Elaborate essay on MAST cell stabilizers , Flow charts and diagrams
Reading File
search_medical_text
mast cell stabilizers mechanism of action pharmacology cromolyn nedocromil
search_medical_text
mast cell stabilizers clinical uses asthma allergic rhinitis conjunctivitis indications contraindications
search_medical_image
mast cell degranulation mechanism allergy pathway diagram
GradeResponse
GradeResponse
GradeResponse
search_medical_text
mast cell IgE degranulation histamine serotonin leukotrienes prostaglandins mediators allergic response
search_medical_image
mast cell stabilizer cromolyn sodium asthma pharmacology
GradeResponse
Now I'll compose a comprehensive, well-structured essay with flow charts rendered in text/markdown format, integrating the retrieved content with my own pharmacological knowledge.
Mast Cell Stabilizers: A Comprehensive Essay
1. Introduction
Mast cell stabilizers (MCS) constitute a unique class of anti-inflammatory drugs whose primary action is to prevent the activation and degranulation of mast cells, thereby blocking the release of preformed and newly synthesized mediators of the allergic and inflammatory cascade. Unlike bronchodilators, corticosteroids, or antihistamines, mast cell stabilizers act upstream — they prevent the allergic response from being initiated at the cellular level rather than antagonizing its downstream effects.
The two prototypical agents are:
- Cromolyn sodium (sodium cromoglycate) — a chromone derivative introduced in the 1960s by Roger Altounyan
- Nedocromil sodium — a pyranoquinoline dicarboxylic acid introduced in the 1980s
Although structurally unrelated, these compounds share broadly similar profiles of bronchoprotective and anti-allergic activity (Exercise-induced Bronchoconstriction, p. 23).
2. The Mast Cell: A Brief Overview
Mast cells are tissue-resident immune cells derived from CD34⁺ hematopoietic progenitors. They are particularly abundant at host-environment interfaces:
| Location | Subtype |
|---|---|
| Lung mucosa | MCT (tryptase only) |
| Skin, GI submucosa | MCTC (tryptase + chymase) |
| Conjunctiva, nasal mucosa | MCT predominant |
| Peritoneum | MCTC predominant |
Their surfaces are densely coated with high-affinity IgE receptors (FcεRI), enabling them to be sensitized by antigen-specific IgE antibodies.
3. Mast Cell Activation and Degranulation
Understanding mast cell stabilizers requires a firm grasp of the underlying degranulation pathway.
FLOW CHART 1: IgE-Mediated Mast Cell Degranulation
┌─────────────────────────────────────────────────────────────────┐
│ SENSITIZATION PHASE (First Exposure) │
│ │
│ Allergen → B Cell Activation → IgE Production │
│ ↓ │
│ IgE binds to FcεRI on Mast Cell Surface │
│ (Mast cell is now sensitized — no symptoms yet) │
└─────────────────────────────────────────────────────────────────┘
│
▼
┌─────────────────────────────────────────────────────────────────┐
│ ACTIVATION PHASE (Re-exposure) │
│ │
│ Allergen Re-enters → Crosslinks Two Adjacent IgE–FcεRI │
│ Complexes on Mast Cell │
│ ↓ │
│ FcεRI Aggregation / Receptor Clustering │
│ ↓ │
│ Activation of Src-family kinases (Lyn, Fyn) │
│ ↓ │
│ Phosphorylation of ITAM motifs on FcεRIβ/γ │
│ ↓ │
│ Recruitment & activation of Syk kinase │
