The role of glucocorticosteroids in the treatment of rheumatoid arthritis.
glucocorticosteroids rheumatoid arthritis mechanism anti-inflammatory
glucocorticoids RA treatment dosing bridging therapy disease modification DMARD
glucocorticoid mechanism action NF-kB cytokines inflammation molecular
glucocorticoids side effects adverse effects osteoporosis RA long-term use
Glucocorticosteroids in the Treatment of Rheumatoid Arthritis
1. Mechanisms of Action
Genomic (Classical) Pathway
- GCs diffuse across cell membranes and bind to intracytoplasmic glucocorticoid receptors (GR-α), forming a ligand-receptor complex.
- This complex translocates to the nucleus and:
- Transrepresses pro-inflammatory transcription factors — most critically NF-κB and AP-1 — reducing expression of cytokines (TNF-α, IL-1β, IL-6, IL-17), chemokines, adhesion molecules, and enzymes (COX-2, iNOS, phospholipase A2).
- Transactivates anti-inflammatory genes encoding lipocortin-1 (annexin A1), IL-10, and IκBα (an NF-κB inhibitor).
Non-Genomic Pathway (rapid effects, relevant at high doses)
- Direct physicochemical membrane interactions and cytosolic GR signalling produce rapid vasoconstrictive and anti-inflammatory effects within minutes, bypassing gene transcription.
Key Cellular Effects in RA
| Cell Type | Effect |
|---|---|
| Macrophages | Reduced cytokine synthesis (TNF-α, IL-1, IL-6, IL-12) |
| T lymphocytes | Reduced proliferation and cytokine release |
| Neutrophils | Reduced migration into synovium |
| Synoviocytes | Reduced prostaglandin and metalloproteinase production |
| Osteoclasts | Indirect promotion (long-term adverse effect) |
2. Place in RA Management
3. Clinical Roles
A. Bridging Therapy (Short-Term)
"Consider using a short course of glucocorticoids in people with active rheumatoid arthritis who are initiating, switching or adding DMARD therapy, using the lowest effective dose until DMARDs take effect." (Pharmacological Management of Inflammatory Arthritis, p. 8 — Conditional recommendation)
- Rationale: csDMARDs (particularly methotrexate) have a latency of 4–12 weeks. GCs suppress inflammation during this gap.
- Duration: typically 4–12 weeks, tapering as DMARD effect becomes apparent.
- Key principle: inability to reach treatment target should trigger DMARD escalation, not prolonged GC use (Pharmacological Management of Inflammatory Arthritis, p. 8).
B. Flare Management
- Short courses of oral prednisone (e.g., 20–40 mg/day, rapidly tapered) or intra-articular GC injections (triamcinolone acetonide, methylprednisolone acetate) are used to manage acute flares, especially when 1–3 joints are predominantly affected.
- Intra-articular injections minimise systemic exposure while delivering high local concentrations.
C. Chronic Low-Dose GC
- Some patients with refractory or erosive RA may use chronic low-dose prednisone (≤7.5 mg/day).
- EULAR and ACR guidelines acknowledge this may have a disease-modifying effect at low doses — reducing radiographic progression in some trials (e.g., the COBRA and BeSt trials showed combination GC strategies slowed joint erosion).
- However, this must be balanced against cumulative toxicity; guidelines recommend aiming for discontinuation whenever possible.
D. IM/IV Pulse Therapy
- High-dose IV methylprednisolone (0.5–1 g/day × 3 days) may be used for severe systemic RA (e.g., vasculitis, pleuropericarditis) or to induce rapid remission.
