Give the detailed long note on Nevus cutaneous lipomatosus superficialis for ms general surgery professor and cross questions and answers for the poster presentation on the same
Nevus cutaneous lipomatosus superficialis clinical features histopathology diagnosis treatment
cutaneous lipomatosus superficialis differential diagnosis surgery management excision
nevus cutaneous lipomatosus superficialis skin lesion histopathology
Nevus Cutaneous Lipomatosus Superficialis (NCLS)
Detailed Clinical Note for MS General Surgery Poster Presentation
1. DEFINITION & NOMENCLATURE
- First described by Hoffmann and Zurhelle in 1921
- Classified under connective tissue hamartomas / benign mesenchymal neoplasms
- ICD-10: D22.9 / Q82.8 (benign melanocytic nevus / other specified congenital malformations of skin)
2. EPIDEMIOLOGY
| Parameter | Details |
|---|---|
| Prevalence | Rare; exact incidence unknown |
| Age of onset | Congenital or appears in 1st–2nd decade (classic form); any age (solitary form) |
| Sex predilection | Slight female predominance in some series |
| Race | No racial predilection |
| Hereditary pattern | Sporadic; rarely familial |
3. PATHOGENESIS & ETIOLOGY
- Hamartomatous differentiation — aberrant differentiation of pluripotent mesenchymal cells in the dermis into mature adipocytes
- Ectopic lipogenesis — perivascular connective tissue or fibroblast precursors undergo lipogenesis under unknown stimuli
- Nevoid malformation — considered a localized developmental error (nevoid origin) rather than a true neoplasm
- Embryological arrest theory — arrest of maturation of dermal mesenchymal cells during fetal development, retaining adipogenic potential
- Some authors postulate a somatic mutation in PTEN or PIK3CA pathway (analogous to other hamartomas), though not definitively proven
No proven association with systemic lipomatosis, chromosomal abnormalities, or malignant transformation.
4. CLASSIFICATION
Type I — Classic / Hoffmann-Zurhelle Type (Zosteriform / Multiple)
- Multiple soft, yellowish, skin-colored papules and nodules grouped in a cerebriform, zosteriform, or clustered configuration
- Present at birth or appears in childhood/early adulthood (before age 30)
- Typically located on the lower trunk (lumbo-gluteal region, buttocks, flanks, thighs)
- Lesions may coalesce forming plaques with a cobblestone or cerebriform surface
- Does not cross the midline (follows Blaschko's lines in some cases)
Type II — Solitary / Acquired Type
- Single, dome-shaped soft pedunculated nodule or papule
- Appears in adults (3rd–5th decade)
- Can occur anywhere — scalp, face, ear (tragus), trunk, extremities
- Smaller, more discrete than Type I
- The histopathology image above shows this type — a pedunculated nodule on the tragus
A third rare variant — associated with folliculosebaceous cystic hamartoma and comedone-like lesions has been described.
5. CLINICAL FEATURES
Symptoms
- Usually asymptomatic
- Occasionally: mild pruritus, tenderness on pressure, cosmetic concern
- No constitutional symptoms
Signs
| Feature | Classic Type I | Solitary Type II |
|---|---|---|
| Number | Multiple (5–100+) | Single |
| Size | 0.5–3 cm (individual) | 0.5–5 cm |
| Shape | Papules/nodules → plaques | Dome-shaped, pedunculated nodule |
| Color | Skin-colored to pale yellow | Skin-colored to yellowish |
| Surface | Smooth, cerebriform, or verrucous | Smooth |
| Consistency | Soft, doughy, non-tender | Soft, compressible |
| Location | Buttocks, flanks, lower trunk | Scalp, face, ear, any site |
| Onset | Congenital/childhood | Adult |
| Growth | Slow, stable after puberty | Slow progressive |
| Comedones | May be present (hair follicle obliteration) | Absent |
Associated Features (Type I)
- Telangiectasia within plaques
- Hyperpigmentation or hypopigmentation
- Comedone-like plugs at follicular ostia
- Focal alopecia over lesional skin
6. HISTOPATHOLOGY
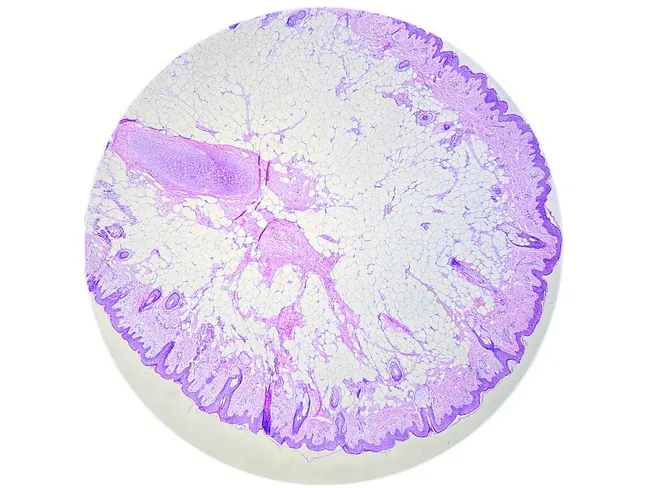
Microscopic Features:
| Layer | Finding |
|---|---|
| Epidermis | Normal or mildly acanthotic; sometimes papillomatous |
| Papillary dermis | Usually normal |
| Reticular dermis (upper & mid) | Mature adipocytes in lobules — pathognomonic finding |
| Adipocytes | Uniform, clear cytoplasm, small compressed nuclei — NO atypia, NO mitoses |
| Stroma | Fibrous bands separating lobules |
| Vasculature | Dilated capillaries (sometimes); no angiomatosis |
| Adnexae | May be displaced or diminished |
| Deep dermis/subcutis | No infiltration (distinguishes from lipoma) |
| Inflammatory infiltrate | Absent or sparse |
Key Histological Diagnostic Criterion:
Mature adipocytes occupying >10% of the dermis, present in the upper/mid reticular dermis without connection to subcutaneous fat — Diagnostic hallmark
7. DERMOSCOPY
- Yellowish-white structureless areas corresponding to dermal fat deposits
- Dilated follicular ostia
- Fine vessels in a reticular pattern
- No pigment network (differentiates from melanocytic lesions)
- Helpful non-invasive pre-operative tool
8. IMAGING
| Modality | Findings |
|---|---|
| Ultrasound | Hyperechoic dermal mass; sharply circumscribed; no subcutaneous extension |
| MRI | T1 hyperintense (fat signal); T2 hyperintense; no enhancement; restricted to dermis |
| CT | Hypodense lesion within dermis |
Imaging is rarely needed but useful for large lesions or when liposarcoma is a concern.
