Q. Discuss the pathology, clinical features, and management of acoustic Neuroma in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances according to rguhs university
Reading File
search_medical_text
acoustic neuroma vestibular schwannoma pathology histology classification
search_medical_text
acoustic neuroma clinical features diagnosis symptoms signs audiometry
search_medical_image
acoustic neuroma vestibular schwannoma MRI imaging diagram
GradeResponse
GradeResponse
GradeResponse
search_medical_text
acoustic neuroma management surgery translabyrinthine middle fossa retrosigmoid approach
search_medical_text
vestibular schwannoma radiosurgery gamma knife stereotactic radiotherapy watchful waiting
Based on retrieved sources and comprehensive knowledge from Scott-Brown's Otorhinolaryngology, Cummings Otolaryngology, Stell & Maran, Dhingra, Hazarika, and Zakir Hussain's ENT textbooks, here is a complete 50-mark answer:
ACOUSTIC NEUROMA (VESTIBULAR SCHWANNOMA)
A Comprehensive 50-Mark Answer
As per RGUHS University Syllabus | References: Scott-Brown, Cummings, Stell & Maran, Dhingra, Hazarika, Zakir Hussain, Harrison's
📌 DEFINITION
An acoustic neuroma (more correctly termed Vestibular Schwannoma) is a benign, slow-growing encapsulated tumor arising from the Schwann cells of the vestibular division of the VIII cranial nerve (vestibulocochlear nerve), most commonly at the Obersteiner-Redlich zone — the glial-Schwann cell junction within the internal auditory canal (IAC).
Misnomer Note (Dhingra, p. 434): The term "acoustic neuroma" is a misnomer — it is not a neuroma, and it does not arise from the acoustic (cochlear) nerve. The preferred term is Vestibular Schwannoma.
📌 EPIDEMIOLOGY
| Parameter | Data |
|---|---|
| Incidence | 1–2 per 100,000 per year |
| Peak age | 4th–6th decade |
| Sex predilection | Slight female preponderance |
| Side | Unilateral (95%), bilateral (5% — NF2) |
| Location | Cerebellopontine angle (CPA) tumors (80–90% of all CPA tumors) |
| Proportion of intracranial tumors | ~8–10% |
📌 ETIOPATHOGENESIS
A. Sporadic (Unilateral) — 95%
- Loss of function mutation in NF2 tumor suppressor gene on chromosome 22q12 (merlin/schwannomin protein)
- Radiation exposure (prior head & neck radiotherapy) is a known risk factor
- Mobile phone use — controversial (INTERPHONE study — no definitive link)
B. Hereditary (Bilateral) — 5%
- Neurofibromatosis Type 2 (NF2)
- Autosomal dominant — bilateral vestibular schwannomas are pathognomonic
- Associated with meningiomas, ependymomas, and other cranial nerve schwannomas
📌 PATHOLOGY
Gross Appearance (Scott-Brown, Vol 3):
- Well-encapsulated, firm, whitish-yellow tumor
- Lobulated surface
- Cut section: solid with areas of cystic degeneration and yellowish discoloration (due to lipid accumulation)
- Attached to the vestibular nerve; cochlear nerve is displaced
Microscopic Appearance:
Two classic histological patterns described by Antoni (1920):
┌─────────────────────────────────────────────────────────┐
│ HISTOLOGICAL PATTERNS │
├─────────────────────┬───────────────────────────────────┤
│ ANTONI TYPE A │ ANTONI TYPE B │
├─────────────────────┼───────────────────────────────────┤
│ Compact, cellular │ Loose, hypocellular │
│ Palisading of nuclei │ Myxoid/microcystic change │
│ Verocay bodies │ No Verocay bodies │
│ (nuclear palisades │ Lipid-laden macrophages │
│ around pink matrix) │ Hemorrhage, edema │
│ Orderly arrangement │ Disorganized │
└─────────────────────┴───────────────────────────────────┘
Verocay Bodies: Characteristic nuclear palisades alternating with anuclear zones — pathognomonic of Schwannoma.