4. Dosing Principles
| Scenario | Typical Regimen |
|---|---|
| Bridging (DMARD initiation) | Prednisone 10–20 mg/day, taper over 4–12 weeks |
| Acute flare (systemic) | Prednisone 20–40 mg/day, taper over 2–4 weeks |
| Intra-articular (single joint flare) | Triamcinolone 20–40 mg, or methylprednisolone 20–40 mg |
| Chronic low-dose | Prednisone ≤7.5 mg/day (aim to discontinue) |
| Severe systemic disease | IV methylprednisolone 500–1000 mg × 1–3 days |
5. Adverse Effects
| System | Adverse Effect |
|---|---|
| Metabolic | Hyperglycaemia / steroid-induced diabetes, dyslipidaemia, weight gain, Cushingoid features |
| Skeletal | Osteoporosis, fragility fractures (highest risk in first 3–6 months), osteonecrosis (avascular necrosis of femoral head) |
| Cardiovascular | Hypertension, accelerated atherosclerosis, fluid retention |
| Immunological | Increased infection risk (bacterial, fungal, viral including VZV reactivation), impaired wound healing |
| Adrenal | HPA axis suppression, adrenal insufficiency on abrupt withdrawal |
| Ocular | Posterior subcapsular cataracts, raised intraocular pressure / glaucoma |
| Neuropsychiatric | Mood disturbance, insomnia, psychosis (rare, high-dose) |
| Musculoskeletal | Steroid myopathy (proximal weakness), skin atrophy |
| GI | Peptic ulceration (especially combined with NSAIDs) |
6. Preventive Co-prescribing
- Osteoporosis prevention: calcium (1000–1200 mg/day) + vitamin D (800–1000 IU/day); add bisphosphonate (e.g., alendronate, risedronate) if expected duration >3 months or fracture risk is elevated (FRAX-guided).
- PPI for GI protection, especially if co-prescribed with NSAIDs or aspirin.
- Blood glucose monitoring in patients with or at risk of diabetes.
- Blood pressure monitoring.
- Vaccinations (influenza, pneumococcal, shingles/VZV) before or early in therapy.
7. Summary
morphology of neuron
neuron morphology structure cell body dendrites axon myelin sheath
neuron morphology structure diagram labeled
Morphology of the Neuron
1. Cell Body (Soma / Perikaryon)
- Size: ranges from ~5 µm (cerebellar granule cells) to ~100 µm (anterior horn motor neurons).
- Nucleus: large, round, euchromatic (pale-staining), with a prominent nucleolus — reflecting high transcriptional activity. Neurons are largely post-mitotic.
- Nissl substance (Nissl bodies): stacks of rough endoplasmic reticulum (rER) + free ribosomes; responsible for intensive protein synthesis. They extend into dendrites but are absent from the axon hillock.
- Cytoskeleton: contains neurofilaments (intermediate filaments), microtubules, and actin filaments, which maintain shape and support axonal transport.
- Cytoplasmic organelles: Golgi apparatus (prominent, juxtanuclear), mitochondria, lysosomes, and lipofuscin granules (aging pigment accumulating with time).
- Inclusions: melanin granules in substantia nigra neurons; lipofuscin in older neurons.
2. Dendrites
- Arise as multiple tapering branches from the soma ("dendritic tree").
- Contain Nissl substance, mitochondria, microtubules, and ribosomes.
- Dendritic spines: small protrusions along the dendritic shaft; sites of excitatory synaptic contact.
- Thin spines (filopodia-like): long slender neck, small head; associated with silent synapses containing NMDA receptors.
- Mushroom-shaped spines: bulbous head, shorter neck; mature, active synapses with high AMPA receptor density — the structural basis of long-term potentiation (LTP).
