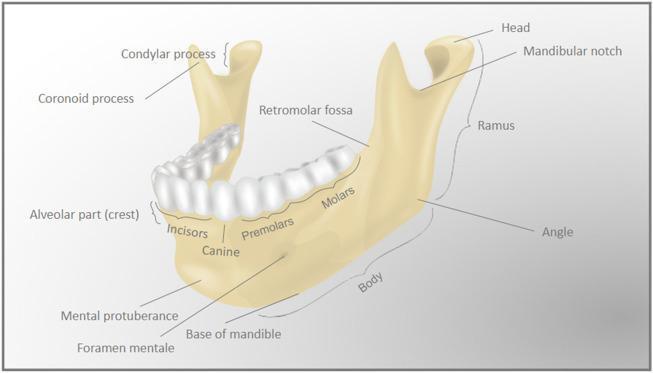
The mandible is the bone of the lower jaw — the only movable bone of the skull, and the most inferior structure visible in the anterior view of the skull.
Lateral (External) View
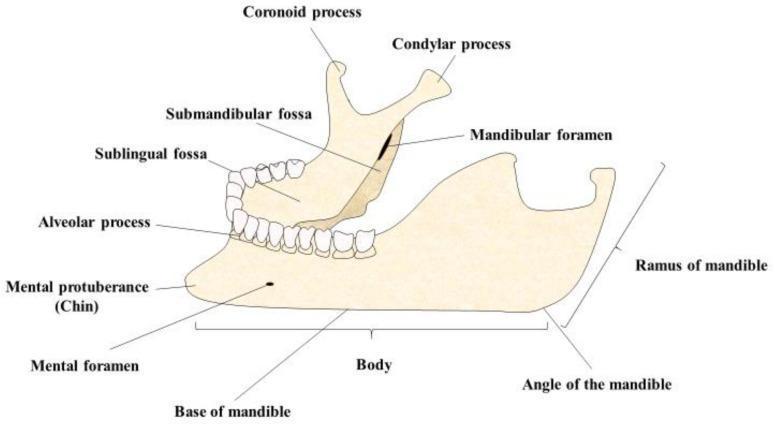
Medial (Internal) View
Overall Structure
The mandible consists of:
- A body — formed by the fusion of right and left halves at the mandibular symphysis anteriorly (marked by a small vertical ridge externally)
- Two rami — quadrangular vertical plates posteriorly
The body and ramus meet at the angle of the mandible on each side.
Body of the Mandible
The body is divided into two parts:
| Part | Description |
|---|---|
| Base of mandible | Lower border; persists throughout life |
| Alveolar part | Upper border; contains the sockets (alveoli) for the lower teeth; resorbed when teeth are removed |
External (Lateral) Surface
- Mental protuberance — midline chin prominence where the two halves meet
- Mental tubercles — small bumps just lateral to the mental protuberance
- Mental foramen — opens midway between the upper and lower borders, usually below the 2nd premolar; transmits the mental nerve and vessels (terminal branches of the inferior alveolar nerve/vessels)
- Oblique line — ridge running from the front of the ramus onto the body; attachment for muscles that depress the lower lip
Internal (Medial) Surface
- Superior & inferior mental spines (genial spines) — two pairs of small bony projections posterior to the symphysis:
- Superior pair → attachment for genioglossus (tongue muscle)
- Inferior pair → attachment for geniohyoid (connects mandible to hyoid)
- Mylohyoid line — oblique ridge running posterosuperiorly from near the midline to just below the last molar; origin of the mylohyoid muscle (floor of mouth)
- Sublingual fossa — shallow depression above the anterior 1/3 of the mylohyoid line; lodges the sublingual salivary gland
- Submandibular fossa — depression below the posterior 2/3 of the mylohyoid line; lodges the submandibular salivary gland
- Shallow groove for the lingual nerve — between the last molar and the mylohyoid line
- Retromolar triangle — small triangular depression behind the last molar; attachment site for the pterygomandibular raphe
Ramus of the Mandible
The ramus is quadrangular, oriented in the sagittal plane, one on each side.