│ ↓ │
│ ┌───────────────────┴──────────────────────┐ │
│ ▼ ▼ │
│ PI3K pathway LAT scaffold │
│ (PIP2 → PIP3) (PLCγ activation) │
│ ↓ ↓ │
│ PKC, Akt activation IP3 → Ca²⁺ release from ER │
│ ↓ DAG → PKC activation │
│ └──────────────────┬────────────────────────┘ │
│ ↓ │
│ ELEVATED INTRACELLULAR Ca²⁺ │
│ ↓ │
│ ┌──────────────────┼──────────────────────┐ │
│ ▼ ▼ ▼ │
│ Degranulation Arachidonic Acid Gene Transcription │
│ (Preformed Metabolism (NF-κB, NFAT) │
│ mediators) (New mediators) (Cytokines) │
└─────────────────────────────────────────────────────────────────┘
FLOW CHART 2: Mediators Released During Mast Cell Degranulation
MAST CELL DEGRANULATION
│
┌──────────────────┼─────────────────────┐
▼ ▼ ▼
PREFORMED MEDIATORS LIPID MEDIATORS CYTOKINES/CHEMOKINES
(Released in seconds) (Minutes) (Hours)
│ │ │
┌──────┴──────┐ ┌──────┴──────┐ ┌─────────┴──────────┐
│ Histamine │ │Prostaglandin│ │ IL-4, IL-5, IL-13 │
│ Tryptase │ │ D2 (PGD2) │ │ TNF-α │
│ Chymase │ │LTC4, LTD4, │ │ IL-8, MCP-1 │
│ Heparin │ │LTE4 (CysLTs)│ │ GM-CSF │
│ Serotonin │ │PAF │ └────────────────────┘
└──────┬──────┘ └──────┬──────┘
│ │
▼ ▼
Vasodilation, Bronchoconstriction,
Itch, urticaria, Mucus hypersecretion,
bronchospasm, Eosinophil recruitment,
angioedema Late-phase inflammation
4. Mechanism of Action of Mast Cell Stabilizers
4.1 Molecular and Cellular Mechanisms
Mast cell stabilizers exert their effects through multiple complementary mechanisms:
A. Chloride Channel Blockade
The primary mechanism: MCS block a specific type of voltage-gated chloride channel on the mast cell membrane. Chloride ion influx is an early and essential step in the signal transduction cascade leading to degranulation. By blocking this channel, MCS prevent the downstream rise in intracellular calcium and halt degranulation.
B. Inhibition of Calcium Influx
Even beyond chloride channels, MCS reduce the net rise in intracellular Ca²⁺ from both intracellular stores (ER) and transmembrane influx, thereby preventing the calcium-dependent exocytosis of granules.
C. Inhibition of Mediator Release
Specifically, both cromolyn and nedocromil inhibit the release of prostaglandin D2 (PGD2), a potent bronchoconstrictor, along with histamine, tryptase, and leukotrienes (Exercise-induced Bronchoconstriction, p. 23).
D. Inhibition of Sensory Nerve Activation
Nedocromil, in particular, suppresses the activation of sensory (C-fiber) nerves in the airways, reducing neurogenic inflammation, cough, and reflex bronchoconstriction — an effect distinct from pure mast cell stabilization.
E. Inhibition of Eosinophil Activation
Both agents reduce eosinophil recruitment and activation in late-phase allergic responses, partly by suppressing mast cell–derived cytokines (IL-5, GM-CSF) that recruit and sustain eosinophils.
F. Inhibition of Non-IgE-Mediated Degranulation
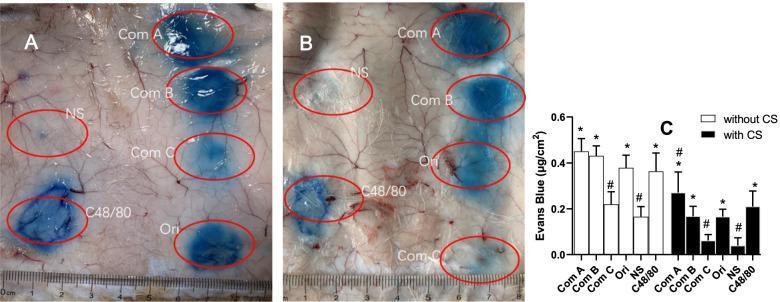
Cromolyn sodium also blocks non-immunological (anaphylactoid) mast cell activation — for example, degranulation triggered by compound 48/80 or certain drugs — as demonstrated in guinea pig PCA models where cromolyn pretreatment visibly attenuated Evans Blue dye leakage from vascular permeability responses (PMC Clinical VQA).