9. DIAGNOSIS
Clinical Diagnosis:
- Based on characteristic morphology and location
- Type I (zosteriform multiple lesions over buttocks in a young patient) is often clinically diagnosed
Definitive Diagnosis:
- Excisional biopsy + Histopathology — required for confirmation, especially solitary lesions
Diagnostic Criteria (Proposed):
- Soft, skin-colored/yellowish papules/nodules in dermis
- No connection to subcutaneous fat clinically or histologically
- Mature adipocytes in the dermis on histology
- No atypia or malignant features
10. DIFFERENTIAL DIAGNOSIS
| Condition | Differentiating Features |
|---|---|
| Intradermal lipoma | Deeper, not in dermis proper; larger; subcutaneous |
| Angiofibromas (tuberous sclerosis) | Facial, associated with TSC features, different histology |
| Connective tissue nevus | No adipocytes; collagen/elastic tissue proliferation |
| Folliculo-sebaceous cystic hamartoma | Overlapping; differentiated by follicular cysts on histology |
| Epidermal nevus | Epidermal proliferation predominates |
| Pedunculated lipofibroma | Fibrous component > adipose; firm |
| Fibrolipoma | More fibrous stroma; subcutaneous origin |
| Well-differentiated liposarcoma | Nuclear atypia, lipoblasts, MDM2/CDK4 amplification |
| Neurofibroma | S100+, neural origin; no mature adipocytes |
| Focal dermal hypoplasia (Goltz syndrome) | X-linked, skin atrophy with herniation of fat, skeletal anomalies |
| Michelin tire baby syndrome | Diffuse skin folds, generalized smooth muscle hamartoma |
11. MANAGEMENT
Indications for Surgery:
- Cosmetic disfigurement / patient request
- Mechanical symptoms (friction, difficulty wearing clothing)
- Diagnostic uncertainty (to rule out malignancy)
- Pedunculated lesion prone to trauma/torsion
- Rapid increase in size (though rare)
Surgical Treatment:
Wide Local Excision (Treatment of Choice)
- Excision with adequate margins down to and including upper subcutaneous fat
- Full-thickness skin excision ensures complete removal of all ectopic fat lobules
- Orientation of incision along Langer's lines for optimal cosmesis
- Closure: primary closure for small lesions; flap or skin graft for large plaques
Liposuction (Adjunctive)
- Used for large, extensive Type I lesions (zosteriform plaques)
- Reduces bulk and improves contour
- Risk of recurrence higher than excision alone
- May be combined with excision
CO₂ Laser Ablation
- Reported for small, superficial lesions
- Less scarring but incomplete depth of treatment → higher recurrence
- Not standard of care for MS General Surgery
Nd:YAG Laser
- Limited evidence; experimental
Recurrence:
- Low recurrence rate with complete excision
- Incomplete excision → local recurrence
- No malignant transformation reported
Prognosis:
- Excellent — purely benign lesion
- No systemic involvement
- No malignant potential
- Psychosocial impact addressed by surgical correction
12. COMPLICATIONS
| Complication | Details |
|---|---|
| Surgical wound infection | Standard post-op risk |
| Seroma/hematoma | Especially for large excisions |
| Scar hypertrophy/keloid | Depending on patient tendency |
| Recurrence | With incomplete excision |
| Cosmetic dissatisfaction | For large lesions requiring grafts |
13. SPECIAL CONSIDERATIONS FOR SURGEONS
- Always send specimen for HPE — mandatory to confirm benignity
- For gluteal/lower trunk lesions: position patient prone, use tourniquet if needed for hemostasis, plan incision for ease of closure
- For solitary lesions on face/ear: consider cosmetic implications, use loupe magnification, ensure clean margins
- Rule out liposarcoma in any atypical or rapidly growing lesion — send for MDM2/CDK4 FISH if histologically borderline
- No role for chemotherapy, radiotherapy, or immunotherapy — purely surgical condition
14. SUMMARY TABLE
| Feature | Details |
|---|---|
| Type | Benign hamartoma |
| Tissue | Ectopic mature dermis adipocytes |
| Classic site | Buttocks, lower trunk (Type I); anywhere (Type II) |
| Age | Childhood (Type I); adults (Type II) |
| Diagnosis | Histopathology |
| Treatment | Wide local excision |
| Recurrence | Low (with complete excision) |
| Malignant potential | None |
CROSS QUESTIONS & ANSWERS FOR POSTER PRESENTATION
(Expected from MS General Surgery Professor)
| Parameter | Type I (Classic) | Type II (Solitary) |
|---|---|---|
| Number | Multiple | Single |
| Age | Birth/childhood | Adults (3rd–5th decade) |
| Location | Buttocks, lower trunk | Any site (scalp, ear, face) |
| Configuration | Zosteriform/grouped | Isolated nodule |
| Prognosis | Same — benign | Same — benign |
- Perform a preoperative MRI to delineate extent and rule out deep infiltration
- Plan wide local excision with margins under GA, patient in prone position
- Ensure full-thickness skin excision to the upper subcutaneous fat
- For very large lesions, consider staged excision or liposuction for debulking followed by excision of residual skin
- Closure with rotation flap or split-thickness skin graft if primary closure is not possible
- Send entire specimen for histopathological examination
- Goltz syndrome is an X-linked condition with widespread skin atrophy, telangiectasias, skeletal abnormalities (syndactyly, osteopathia striata), ocular anomalies, and dental defects. The fat appears through atrophic skin as a herniation.
