Biochemistry of renal system/urinary
Reading File
search_medical_text
renal biochemistry glomerular filtration tubular reabsorption secretion
search_medical_text
kidney acid-base regulation bicarbonate ammoniagenesis hydrogen ion excretion
search_medical_text
renin angiotensin aldosterone system RAAS biochemistry kidney
search_medical_text
renal handling of glucose amino acids proteins urine composition biochemistry
search_medical_image
nephron biochemistry renal tubule transport diagram
GradeResponse
GradeResponse
GradeResponse
GradeResponse
GradeResponse
search_medical_text
ADH vasopressin aquaporin water reabsorption collecting duct urine concentration
search_medical_text
erythropoietin vitamin D activation 1-alpha hydroxylase kidney endocrine function
search_medical_text
GFR measurement creatinine urea BUN clearance renal function biochemical markers
GradeResponse
Here is a comprehensive overview of the Biochemistry of the Renal/Urinary System:
Biochemistry of the Renal/Urinary System
1. Glomerular Filtration
The nephron begins with ultrafiltration at the glomerulus. The glomerular filtration barrier consists of:
- Fenestrated capillary endothelium
- Glomerular basement membrane (type IV collagen, laminin, heparan sulfate)
- Podocytes with filtration slits
Starling forces govern filtration:
Net Filtration Pressure = (Pcap − Pbow) − (πcap − πbow)
- Normal GFR ≈ 125 mL/min (~180 L/day of ultrafiltrate)
- The filtrate is essentially plasma minus large proteins
Biochemical markers of GFR:
| Marker | Notes |
|---|---|
| Creatinine | Freely filtered; small tubular secretion; varies with muscle mass |
| Urea (BUN) | Freely filtered; ~50% reabsorbed; influenced by protein intake & hydration |
| Inulin | Gold-standard exogenous marker; freely filtered, neither secreted nor reabsorbed |
| Cystatin C | Less influenced by muscle mass; rises earlier in AKI |
2. Proximal Convoluted Tubule (PCT) — Bulk Reabsorption
The PCT reabsorbs ~65–70% of filtered water, Na⁺, and virtually all filtered glucose and amino acids.
Key Transporters
| Substance | Transporter | Mechanism |
|---|---|---|
| Glucose | SGLT-2 (S1 segment), SGLT-1 (S3) | Na⁺-coupled cotransport (secondary active) |
| Amino acids | Various Na⁺-coupled transporters | Secondary active |
| Na⁺ | NHE3 (Na⁺/H⁺ exchanger) | Antiport |
| HCO₃⁻ | NBC1 (Na⁺/HCO₃⁻ cotransporter) | Basolateral exit |
| Phosphate | NaPi-IIa, NaPi-IIc | Na⁺-coupled; inhibited by PTH |
| Water | AQP1 | Osmotic, follows Na⁺ |
Renal glucose threshold: ~180 mg/dL (10 mmol/L). Below this, all filtered glucose is reabsorbed. Above it, glucosuria occurs. Urine glucose does not reliably distinguish euglycemia from hypoglycemia (Laboratory Analysis in DM, p. 22).
Organic acids and uric acid are both secreted and reabsorbed in the PCT. Hyperuricemia in renal insufficiency results from decreased tubular secretion plus reduced GFR (Harrison's, p. 11836).
3. Loop of Henle — Concentration Gradient
The loop generates the medullary hypertonicity essential for concentrating urine via the countercurrent multiplier.
Transport at each segment
| Segment | Permeability | Transporter | Function |
|---|---|---|---|
| Thin descending limb | H₂O permeable (AQP1), impermeable to solute | — | Water moves out by osmosis |
| Thin ascending limb | Impermeable to water | Passive NaCl diffusion | NaCl leaves → dilutes filtrate |
| Thick ascending limb (TAL) | Impermeable to water | NKCC2 (Na⁺-K⁺-2Cl⁻) | Actively pumps NaCl out; dilutes filtrate |
NKCC2 in the TAL is the target of loop diuretics (furosemide).
ROMK channels recycle K⁺ to the lumen, essential for NKCC2 function. CLC-K1 channels on the basolateral side facilitate Cl⁻ exit. (Harrison's, p. 1386)
Urea recycling (UT-A1, UT-A2, UT-A3 in collecting duct; UT-B in vasa recta) contributes ~400–500 mOsm to medullary interstitium.