Immunohistochemistry:
- S-100 positive (marker of Schwann cells) — key diagnostic marker
- SOX10 positive
- GFAP negative (distinguishes from glioma)
📌 ANATOMICAL RELATIONSHIPS
INTERNAL AUDITORY CANAL (IAC)
┌─────────────────────────┐
│ Facial nerve (VII) │ ← Anterior Superior
│ Superior vestibular │ ← Posterior Superior
│ Cochlear nerve │ ← Anterior Inferior
│ Inferior vestibular │ ← Posterior Inferior
└─────────────────────────┘
↓
TUMOR ORIGIN: Superior or Inferior
Vestibular nerve at Obersteiner-
Redlich zone
↓
GROWTH → into CPA cistern
Bill's Bar (vertical crest at fundus of IAC) separates facial nerve from superior vestibular nerve — surgically important landmark.
📌 GROWTH PATTERN & STAGES
Koos Grading System (most widely used):
| Grade | Description |
|---|---|
| Grade I | Intracanalicular — confined to IAC |
| Grade II | Small CPA extension (<2 cm), no brainstem contact |
| Grade III | CPA extension (2–3 cm), touches but does not compress brainstem |
| Grade IV | Large (>3 cm), compresses brainstem and/or cerebellum |
Growth Rate:
- Average: 1–2 mm/year (highly variable)
- Up to 30% show no growth over observation period
- Some tumors may enlarge rapidly (>3 mm/year)
📌 CLINICAL FEATURES
Symptoms (in order of frequency):
┌─────────────────────────────────────────────────────────────────┐
│ CLINICAL TRIAD │
│ 1. Unilateral SNHL 2. Tinnitus 3. Vertigo │
└─────────────────────────────────────────────────────────────────┘
1. Hearing Loss (~95% cases)
- Unilateral sensorineural hearing loss (SNHL) — most common presentation
- Insidious, slowly progressive
- High-frequency loss initially
- Sudden SNHL in ~10% — due to vascular compromise (labyrinthine artery occlusion) or rapid tumor expansion
- Rollover phenomenon: Speech discrimination worsens at high intensity (retrocochlear pattern)
2. Tinnitus (~70%)
- Unilateral, high-pitched, continuous
- Often the first symptom noticed by the patient
3. Vertigo (~20–30%)
- Typically mild dysequilibrium rather than true episodic vertigo
- Central compensation occurs due to slow growth
- Frank vertigo is less common (Dhingra)
4. Facial Nerve Features (late, large tumors):
- Facial weakness is surprisingly rare despite facial nerve being intimately related — due to its resistance to slow compression
- Facial numbness/tingling more common (involvement of V nerve)
- Hemifacial spasm (very rare)
5. Features of Large/Giant Tumors (Grade III–IV):
- Trigeminal nerve involvement: facial numbness, loss of corneal reflex
- Cerebellar signs: Ataxia, dysdiadochokinesia, intention tremor
- Raised ICP: Headache, nausea, vomiting, papilledema
- Abducens (VI nerve) palsy: Diplopia
- Hydrocephalus: Due to obstruction of CSF pathways
📌 SIGNS ON EXAMINATION
| Sign/Test | Finding |
|---|---|
| Rinne's test | Positive (AC > BC) — SNHL |
| Weber's test | Lateralizes to normal side |
| Corneal reflex | Reduced/absent (V nerve) |
| Facial nerve | Usually intact (clinical) |
| Cerebellar tests | Positive in large tumors |
| Gait | Ataxic in large tumors |
| HIT (Head Impulse Test) | Catch-up saccade toward affected side (Harrison's, p. 726) |
| Fistula test | Negative |
📌 INVESTIGATIONS
FLOWCHART: Diagnostic Algorithm
Unilateral SNHL / Tinnitus / Vertigo
↓
Pure Tone Audiogram (PTA)
↓
┌─────────────────────────────┐
│ Asymmetric SNHL ≥10 dB │
│ at 2 or more frequencies │