- NCLS is localized, sporadic, has no systemic associations, no skin atrophy, and the fat is truly within the dermis as a hamartoma, not herniating from subcutaneous tissue.
- H&E: Shows mature adipocytes — large cells with clear vacuolated cytoplasm and peripherally compressed nuclei
- Oil Red O / Sudan IV: Positive staining for lipid in fresh/frozen sections
- S100: Negative — differentiates from neural tumors
- MDM2/CDK4 immunohistochemistry or FISH: Negative — rules out well-differentiated liposarcoma
- CD34: May show stromal positivity (non-specific)
- Ensure full-thickness excision down to and including the superficial subcutaneous fat
- Confirm clear margins on frozen section if clinically uncertain
- Avoid superficial shave excision or incomplete removal
- For liposuction-based treatment of large lesions, counsel patient about higher recurrence rates (~20–30%) compared to excision
- Clinical diagnosis — History (since childhood, buttock, multiple, soft, yellow) is strongly suggestive of Type I NCLS
- Dermoscopy — Yellow-white structureless areas, no pigment network
- Excisional biopsy of representative lesion — Confirm with HPE
- Once confirmed benign: counseling — inform patient of benign nature, no malignant potential
- Surgical excision if cosmetically distressing or symptomatic
- No systemic workup needed — NCLS has no systemic associations
- Review slides with a dedicated dermatopathologist/surgical pathologist
- Order MDM2 and CDK4 immunohistochemistry — positive in ALT/WDLPS
- Send for MDM2 FISH — amplification of chromosome 12q13-15 confirms WDLPS
- If confirmed malignant: re-excision with wide clear margins, oncology referral
- If negative for MDM2/CDK4: likely benign atypical NCLS — close follow-up with re-excision if incomplete margins
- Identify the yellowish structureless areas representing dermal fat
- Rule out melanocytic pathology (absence of pigment network)
- Identify dilated follicular openings and reticular vessels
- Guide decision for biopsy (helps clinician avoid unnecessary biopsy of classic cases)
- Not a substitute for histopathology in solitary or atypical lesions
- NCLS = benign hamartoma — ectopic mature adipocytes in dermis
- Two types: Type I (multiple, congenital, buttocks) and Type II (solitary, adult, any site)
- Diagnosis confirmed by histopathology — mature adipocytes in dermis, no atypia
- Wide local excision is treatment of choice — curative, low recurrence
- No malignant potential — reassure patient
- Exclude well-differentiated liposarcoma in atypical cases via MDM2/CDK4
- Rare condition — thorough clinico-pathological correlation essential
QUICK REVISION MNEMONICS
-
Dermis — location of ectopic fat
-
Ectopic mature adipocytes
-
Rare hamartoma (Hoffmann-Zurhelle)
-
Multiple (Type I) or Mono/solitary (Type II)
-
Innocuous — no malignant potential
-
Surgery (wide excision) = cure
-
Fat in dermis = pathognomonic
-
Adult onset for Type II
-
Type I = congenital, zosteriform, buttocks
Note for Poster: The lesion is purely benign and surgical excision is curative. Always send HPE. The key message for a General Surgery audience is the distinction from liposarcoma and the confirmatory role of histopathology + MDM2/CDK4 testing when in doubt.
Layers of abdomen
layers of anterior abdominal wall anatomy surgical
layers of anterior abdominal wall cross section anatomy
anterior abdominal wall layers skin subcutaneous fat fascia muscles peritoneum rectus sheath
rectus sheath anatomy above below arcuate line cross section
Layers of the Abdomen
ANTERIOR ABDOMINAL WALL — Layers (Superficial to Deep)
Mnemonic: "Some Surgeons Can Find All Patients Extremely Pale, Inspite Prolonged Effort"
Skin → Superficial fascia → Camper's → Scarpa's → Fat → Aponeuroses/Muscles → Fascia Transversalis → Extraperitoneal fat → Peritoneum
Layer by Layer:
| # | Layer | Key Details |
|---|---|---|
| 1 | Skin | Thin, mobile; Langer's lines run transversely |
| 2 | Superficial Fascia (Camper's) | Fatty layer; continuous with superficial fat of thigh |
| 3 | Deep Fascia of Superficial Fascia (Scarpa's) | Membranous layer; fuses with fascia lata below inguinal ligament; forms dartos of scrotum |
| 4 | External Oblique Muscle & Aponeurosis | Fibers run downward and medially ("hands in pocket"); forms inguinal ligament, anterior rectus sheath, linea alba |
| 5 | Internal Oblique Muscle & Aponeurosis | Fibers run upward and medially; forms both anterior and posterior rectus sheath above arcuate line |
| 6 | Transversus Abdominis Muscle & Aponeurosis | Horizontal fibers; forms posterior rectus sheath above arcuate line |
| 7 | Transversalis Fascia | Deep investing fascia; forms internal spermatic fascia at deep inguinal ring |
