Symptoms of aortic dissection
aortic dissection symptoms clinical presentation
aortic dissection classification types
Aortic Dissection: Symptoms & Clinical Presentation
Cardinal Symptom: Pain
- Tearing or ripping in quality
- Abrupt onset — maximal intensity at the very start (unlike MI, which builds gradually)
- Associated with diaphoresis
- Migratory — moves as the dissection propagates along the aorta
| Location of Pain | Likely Segment Involved |
|---|---|
| Anterior chest | Ascending aorta (Type A) |
| Interscapular / back | Descending aorta (Type B) |
| Abdomen / flank | Extension into abdominal aorta |
Other Symptoms
- Syncope — due to pain, cardiac tamponade, or cerebral hypoperfusion
- Dyspnea — pulmonary edema from acute aortic regurgitation
- Weakness / neurologic deficits — from carotid or spinal artery involvement
Physical Examination Findings
| Finding | Mechanism |
|---|---|
| Hypertension | Most common; especially in Type B |
| Hypotension / shock | Cardiac tamponade, aortic rupture |
| Pulse deficit (asymmetric pulses) | Branch vessel occlusion |
| Aortic regurgitation murmur | Involvement of aortic root (Type A) |
| Pulmonary edema | Acute AR leading to LV volume overload |
| Hemiplegia / hemianesthesia | Carotid artery obstruction |
| Paraplegia | Spinal cord ischemia (intercostal/spinal artery occlusion) |
| Bowel ischemia | Mesenteric artery occlusion |
| Hematuria | Renal artery involvement |
| Myocardial ischemia | Coronary ostium involvement (RCA > LCA) |
Classification (DeBakey & Stanford)
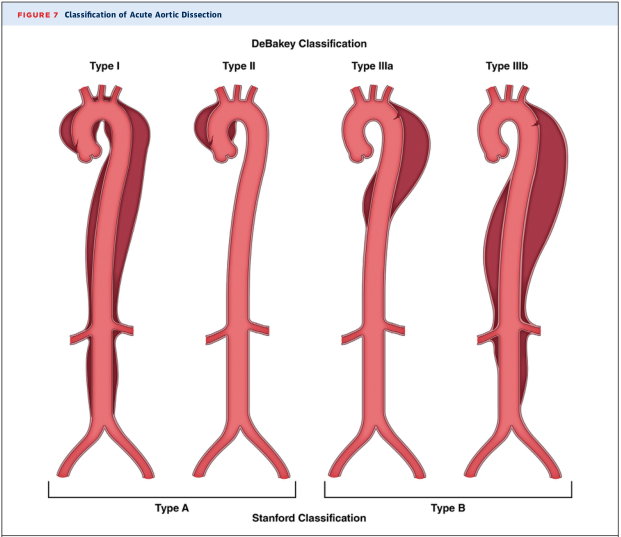
- Stanford Type A (DeBakey I & II) — involves ascending aorta → higher risk of AR, tamponade, coronary/carotid occlusion, stroke
- Stanford Type B (DeBakey IIIa & IIIb) — descending aorta only → back/abdominal pain, visceral/renal/limb ischemia
Epidemiology Note
- Peak incidence: 6th–7th decade
- Male predominance (2:1 ratio)
- Classic risk factors include hypertension, connective tissue disorders (Marfan, Ehlers-Danlos), bicuspid aortic valve, and prior aortic surgery
Key clinical pearl: A patient with sudden severe tearing chest/back pain + pulse deficit + hypertension should be treated as aortic dissection until proven otherwise. CT angiography of the chest/abdomen/pelvis is the diagnostic standard.