FLOW CHART 3: Where Mast Cell Stabilizers Intervene
Allergen + IgE–FcεRI Crosslinking
│
▼
FcεRI Aggregation
│
▼
Src kinase / Syk activation
│
▼
┌─────────────────────────────┐
│ CHLORIDE CHANNEL OPENS │◄──── ✦ MCS BLOCK HERE
└─────────────────────────────┘
│
▼
Intracellular Ca²⁺ Rise ◄──────── ✦ MCS ALSO REDUCE Ca²⁺
│
▼
Granule Fusion & Exocytosis ◄─── ✦ MCS PREVENT THIS STEP
│
▼
Mediator Release (Histamine,
PGD2, Leukotrienes, etc.) ◄──────── ✦ MCS INHIBIT RELEASE
RESULT: ✦ No/Minimal Allergic Response
5. Individual Agents
5.1 Cromolyn Sodium (Sodium Cromoglycate)
| Property | Detail |
|---|---|
| Chemical class | Chromone dicarboxylic acid |
| Route of administration | Inhaled (MDI, nebulizer), Intranasal, Ophthalmic, Oral |
| Onset of action | No acute bronchodilation; requires 2–4 weeks for full prophylactic effect |
| Duration of bronchoprotection | 1–2 hours per dose (Exercise-induced Bronchoconstriction, p. 23) |
| Bioavailability (oral) | < 1% — almost entirely unabsorbed |
| Metabolism | Not metabolized; excreted unchanged |
| Half-life | ~80 minutes |
| Safety | Extremely safe; among the safest drugs in medicine |
5.2 Nedocromil Sodium
| Property | Detail |
|---|---|
| Chemical class | Pyranoquinoline dicarboxylic acid |
| Route of administration | Inhaled, Ophthalmic |
| Potency vs Cromolyn | 4–10× more potent on a molar basis |
| Additional effects | Inhibits sensory nerve activation (C-fibers) |
| Side effects | Unpleasant bitter taste (major complaint), headache |
| Bioavailability | ~2–3% systemic absorption when inhaled |
5.3 Other Agents with Mast Cell Stabilizing Properties
Several drugs, while primarily classified differently, exert significant mast cell stabilization as part of their mechanism:
| Drug | Primary Class | MCS Relevance |
|---|---|---|
| Ketotifen | Antihistamine | Strong MCS + H1 antagonist; used in asthma, conjunctivitis |
| Olopatadine | Antihistamine | H1 blocker + MCS; commonly used topically for allergic conjunctivitis (Harrison's, p. 947) |
| Lodoxamide | Pure MCS | More potent than cromolyn for ocular use |
| Pemirolast | MCS | Ophthalmic use in allergic conjunctivitis |
| Amlexanox | MCS/anti-inflammatory | Oral aphthous ulcers |
6. Clinical Indications
6.1 Bronchial Asthma
MCS are used exclusively as prophylactic/controller agents — never for acute bronchospasm relief.
Specific roles include:
- Mild persistent asthma: As a second-line controller alternative to low-dose inhaled corticosteroids (ICS), especially in children where steroid side effects are a concern
- Exercise-induced bronchoconstriction (EIB): Cromolyn/nedocromil, taken 10–15 minutes before exercise, provide rapid-onset bronchoprotection lasting 1–2 hours. They can be used alone or in combination with other EIB-preventive drugs for enhanced protection (Exercise-induced Bronchoconstriction, p. 23)
- Allergen-induced asthma: Pre-treatment before unavoidable allergen exposure
- Occupational asthma: Prophylaxis when allergen avoidance is impossible
6.2 Allergic Rhinitis
Intranasal cromolyn sodium 2–4% solution:
- Prevents seasonal and perennial allergic rhinitis
- Most effective when started 1–2 weeks before the allergy season
- Requires dosing 4–6 times daily — a major adherence challenge
- Safety makes it the preferred agent in pregnancy and infants
6.3 Allergic Conjunctivitis
Topical cromolyn, lodoxamide, pemirolast, and ketotifen/olopatadine (dual-action) are standard treatments. According to Harrison's Principles of Internal Medicine (p. 947), "symptoms caused by allergic conjunctivitis can be alleviated with cold compresses, topical vasoconstrictors, antihistamines (olopatadine), and mast cell stabilizers (cromolyn)." They address:
- Itching, redness, tearing (epiphora)
- Cobblestone papillae (giant papillary conjunctivitis)
- Atopic conjunctivitis in patients with atopic dermatitis or asthma
6.4 Mastocytosis and Mast Cell Activation Syndrome (MCAS)
Oral cromolyn sodium is a cornerstone of therapy for:
- Systemic mastocytosis (GI symptoms: diarrhea, cramping, nausea)
- Mast cell activation syndrome
- Food-induced allergic enteropathy
- Mechanism: Even though oral bioavailability is < 1%, local luminal effects on intestinal mast cells suffice
6.5 Food Allergy
Oral cromolyn is used as prophylaxis before exposure to food allergens (e.g., in patients with mastocytosis or recurrent GI allergy).