| 8 | Extraperitoneal (Preperitoneal) Fat | Loose areolar tissue + fat; TEP/TAPP hernia repair works in this plane |
| 9 | Peritoneum (Parietal) | Innermost layer; innervated (sensitive to pain) |
MUSCLES IN DETAIL
Lateral Group (3 flat muscles):
| Muscle | Origin | Insertion | Nerve | Action |
|---|---|---|---|---|
| External Oblique | Lower 8 ribs | Iliac crest, inguinal ligament, linea alba | T7–T12 | Compresses abdomen, trunk rotation to opposite side |
| Internal Oblique | Inguinal ligament, iliac crest, thoracolumbar fascia | Lower 3 ribs, linea alba | T7–T12, L1 | Compresses abdomen, trunk rotation to same side |
| Transversus Abdominis | Inguinal ligament, iliac crest, lower 6 costal cartilages | Linea alba, pubic crest | T7–T12, L1 | Compresses abdomen (principal compressor) |
Medial Group:
| Muscle | Origin | Insertion | Nerve | Action |
|---|---|---|---|---|
| Rectus Abdominis | Pubic symphysis & crest | Xiphisternum, costal cartilages 5–7 | T7–T12 | Flexes trunk, compresses abdomen |
| Pyramidalis | Pubic body | Linea alba | T12 | Tenses linea alba (absent in ~20%) |
RECTUS SHEATH — Surgical Anatomy
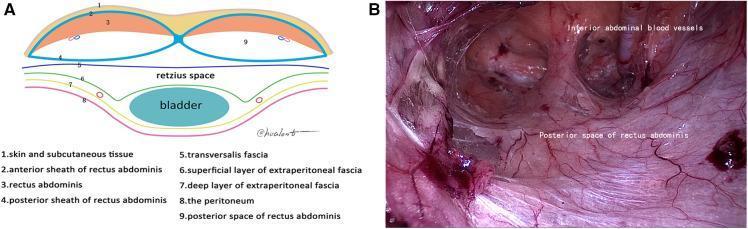
Above Arcuate Line (Upper ¾ of rectus):
| Sheath | Composition |
|---|---|
| Anterior | Aponeurosis of external oblique + anterior lamina of internal oblique |
| Posterior | Posterior lamina of internal oblique + aponeurosis of transversus abdominis |
Below Arcuate Line (Lower ¼, below level of ASIS):
| Sheath | Composition |
|---|---|
| Anterior | All three aponeuroses (EO + IO + TA) pass anterior to rectus |
| Posterior | ABSENT — only transversalis fascia + peritoneum |
The Arcuate line (linea semicircularis / Douglas's line) marks this transition — a surgically important landmark for TEP hernia repair and rectus sheath hematoma.
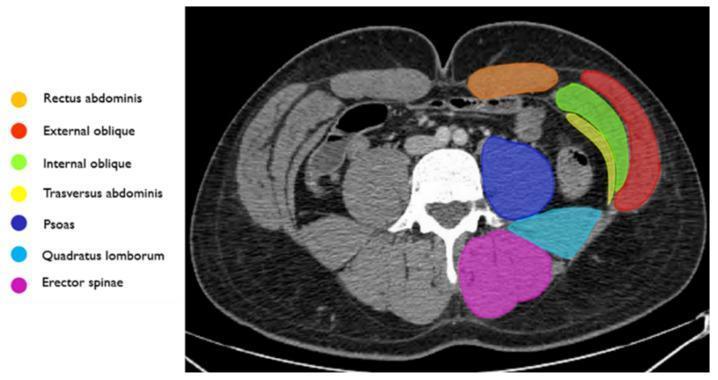
POSTERIOR ABDOMINAL WALL — Layers
| # | Layer |
|---|---|
| 1 | Skin |
| 2 | Superficial fascia |
| 3 | Thoracolumbar fascia (posterior, middle, anterior layers) |
| 4 | Erector spinae (posteriorly) |
| 5 | Quadratus lumborum |
| 6 | Psoas major (medially, along lumbar vertebrae) |
| 7 | Iliacus (laterally, in iliac fossa) |
| 8 | Transversalis fascia |
| 9 | Retroperitoneal fat |
| 10 | Parietal peritoneum |
NERVE SUPPLY OF ANTERIOR ABDOMINAL WALL
| Nerve | Segments | Region |
|---|---|---|
| Thoracoabdominal nerves | T7–T11 | Upper and mid abdomen |
| Subcostal nerve | T12 | Just above iliac crest |
| Iliohypogastric nerve | L1 | Hypogastric / inguinal region |
| Ilioinguinal nerve | L1 | Scrotum/labia, medial thigh |
McBurney's point (appendix) — T10 dermatome
Umbilicus — T10 dermatome (landmark for spinal block level)
BLOOD SUPPLY
| Vessel | Territory |
|---|---|
| Superior epigastric (from internal thoracic) | Upper rectus |
| Inferior epigastric (from external iliac) | Lower rectus; anastomoses with superior |
| Deep circumflex iliac | Lateral wall |
| Superficial epigastric / circumflex iliac | Superficial fascia layers |
| Lower intercostal & subcostal vessels | Lateral muscles |
The inferior epigastric artery is the medial boundary of the deep inguinal ring (lateral to Hesselbach's triangle). Critical landmark in laparoscopic inguinal hernia repair.
SURGICAL INCISIONS & LAYERS ENCOUNTERED
| Incision | Layers Cut |
|---|---|
| Midline (Linea Alba) | Skin → Camper's → Scarpa's → Linea alba → Extraperitoneal fat → Peritoneum |
| Paramedian | Skin → fat → Anterior rectus sheath → Rectus retracted → Posterior sheath → Peritoneum |
| Pfannenstiel | Skin → fat → Anterior rectus sheath (transverse) → Rectus split vertically → Peritoneum |
| Gridiron (McBurney's) | Skin → fat → EO aponeurosis → IO (split) → TA (split) → Peritoneum |
| Kocher's (subcostal) | Skin → fat → Rectus sheath → Rectus (cut) → Peritoneum |
| Lanz | Similar to gridiron but cosmetically better |
QUICK REVISION
| Structure | Surgical Significance |
|---|---|
| Linea alba | Avascular midline — preferred incision site |
| Arcuate line | Absent posterior rectus sheath below it — TEP plane |
| Scarpa's fascia | Sutured separately in Pfannenstiel to reduce dead space |
| Transversalis fascia | Defect here = direct inguinal hernia |
| Inferior epigastric a. | Medial boundary of deep ring; marks indirect vs direct hernia |
| Hesselbach's triangle | Boundaries: inguinal ligament, inferior epigastric, lateral rectus sheath |
Layers at camera port in lap cholecystectomy
laparoscopic cholecystectomy umbilical port insertion layers trocar anatomy
umbilical port trocar layers laparoscopic cholecystectomy anatomy
Layers at the Camera Port in Laparoscopic Cholecystectomy
Why the Umbilicus?