Any ecg holter ecg changes?
aortic dissection ECG electrocardiogram changes findings
aortic dissection ECG findings electrocardiogram
ECG Changes in Aortic Dissection
Typical ECG Findings
| Finding | Frequency | Mechanism |
|---|---|---|
| Normal ECG | ~30–40% of cases | No coronary/pericardial involvement |
| Non-specific ST/T changes | Most common abnormality | Pressure, stress response |
| LVH pattern | Common | Underlying hypertension (major risk factor) |
| ST elevation (inferior leads II, III, aVF) | ~1–2% | RCA ostium involvement by dissection flap |
| ST depression + diffuse ischemic changes | Present in coronary involvement | LMCA or other coronary ostium compression |
| Electrical alternans / low voltage | If pericardial effusion/tamponade present | Hemopericardium compressing the heart |
| Sinus tachycardia | Common | Pain, hemodynamic stress |
The Critical Trap: Dissection Mimicking STEMI
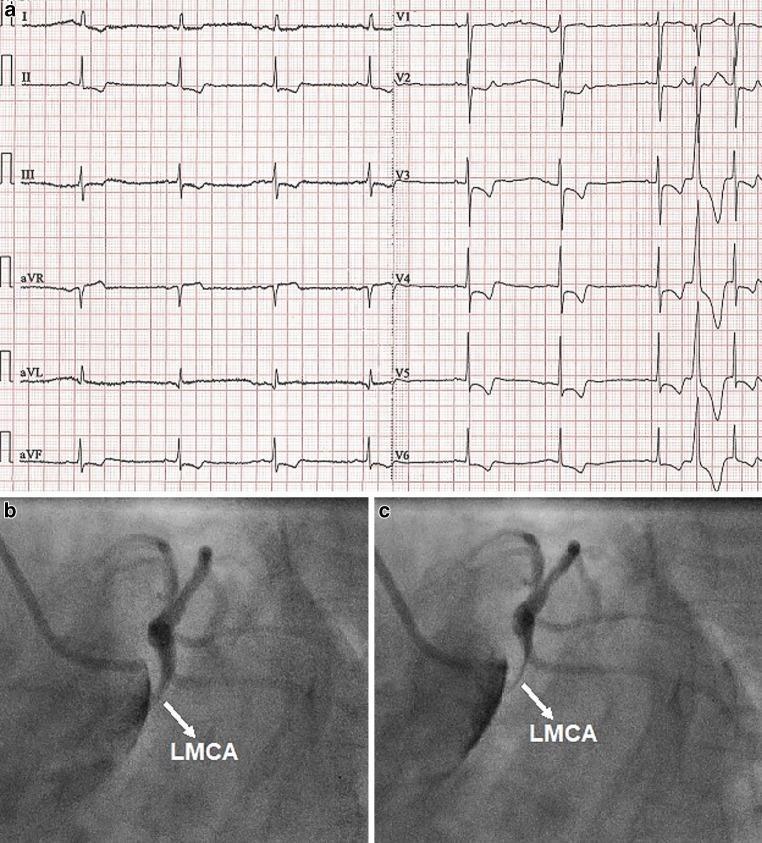
- (a) 12-lead ECG with widespread ST depression + negative T waves in inferior and precordial leads (V2–V6), with ST elevation in aVR — indicating global subendocardial ischemia from LMCA compromise
- (b & c) Coronary angiography showing dynamic compression of the LMCA by a false lumen — the lumen becomes slit-like during diastole
This is a fatal diagnostic trap: giving thrombolytics or proceeding to PCI without recognizing the underlying dissection can cause aortic rupture and death.
Holter ECG Relevance
- Aortic dissection is an acute, time-critical emergency — diagnosis relies on CT angiography, not prolonged rhythm monitoring
- Holter is designed for paroxysmal arrhythmia detection over 24–48 hours, not acute structural vascular catastrophe
- However, post-dissection, Holter may be used if the patient develops arrhythmias (e.g., from myocardial ischemia, pericarditis, or surgical repair sequelae)
Key Clinical Pearl
An ECG showing inferior ST elevation in a patient with tearing back pain should raise immediate suspicion for Type A dissection with RCA involvement — not primary ACS. The combination of pain character + pulse differential + CXR (widened mediastinum) should guide toward CT angiography before any reperfusion therapy.