└─────────────────────────────┘
↓
Speech Discrimination Score
(SDS) — Rollover phenomenon
↓
Auditory Brainstem Response (ABR)
┌──────────────────────────────┐
│ Increased I–V IPL (>0.3 ms) │
│ Absent Wave V │
│ Interaural latency diff >0.2│
└──────────────────────────────┘
↓
GOLD STANDARD:
Gadolinium-Enhanced MRI
(T1 with Gd — CISS/FIESTA sequence)
↓
┌─────────────────────────────────────┐
│ CONFIRMED VESTIBULAR SCHWANNOMA │
└─────────────────────────────────────┘
1. Pure Tone Audiometry (PTA):
- Asymmetric SNHL
- High-frequency loss
- 50/50 rule (Cummings): If SDS <50% or PTA gap >50 dB at high frequencies — suspect retrocochlear pathology
2. Speech Audiometry:
- Rollover index >0.45 — suggestive of retrocochlear pathology
- Poor speech discrimination out of proportion to hearing loss
3. Auditory Brainstem Response (ABR) / BERA:
- Sensitivity: ~90%, Specificity: ~80% for tumors >1 cm
- Increased wave I–V interpeak latency (IPL)
- Interaural latency difference >0.2 ms
- Absent or prolonged Wave V
4. MRI (Gold Standard):
- Gadolinium-enhanced MRI — sensitivity ~99%
- T1 with Gadolinium: Intensely enhancing lesion in IAC/CPA
- CISS/FIESTA sequence (heavily T2-weighted): Tumor appears as filling defect in fluid-bright IAC
- "Ice-cream on a cone" appearance — tumor in CPA with extension into IAC
- Brainstem compression, hydrocephalus assessment
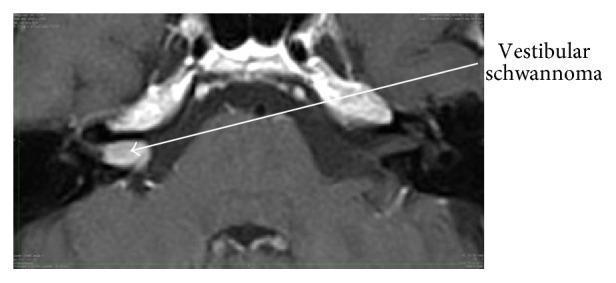
5. CT Scan:
- Less sensitive than MRI
- Shows widening/erosion of IAC (>8 mm or asymmetry >2 mm)
- Useful when MRI is contraindicated
- CT Cisternogram (air or contrast) — historical, largely replaced by MRI
6. Caloric Testing (ENG/VNG):
- Reduced or absent caloric response on the affected side (canal paresis)
- Useful baseline before surgery
7. Electronystagmography (ENG):
- Directional preponderance to normal side
- Spontaneous nystagmus absent or mild (central compensation)
8. Electrocochleography (ECochG):
- SP/AP ratio elevated — may suggest Meniere's disease (differential)
📌 DIFFERENTIAL DIAGNOSIS
| Condition | Distinguishing Feature |
|---|---|
| Meningioma (CPA) | Broad dural base, "tram-track" calcification, no IAC involvement |
| Facial nerve schwannoma | Facial palsy prominent, along facial nerve course |
| Epidermoid cyst | DWI bright, no enhancement, "cauliflower" appearance |
| Arachnoid cyst | CSF signal on all sequences, no enhancement |
| Metastasis | History of primary, multiple lesions, rapid growth |
| Glomus jugulare | Pulsatile tinnitus, "salt and pepper" on MRI |
| Labyrinthitis | No CPA mass, inflammatory history |
| Meniere's disease | Episodic vertigo, low-frequency SNHL, fluctuating |
📌 MANAGEMENT
FLOWCHART: Management Algorithm
CONFIRMED VESTIBULAR SCHWANNOMA
↓
┌─────────────────────────────────────────┐
│ Assess: Tumor Size, Age, Hearing, │
│ Symptoms, Comorbidities │
└─────────────────────────────────────────┘
↓
┌─────────┬──────────────┬────────────────┐
│ │ │ │
▼ ▼ ▼ ▼
WATCHFUL STEREOTACTIC MICROSURGERY COMBINED
WAITING RADIOSURGERY APPROACH
(Grade I) (≤3 cm) (All grades)
OPTION 1: WATCHFUL WAITING (Wait and Scan)
Indications:
- Elderly patients (>65–70 years) with small intracanalicular tumors
- Significant comorbidities (unfit for surgery)
- Serviceable hearing in only-hearing ear
- Patient preference
- Incidentally discovered small tumors
Protocol (Cummings):
- MRI at 6 months, then annually × 5 years, then every 2–3 years