Layers Traversed at the Umbilical Camera Port
(Superficial → Deep)
| # | Layer | Surgical Note |
|---|---|---|
| 1 | Skin | Incision made just inferior to the umbilical cicatrix (transverse or vertical, 10–12 mm) |
| 2 | Subcutaneous fat (Camper's fascia) | Thin at umbilicus; dissected with sharp/blunt dissection |
| 3 | Scarpa's fascia | Membranous layer — may be encountered but is thin/fused at umbilicus |
| 4 | Umbilical cicatrix / Umbilical stalk | The fibrous remnant of the umbilical cord; directly adherent to linea alba below |
| 5 | Linea alba (Decussation of aponeuroses) | EO + IO + TA aponeuroses fuse here; incised vertically 5 mm — the actual fascial barrier; no rectus muscle here |
| 6 | Extraperitoneal (Preperitoneal) fat | Thin loose areolar layer; traversed with blunt haemostat |
| 7 | Parietal Peritoneum | Final layer — "pop" felt as instrument enters; entered with blunt haemostat or trocar |
| 8 | Peritoneal cavity | Trocar confirmed inside; CO₂ insufflation commenced |
Key Anatomical Points
- No muscle layer is cut at the umbilicus — the rectus muscles lie lateral; only the linea alba (aponeurosis) is incised
- The skin at the umbilicus is directly fused to the linea alba — hence it is everted and grasped at the cicatrix to get closest to the fascia
- The decussation of fibres just above the linea alba junction is the fascial layer that is incised
- Below the umbilicus: the posterior rectus sheath is absent (below arcuate line) — only transversalis fascia + peritoneum lie deep to the muscles
- The inferior epigastric vessels lie lateral — safe at the midline umbilical entry
Two Techniques of Umbilical Entry
1. Veress Needle (Closed Technique)
- 1st pop = Linea alba / fascia
- 2nd pop = Peritoneum Then insufflate → blind trocar insertion
2. Hasson's Open Technique (Preferred, Safer)
- Evert umbilical cicatrix with toothed forceps
- Dissect down to expose decussation of fibres above linea alba
- 5 mm vertical incision through fascia only
- Blunt haemostat pushed through preperitoneal fat + peritoneum — "pop" felt
- Blunt-tipped 10/12 mm trocar inserted under direct vision
- Camera confirms intraperitoneal placement → CO₂ insufflation begins
Hasson's technique is preferred in obese patients, previous abdominal surgeries, and beginners — avoids risk of vascular/bowel injury from blind Veress needle insertion.
Summary — Layers at a Glance
SKIN
↓
Subcutaneous fat (Camper's fascia)
↓
Scarpa's fascia
↓
Umbilical cicatrix / stalk
↓
LINEA ALBA ← only fascial layer incised (no muscle here)
↓
Extraperitoneal fat
↓
PARIETAL PERITONEUM ← "pop"
↓
PERITONEAL CAVITY ✓
Complications Related to Port Insertion at This Site
| Complication | Reason |
|---|---|
| Bowel injury | Adherent bowel to anterior abdominal wall (previous surgeries) — use Palmer's point in such cases |
| Vascular injury (aorta, IVC) | Excessive trocar angulation in thin patients |
| Port-site hernia | Failure to close linea alba (fascial defect) after 10 mm+ ports — Richter's hernia risk |
| Insufflation into preperitoneal space | Veress needle not fully through peritoneum |
| Omental/bowel emphysema | Veress needle in omentum or bowel |
Always close fascial defects ≥10 mm at the umbilical port — risk of port-site hernia is significant if linea alba is not sutured.
Ports of laproscopic cholecystectomy
laparoscopic cholecystectomy port placement positions trocar sites 4 port technique
laparoscopic cholecystectomy port placement diagram trocar positions
laparoscopic cholecystectomy epigastric port working ports instruments used trocar size mm
single port SILS reduced port laparoscopic cholecystectomy three port technique
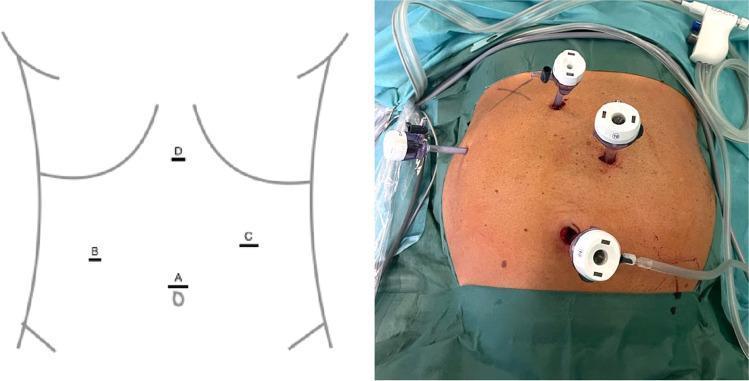
Ports of Laparoscopic Cholecystectomy
Standard 4-Port Technique (American/French)

Port Details — One by One
PORT 1 — Camera Port (Primary Port)
| Feature | Details |
|---|---|
| Site | Umbilicus (infra- or supra-umbilical) |
| Size | 10–12 mm |
| Purpose | 30° laparoscopic camera / telescope |
| Technique | Hasson's open (preferred) or Veress needle |
| Layers | Skin → Camper's → Scarpa's → Linea alba → Extraperitoneal fat → Peritoneum |
| Inserted by | Surgeon; first port placed |
| Note | Umbilicus is thinnest, most avascular point; no muscle here — only linea alba |
PORT 2 — Epigastric Port (Surgeon's Right Hand)
| Feature | Details |
|---|---|
| Site | Epigastric region — 5 cm below xiphisternum, slightly right of midline |
| Size | 10 mm |
| Purpose | Clip applicator (10 mm titanium clips), dissector, hook cautery, scissors |
| Technique | Inserted under direct vision after pneumoperitoneum |
| Structures avoided | Falciform ligament (pass to right of it), left lobe of liver |
| Note | Most important working port — cystic duct and artery are clipped through this port; must close fascia (≥10 mm) |