31-year-old active male presented with tearing, left-sided chest pain, lasting for 1 to 2 seconds, accompanied with lightheadedness. When he checked his BP, it was 152 by 82 millimeters of mercury, and there was tachycardia at 110 beats per minute. The episode lasted for 15 minutes. The pain was there only for 1 to 2 seconds. Occasionally, he also experiences ectopic beats, that is a missed beat feeling. So cardiologist ordered Holter ECG, which showed less than 0.1% ectopic burden and no other abnormalities. Six months back, his echo was normal. Cardiac stress test up to 186 beats per minute was normal.
young male tearing chest pain brief seconds lightheadedness tachycardia differential diagnosis
musculoskeletal chest pain vs cardiac causes young adults pleuritic costochondritis
Why This Is Almost Certainly NOT Aortic Dissection
| Feature | This Patient | Typical Aortic Dissection |
|---|---|---|
| Age | 31, active male | Peak: 60s–70s |
| Pain duration | 1–2 seconds | Sustained, minutes to hours |
| Pain character | Tearing, left-sided | Tearing/ripping, but persistent and migratory |
| Episode duration | 15 minutes total | Does not self-resolve spontaneously |
| Echo (6 months ago) | Normal | Often shows aortic root dilation, AR |
| Stress test to 186 bpm | Normal | Would not be safely completed |
| Holter | Benign (<0.1% ectopics) | Not relevant to dissection |
More Likely Diagnoses
1. Musculoskeletal / Costochondral Pain (Most likely)
- Sharp, fleeting, left-sided chest pain lasting seconds is a classic descriptor
- Can be positional, worse with movement, deep breath, or palpation
- Aggravated by exercise in an active individual
- (Harrison's, p. 10483): Costochondritis/Tietze syndrome frequently mimics cardiac pain
2. Benign Precordial Catch Syndrome (Texidor's Twinge)
- Extremely common in young, athletic adults
- Sharp, stabbing left-sided chest pain lasting 1–3 seconds, often at rest or with position change
- Completely benign, no structural correlate
- Self-limiting; no treatment needed
3. Vasovagal / Autonomic Response
- The tachycardia + lightheadedness + brief episode pattern fits an autonomic surge (e.g., anxiety, exertion-triggered adrenergic response)
- BP of 152/82 in a young active male during a symptomatic episode suggests sympathetic activation, not hemodynamic compromise
4. Ectopic Beats (PACs/PVCs)
- The "missed beat" sensation he reports is confirmed by Holter
- PVCs/PACs can cause a brief sharp chest sensation and lightheadedness due to the post-ectopic pause and compensatory beat
- <0.1% burden is entirely benign — well below the 10–24% threshold associated with cardiomyopathy risk
5. Pleuritis / Pleural irritation
- Sharp, fleeting, left-sided pain can reflect parietal pleural irritation
- Especially relevant in an active individual post-exertion
What the Workup Tells Us
| Test | Result | Interpretation |
|---|---|---|
| Echo (6 months ago) | Normal | No structural heart disease, normal aortic root |
| Stress test (186 bpm) | Normal | No inducible ischemia, no arrhythmia under maximal exertion |
| Holter ECG | <0.1% ectopics, no other abnormality | Benign; no significant arrhythmia, no SVT, no pauses |
- Obstructive coronary artery disease
- Hypertrophic cardiomyopathy
- Significant arrhythmia
- Structural aortic pathology
Suggested Next Steps (if symptoms persist)
- Chest wall palpation — reproduce the pain? → confirms musculoskeletal
- Review medication/stimulant use — caffeine, pre-workout supplements are very common triggers of tachycardia + ectopics in young active males
- Ambulatory BP monitoring — to assess whether the BP elevation is episodic/anxiety-driven
- Reassurance — the most important intervention; benign precordial catch and musculoskeletal pain in young active men are extremely common and carry no adverse prognosis
- Repeat echo only if symptoms change in character, frequency, or severity
Bottom line: This presentation is highly reassuring. The pain characteristics (1–2 seconds, self-limiting) combined with a thoroughly normal cardiac workup point strongly toward a benign musculoskeletal or autonomic etiology — not aortic dissection or any serious cardiac condition.