Anatomy of female reproductive system
female reproductive system anatomy organs structure
female reproductive system anatomy diagram
Anatomy of the Female Reproductive System
Internal Organs
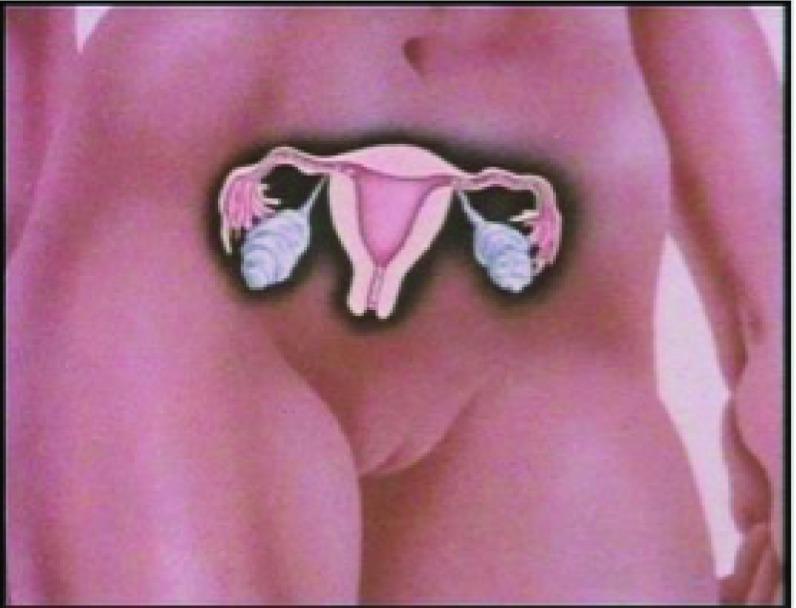
1. Ovaries
- Paired, almond-shaped gonads (~3 × 2 × 1 cm)
- Located on the lateral pelvic wall, one on each side
- Functions: Produce oocytes (eggs) and secrete sex hormones (estrogen, progesterone)
- Consist of an outer cortex (contains follicles) and inner medulla (vasculature/stroma)
- Blood supply: Ovarian arteries (from abdominal aorta) and branches of uterine arteries
2. Fallopian Tubes (Uterine Tubes)
-
~10–12 cm long; connect ovaries to the uterus
-
Four parts (lateral to medial):
Part Feature Infundibulum Funnel-shaped; ends in fimbriae that sweep the oocyte Ampulla Widest, longest segment; site of fertilization Isthmus Narrow segment near uterus Intramural (interstitial) Passes through uterine wall -
Lined by ciliated columnar epithelium that propels the ovum toward the uterus
3. Uterus
- Hollow, muscular, pear-shaped organ; ~7.5 cm long in nulliparous women
- Parts:
- Fundus – dome-shaped top above the fallopian tube openings
- Body (corpus) – main bulk of the uterus
- Isthmus – narrow junction between body and cervix
- Cervix – lower cylindrical portion opening into the vagina
- Layers of the uterine wall:
Layer Description Endometrium Innermost mucosa; cyclically shed during menstruation Myometrium Thick smooth muscle; contracts during labor Perimetrium Outer serosal layer (peritoneum) - Normal position: Anteverted and anteflexed
- Blood supply: Uterine arteries (from internal iliac arteries)
4. Cervix
- Lower cylindrical segment of uterus, ~2.5–3 cm long
- External os – opening into the vagina
- Internal os – opening into the uterine cavity
- Cervical canal – connects internal and external os; lined by mucus-secreting columnar epithelium
- The transformation zone (squamocolumnar junction) is clinically important — site of cervical dysplasia and carcinoma
5. Vagina
- Fibromuscular canal, ~8–10 cm long
- Extends from the cervix to the external genitalia
- Upper end surrounds the cervix forming vaginal fornices (anterior, posterior, and two lateral)
- The posterior fornix is deepest and relates to the pouch of Douglas (rectouterine pouch)
- Lower end opens at the vulva (covered by the hymen in virgins)
- Wall layers: Mucosa (non-keratinized stratified squamous epithelium), muscularis, adventitia
External Genitalia (Vulva)
| Structure | Description |
|---|---|
| Mons pubis | Fatty eminence overlying pubic symphysis; covered by pubic hair |
| Labia majora | Two prominent skin folds; homologous to male scrotum |
| Labia minora | Inner, hairless folds; enclose the vestibule |
| Clitoris | Erectile organ at the anterior junction of labia minora; homologous to the penis |
| Vestibule | Space between the labia minora; contains urethral and vaginal openings |
| Bartholin's glands | Paired mucus-secreting glands at the posterior vestibule; homologous to Cowper's glands |
| Hymen | Thin membrane partially covering the vaginal opening |
Supporting Ligaments
| Ligament | Attachment | Function |
|---|---|---|