PORT 3 — Right Mid-Abdomen Port (Surgeon's Left Hand / Retraction)
| Feature | Details |
|---|---|
| Site | Right mid-clavicular line, at or just below the right costal margin (2–3 cm below) |
| Size | 5 mm |
| Purpose | Fundus retraction (grasper to hold gallbladder fundus — Hartmann's pouch retraction), irrigation/suction |
| Technique | Under direct vision; lateral to rectus sheath to avoid epigastric vessels |
| Note | Creates upward and lateral traction on gallbladder to expose Calot's triangle |
PORT 4 — Right Anterior Axillary Line Port (Assistant's Port)
| Feature | Details |
|---|---|
| Site | Right anterior axillary line (or right flank), below costal margin |
| Size | 5 mm |
| Purpose | Assistant retracts gallbladder upward over liver (fundus held here); also used for suction |
| Technique | Under direct vision |
| Note | Provides countertraction — essential for opening Calot's triangle safely; some surgeons omit this in 3-port technique |
Summary Table
| Port | Site | Size | Used By | Instrument |
|---|---|---|---|---|
| 1 — Camera | Umbilicus | 10–12 mm | Surgeon | 30° Telescope |
| 2 — Epigastric | 5 cm below xiphoid, right of midline | 10 mm | Surgeon (R hand) | Clip applicator, Hook, Dissector |
| 3 — R. Mid-clavicular | Right MCL, subcostal | 5 mm | Surgeon (L hand) | Grasper (Hartmann's pouch) |
| 4 — R. Ant. Axillary | Right AAL, subcostal | 5 mm | Assistant | Grasper (Fundus retraction) |
Port Placement Principles (Bailey & Love, p. 124)
- All secondary ports inserted under direct vision — never blind
- Trocars always inserted perpendicular to abdominal wall
- Non-midline ports placed lateral to rectus sheath (mid-clavicular line) to avoid inferior epigastric vessels
- Use smallest diameter trocar possible — less pain, less hernia risk
- All ports >5 mm require fascial closure to prevent port-site hernia
- Remove all ports under direct vision — check for port-site bleeding
American vs French Technique
| Feature | American Technique | French Technique |
|---|---|---|
| Patient position | Supine | Lithotomy / legs split |
| Surgeon position | Left side of patient | Between legs |
| Port 3 & 4 placement | Right side (as above) | More cephalad / subcostal |
| Camera port | Umbilical | Umbilical |
| Advantage | Common, simple | Better liver retraction, obese patients |
Alternative Techniques
3-Port Technique
- Port 4 (assistant's port) omitted
- Surgeon provides retraction through Port 3
- Suitable for: thin patients, experienced surgeons, non-complicated gallbladders
Single Incision Laparoscopic Surgery (SILS)
- All ports through one umbilical incision using a multi-channel port
- Better cosmesis ("scarless")
- Technical difficulty higher; longer operative time
- Not standard of care
Robotic Cholecystectomy
- 4 ports (3 robotic arms + 1 assistant)
- Placed more superiorly to accommodate robotic arm geometry
Critical View of Safety (CVS) — Why Port Positions Matter
- Port 3 retracts Hartmann's pouch laterally → opens hepatocystic triangle
- Port 4 retracts fundus upward → stretches cystic duct
- Port 2 dissects Calot's triangle from epigastric approach at correct angle
CVS requires: (1) Hepatocystic triangle cleared of fat/fibrotic tissue, (2) Lower gallbladder dissected off liver bed, (3) Only two structures entering gallbladder visible — before any clipping.
Port-Site Complications
| Complication | Port Most at Risk | Prevention |
|---|---|---|
| Port-site hernia | Umbilical (10–12 mm), Epigastric (10 mm) | Close fascia for all ≥10 mm ports |
| Epigastric vessel injury | Port 3 & 4 if placed medially | Stay lateral to rectus — MCL/AAL |
| Bowel injury | Any port, especially in adhesions | Direct vision insertion, Hasson at umbilicus |
| Port-site metastasis | Umbilical (in malignancy) | Extract specimen in bag |
| Subcutaneous emphysema | Any port | Correct fascial placement confirmed |
Sage guidlines
SAGE guidelines laparoscopic cholecystectomy safe cholecystectomy
SAGE Guidelines — Safe Cholecystectomy
Bile duct injury occurs in 0.3–0.6% of laparoscopic cholecystectomies — the most feared complication. SAGES SAFE guidelines were created to systematically reduce this.
The SAGES Safe Cholecystectomy Program — 6 Strategies
"The Fundamental Principles of Safe Laparoscopic Cholecystectomy"
1. ACHIEVE THE CRITICAL VIEW OF SAFETY (CVS)
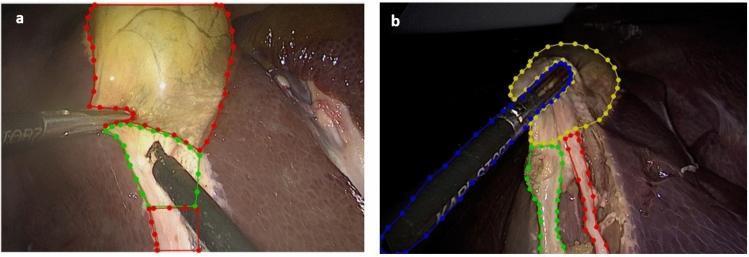
| Criterion | Description |
|---|---|
| 1 | Hepatocystic triangle completely cleared of fat and fibrotic tissue |
| 2 | Lower one-third of gallbladder dissected off the liver bed (cystic plate visible) |
| 3 | Only two structures seen entering the gallbladder — cystic duct and cystic artery |
CVS was described by Strasberg et al. (1995) — the gold standard before clipping any structure.