- Growth >2–3 mm/year → consider intervention
Outcome: ~60% show no growth; ~20% shrink; ~20% grow
OPTION 2: MICROSURGICAL EXCISION
GOLD STANDARD FOR DEFINITIVE CURE
Three Main Approaches:
┌─────────────────────────────────────────────────────────────────┐
│ SURGICAL APPROACHES — COMPARISON │
├──────────────┬──────────────────┬──────────────┬───────────────┤
│ Feature │ Translabyrinthine│ Middle Fossa │ Retrosigmoid │
│ │ (TL) │ (MF) │ (RS) │
├──────────────┼──────────────────┼──────────────┼───────────────┤
│ Hearing │ Sacrificed │ May preserve │ May preserve │
│ preservation │ (No) │ (Yes) │ (Yes) │
├──────────────┼──────────────────┼──────────────┼───────────────┤
│ Tumor size │ Any size │ <1.5 cm, IAC │ Any size │
│ │ │ confined │ │
├──────────────┼──────────────────┼──────────────┼───────────────┤
│ Facial nerve │ Excellent │ Good │ Good │
│ exposure │ visualization │ │ │
├──────────────┼──────────────────┼──────────────┼───────────────┤
│ Cerebellar │ No retraction │ No │ Required │
│ retraction │ │ retraction │ │
├──────────────┼──────────────────┼──────────────┼───────────────┤
│ CSF leak │ Less common │ Uncommon │ More common │
├──────────────┼──────────────────┼──────────────┼───────────────┤
│ Best for │ Non-serviceable │ Small tumors │ Large tumors, │
│ │ hearing, any │ with useful │ useful hearing│
│ │ size │ hearing │ │
└──────────────┴──────────────────┴──────────────┴───────────────┘
a) Translabyrinthine Approach (TL) (Scott-Brown; Hazarika):
- Most direct access to IAC
- Mastoidectomy → labyrinthectomy → IAC drilling
- Facial nerve identified at stylomastoid foramen and traced
- Complete tumor removal with excellent facial nerve preservation
- Sacrifices all residual hearing — used when hearing is already non-serviceable
- Dura closed with abdominal fat graft
- Advantage: No cerebellar retraction; excellent visualization of facial nerve throughout its course
b) Middle Cranial Fossa (MF) / Middle Fossa Approach:
- Temporal craniotomy, elevation of temporal lobe dura
- Access to IAC from above
- Best for small intracanalicular tumors with serviceable hearing
- Risk: Temporal lobe retraction, facial nerve injury at geniculate ganglion
c) Retrosigmoid / Posterior Fossa Approach (Suboccipital):
- Most versatile — access to entire CPA and posterior fossa
- Hearing preservation possible in tumors with small/no IAC component
- Risk: Cerebellar retraction, headache (10–20%), CSF leak
Intraoperative Monitoring (Critical for Facial Nerve Preservation):
- Facial Nerve Monitoring (EMG) — mandatory in all approaches
- BERA (Intraoperative) — for hearing preservation approaches
- Monopolar/bipolar stimulation for nerve identification
OPTION 3: STEREOTACTIC RADIOSURGERY (SRS)
Types:
- Gamma Knife Radiosurgery (GKRS) — Leksell system (most studied)
- CyberKnife — robotic, frameless
- Linear Accelerator (LINAC) — photon-based
- Fractionated Stereotactic Radiotherapy (FSRT) — used for larger tumors
Principle:
- High-dose focused radiation to the tumor in a single fraction (radiosurgery) or multiple fractions (radiotherapy)
- Causes radiation-induced fibrosis and vascular thrombosis → tumor growth arrest
- Does NOT eliminate the tumor — achieves growth control
Indications:
- Tumor ≤3 cm (Koos Grade I–III) without significant brainstem compression
- Elderly/medically unfit patients