| Broad ligament | Uterus to lateral pelvic walls (peritoneal fold) | Contains uterine tubes, ovaries, blood vessels |
| Round ligament | Uterine fundus to labia majora | Maintains anteverted position |
| Uterosacral ligament | Cervix to sacrum | Primary support; prevents uterine prolapse |
| Cardinal (Mackenrodt's) ligament | Cervix/vagina to lateral pelvic wall | Main structural support of the uterus |
| Ovarian ligament | Ovary to uterine wall | Anchors ovary |
Blood Supply Summary
| Structure | Arterial Supply |
|---|---|
| Ovaries | Ovarian arteries (aorta) |
| Uterus & tubes | Uterine arteries (internal iliac) |
| Vagina (upper) | Uterine + vaginal arteries |
| Vagina (lower) | Internal pudendal artery |
| Vulva | Internal & external pudendal arteries |
Lymphatic Drainage (Clinically Important)
| Structure | Drains to |
|---|---|
| Ovaries | Para-aortic (lumbar) nodes |
| Uterine body (upper) | Para-aortic nodes |
| Uterine body (lower) & cervix | Internal iliac, external iliac, obturator nodes |
| Vagina (upper) | Internal & external iliac nodes |
| Vagina (lower) / vulva | Superficial inguinal nodes |
Nerve Supply
- Sympathetic: T10–L2 (via hypogastric plexus)
- Parasympathetic: S2–S4 (pelvic splanchnic nerves)
- Somatic (vulva/perineum): Pudendal nerve (S2–S4)
Chronic hypertension in pregnancy
chronic hypertension in pregnancy definition classification management
chronic hypertension pregnancy complications superimposed preeclampsia fetal outcomes
hypertension in pregnancy classification management
antihypertensive drugs safe in pregnancy labetalol nifedipine methyldopa
chronic hypertension pregnancy monitoring antenatal care delivery timing
Chronic Hypertension in Pregnancy
Definition & Classification
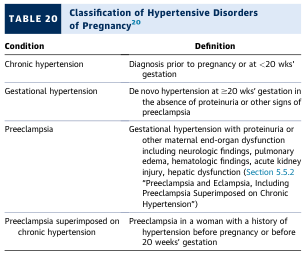
| Condition | Definition |
|---|---|
| Chronic hypertension | Diagnosed before pregnancy or at <20 weeks' gestation |
| Gestational hypertension | De novo hypertension at ≥20 weeks; no proteinuria or end-organ dysfunction |
| Preeclampsia | Gestational hypertension + proteinuria or end-organ dysfunction |
| Superimposed preeclampsia | Preeclampsia in a woman with pre-existing chronic hypertension |
Severity Classification
| Category | Systolic (mmHg) | Diastolic (mmHg) |
|---|---|---|
| Mild/non-severe | 140–159 | 90–109 |
| Severe | ≥160 | ≥110 |
Maternal & Fetal Risks
- Superimposed preeclampsia — occurs in 20–50% of women with chronic hypertension (ACC/AHA Guidelines, p. 67)
- HELLP syndrome (hemolysis, elevated liver enzymes, low platelets)
- Eclampsia
- Placental abruption
- Stroke / hypertensive crisis
- Acute kidney injury
- Pulmonary edema
- Intrauterine growth restriction (IUGR)
- Preterm birth
- Placental insufficiency
- Stillbirth
- Neonatal intensive care admission
Superimposed Preeclampsia
- A sudden increase in baseline blood pressure occurs
- New-onset or worsening proteinuria develops
- New end-organ dysfunction appears (thrombocytopenia, elevated liver enzymes, renal impairment, neurological symptoms, pulmonary edema)
Preconception & Early Pregnancy Assessment
- Identify remediable/secondary causes (renal artery stenosis, primary aldosteronism, pheochromocytoma, etc.)