- CVS must be achieved from both anterior and posterior views
- If CVS cannot be achieved → do NOT clip → move to bail-out strategies
- Document CVS with a photograph intraoperatively
2. KNOW THE HEPATOCYSTIC TRIANGLE ANATOMY & ITS VARIATIONS
- Superiorly — inferior surface of liver
- Medially — common hepatic duct
- Laterally — cystic duct
Important Anatomical Variations to Know:
| Structure | Normal | Variation |
|---|---|---|
| Cystic artery | From right hepatic artery, within Calot's triangle | Dual cystic arteries; from GDA, left hepatic, proper hepatic |
| Cystic duct | Joins CHD at mid-level, right lateral | Low/high junction, parallel course, medial insertion, short cystic duct |
| Right hepatic artery | Posterior to CHD | Caterpillar/Moynihan's hump — loops anteriorly, mimics cystic artery |
| Aberrant bile duct | Absent | Duct of Luschka — small duct from GB bed to CHD; if not recognized → bile leak |
3. RECOGNIZE THE INFUNDIBULAR TECHNIQUE IS UNRELIABLE
- In inflammation/fibrosis, the CBD can be tented up and mistaken for the cystic duct
- The parallel course variant of the cystic duct running alongside the CBD is especially treacherous
- SAGES explicitly states: the infundibular technique alone should NOT be used — CVS must be achieved
4. IF IN DOUBT — STOP AND OBTAIN A CHOLANGIOGRAM / BAIL OUT
Bail-out Strategies (in order of preference):
| Strategy | When to Use |
|---|---|
| Intraoperative Cholangiogram (IOC) | Anatomical uncertainty; visualizes biliary tree in real time |
| Laparoscopic Ultrasound | Identifies CBD, hepatic artery without radiation |
| Fundus-first / Top-down dissection | Severe Calot's triangle inflammation — dissect GB off liver from fundus downward |
| Subtotal cholecystectomy (reconstituting or fenestrating) | Cannot safely dissect Calot's triangle — leave posterior GB wall on liver |
| Conversion to open | Loss of orientation, uncontrolled bleeding, dense adhesions |
| Drain and refer | Damage control — drain biliary field, close, refer to HPB surgeon |
The most important bail-out is conversion to open — this is NOT a failure; it is a safe, deliberate decision.
5. KNOW WHEN AND HOW TO OBTAIN HELP
- Involve a more experienced colleague intraoperatively if uncertain
- Refer to an HPB (Hepatopancreatobiliary) surgeon if BDI is suspected
- Do not attempt repair of a bile duct injury if you are not trained in biliary reconstruction — premature repair worsens outcomes
- Intraoperative recognition of BDI → immediate referral to HPB center
6. AVOID CLASSIC BILE DUCT INJURY MECHANISMS
The "Visual Illusion" — Most Common Cause of BDI:
- Excessive cephalad traction on the gallbladder aligns the CBD with the cystic duct
- Creates a false impression of a "cystic duct" that is actually the CBD
- Compounded by failure to achieve CVS
Strasberg Classification of BDI:
| Type | Injury |
|---|---|
| A | Bile leak from cystic duct stump or Duct of Luschka |
| B | Occlusion of aberrant right hepatic duct |
| C | Transection (not ligated) of aberrant right hepatic duct |
| D | Lateral injury to CBD |
| E1–E5 | Transection of CBD at varying levels (Bismuth classification) |
Type E injuries (CBD transection) are the most devastating — require Roux-en-Y hepaticojejunostomy.
SAGES SAFE CHOLECYSTECTOMY — The 5 Pillars (Simplified for Exam)
| Pillar | Key Message |
|---|---|
| 1. CVS | Achieve and document Critical View of Safety before clipping |
| 2. Anatomy | Know normal and variant hepatobiliary anatomy |
| 3. Cholangiogram | Use IOC liberally when in doubt |
| 4. Bail-out | Know when to stop, convert, or perform subtotal cholecystectomy |
| 5. Culture | Promote a culture of safety — no hesitation to ask for help |
Critical View of Safety vs Other Methods
| Method | Safety | Reliability |
|---|---|---|
| CVS (Strasberg) | Highest | Gold standard |
| Infundibular technique | Lower | Unreliable in inflammation |
| IOC | High | Requires expertise/equipment |
| Laparoscopic ultrasound | High | Operator dependent |
| Subtotal cholecystectomy | Safe bail-out | For hostile Calot's |
"Difficult Cholecystectomy" — When to Anticipate Problems
| Risk Factor | Strategy |
|---|---|
| Acute cholecystitis | Early surgery (<72 hrs); expect inflammation |
| Mirizzi syndrome | Always do IOC; high CBD injury risk |
| Previous upper abdominal surgery | Open Hasson + adhesiolysis |
| Cirrhosis | Coagulopathy; bleeding risk; consider open |
| Obesity (BMI >40) | Longer trocars; may need extra port |
| Contracted/fibrotic GB | Subtotal cholecystectomy likely |
| Suspected malignancy | Conversion to open; staging first |
Subtotal Cholecystectomy — The Ultimate Bail-Out
| Type | Description |
|---|---|
| Fenestrating | Open GB anteriorly, remove stones, ablate mucosa, leave posterior wall on liver — leave cystic duct open, drain |
| Reconstituting | Remove all of GB except posterior wall adherent to liver; close cystic duct stump; reconstruct |
Fenestrating type has higher bile leak rate but is safer in hostile abdomen. Both are safer than risking BDI.
Quick Revision — SAGE in One Line Each
- S — Stop if unsafe; Subtotal if Calot's hostile
- A — Achieve CVS (all 3 criteria) before clipping
- G — Get help / Get a cholangiogram when in doubt
- E — Examine anatomy carefully; know variants
Bottom line: The single most important safety measure in laparoscopic cholecystectomy is achieving and documenting the Critical View of Safety before placing any clip. When in doubt — bail out.
Carter thomson suture passer
Carter Thomson suture passer device laparoscopic port closure
Carter-Thomason Suture Passer
What Is It?
