clipping vs coiling vs flow diversion and other aneurysm fixes, when to do what?
Reading File
search_medical_text
intracranial aneurysm surgical clipping vs endovascular coiling indications outcomes
search_medical_text
flow diverter pipeline embolization device intracranial aneurysm treatment
GradeResponse
GradeResponse
search_medical_text
ISAT trial aneurysm coiling clipping randomized controlled trial results
search_medical_text
unruptured intracranial aneurysm treatment decision factors size location morphology
I have enough retrieved material to anchor the answer. I'll now synthesize a comprehensive response drawing on the retrieved sources plus my own knowledge.
Intracranial Aneurysm Treatment: When to Use What
The Core Modalities at a Glance
| Modality | Approach | Mechanism | Best Suited For |
|---|---|---|---|
| Surgical clipping | Open craniotomy | Metal clip across aneurysm neck | Wide-neck, complex, MCA, young patients |
| Endovascular coiling | Catheter via femoral artery | Platinum coils pack the sac → thrombosis | Posterior circulation, elderly, narrow-neck |
| Stent-assisted coiling | Endovascular + stent | Stent scaffolds coils in wide-neck aneurysms | Wide-neck where coiling alone fails |
| Flow diversion (PED) | Endovascular | Redirects flow away from sac → thrombosis | Large/giant fusiform, cavernous/paraclinoid ICA |
| Balloon-assisted coiling | Endovascular | Balloon remodels neck during coiling | Wide-neck, short-term remodeling |
| Intrasaccular devices (WEB) | Endovascular | Braided mesh fills the sac | Bifurcation wide-neck (MCA, AcomA, basilar) |
| Observation | — | Serial imaging | Small unruptured, high surgical risk |
1. Surgical Clipping
A metal clip is placed directly across the aneurysm neck via craniotomy, immediately eliminating rebleed risk (Harrison's, p. 12200).
Prefer clipping when:
- Middle cerebral artery (MCA) aneurysms — surgically accessible, often wide-necked or have branch incorporation
- Young patients (<40–50 yrs) — lifelong durability without retreatment, no antiplatelet requirement
- Wide neck (dome:neck ratio <2, neck >4 mm) not amenable to coiling
- Associated hematoma requiring surgical evacuation — clip + drain in one operation
- Aneurysm morphology with incorporated branches best seen surgically
- Failed endovascular treatment or coil compaction
- Aneurysms at anterior communicating artery with favorable geometry in young patients
Drawbacks: Craniotomy and brain retraction carry neurologic morbidity; more challenging with post-SAH brain edema.
2. Endovascular Coiling
Platinum coils are delivered via femoral artery catheter to pack the sac, promoting thrombosis and walling it off from circulation (Harrison's, p. 12200). Bailey & Love (p. 731) note that Class I evidence favors coiling where feasible, referencing the landmark ISAT trial.
ISAT (International Subarachnoid Aneurysm Trial): In ruptured aneurysms treatable by both methods, coiling reduced death or dependency at 1 year from ~30% (clipping) to ~24% (coiling) — an absolute risk reduction of ~7%. Long-term follow-up showed durable benefit, though coiled aneurysms have higher retreatment rates (~17% vs ~4% for clipping).
Prefer coiling when:
- Posterior circulation aneurysms (basilar tip, PCA) — surgically deep and hazardous
- Elderly patients or those with significant comorbidities — lower procedural morbidity
- Narrow-neck aneurysms (neck <4 mm, dome:neck ≥2)
- Patient is in poor neurological grade (Hunt-Hess 4–5) — minimally invasive preferable
- Cavernous ICA aneurysms (below the dural ring) — not truly intracranial, coiling/flow diversion preferred
- Aneurysms in surgically inaccessible locations
Drawbacks: Higher recanalization/retreatment rate vs clipping; requires antiplatelet therapy if stent used; not ideal for wide-neck aneurysms without adjuncts.
3. Stent-Assisted Coiling
A self-expanding intracranial stent (e.g., Neuroform, LVIS) is deployed across the aneurysm neck to act as scaffolding, preventing coil herniation.
Use when: Wide-neck aneurysm where coiling alone would prolapse coils into parent artery. Requires dual antiplatelet therapy (DAPT) for ~3–6 months, which is a relative contraindication in acute SAH (bleeding risk with heparinization and antiplatelets).
4. Flow Diversion (Pipeline Embolization Device — PED)
A high-mesh-density stent (>30% metal coverage) is deployed in the parent artery across the aneurysm neck. Flow is redirected, promoting intra-aneurysmal thrombosis over weeks to months. The aneurysm gradually involutes.