6.6 Vernal/Atopic Keratoconjunctivitis
Lodoxamide (0.1% ophthalmic) is considered superior to cromolyn for severe forms of vernal keratoconjunctivitis.
7. Pharmacokinetics: Comparative Summary
ROUTE → ABSORPTION → DISTRIBUTION → METABOLISM → EXCRETION
│
├─ INHALED CROMOLYN
│ ~10% reaches airways
│ ~1% systemic absorption
│ No hepatic metabolism
│ Excreted unchanged in urine/bile
│
├─ INHALED NEDOCROMIL
│ ~2–3% systemic
│ Protein binding ~89%
│ Excreted unchanged
│
├─ INTRANASAL CROMOLYN
│ Local action; minimal systemic absorption
│
└─ ORAL CROMOLYN
<1% absorbed
Acts locally on gut mast cells
No significant systemic effect
8. Adverse Effects and Safety Profile
Mast cell stabilizers are among the safest pharmacological agents available.
| Adverse Effect | Drug | Frequency |
|---|---|---|
| Throat irritation, cough | Inhaled cromolyn | Common (device-related) |
| Bronchospasm (paradoxical) | Inhaled cromolyn | Rare; use bronchodilator pre-treatment |
| Bitter taste | Nedocromil | Very common (~20%) |
| Headache | Nedocromil | Occasional |
| Local burning/stinging | Ophthalmic preparations | Mild, transient |
| Nasal stinging | Intranasal cromolyn | Mild |
| Nausea, abdominal pain | Oral cromolyn | Occasional |
| Systemic side effects | Both | Virtually absent |
No significant drug interactions have been reported with either agent.
Pregnancy Category B: Both cromolyn and nedocromil are considered safe in pregnancy. Intranasal cromolyn is the preferred first-line agent for allergic rhinitis in pregnant patients.
9. Comparison with Other Anti-Allergic Drug Classes
┌────────────────┬──────────────┬──────────────┬──────────────┬──────────────┐
│ Property │ Mast Cell │ Inhaled │Antihistamines│Leukotriene │
│ │ Stabilizers │Corticosteroids│(H1 blockers) │ Modifiers │
├────────────────┼──────────────┼──────────────┼──────────────┼──────────────┤
│ Mechanism │Prevent mast │Broad anti- │Block H1 │Block/Inhibit │
│ │cell release │inflammatory │receptor │CysLT receptor│
├────────────────┼──────────────┼──────────────┼──────────────┼──────────────┤
│ Acute Relief │ No │ No │ Yes │ Partial │
├────────────────┼──────────────┼──────────────┼──────────────┼──────────────┤
│ Prophylaxis │ Yes ✓✓ │ Yes ✓✓✓ │ Yes ✓ │ Yes ✓✓ │
├────────────────┼──────────────┼──────────────┼──────────────┼──────────────┤
│ Onset (days) │ 7–14 days │ 2–3 days │ Hours │ 1–3 days │
├────────────────┼──────────────┼──────────────┼──────────────┼──────────────┤
│ Safety │ Excellent │ Good (local) │ Good │ Good │
├────────────────┼──────────────┼──────────────┼──────────────┼──────────────┤
│ Steroid-sparing│ Yes │ N/A │ Partial │ Yes │
├────────────────┼──────────────┼──────────────┼──────────────┼──────────────┤
│ 1st Line? │ 2nd line │ 1st line │ 1st line │ 2nd line │
│ (Asthma) │ │ │ (rhinitis) │ │
└────────────────┴──────────────┴──────────────┴──────────────┴──────────────┘
10. Clinical Decision Algorithm: When to Use Mast Cell Stabilizers
PATIENT PRESENTS WITH ALLERGIC/INFLAMMATORY SYMPTOMS
│
┌─────────────┴──────────────┐
▼ ▼
ACUTE EPISODE? CHRONIC/PROPHYLAXIS?