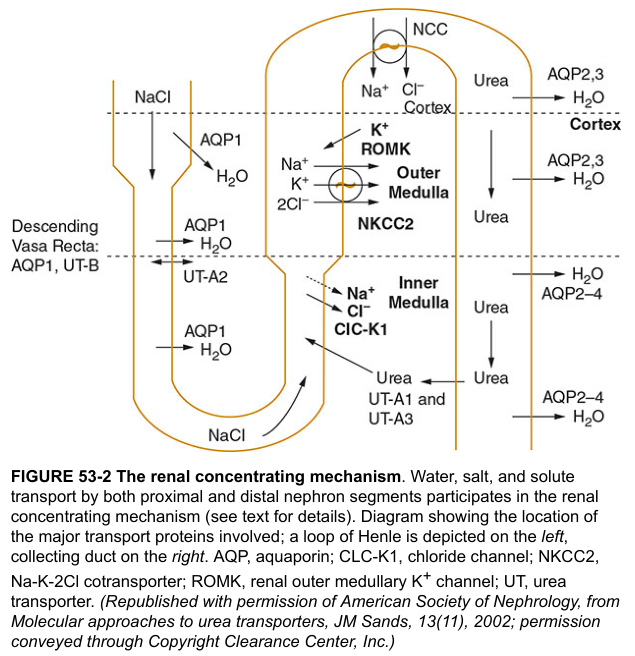
4. Distal Convoluted Tubule (DCT)
- NCC (Na⁺-Cl⁻ cotransporter) is the dominant transporter; target of thiazide diuretics
- Ca²⁺ reabsorption: active, regulated by PTH and vitamin D (via TRPV5 channels)
- Mg²⁺ reabsorption occurs primarily in the TAL and DCT
- Fine-tuning of K⁺ begins here under aldosterone influence
5. Collecting Duct — Final Urine Composition
The collecting duct makes final adjustments to urine concentration and composition (Harrison's, p. 8426):
Vasopressin (ADH) Signaling — Water Reabsorption
- ADH binds V2 receptor (basolateral) on principal cells
- G-protein → adenylyl cyclase → ↑cAMP → PKA activation
- PKA phosphorylates AQP2 vesicles → insertion into apical membrane
- Water reabsorbs via AQP2 (apical), exits via AQP3/AQP4 (basolateral)
- Absence of ADH → water-impermeable → dilute urine (diabetes insipidus if ADH absent)
Aldosterone — Na⁺ Reabsorption / K⁺ Secretion
- Binds mineralocorticoid receptor → ↑ transcription of ENaC and Na⁺/K⁺-ATPase
- Net: Na⁺ (and water) retained; K⁺ secreted via ROMK
H⁺ Secretion — Intercalated Cells
- α-intercalated cells: apical H⁺-ATPase and H⁺/K⁺-ATPase secrete H⁺ (urine acidification)
- β-intercalated cells: secrete HCO₃⁻ (alkaline states)
6. Renal Acid-Base Regulation
The kidney is the only organ that regenerates HCO₃⁻ and excretes net acid. (Harrison's, p. 8421)
Mechanisms of Acid Excretion
A. Bicarbonate reclamation (proximal tubule):
- Filtered HCO₃⁻ is not directly reabsorbed; instead:
- H⁺ secreted via NHE3 combines with luminal HCO₃⁻ → H₂CO₃ → CO₂ + H₂O (catalyzed by carbonic anhydrase IV, luminal)
- CO₂ diffuses into cell → reformed to H₂CO₃ by CA II → HCO₃⁻ exits basolaterally via NBC1
B. Titratable acid excretion:
- Secreted H⁺ + HPO₄²⁻ → H₂PO₄⁻ (excreted)
- Phosphate is the major urinary buffer
C. Ammoniagenesis:
- Glutamine is metabolized in PCT mitochondria → α-ketoglutarate + 2 NH₄⁺
- NH₃ diffuses into lumen; trapped as NH₄⁺ by secreted H⁺
- Hypokalemia → ↑ ammoniagenesis; Hyperkalemia/hypoaldosteronism → ↓ ammoniagenesis → Type IV RTA
Summary of Acid Secretion
| Site | Mechanism | Buffer |
|---|---|---|
| PCT | NHE3; CA II and CA IV | Bicarbonate, phosphate, ammonia |
| TAL | Some H⁺ secretion | — |
| Collecting duct (α-IC) | H⁺-ATPase; H⁺/K⁺-ATPase | Ammonia, titratable acid |
7. Renin-Angiotensin-Aldosterone System (RAAS)
The RAAS functions as both a hormonal and paracrine system (Harrison's, p. 1154):
↓ Renal perfusion pressure / ↓ NaCl at macula densa / ↑ Sympathetic tone
↓
Juxtaglomerular (JG) cells → release RENIN
↓
Angiotensinogen (liver) → Angiotensin I (10 aa)
↓ [ACE — lung, endothelium]
Angiotensin II (8 aa)
↓
├── AT1 receptor → Vasoconstriction, Na⁺ retention, aldosterone release, ADH release
└── AT2 receptor → Vasodilation, antiproliferative
↓
Adrenal cortex (zona glomerulosa) → ALDOSTERONE
↓
ENaC ↑, Na⁺/K⁺-ATPase ↑ → Na⁺ and water retained, K⁺ excreted
Angiotensin II also directly stimulates NHE3 in the PCT, increasing Na⁺ and HCO₃⁻ reabsorption.