FDA-approved / strongly preferred for:
- Large (10–25 mm) and giant (>25 mm) aneurysms of the ICA (cavernous, paraclinoid, ophthalmic) — landmark PUFS trial showed 73% occlusion at 6 months, ~87% at 5 years
- Fusiform / dissecting aneurysms without a defined neck (coiling/clipping not feasible)
- Recurrent aneurysms after prior coiling
- Aneurysms unsuitable for clipping or coiling
Key caveats:
- Not ideal in acute SAH — requires prolonged DAPT (aspirin + clopidogrel ≥6 months), risky in the acute hemorrhagic period
- Delayed occlusion (weeks–months) means not immediately protective against rebleed
- Risk of perforator infarcts, thromboembolic events (~5%), vessel occlusion
- Posterior circulation use is debated — higher complication rates with perforators at risk (basilar perforators)
- Newer devices: FRED (Flow Re-Direction Endoluminal Device), Surpass, p64 — similar indications
5. Intrasaccular Flow Disruption (WEB Device)
A braided nitinol mesh is deployed inside the aneurysm sac at bifurcation points. Unlike stent-based approaches, it does not require DAPT long-term.
Ideal for:
- Wide-neck bifurcation aneurysms: MCA bifurcation, anterior communicating artery (AcomA), basilar apex
- Situations where DAPT is undesirable (e.g., SAH, planned surgery)
WEBCAST and French Observatory trials showed ~53–85% adequate occlusion at 1 year.
6. Balloon-Assisted Coiling (BAC)
An inflatable balloon temporarily occludes the parent artery during coiling to allow coil placement in wide-neck aneurysms without a stent. No long-term antiplatelet needed (short-term heparin only).
Use when: Wide-neck, acutely ruptured aneurysm where stent/DAPT is contraindicated.
7. Observation ("Watch and Wait")
Not all aneurysms need immediate treatment.
Observation is appropriate for:
- Small unruptured aneurysms (<7 mm) in low-risk locations (cavernous ICA, distal MCA) with no symptoms
- PHASES score and UIATS (Unruptured Intracranial Aneurysm Treatment Score) help risk-stratify
- High surgical/endovascular risk patients
- Incidental cavernous sinus aneurysms (rarely rupture intracranially)
Risk factors favoring treatment of unruptured aneurysms:
- Size >7–10 mm (risk rises sharply)
- Posterior circulation or posterior communicating artery location
- Prior SAH from another aneurysm
- Familial aneurysm history
- Irregular/lobulated morphology ("daughter sac")
- Younger patient with long life expectancy
- Symptomatic (oculomotor palsy, headache)
Decision Framework Summary
Ruptured aneurysm?
├── YES → Early intervention (<24h if stable) (Bailey & Love, p. 731)
│ ├── Posterior circulation, narrow-neck, elderly → COILING preferred (ISAT)
│ ├── MCA, hematoma, wide-neck, young → CLIPPING preferred
│ ├── Wide-neck, no stent possible → Balloon-assisted coiling or WEB
│ └── Giant/fusiform, acute setting → Clipping or staged flow diversion
│
└── NO (Unruptured)
├── Risk stratify (PHASES score, UIATS)
├── Small, low-risk location → Observe + serial imaging
├── Large/giant, ICA → Flow diversion (PED) preferred
├── Bifurcation, wide-neck → WEB device or stent-assisted coiling
├── MCA, young, surgical candidate → Clipping (durability)
└── Posterior circulation, deep → Coiling or flow diversion
Key Principle: Multidisciplinary Decision
As Bailey & Love (p. 731) emphasizes: these decisions are shared between neurosurgeons, neurointerventional radiologists, and the patient. Aneurysm treatment is among the clearest examples of mandatory multidisciplinary team (MDT) discussion — anatomy, patient age, comorbidities, rupture status, and operator expertise all weigh into the choice. No single modality is universally superior.
Rebleed risk context: Untreated ruptured aneurysms carry a 4% rebleed risk in the first 24 hours, then ~1.5%/day thereafter — 80% of reblee cases end in poor outcome — which is why early intervention is the current consensus regardless of modality chosen (Bailey & Love, p. 731).