│ │
┌──────┴──────┐ ▼
│ NOT FOR MCS │ What is the clinical condition?
│ Use: SABAs, │ │
│antihistamines│ ┌────────────────┼─────────────────┐
└─────────────┘ ▼ ▼ ▼
ASTHMA RHINITIS CONJUNCTIVITIS
│ │ │
Severity? Season/Perennial? Severity?
│ │ │
┌─────────┴──────┐ ┌────┴────┐ ┌─────┴─────┐
▼ ▼ ▼ ▼ ▼ ▼
MILD MOD/ Seasonal Perennial Mild Severe
Persistent SEVERE │ │ │ │
│ │ Cromolyn Prefer Cromolyn/ Lodoxamide/
Consider MCS Prefer (start intranasal Ketotifen Olopatadine
(esp. children) ICS 2wks early) steroid (topical) + consider
before EIB ±MCS spray steroids
or allergen
exposure
11. Treatment Algorithm for Exercise-Induced Bronchoconstriction (EIB)
PATIENT WITH EXERCISE-INDUCED BRONCHOCONSTRICTION
│
┌──────────────┴──────────────┐
▼ ▼
Mild/Infrequent EIB Frequent/Severe EIB
│ │
Pre-exercise dose of Is patient on controller?
SABA (albuterol) OR │
CROMOLYN/NEDOCROMIL ┌────────┴────────┐
10–15 min before ▼ ▼
exercise NO YES
│ │
Add low-dose Optimize ICS;
ICS or LTRA add pre-exercise
│ SABA + MCS
If EIB persists, combination
add MCS or LABA
(per EIB guidelines,
p. 23)
12. Mast Cell Stabilizers in Special Populations
12.1 Pediatrics
- MCS are among the safest options for children with asthma and allergic rhinitis
- Cromolyn nebulizer solution is used in infants with recurrent wheezing
- Considered when parents/clinicians wish to avoid inhaled corticosteroids in very young children
- Inhaled cromolyn was previously a first-line agent for childhood asthma but has been largely replaced by ICS due to greater efficacy
12.2 Pregnancy
- Preferred agents for allergic rhinitis in pregnancy (intranasal cromolyn: Category B)
- Cromolyn is compatible with breastfeeding
- Minimal systemic absorption ensures fetal safety
12.3 Elderly
- Generally safe
- The low systemic side effect profile makes MCS attractive in elderly patients on multiple medications
- Device training important for inhaled formulations
13. Formulations and Dosing Guide
| Agent | Formulation | Indication | Typical Dose |
|---|---|---|---|
| Cromolyn sodium | MDI (800 mcg/puff) | Asthma prophylaxis | 2 puffs QID |
| Cromolyn sodium | Nebulizer (20 mg/2 mL) | Asthma (infants/severe) | 20 mg QID |
| Cromolyn sodium | Nasal spray (5.2 mg/spray) | Allergic rhinitis | 1 spray each nostril 3–6×/day |
| Cromolyn sodium | Ophthalmic (4%) | Allergic conjunctivitis | 1–2 drops 4–6×/day |
| Cromolyn sodium | Oral (100 mg capsules) | Mastocytosis, GI allergy | 200 mg QID (before meals) |
| Nedocromil sodium | MDI (1.75 mg/puff) | Asthma prophylaxis | 2 puffs QID |
| Nedocromil sodium | Ophthalmic (2%) | Allergic conjunctivitis | 1–2 drops twice daily |
| Lodoxamide | Ophthalmic (0.1%) | Vernal keratoconjunctivitis | 1–2 drops QID |
| Ketotifen | Oral/ophthalmic | Asthma, conjunctivitis | 1 mg BD (oral) |
14. Limitations and Declining Use
Despite their excellent safety profile, mast cell stabilizers have declined in clinical usage due to:
- Inferior efficacy compared to ICS: Inhaled corticosteroids provide superior asthma control
- Frequent dosing: 4× daily dosing reduces adherence compared to once-daily ICS
- Short duration: Bronchoprotection lasts only 1–2 hours (Exercise-induced Bronchoconstriction, p. 23)
- Slow onset: Full anti-inflammatory effect takes 2–4 weeks