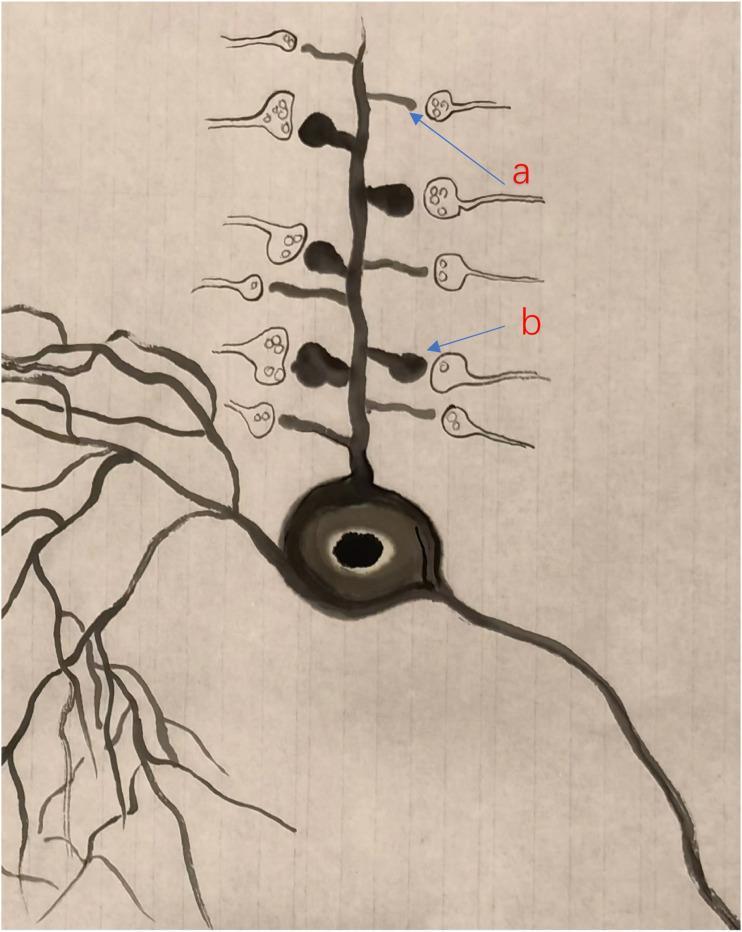
- Surface area of dendrites is greatly amplified by branching and spines, allowing convergence of thousands of synaptic inputs.
3. Axon Hillock
- The cone-shaped region of the soma from which the axon emerges.
- Lacks Nissl substance — a key histological landmark.
- The site of action potential initiation: the highest concentration of voltage-gated Na⁺ channels is at the initial segment (just distal to the hillock), giving it the lowest threshold for firing.
4. Axon
- Length: from <1 mm (interneurons) to >1 m (lower motor neurons to the feet).
- Diameter: correlates with conduction velocity — larger diameter → faster conduction.
- Axoplasm: contains microtubules, neurofilaments, mitochondria, and smooth ER, but no ribosomes or rER (all proteins are synthesized in the soma and transported).
- Axonal transport:
- Anterograde (soma → terminal): kinesin motor proteins; fast (~400 mm/day) for vesicles/organelles, slow (~1–4 mm/day) for cytoskeletal elements.
- Retrograde (terminal → soma): dynein motor proteins; returns degraded organelles and trophic signals (e.g., NGF).
- Collateral branches: axons may branch (collaterals) to contact multiple targets.
- Axon terminal (synaptic bouton): the distal end, containing synaptic vesicles packed with neurotransmitters, ready for exocytosis upon Ca²⁺ influx.
5. Myelin Sheath
| Feature | CNS | PNS |
|---|---|---|
| Producing cell | Oligodendrocyte | Schwann cell |
| Axons per cell | One oligodendrocyte sheaths multiple axons | Each Schwann cell covers one axon segment |
| Regeneration | Poor | Good |
- Nodes of Ranvier: periodic gaps (~1–2 µm) in the myelin sheath where Na⁺ and K⁺ channels are clustered. Action potentials "jump" node-to-node — saltatory conduction — achieving velocities up to 70–120 m/s in large myelinated fibers.
- Internodal segments: myelinated lengths between nodes (~1–2 mm).
- Oligodendrocytes contact axons at paranodes, where channel clustering and axon-myelin molecular interactions are required for structural stability and normal axon lifespan (Harrison's, p. 11977).