- Assess end-organ damage: renal function, proteinuria (baseline), echocardiogram (LVH), ophthalmology (hypertensive retinopathy)
- Transition off teratogenic antihypertensives — particularly ACE inhibitors and ARBs (contraindicated; associated with fetal renal dysgenesis and oligohydramnios)
Antihypertensive Treatment in Pregnancy
First-Line Agents
| Drug | Mechanism | Notes |
|---|---|---|
| Labetalol (oral) | α + β blocker | Most commonly used; well-studied safety profile |
| Nifedipine XL | Calcium channel blocker | Extended-release; effective and well-tolerated |
| Methyldopa | Central α2-agonist | Long safety record; less preferred due to side effects (sedation) |
For Acute/Severe Hypertension (≥160/110 mmHg)
| Drug | Route | Dose |
|---|---|---|
| Labetalol IV | IV bolus | 20 mg IV, repeat 40–80 mg q10–30 min; max 300 mg |
| Hydralazine IV | IV bolus | 5–10 mg IV q20 min |
| Nifedipine (immediate-release) | Oral | 10 mg PO, repeat in 30 min |
| Nicardipine IV | IV infusion | Used in ICU settings |
Contraindicated Drugs
| Drug | Reason |
|---|---|
| ACE inhibitors (e.g., enalapril) | Fetal renal tubular dysplasia, oligohydramnios, skull hypoplasia (2nd/3rd trimester) |
| ARBs (e.g., losartan) | Same as ACEi |
| Direct renin inhibitors (aliskiren) | Same as ACEi/ARBs |
| Atenolol | IUGR, neonatal bradycardia |
Aspirin Prophylaxis
- Low-dose aspirin (81 mg/day) started at 12–16 weeks is the only routinely recommended intervention to reduce the risk of superimposed preeclampsia in high-risk women (ACC/AHA Guidelines, p. 67).
Antenatal Monitoring
| Frequency | Assessment |
|---|---|
| Every 2–4 weeks (early) | BP monitoring, symptoms |
| Each visit | Urine protein (dipstick or PCR) |
| 28–32 weeks | Fetal growth ultrasound (for IUGR) |
| 32–36 weeks | Umbilical artery Doppler if growth restriction suspected |
| 32–37 weeks | Fetal surveillance (non-stress test / biophysical profile) |
Timing of Delivery
| Clinical Scenario | Recommended Delivery Timing |
|---|---|
| Well-controlled, no complications | 38–39 weeks |
| Difficult to control, end-organ damage | 37 weeks |
| Superimposed preeclampsia without severe features | 37 weeks |
| Superimposed preeclampsia with severe features | 34–37 weeks (individualized) |
| IUGR or deteriorating fetal status | Earlier delivery based on clinical status |
Postpartum Considerations
- BP often rises in the first 3–5 days postpartum — close monitoring is essential
- Antihypertensives should be continued postpartum; most are compatible with breastfeeding (labetalol, nifedipine, enalapril are considered safe for lactation)
- ACE inhibitors/ARBs can be restarted postpartum if not breastfeeding, or enalapril/captopril if breastfeeding (low milk transfer)