Medial Surface
- Mandibular foramen — large opening for the inferior alveolar nerve and vessels entering the mandibular canal
- Lingula — small bony spine overhanging the mandibular foramen anteriorly; attachment for the sphenomandibular ligament
Superior Border
The ramus terminates superiorly in two processes separated by the mandibular notch (sigmoid notch):
| Process | Position | Articulation/Function |
|---|---|---|
| Coronoid process | Anterior | Insertion of temporalis muscle |
| Condylar process | Posterior | Articulates with temporal bone at the temporomandibular joint (TMJ) |
The condylar process has a head (rounded articular surface) and a neck below it.
Key Foramina & Their Contents
| Foramen | Location | Contents |
|---|---|---|
| Mental foramen | External body, below 2nd premolar | Mental nerve & vessels |
| Mandibular foramen | Medial ramus | Inferior alveolar nerve & vessels |
Muscle Attachments (Summary)
| Muscle | Attachment Site |
|---|---|
| Masseter | Angle & lower lateral ramus (insertion) |
| Medial pterygoid | Medial angle of ramus (insertion) |
| Lateral pterygoid | Neck of condyle / articular disc (insertion) |
| Temporalis | Coronoid process & anterior ramus (insertion) |
| Genioglossus | Superior mental spines (origin) |
| Geniohyoid | Inferior mental spines (origin) |
| Mylohyoid | Mylohyoid line (origin) |
| Digastric (anterior belly) | Digastric fossa near midline base (origin) |
| Buccinator | Alveolar part (lateral) posterior region (origin) |
Clinical Relevance
- Inferior alveolar nerve block: Local anaesthetic is injected near the mandibular foramen, on the medial surface of the ramus, to anaesthetise the teeth, lower lip, and chin on that side.
- Mental foramen: The mental nerve exits here; the foramen migrates posteriorly with age (from the deciduous incisor region toward the premolar region).
- Edentulous mandible: Loss of teeth causes resorption of the alveolar part, reducing overall mandibular height and potentially exposing the mental foramen to the upper border — relevant in denture fitting.
- Mandible fractures: Common at the condylar neck (most frequent), angle, parasymphysis/canine region; often bilateral because force is transmitted around the arch.