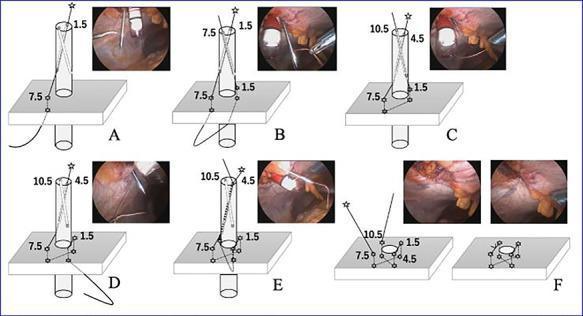
Indications
| Indication | Details |
|---|---|
| Port-site fascial closure | All trocar sites ≥10 mm (mandatory) |
| ≥5 mm in high-risk patients | Obese, elderly, immunocompromised, steroid use |
| Midline 5 mm ports | Higher hernia risk than lateral ports — consider closure |
| Prevention of port-site hernia | Especially umbilical and epigastric 10–12 mm ports |
| Prevention of Richter's hernia | Partial bowel wall herniation through fascial defect |
Components of the Carter-Thomason Device
| Component | Description |
|---|---|
| Outer cannula/guide | Hollow cylinder passed alongside/through trocar site into peritoneal cavity; has two diagonal channels at 180° to each other |
| Suture passer (inner needle) | Long, sharp, hollow needle with a hook/eye at tip; passes through the channels of the guide |
| Suture | Absorbable (Vicryl 0 or 1) or non-absorbable (PDS) depending on surgeon preference |
How It Works — Step by Step
Principle:
Steps:
- Pneumoperitoneum maintained; 30° camera observing from adjacent port
- Trocar to be closed still in situ (or partially withdrawn)
- Carter-Thomason guide inserted through/alongside the trocar into peritoneal cavity
- Suture passer needle advanced through one diagonal channel (e.g., 1:30 o'clock position)
- Needle tip seen inside peritoneal cavity by camera
- Free end of suture loaded into the needle hook and pulled back through the channel — suture now passes through fascia on one side
- Guide rotated 180° (to 7:30 o'clock position — opposite side of defect)
- Suture passer advanced through second channel
- Loop of the same suture captured and pulled back through — suture now passes through fascia on both sides of the defect
- Trocar removed while maintaining suture ends
- Both ends of the suture now exit through the skin on either side
- Fascia is pulled together as suture is tied
- Creates a figure-of-8 closure of the full fascial defect
- Skin closed separately with absorbable subcuticular suture or skin glue
Types of Suture Patterns Achieved
| Pattern | Technique | Use |
|---|---|---|
| Figure-of-8 | Two passes through opposite sides | Standard, most common |
| Z-suture | Four passes at 1:30, 7:30, 10:30, 4:30 positions | Larger defects, more secure |
| Simple interrupted | Single pass each side | Thin patients, small defects |
Advantages
| Advantage | Details |
|---|---|
| Direct vision closure | Camera confirms full-thickness fascial bite on both sides — no blind suturing |
| Safe in obese patients | Standard closure impossible in thick abdominal walls — device bridges the depth |
| Avoids Richter's hernia | Bowel not trapped in partially closed defect |
| Quick | Entire closure takes 1–2 minutes per port |
| Minimal trauma | No large skin incision needed for fascial closure |
| Prevents port-site hernia | Gold standard for fascial closure in laparoscopic surgery |
Disadvantages / Complications
| Issue | Details |
|---|---|
| Bowel entrapment | If suture passed too deeply without camera visualization |
| Vessel injury | Epigastric vessels in the needle path — stay midline or use camera guidance |
| Suture breakage | If tied under excessive tension |
| Inadequate bite | If only peritoneum caught and not fascia — defect not truly closed |
| Cost | Disposable device adds to operative cost |
Alternatives to Carter-Thomason
| Device/Method | Description |
|---|---|
| Veress needle closure technique | Suture loaded on Veress needle — cheap but blind |
| Standard J-needle (Keith needle) | Long straight needle passed through skin under vision — requires incision |
| Endoclose device | Similar principle — hook needle passed under vision |
| Open fascial closure | Extending skin incision, directly suturing fascia under vision — gold standard in open surgery |
| Zero-closure trocars | Radially dilating trocars (Versaport, Endopath XCEL) — fascial defect smaller, may not need closure for 10–12 mm |
Important Rules for Port-Site Fascial Closure
| Rule | Guideline |
|---|---|
| ≥10 mm ports | Always close fascia — mandatory (SAGES, Bailey & Love p.124) |
| 5 mm lateral ports | Generally safe without closure |
| 5 mm midline ports | Consider closure |
| Suture material | PDS (polydioxanone) No.1 preferred — delayed absorbable, strong, low infection risk |
| Skin closure | Separate from fascial closure — absorbable subcuticular stitch or tissue glue |
| Timing | Close under vision before complete deflation of pneumoperitoneum |
Port-Site Hernia — Why It Matters
| Feature | Details |
|---|---|
| Incidence | 0.65–2.8% overall; up to 5% at umbilical 10–12 mm sites |
| Most common site | Umbilical port (camera port) |
| Most dangerous type | Richter's hernia — partial bowel wall herniation, can strangulate without obstruction |
| Risk factors | Obesity, ≥10 mm port, midline location, fascial defect not closed, wound infection |
| Prevention | Carter-Thomason or equivalent device for all ≥10 mm ports |
Quick Summary
Carter-Thomason Suture Passer
↓
Guided cannula → peritoneal cavity
↓
Suture passed through fascia
(both sides of trocar defect)
under direct laparoscopic vision
↓
Trocar removed
↓
Figure-of-8 fascial closure tied
↓
Port-site hernia PREVENTED
Key teaching point: The Carter-Thomason device converts a previously blind and technically difficult fascial closure into a safe, direct-vision, reproducible procedure — essential for all laparoscopic ports ≥10 mm.