- Screen for underlying secondary hypertension if BP does not normalize by 12 weeks postpartum
Key Takeaways
- Chronic hypertension complicates ~1–5% of pregnancies and carries significant maternal and fetal risks
- The most feared complication is superimposed preeclampsia (20–50% risk)
- Labetalol and extended-release nifedipine are first-line oral agents; target BP 130–150/80–100 mmHg
- ACE inhibitors and ARBs are contraindicated throughout pregnancy
- Low-dose aspirin from 12–16 weeks reduces superimposed preeclampsia risk
- Severe hypertension (≥160/110) requires urgent IV treatment within 30–60 minutes
Cause of preeclampsia
preeclampsia etiology pathophysiology causes risk factors
preeclampsia pathophysiology placental dysfunction diagram
Causes & Pathophysiology of Preeclampsia
The Two-Stage Model
Stage 1: Abnormal Placentation
- Shallow trophoblast invasion of spiral arteries
- Spiral arteries remain narrow, high-resistance, and reactive
- Chronic uteroplacental ischemia and ischemia-reperfusion injury
- Excessive oxidative stress and reactive oxygen species (ROS) generation
Stage 2: Systemic Maternal Endothelial Dysfunction
Angiogenic Imbalance — Key Mechanism
| Factor | Role in Preeclampsia |
|---|---|
| sFlt-1 (soluble FMS-like tyrosine kinase-1) | Anti-angiogenic; binds and neutralizes free VEGF and PlGF → endothelial dysfunction |
| Soluble endoglin (sEng) | Antagonizes TGF-β signaling → impairs vasodilation |
| VEGF (vascular endothelial growth factor) | Normally promotes endothelial health; decreased in preeclampsia |
| PlGF (placental growth factor) | Angiogenic; markedly reduced → used as a biomarker |
| TGF-β (transforming growth factor-β) | Vasodilatory; blocked by excess sEng |
Excessive placental production of sFlt-1 and sEng creates an anti-angiogenic state, causing systemic endothelial injury (Harrison's, p. 13632).
Pathophysiology Cascade
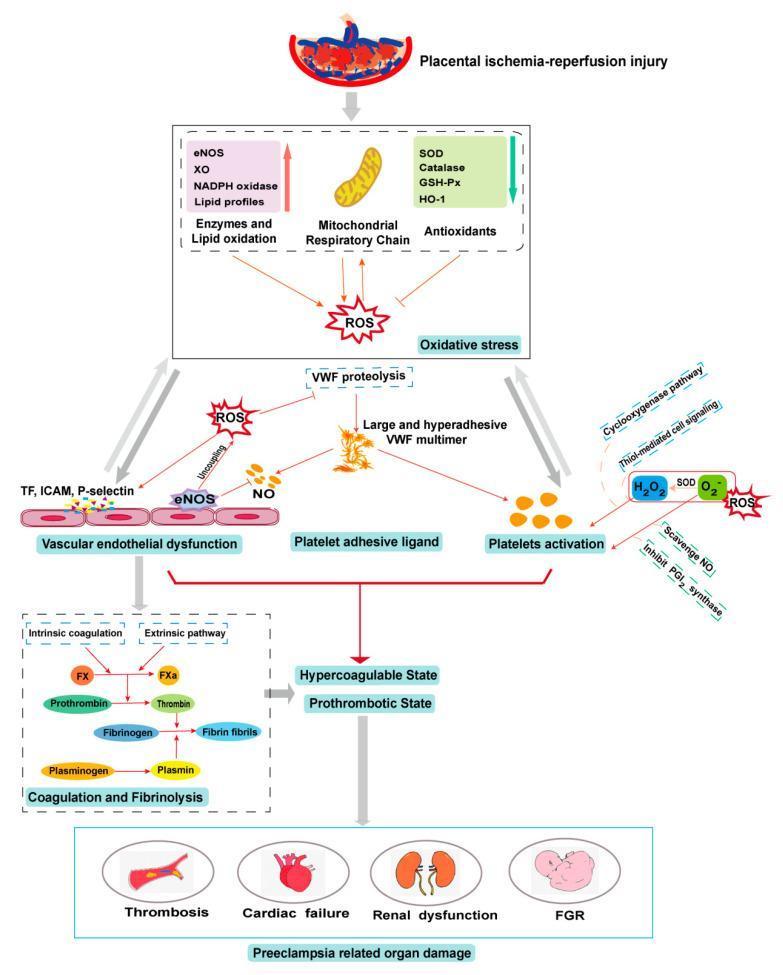
- Placental ischemia-reperfusion injury → oxidative stress (ROS generation via eNOS uncoupling, NADPH oxidase, xanthine oxidase)
- Vascular endothelial dysfunction → upregulation of tissue factor (TF), ICAM, P-selectin
- Large VWF multimer formation → platelet activation
- Nitric oxide (NO) scavenging → loss of vasodilation → hypertension
- Coagulation cascade activation → hypercoagulable, prothrombotic state
- End-organ damage → renal dysfunction, cardiac failure, fetal growth restriction (FGR)
Neurological Manifestations
Contributing Mechanisms Summary
| Mechanism | Description |
|---|---|
| Uteroplacental ischemia | Failed spiral artery remodeling → reduced perfusion |
| Exaggerated maternal inflammatory response | Systemic activation of innate immunity |