- Mandible dislocation: The condyle moves anterior to the articular eminence; associated with wide opening, trauma, or yawning.
— Gray's Anatomy for Students, pp. 993, 1229–1230
Smooth muscle and cardiac muscle
Reading File
Finding Sources
Finding Sources
Searching Images
smooth muscle histology microscopy
Searching Images
cardiac muscle histology intercalated disc
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
Reading File
Loading Image
Loading Image
I now have everything needed for a thorough, well-illustrated answer. Here it is:
Smooth Muscle & Cardiac Muscle
SMOOTH MUSCLE
Histology & General Characteristics
Smooth muscle lacks striations — unlike skeletal and cardiac muscle, its thick and thin filaments are not organized into sarcomeres, so no banding pattern is visible under light microscopy. Each fiber is a long, tapering, uninucleate cell with a centrally located, elongated nucleus at the cell's widest point.
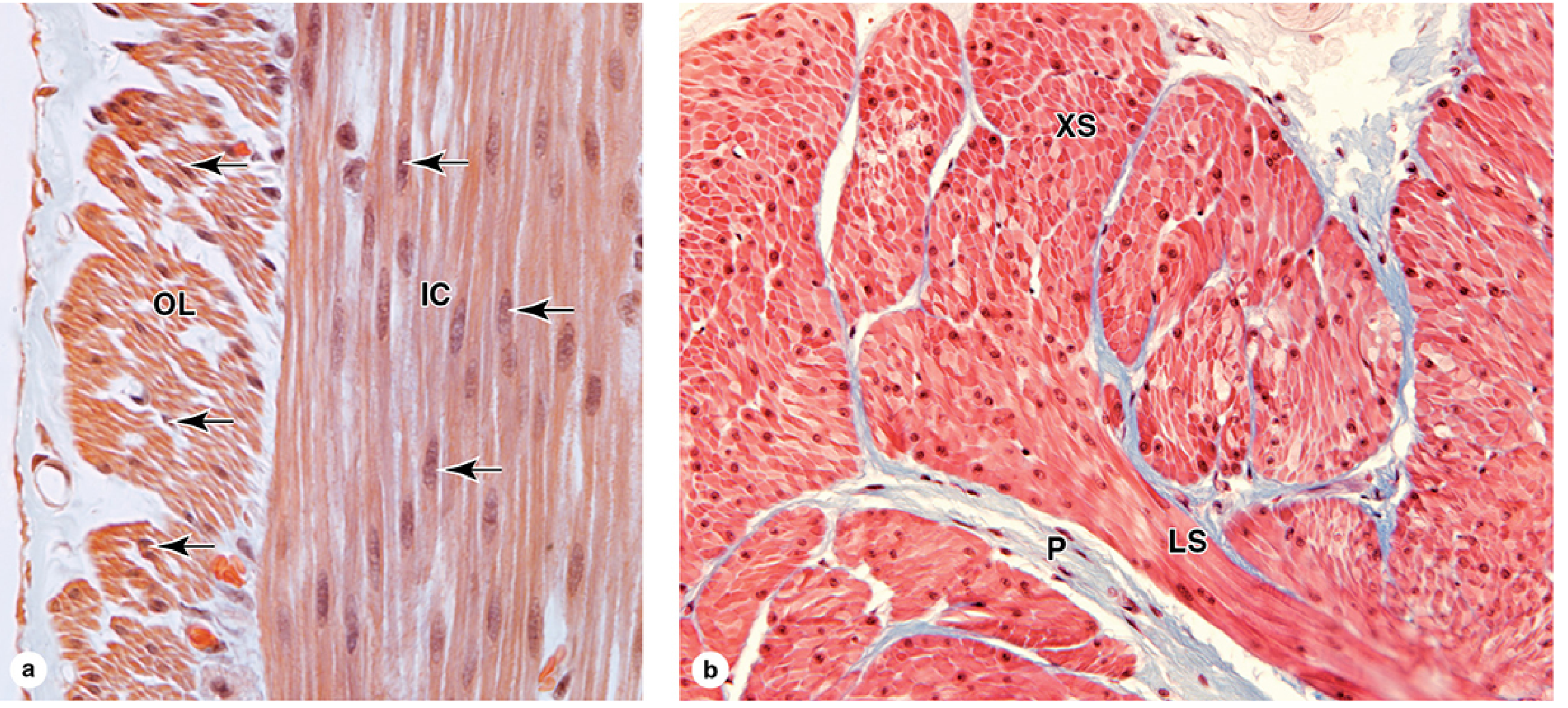
Smooth muscle of the GI tract (a) and bladder (b). (Junqueira's Basic Histology, p. 526–527)
Each fiber is enclosed by an external lamina and endomysium of type I and III collagen. The sarcoplasmic reticulum (SR) is less developed than in striated muscle; instead, shallow membrane invaginations called caveolae (analogous to T-tubules) cluster near the peripheral SR to regulate Ca²⁺.
Location & Function
| Location | Function |
|---|---|
| GI tract, bladder, uterus, ureter | Motility (peristalsis, emptying) |
| Blood vessels | Vasoconstriction/dilation, maintaining tone |
| Bronchioles, iris, ciliary muscle, vas deferens | Luminal diameter regulation |
Types of Smooth Muscle
| Type | Coupling | Location | Properties |
|---|---|---|---|
| Unitary (single-unit) | Gap junctions present | GI tract, bladder, uterus, ureter | Electrically coupled; synchronized contraction; spontaneous pacemaker/slow waves |
| Multiunit | Little or no coupling | Iris, ciliary muscle, vas deferens | Each fiber acts independently; densely innervated by ANS |
| Vascular | Intermediate | Blood vessel walls | Combination of unitary and multiunit properties |
Excitation–Contraction Coupling
The key difference from skeletal muscle: smooth muscle has no troponin. Instead, Ca²⁺ acts via calmodulin → myosin-light-chain kinase (MLCK).