make a small table, like anterior circulation + wide neck anerurysm + young pateint is clipping, posterior circualtion + old patient is coiling for example
search_medical_image
intracranial aneurysm clipping coiling treatment decision
GradeResponse
| Location | Neck | Patient Profile | Modality |
|---|---|---|---|
| Anterior (MCA) | Wide (>4 mm) | Young, fit | Clipping |
| Anterior (MCA) | Wide (>4 mm) | Elderly / high surgical risk | Stent-assisted coiling or WEB |
| Anterior (AcomA, ICA) | Narrow | Any | Coiling (first-line) |
| Anterior, large/giant (ICA paraclinoid/cavernous) | Any / fusiform | Any | Flow diversion (PED) |
| Anterior, bifurcation wide-neck (MCA, AcomA) | Wide | Any, DAPT undesirable | WEB device |
| Posterior (basilar tip, PCA) | Narrow | Any | Coiling (surgery too deep) |
| Posterior (basilar tip) | Wide | Any | Stent-assisted coiling or WEB |
| Any + associated hematoma needing evacuation | Any | Any | Clipping (drain + clip in one) |
| Any, ruptured, acute SAH | Wide | Any | Balloon-assisted coiling (avoids DAPT) |
| Small unruptured (<7 mm), low-risk location | — | Elderly / high risk | Observe |
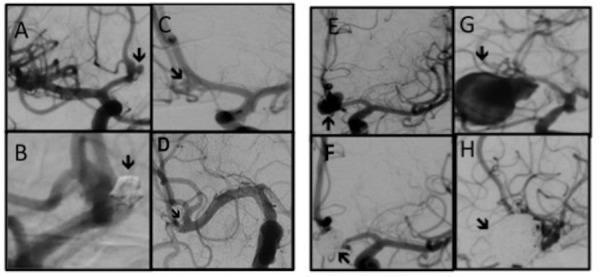
The DSA above shows the contrast between surgical clipping (Panel D — metal clip across the neck, sac no longer fills) and coiling (Panels B/F/H — dense coil packing inside the sac) across aneurysms from 2–26 mm at AcomA and MCA.
Quick rules of thumb:
- Young + MCA + wide neck = clip (durable, no lifelong antiplatelet)
- Posterior circulation + any age = coil (too deep to clip safely)
- Large/giant + fusiform + ICA = flow divert
- Ruptured + wide neck + no DAPT = balloon-assisted coiling or WEB
- Tiny incidental + old/frail = watch
what to do if theres an Subarqacnoid hemmorhage tho,
SAH changes the calculus significantly — the rebleed risk forces your hand on timing and limits some options.
SAH-Specific Decision Rules
Timing first: Intervene within 24 hours if the patient is stable enough (Hunt-Hess 1–3). Rebleed risk is 4% in the first 24h, then ~1.5%/day — 80% of reblee cases end badly. Don't wait.
Treatment Choice in Acute SAH
| Scenario | Preferred Modality | Reason |
|---|---|---|
| Posterior circulation (basilar, PCA), any neck | Coiling | Too deep to clip; ISAT favors coiling overall |
| Anterior circulation, narrow neck | Coiling | Less invasive, faster recovery, ISAT benefit |
| MCA + wide neck + hematoma | Clipping | Evacuate clot + clip in one operation |
| MCA + wide neck, no hematoma, young | Clipping | Durable, DAPT-free |
| Wide neck, no stent feasible, acute SAH | Balloon-assisted coiling | Avoids DAPT (stents require it — dangerous acutely) |
| Wide neck bifurcation (MCA, basilar, AcomA) | WEB device | No DAPT needed, works in acute setting |
| Large/giant fusiform, acute SAH | Clipping or staged | Flow diversion avoided acutely (see below) |
| Poor-grade SAH (Hunt-Hess 4–5) | Coiling if possible | Minimally invasive; clipping high risk in comatose pt |
Why Flow Diversion is Avoided in Acute SAH
This is the big one. Flow diversion (PED) is essentially contraindicated in acute SAH because:
- Requires 6–12 months of DAPT (aspirin + clopidogrel) → catastrophic rebleed risk
- Delayed occlusion (weeks to months) → no immediate protection against rebleed
- You need the aneurysm sealed now
Reserve PED for unruptured or chronic/remote-SAH cases.
What About Stent-Assisted Coiling in SAH?
Relatively contraindicated acutely — stents require DAPT loading, which conflicts with the hemorrhagic state. It's used only as a last resort when the aneurysm is otherwise untreatable, accepting the bleeding risk.
Post-Securing the Aneurysm: The SAH Isn't Over
Once the aneurysm is secured, the fight shifts to vasospasm and delayed cerebral ischemia (DCI):
- Peaks at days 4–14 after SAH
- Treat with nimodipine (60 mg q4h orally × 21 days — reduces DCI, not vasospasm itself)
- Induced hypertension (permissible only after aneurysm is secured)
- Monitor with TCD, CT perfusion, or DSA
- Refractory vasospasm → intra-arterial vasodilators (verapamil, nicardipine) or balloon angioplasty
Bottom line for SAH: Coil if you can, clip if you must (hematoma, wide-neck MCA, young patient), avoid stents and flow diversion acutely, and get it done within 24 hours.
This is a shared conversation. Sign in to Orris to start your own chat.