6. Classification by Morphology
| Type | Structure | Example |
|---|---|---|
| Multipolar | One axon + multiple dendrites | Motor neurons, pyramidal cells |
| Bipolar | One axon + one dendrite | Retinal ganglion cells, cochlear neurons |
| Unipolar (pseudounipolar) | Single process divides into central + peripheral branches | Dorsal root ganglion sensory neurons |
| Anaxonic | No distinct axon | Amacrine cells of retina |
7. Summary Table of Neuronal Components
| Component | Key Features | Function |
|---|---|---|
| Soma | Nucleus, Nissl bodies, organelles | Metabolic center, protein synthesis |
| Dendrites | Multiple, tapering, spines present | Receive synaptic input |
| Axon hillock | No Nissl, high Na⁺ channel density | Action potential initiation |
| Axon | Single, long, no ribosomes | Signal transmission |
| Myelin sheath | Oligodendrocyte (CNS) / Schwann cell (PNS) | Insulation, saltatory conduction |
| Nodes of Ranvier | Gaps in myelin, Na⁺/K⁺ channels | Saltatory conduction |
| Synaptic terminal | Synaptic vesicles, Ca²⁺-dependent release | Neurotransmitter release |
morphology of nephron
nephron morphology structure glomerulus tubules loop of Henle histology
nephron structure diagram labeled parts
glomerulus Bowman capsule podocytes filtration barrier anatomy
proximal convoluted tubule distal tubule collecting duct epithelium function histology
Morphology of the Nephron
Nephron Diagram
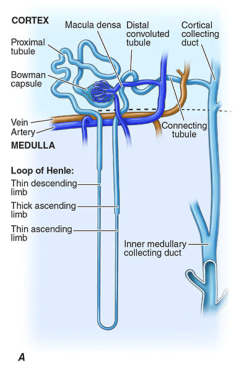
Types of Nephrons
| Feature | Cortical Nephron | Juxtamedullary Nephron |
|---|---|---|
| Glomerulus location | Mid-to-outer cortex | Cortex–medulla junction |
| Loop of Henle | Short | Long (extends deep into medulla) |
| Blood supply | Shared peritubular capillaries | Individual vasa recta running alongside the loop |
| Proportion | ~85% of nephrons | ~15% of nephrons |
| Role | General filtration | Urinary concentration (countercurrent mechanism) |
Components of the Nephron
1. Renal Corpuscle (Malpighian Corpuscle)
A. Glomerulus
- A tuft of fenestrated capillaries derived from the afferent arteriole, forming a branching capillary bed embedded in mesangial matrix, draining into the efferent arteriole (Harrison's, p. 8559).
- The two kidneys together harbor nearly 1.8 million glomerular capillary tufts.
- The efferent arteriole subsequently feeds the peritubular capillaries (cortical nephrons) or vasa recta (juxtamedullary nephrons).
Glomerular Filtration Barrier (3 layers)
| Layer | Structure | Function |
|---|---|---|
| Fenestrated endothelium | Pores ~70–100 nm; negatively charged glycocalyx | Prevents cellular elements from passing |
| Glomerular basement membrane (GBM) | Type IV collagen, laminin, heparan sulfate; negatively charged | Size and charge barrier |
| Podocytes (visceral epithelium) | Foot processes (pedicels) with filtration slits bridged by slit diaphragm (nephrin, podocin) | Final barrier; most critical for protein retention |
- Mesangial cells: specialized pericyte-like cells embedded in mesangial matrix; provide structural support, regulate capillary flow, and have phagocytic function.
B. Bowman's Capsule
- A double-walled cup enclosing the glomerulus (Harrison's, p. 8559).
- Parietal layer: simple squamous epithelium; transitions into the proximal tubule epithelium.
- Visceral layer: podocytes with foot processes covering the outer surface of glomerular capillaries.
- Bowman's space (urinary space): between the two layers — collects the glomerular filtrate (primary urine, ~180 L/day).
2. Proximal Convoluted Tubule (PCT)
- Located in the cortex, close to the glomerulus.
- Epithelium: simple cuboidal cells with:
- Brush border (microvilli): massively amplifies apical surface area for reabsorption.
- Abundant mitochondria: basolateral infoldings packed with mitochondria to power active transport.
- Prominent nucleolus and acidophilic cytoplasm.