![Smooth muscle E-C coupling cascade: depolarization/hormones/IP₃ → ↑[Ca²⁺] → Ca²⁺-calmodulin → MLCK → myosin-light-chain phosphorylation → cross-bridge cycling → tension](https://cdn.orris.care/cdss_images/01552b35ff2aca62ce0fdd91575e9396d92b7f8facbfaaefecd29dc0b0403d8d.png)
Fig. 1.29 — Costanzo Physiology, p. 49
Steps:
- Depolarization opens sarcolemmal voltage-gated Ca²⁺ channels → Ca²⁺ enters from ECF
- Entered Ca²⁺ triggers Ca²⁺-induced Ca²⁺ release (CICR) from SR via ryanodine receptor (RYR2)
- Two additional routes raise [Ca²⁺]:
- Ligand-gated Ca²⁺ channels — opened by hormones/neurotransmitters via G proteins
- IP₃-gated SR channels — hormones → G protein → PLC → PIP₂ → IP₃ → SR Ca²⁺ release
- Ca²⁺ binds calmodulin (4 ions cooperatively)
- Ca²⁺-calmodulin activates MLCK, which phosphorylates myosin light chain
- Phosphorylated myosin has increased ATPase activity → binds actin → cross-bridge cycling → tension
- Ca²⁺-calmodulin also phosphorylates calponin and caldesmon (thin-filament proteins), relieving their inhibition of myosin ATPase
Relaxation occurs when [Ca²⁺] falls via:
- Hyperpolarization (closes voltage-gated Ca²⁺ channels)
- cAMP/cGMP-mediated Ca²⁺ channel inhibition
- Reduced IP₃ production
- Increased SR Ca²⁺-ATPase activity
- Myosin-light-chain phosphatase dephosphorylates myosin light chain → cross-bridges stop
Ca²⁺ Sources (Comparison)
| Muscle Type | Main Ca²⁺ Source |
|---|---|
| Skeletal | SR only (via direct mechanical coupling of DHP → RYR1) |
| Smooth | Both ECF (voltage-gated + ligand-gated channels) and SR (CICR + IP₃-gated) |
| Cardiac | Both ECF and SR (CICR via Cav1.2 → RYR2) |
CARDIAC MUSCLE
Histology & Ultrastructure