8. Endocrine Functions of the Kidney
A. Erythropoietin (EPO)
- Produced by peritubular interstitial cells (inner cortex/outer medulla)
- Stimulated by hypoxia via HIF-1α/HIF-2α transcription factors
- Stimulates erythropoiesis in bone marrow
- CKD → fibrosis of interstitium → ↓ EPO → normochromic normocytic anemia
B. Vitamin D Activation
| Step | Location | Enzyme |
|---|---|---|
| Cholecalciferol (D3) | Skin (UV) / diet | — |
| 25-hydroxyvitamin D [25(OH)D] | Liver | 25-hydroxylase (CYP2R1) |
| 1,25(OH)₂D (Calcitriol — active) | Kidney (PCT) | 1α-hydroxylase (CYP27B1) — stimulated by PTH, ↓ PO₄, ↓ Ca²⁺ |
CKD → loss of 1α-hydroxylase → ↓ calcitriol → ↓ Ca²⁺ absorption → secondary hyperparathyroidism → renal osteodystrophy
C. Prostaglandins and Kinins
- Renal medulla produces PGE₂ and PGI₂ → vasodilatory, natriuretic
- Kallikrein-kinin system in the kidney also modulates Na⁺ excretion and vasodilation
- NSAIDs block prostaglandin synthesis → ↓ GFR (especially in states of renal hypoperfusion)
9. Potassium Homeostasis
- ~90% of filtered K⁺ reabsorbed by PCT (~65%) and TAL (~25%)
- Collecting duct principal cells are the main site of K⁺ secretion (regulated)
- Aldosterone → ↑ apical ROMK → ↑ K⁺ secretion
- Alkalosis → K⁺ shifts intracellularly → ↑ renal K⁺ secretion
- Hyperkalemia → depolarizes cells → directly stimulates aldosterone release
10. Calcium and Phosphate Handling
| Mineral | PCT | TAL | DCT |
|---|---|---|---|
| Ca²⁺ | ~65% paracellular | ~25%, PTH-sensitive | ~10%, active, TRPV5; PTH/Vit D stimulate |
| PO₄³⁻ | ~85% via NaPi-IIa/IIc; PTH inhibits | Minimal | Minimal |
FGF-23 (from osteocytes): inhibits NaPi-IIa → phosphaturia; also inhibits 1α-hydroxylase → ↓ calcitriol. Elevated in CKD.
11. Urine Composition — Normal Biochemistry
| Component | Normal range | Significance |
|---|---|---|
| Na⁺ | 40–220 mEq/day | Reflects dietary intake + RAAS |
| K⁺ | 25–125 mEq/day | Reflects aldosterone, dietary K⁺ |
| Creatinine | 0.6–1.8 g/day | Constant (muscle mass dependent) |
| Urea nitrogen | 6–17 g/day | Reflects protein catabolism |
| Glucose | 0 (trace) | Glucosuria if serum > 180 mg/dL |
| Protein | <150 mg/day | Microalbuminuria = 30–300 mg/day |
| pH | 4.5–8.0 | Reflects acid-base status |
| Osmolality | 50–1200 mOsm/kg | Reflects ADH, hydration |
Key Integrative Points
- GFR is the central determinant of all renal biochemical functions
- The proximal tubule handles bulk reabsorption (Na⁺, glucose, amino acids, HCO₃⁻, phosphate)
- The loop of Henle builds the osmotic gradient; the collecting duct exploits it under ADH
- Aldosterone (via RAAS) and ADH are the two dominant hormonal axes for final urine composition
- The kidney is an endocrine organ: EPO, calcitriol, prostaglandins, renin
- Acid-base balance is maintained through HCO₃⁻ reclamation, titratable acid, and ammoniagenesis
This is a shared conversation. Sign in to Orris to start your own chat.