- Market withdrawal: Inhaled cromolyn MDI was withdrawn from the US market (2010); nebulizer solution remains available
- Taste (nedocromil): Bitter taste significantly reduces patient acceptance
15. Emerging Perspectives and Future Directions
- Mast cell stabilization in anaphylaxis prevention: Cromolyn's demonstrated ability to block non-IgE-mediated (anaphylactoid) degranulation opens avenues for drug-induced hypersensitivity prevention (PMC Clinical VQA)
- MCAS therapy: Growing recognition of mast cell activation syndrome has renewed interest in oral cromolyn
- Novel MCS: Research into more potent, selective, and better-tolerated MCS continues
- Combination inhalers: Pairing MCS with bronchodilators for EIB management
- Topical applications: Expanded use in food allergy, eosinophilic esophagitis, and IBD
16. Summary Flow Chart
┌──────────────────────────────────────────────────────────┐
│ MAST CELL STABILIZERS — AT A GLANCE │
├──────────────────────────────────────────────────────────┤
│ DRUGS: Cromolyn Sodium | Nedocromil Sodium │
│ Lodoxamide | Ketotifen | Olopatadine │
├──────────────────────────────────────────────────────────┤
│ MECHANISM: │
│ Block Cl⁻ channels → ↓ Ca²⁺ influx → ↓ Degranulation │
│ Inhibit PGD2, histamine, leukotriene release │
│ Nedocromil: also blocks C-fiber nerve activation │
├──────────────────────────────────────────────────────────┤
│ USES: │
│ • Asthma (prophylaxis, EIB prevention) │
│ • Allergic rhinitis │
│ • Allergic conjunctivitis │
│ • Mastocytosis / MCAS (oral cromolyn) │
│ • Food allergy prophylaxis │
├──────────────────────────────────────────────────────────┤
│ SAFETY: ✓ Excellent ✓ Safe in pregnancy ✓ Safe in kids │
├──────────────────────────────────────────────────────────┤
│ LIMITATIONS: Frequent dosing | Slow onset | ICS superior │
│ Short bronchoprotection (1–2 hours) │
└──────────────────────────────────────────────────────────┘
Visual Reference: Cromolyn Sodium in Anaphylactoid Reaction Model
The following image illustrates the experimental pharmacology of cromolyn sodium — demonstrating its ability to attenuate non-IgE-mediated mast cell degranulation. Panel B (cromolyn-pretreated animals) shows markedly reduced Evans Blue leakage compared to Panel A (untreated controls), with the bar graph (Panel C) quantifying the reduction in vascular permeability:
References
- Exercise-induced Bronchoconstriction, p. 23 — Cromolyn and nedocromil: bronchoprotective mechanisms, duration, and combination therapy for EIB
- Harrison's Principles of Internal Medicine, 21st Edition, p. 947 — Allergic conjunctivitis management including mast cell stabilizers (cromolyn) and dual-action agents (olopatadine)
- PMC Clinical VQA — Experimental demonstration of cromolyn sodium attenuating non-IgE-mediated mast cell degranulation in the guinea pig PCA model
This essay covers the complete pharmacological profile of mast cell stabilizers — from molecular mechanisms and cellular pathways, through clinical applications across respiratory, ocular, nasal, and GI domains, to practical prescribing considerations and emerging uses. The flow charts provide visual frameworks for understanding degranulation pathways, drug intervention points, and clinical decision-making.
This is a shared conversation. Sign in to Orris to start your own chat.