Cardiac muscle is striated (like skeletal muscle) but composed of individual cells (cardiomyocytes) joined end-to-end — not fused into multinucleated syncytia.
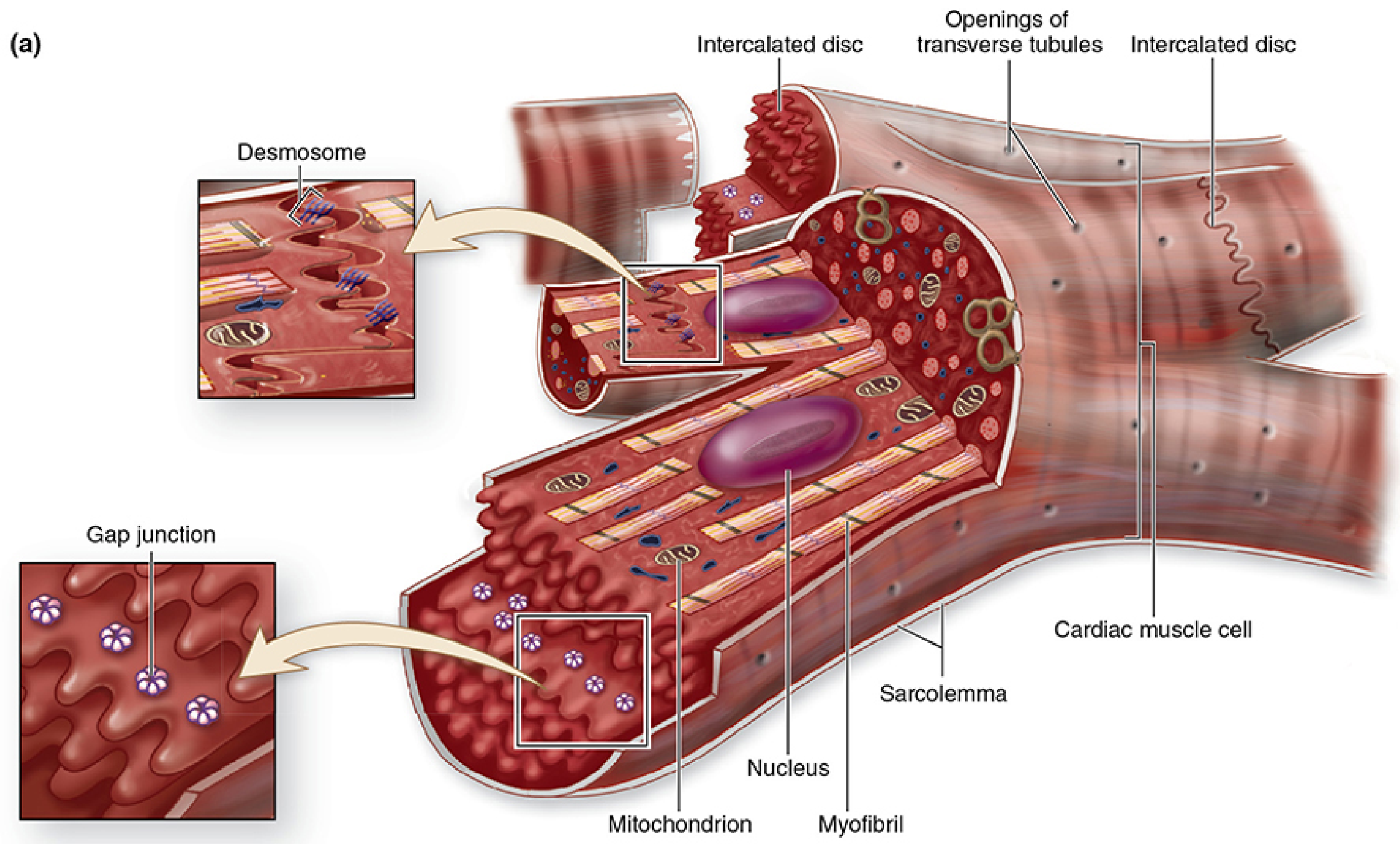
Fig. 10–16 Cardiac muscle cells with intercalated discs — Junqueira's Basic Histology, p. 521
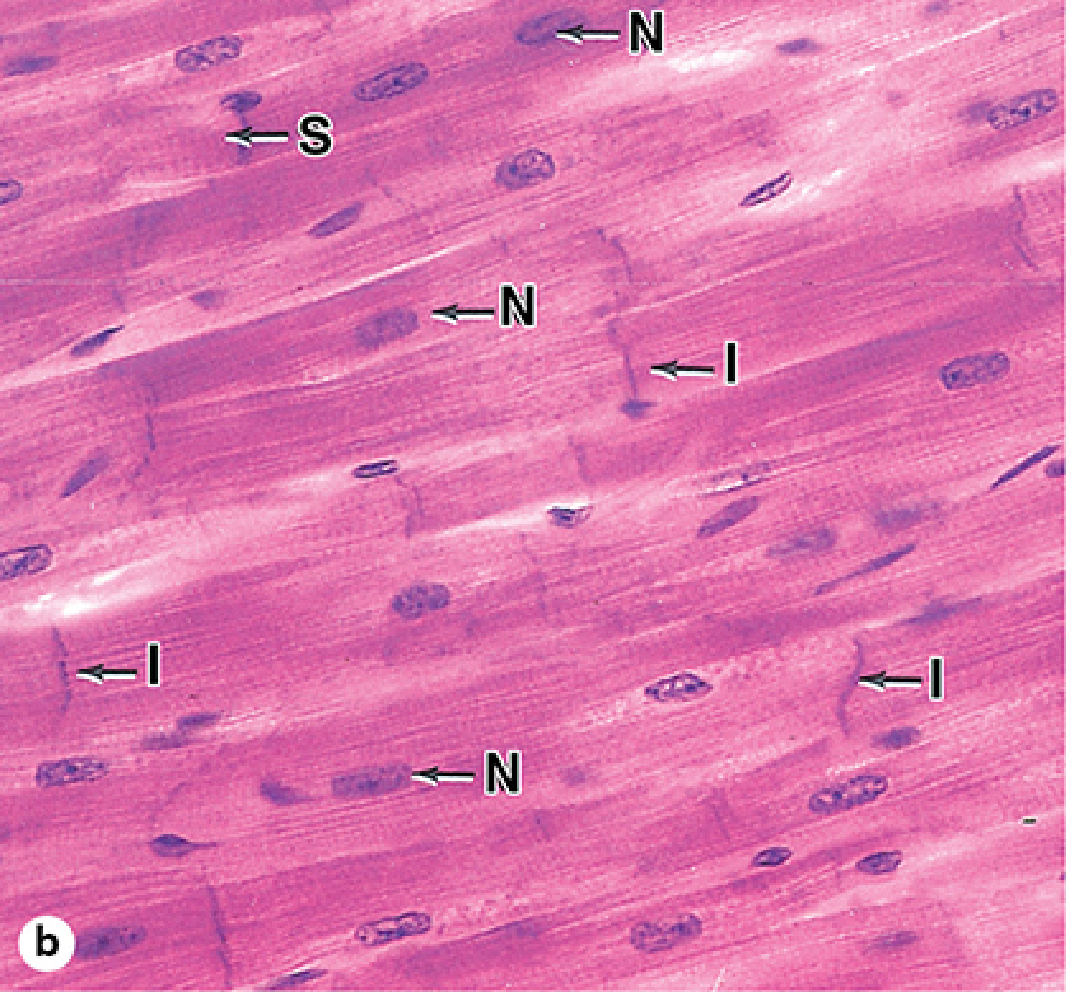
Fig. 10–16b — Junqueira's Basic Histology, p. 522
Key histological features:
| Feature | Cardiac Muscle | Skeletal Muscle |
|---|---|---|
| Striations | Present | Present |
| Nucleus | Single, central | Multiple, peripheral |
| Cell borders | Individual cells | Fused syncytium |
| Cell size | 15–30 µm diameter, 85–120 µm long | Much larger |
| Intercalated discs | Present (pathognomonic) | Absent |
| Branching | Yes | No |
| Mitochondria | Up to 40% of cell volume | Less abundant |
| T-tubules | At Z discs (dyads with SR) | At A-I band junctions (triads) |
| SR | Less organized | Highly organized |
Intercalated Discs — Structure & Function
Intercalated discs are the step-like junctions between adjacent cardiomyocytes. They appear as irregular transverse lines on H&E — do not confuse with the repetitive, closely-spaced sarcomere striations.
Transverse region (perpendicular to fiber axis):
- Desmosomes — mechanical adhesion, resist contractile forces
- Fascia adherens — anchors actin filaments; provides strong intercellular adhesion
Lateral/longitudinal region (parallel to fiber axis):
- Gap junctions — ionic continuity between cells; act as "electrical synapses"; permit rapid impulse conduction so many adjacent cells contract as a functional unit (functional syncytium)
Excitation–Contraction Coupling in Cardiac Muscle