- Function: reabsorbs ~65–70% of filtered Na⁺, water, glucose (100%), amino acids (100%), HCO₃⁻, phosphate, uric acid; secretes organic acids, drugs, and H⁺.
- Highly susceptible to ischemic/toxic injury (ATN).
3. Loop of Henle
A. Thick Descending Limb (Pars Recta / Straight Part of PCT)
- Continuation of PCT; simple cuboidal with a brush border (less prominent than PCT).
B. Thin Descending Limb
- Simple squamous epithelium; highly permeable to water (aquaporin-1), impermeable to solutes.
- Water leaves passively as tubular fluid descends into the hyperosmotic medullary interstitium.
C. Thin Ascending Limb (juxtamedullary nephrons only)
- Simple squamous epithelium; impermeable to water, permeable to NaCl.
D. Thick Ascending Limb (TAL)
- Simple cuboidal/columnar epithelium; no brush border.
- Impermeable to water; actively transports Na⁺-K⁺-2Cl⁻ via the NKCC2 co-transporter (site of action of loop diuretics — furosemide).
- Creates the hyperosmotic medullary interstitium that drives water reabsorption in the collecting duct.
- Contains the macula densa at its terminal portion (see below).
4. Juxtaglomerular Apparatus (JGA)
| Component | Cell Type | Function |
|---|---|---|
| Macula densa | Specialized tall columnar cells of TAL | Senses luminal NaCl concentration; signals to JG cells |
| Juxtaglomerular (granular) cells | Modified smooth muscle of afferent arteriole | Secrete renin (activates RAAS); respond to pressure and macula densa signals |
| Lacis cells (extraglomerular mesangium) | Between macula densa and glomerulus | Structural and signalling role |
5. Distal Convoluted Tubule (DCT)
- Located in the cortex.
- Epithelium: simple cuboidal; no brush border (key histological distinction from PCT), fewer and smaller microvilli, basolateral infoldings with mitochondria.
- Shorter than PCT; lumen appears wider and paler on histology.
- Function: reabsorbs Na⁺ via NCC co-transporter (thiazide diuretic target); reabsorbs Ca²⁺ (PTH/vitamin D regulated); early aldosterone-responsive segment.
6. Collecting Duct (CD)
- Receives filtrate from multiple nephrons; runs from cortex through medulla to the renal papilla.
- Epithelium: simple cuboidal to columnar; two cell types:
| Cell Type | Morphology | Function |
|---|---|---|
| Principal cells | Pale cytoplasm, few organelles | Na⁺ reabsorption (ENaC); K⁺ secretion; water reabsorption via aquaporin-2 (ADH-regulated) |
| Intercalated cells (A & B) | Dark granular cytoplasm, many mitochondria | Acid-base regulation; H⁺ secretion (Type A) or HCO₃⁻ secretion (Type B) |
- ADH (vasopressin) inserts aquaporin-2 channels into principal cell apical membranes → water reabsorption → concentrated urine.
- Aldosterone acts on principal cells → increased ENaC and Na⁺/K⁺-ATPase → Na⁺ retention, K⁺ loss.
Summary: Histological Comparison of Tubular Segments
| Segment | Epithelium | Brush Border | Key Feature |
|---|---|---|---|
| PCT | Simple cuboidal | Prominent | Eosinophilic cytoplasm, narrow lumen |
| Thin limb (Henle) | Simple squamous | Absent | Resembles capillaries |
| Thick ascending limb | Simple cuboidal | Absent | Pale cells, no microvilli |
| DCT | Simple cuboidal | Absent/minimal | Wider lumen, paler than PCT |
| Collecting duct | Cuboidal → columnar | Absent | Principal + intercalated cells, distinct cell borders |
Summary: Functional Zones
CORTEX: Renal corpuscle → PCT → DCT → cortical collecting duct
OUTER MEDULLA: Thick descending limb (pars recta) → Thin descending limb → TAL
INNER MEDULLA: Thin descending + ascending limbs (juxtamedullary only) → inner medullary collecting duct → renal papilla