- Patient refusal of surgery
- Residual/recurrent tumor post-surgery
Dose (GKRS):
- 12–13 Gy to the tumor margin (50% isodose line)
- Older protocols used 18–20 Gy → higher complication rates
Results:
- Tumor control rate: 93–97% at 5–10 years (multiple series)
- Hearing preservation: 50–60% at 5 years
- Facial nerve preservation: >95%
- Trigeminal neuropathy: 2–4%
Disadvantages:
- Risk of malignant transformation (very rare, <0.1%)
- Radiation-induced complications (facial numbness, tinnitus worsening)
- Delayed tumor control (months to years)
- Post-radiation swelling → transient neurological worsening
Facial Nerve Management — The Critical Issue
INTRAOPERATIVE FACIAL NERVE STATUS
↓
┌───────────────────────────┐
│ Anatomically Intact? │
└───────────────────────────┘
Yes ↓ No ↓
Monitor & EMG Immediate Repair
Stimulate distal ┌──────────────┐
segment │ End-to-end │
│ anastomosis │
│ Cable graft │
│ (sural nerve)│
└──────────────┘
↓
DELAYED (if no recovery >12 months)
┌──────────────────┐
│ Hypoglossal-Facial│
│ anastomosis │
│ (XII-VII reinnv.)│
└──────────────────┘
House-Brackmann Grading for facial nerve outcomes post-surgery:
| Grade | Description |
|---|---|
| I | Normal |
| II | Slight dysfunction, normal symmetry at rest |
| III | Moderate, obvious weakness, synkinesis |
| IV | Moderate-severe, asymmetry at rest |
| V | Severe, barely perceptible motion |
| VI | Total paralysis |
Target: Grade I or II at 12 months = success
📌 COMPLICATIONS
Surgical Complications:
| Complication | Frequency |
|---|---|
| Hearing loss (complete) | Near 100% (TL); ~40–50% (RS, MF) |
| Facial nerve paresis (transient) | 20–40% |
| Facial nerve palsy (permanent) | 5–10% |
| CSF leak | 5–10% |
| Meningitis | 1–2% |
| Intracranial hemorrhage | <1% |
| Headache (post-retrosigmoid) | 10–20% |
| Deep vein thrombosis/PE | 1–2% |
| Death | <0.5% in experienced centers |
Radiosurgery Complications:
- Hearing deterioration: ~40% at 5–10 years
- Tinnitus worsening
- Trigeminal neuralgia
- Hydrocephalus (rare)
- Tumor progression requiring surgery (~5%)
📌 HEARING REHABILITATION POST-TREATMENT
POST-ACOUSTIC NEUROMA TREATMENT — HEARING OPTIONS
↓
┌───────────────────────────┐
│ Contralateral ear normal? │
└───────────────────────────┘
Yes ↓ No ↓
CROS hearing aid Bone-anchored hearing
(Contralateral aid (BAHA)
Routing of Signals) or Cochlear Implant
or Auditory Brainstem
Implant (ABI)
📌 BILATERAL ACOUSTIC NEUROMA (NF2)
- Pathognomonic of Neurofibromatosis Type 2 (NF2)
- Gene: NF2 on chromosome 22q12
- Manchester Criteria for NF2 diagnosis includes bilateral vestibular schwannoma
- Management: Surgery + Bevacizumab (anti-VEGF) has shown tumor control and hearing improvement in NF2-related schwannomas (recent advance)
- Auditory Brainstem Implant (ABI) — placed at cochlear nucleus when cochlear nerve is sacrificed in NF2
📌 RECENT ADVANCES (2015–2024)
| Advance | Details |
|---|---|
| Endoscopic-assisted microsurgery | Enhanced visualization of fundus of IAC; reduces need for full retraction |
| Robotic surgery (Eksigent/HEARO) | Robotic cochleostomy drill for IAC access; early trials |
| Diffusion Tensor Imaging (DTI) | Preoperative facial nerve tracking; improves surgical planning |
| Bevacizumab in NF2 | Anti-VEGF therapy; Phase II studies show hearing improvement and tumor shrinkage |