- Depolarization → voltage-gated Cav1.2 channels open → Ca²⁺ enters from ECF
- Entered Ca²⁺ triggers CICR via RYR2 on SR → large amplified Ca²⁺ release
- Ca²⁺ binds troponin C (same as skeletal muscle) → tropomyosin moves → cross-bridge cycling
- T-tubules are located at Z lines and form dyads (one T-tubule + one SR terminal cisterna), vs. triads in skeletal muscle
Special Properties
- Autorhythmicity: Contractions are intrinsic and spontaneous; individual cardiomyocytes in culture continue to beat. Pacemaker nodes regulate and coordinate rhythm. Autonomic innervation modulates rate (sympathetic ↑, parasympathetic ↓) but does not initiate contraction.
- All-or-none contraction: Individual cardiomyocytes contract all-or-none; the gap junctions ensure coordinated contraction of the entire heart wall.
- Helical fiber arrangement: Cells branch and interweave in spiraling layers, producing an efficient wringing contraction that empties the ventricles.
Ventricular vs. Atrial Differences
| Feature | Ventricular | Atrial |
|---|---|---|
| Wall thickness | Much thicker | Thinner |
| T-tubules | Well-developed, large lumen | Absent or very small |
| Secretory granules | Rare | Present (ANF granules) |
Atrial natriuretic factor (ANF): Peptide hormone stored in secretory granules (~600/cell in right atrium); released in response to atrial stretch → acts on kidney to promote natriuresis and diuresis, opposing aldosterone and ADH.
Regenerative Capacity
Adult mammalian cardiac muscle lacks satellite cells and has minimal regenerative capacity after injury. This is why myocardial infarction causes permanent scar (fibrosis) rather than muscle regeneration.
Side-by-Side Comparison
| Feature | Smooth Muscle | Cardiac Muscle |
|---|---|---|
| Striations | No | Yes |
| Nuclei | Single, central | Single, central |
| Sarcomeres | Absent | Present |
| Intercalated discs | Absent | Present |
| Gap junctions | Unitary type only | All cardiac cells |
| T-tubules | Caveolae (rudimentary) | Present (at Z discs; dyads) |
| Ca²⁺ sensor | Calmodulin → MLCK | Troponin C |
| Contraction trigger | ANS, hormones, stretch, pacemakers | Intrinsic pacemaker nodes + ANS modulation |
| Regeneration | Some plasticity | Minimal |
| Mitochondria | Moderate | Very abundant (up to 40% cell volume) |
| Endocrine function | No | Atria secrete ANF |
Sources: Costanzo Physiology 7th Ed., pp. 48–50; Junqueira's Basic Histology 17th Ed., pp. 521–527; Medical Physiology (Boron & Boulpaep), pp. 2014, 2034–2036
For viva question
Here are high-yield viva questions on Smooth Muscle & Cardiac Muscle — with model answers:
Viva Questions: Smooth Muscle & Cardiac Muscle
SMOOTH MUSCLE
Q1. Why does smooth muscle lack striations?
Smooth muscle has thick (myosin) and thin (actin) filaments, but they are not arranged into sarcomeres. Without the regular sarcomeric banding pattern, no cross-striations are visible under light microscopy.
Q2. What are the two types of smooth muscle? Give examples of each.
| Type | Examples |
|---|---|
| Unitary (single-unit) | GI tract, bladder, uterus, ureter |
| Multiunit | Iris, ciliary muscle of lens, vas deferens |
Unitary smooth muscle cells are linked by gap junctions → electrically coupled → coordinated contraction. Multiunit cells are independent, each densely innervated by the ANS.
Q3. What is the key difference in the mechanism of contraction between smooth muscle and skeletal muscle?
- Skeletal muscle: Ca²⁺ binds troponin C → removes tropomyosin inhibition → cross-bridge cycling
- Smooth muscle: No troponin. Ca²⁺ binds calmodulin → Ca²⁺-calmodulin activates myosin-light-chain kinase (MLCK) → MLCK phosphorylates myosin light chain → increases myosin ATPase → cross-bridge cycling
Q4. What are the three sources of Ca²⁺ for smooth muscle contraction?
- Voltage-gated Ca²⁺ channels (sarcolemma) — opened by depolarization
- Ligand-gated Ca²⁺ channels (sarcolemma) — opened by hormones/neurotransmitters via G proteins
- IP₃-gated SR channels — hormone → G protein → PLC → PIP₂ → IP₃ → Ca²⁺ release from SR
Key point: Unlike skeletal muscle (SR only), smooth muscle can raise [Ca²⁺] from both ECF and SR.
Q5. How does smooth muscle relax?
By falling [Ca²⁺] through:
- Hyperpolarization → closes voltage-gated Ca²⁺ channels
- cAMP/cGMP → inhibit Ca²⁺ channels or activate SR Ca²⁺-ATPase
- Reduced IP₃ production → less SR Ca²⁺ release
- Myosin-light-chain phosphatase → dephosphorylates myosin light chain → stops cross-bridge cycling
Q6. What are caveolae? What is their significance in smooth muscle?
Caveolae are shallow sarcolemmal invaginations (cholesterol/sphingolipid-rich lipid raft microdomains) that cluster near peripheral SR. They are the functional equivalent of T-tubules in smooth muscle — they facilitate coupling between membrane depolarization and SR Ca²⁺ release.
Q7. What is the role of calponin and caldesmon in smooth muscle?
At low [Ca²⁺]: calponin and caldesmon bind actin → inhibit myosin ATPase → prevent cross-bridge formation.
At high [Ca²⁺]: Ca²⁺-calmodulin → phosphorylation of calponin and caldesmon → inhibition is relieved → cross-bridges form.
Q8. What is pacemaker (slow wave) activity in smooth muscle?
Unitary smooth muscle shows spontaneous, rhythmic membrane depolarizations called slow waves, generated intrinsically by pacemaker cells (e.g., interstitial cells of Cajal in the GI tract). The frequency of slow waves determines the frequency of action potentials and therefore the rhythm of contractions.