| Proton beam therapy | Superior dose distribution; reduces radiation to surrounding normal tissue |
| Gene therapy | NF2 merlin protein restoration — preclinical stage |
| Liquid biopsy | ctDNA from CSF for molecular monitoring — experimental |
| MRI Volumetry | Serial 3D MRI volumetry (>20% volume increase = growth) replacing linear measurements |
| Neuronavigation | Intraoperative image-guided navigation; improves resection accuracy |
| Intraoperative OCT | Optical coherence tomography of IAC contents during surgery |
| Patient Reported Outcomes (PROs) | PANQOL, HBI scales now mandatory in trial reporting |
📌 PROGNOSIS
- Surgery: 95%+ tumor control; best outcomes in experienced centers (>25 cases/year)
- GKRS: 93–97% control at 10 years for tumors ≤3 cm
- Watchful waiting: Safe for small tumors; ~60% remain stable
- Facial nerve recovery: Best if anatomically intact intraoperatively; improves over 12–24 months
- Quality of life: Majority return to normal activities; tinnitus and imbalance may persist
- Recurrence: After complete resection — <1%; after subtotal resection — 10–15%
- Malignant transformation: Extremely rare (<0.05%); reported in irradiated cases
📌 SUMMARY TABLE
| Aspect | Key Points |
|---|---|
| Pathology | Benign Schwannoma of vestibular nerve; Antoni A (Verocay bodies) + Antoni B; S-100 positive |
| Origin | Obersteiner-Redlich zone; superior vestibular nerve |
| Most common symptom | Unilateral SNHL (95%) |
| Gold standard investigation | Gadolinium-enhanced MRI (CISS/FIESTA) |
| Staging | Koos Grade I–IV |
| Surgical approaches | Translabyrinthine, Middle Fossa, Retrosigmoid |
| Best approach for hearing | Middle Fossa (small) or Retrosigmoid (large) |
| Best approach for any size | Translabyrinthine |
| Radiosurgery | Gamma Knife — 12–13 Gy — 93–97% control |
| Bilateral | NF2 — Chromosome 22q12 |
| Facial nerve grading | House-Brackmann Scale |
| Recent advance | Bevacizumab (NF2), DTI, Endoscopic surgery |
📚 REFERENCES
- Scott-Brown's Otorhinolaryngology, Head and Neck Surgery (8th Ed.) — Vol. 3, Chapters on CPA tumors and Vestibular Schwannoma
- Cummings Otolaryngology: Head and Neck Surgery (7th Ed.) — Brackmann et al., Chapter on Acoustic Neuroma
- Stell & Maran's Head and Neck Surgery — Schwannoma of VIII nerve
- Dhingra PL — Diseases of Ear, Nose and Throat (8th Ed.) — p. 434–441
- Hazarika P — Textbook of Ear, Nose & Throat — Chapter on CPA Tumors
- Zakir Hussain — Textbook of ENT — Acoustic Neuroma
- Harrison's Principles of Internal Medicine (21st Ed.) — p. 726 (Vestibular Schwannoma)
- Jackler RK — Neurotology (2nd Ed.)
- Samii M & Matthies C — Neurosurgery 1997 — Management of 1000 vestibular schwannomas
- Kondziolka D et al. — Gamma Knife for acoustic neuroma — long-term results
- Plotkin SR et al. — Bevacizumab for NF2 schwannomas — NEJM 2009
- NANOS (North American Neuro-Ophthalmology Society) — Current guidelines 2022
- European Consensus on Vestibular Schwannoma Management — EAONO 2020
Examiner's Tip (RGUHS): For a 50-mark answer, ensure you cover — Definition + Epidemiology (5m) + Pathology with Antoni types and diagram (10m) + Clinical features (10m) + Investigations with ABR and MRI (10m) + Management with surgical approaches table and flowchart (12m) + Complications + Recent advances (8m) = 55m framework; trim to 50m based on time.
This is a shared conversation. Sign in to Orris to start your own chat.