CARDIAC MUSCLE
Q9. What is an intercalated disc? What structures does it contain and what is the function of each?
Intercalated discs are step-like junctions at the ends of cardiomyocytes where adjacent cells meet.
| Structure | Region | Function |
|---|---|---|
| Desmosomes | Transverse | Mechanical adhesion — withstands contractile forces |
| Fascia adherens | Transverse | Anchors actin filaments; strong intercellular adhesion |
| Gap junctions | Lateral/longitudinal | Ionic/electrical continuity — allows rapid impulse spread, functional syncytium |
Q10. Why is cardiac muscle called a functional syncytium?
Cardiac muscle cells are anatomically separate, but gap junctions in the intercalated discs allow free passage of ions between cells. An action potential in one cell propagates rapidly to adjacent cells, causing them to contract as a unit — behaving like a syncytium functionally, without being one structurally.
Q11. How does E-C coupling in cardiac muscle differ from skeletal muscle?
| Feature | Skeletal | Cardiac |
|---|---|---|
| Mechanism | Direct mechanical coupling (DHP receptor → RYR1) | CICR (Cav1.2 → Ca²⁺ entry → RYR2 → SR Ca²⁺ release) |
| Ca²⁺ source | SR only | ECF + SR |
| T-tubule location | A-I band junction | Z disc |
| SR junctions | Triads (2 terminal cisternae + 1 T-tubule) | Dyads (1 terminal cisterna + 1 T-tubule) |
| Ca²⁺ sensor | Troponin C | Troponin C |
Q12. Where are T-tubules located in cardiac muscle? What junction do they form?
T-tubules in ventricular cardiac muscle are located at the Z discs (not the A-I junction as in skeletal muscle). They associate with one terminal cisterna of SR, forming dyads (vs. triads in skeletal muscle).
Atrial cardiomyocytes have absent or very small T-tubules.
Q13. Why does cardiac muscle have so many mitochondria?
Mitochondria occupy up to 40% of cardiomyocyte volume — more than any other muscle type. The heart works continuously and relies almost exclusively on aerobic oxidative phosphorylation (primarily of fatty acids) for ATP. This high mitochondrial density reflects the enormous, uninterrupted energy demand.
Q14. What is atrial natriuretic factor (ANF)? Which cardiac cells secrete it?
ANF is a peptide hormone stored in secretory granules near the nuclei of atrial cardiomyocytes (~600 granules/cell in the right atrium). Released in response to atrial stretch (high blood volume) → acts on kidney → promotes Na⁺ and water excretion (natriuresis and diuresis) → reduces blood volume and pressure. It opposes aldosterone and ADH. This makes atrial muscle cells both contractile and endocrine in function.
Q15. Why does cardiac muscle have poor regenerative capacity?
Adult mammalian cardiomyocytes lack satellite cells (muscle stem cells). After injury (e.g., myocardial infarction), lost cardiomyocytes are replaced by fibrous scar tissue (fibrosis) rather than new muscle — this is the basis of post-MI remodeling and heart failure.
Q16. What is the difference between autorhythmicity and autonomic control of the heart?
- Autorhythmicity: The heart's rhythm is intrinsic — pacemaker nodes (SA node, AV node) generate spontaneous action potentials independently. Cardiac muscle cells in culture continue to beat.
- Autonomic control modulates but does not initiate the rhythm:
- Sympathetic → ↑ heart rate and contractility
- Parasympathetic → ↓ heart rate
HIGH-YIELD COMPARISON QUESTION
Q17. Compare smooth, cardiac, and skeletal muscle across key features.
| Feature | Skeletal | Cardiac | Smooth |
|---|---|---|---|
| Striations | Yes | Yes | No |
| Nuclei | Multiple, peripheral | Single, central | Single, central |
| Voluntary control | Yes | No | No |
| T-tubules | A-I junction (triads) | Z disc (dyads) | Caveolae |
| Ca²⁺ sensor | Troponin C | Troponin C | Calmodulin |
| Intercalated discs | No | Yes | No |
| Regeneration | Yes (satellite cells) | Minimal | Yes |
| Pacemaker activity | No | Yes | Unitary type: Yes |
| Endocrine function | No | Yes (ANF) | No |
This is a shared conversation. Sign in to Orris to start your own chat.