| Angiogenic imbalance | Excess sFlt-1/sEng; reduced VEGF/PlGF |
| Endothelial dysfunction | Widespread vascular injury, increased permeability |
| Oxidative stress | ROS generation → lipid peroxidation, NO depletion |
| Prothrombotic state | Platelet activation, coagulation cascade → HELLP, thrombosis |
| Immune maladaptation | Inadequate tolerance to paternal (fetal) antigens |
Risk Factors (Predisposing Causes)
| Category | Risk Factors |
|---|---|
| Obstetric | Nulliparity, multiple gestation, molar pregnancy, previous preeclampsia |
| Medical | Chronic hypertension, pre-existing diabetes, renal disease, autoimmune conditions (SLE, antiphospholipid syndrome) |
| Maternal | Obesity (BMI >30), age >35, Black race (higher risk & severity) |
| Genetic | Family history of preeclampsia |
| Paternal | New partner (reduced immune tolerance), long inter-pregnancy interval |
| Placental | Thrombophilias, placenta previa, hydrops fetalis |
Immune Maladaptation Theory
- Higher risk in first pregnancies (nulliparity)
- Protection in subsequent pregnancies with the same partner
- Increased risk with donor egg IVF (entirely foreign paternal and maternal antigens)
- Reduced risk with longer sexual cohabitation before pregnancy (immune tolerance builds over time)
Why Delivery Cures Preeclampsia
Key Takeaway
Failed trophoblast invasion → uteroplacental ischemia → oxidative stress → angiogenic imbalance (↑sFlt-1, ↓VEGF/PlGF) → endothelial dysfunction → hypertension + proteinuria + end-organ damage
Hypertension disease in pregnancy
hypertensive disorders of pregnancy classification diagnosis criteria
hypertensive disorders pregnancy preeclampsia eclampsia HELLP
preeclampsia eclampsia HELLP syndrome diagnosis management treatment
gestational hypertension management antihypertensive delivery timing pregnancy
Hypertensive Disorders of Pregnancy (HDP)
Classification

| Condition | Definition |
|---|---|
| Chronic Hypertension | Pre-existing diagnosis OR BP ≥140/90 mmHg on 2 occasions ≥4 hours apart before 20 weeks' gestation |
| Gestational Hypertension | De novo hypertension at ≥20 weeks; no proteinuria or end-organ dysfunction |
| Preeclampsia | Gestational hypertension + proteinuria or end-organ dysfunction |
| Superimposed Preeclampsia | Preeclampsia developing in a woman with pre-existing chronic hypertension |
| Eclampsia | New-onset generalized tonic-clonic seizures in a patient with preeclampsia |
1. Chronic Hypertension in Pregnancy
- Defined as BP ≥140/90 mmHg before 20 weeks or pre-existing
- Carries risk of superimposed preeclampsia (20–50%), IUGR, placental abruption
- Treatment: Labetalol, extended-release nifedipine, or methyldopa
- Target BP: 130–150 / 80–100 mmHg
- Contraindicated: ACE inhibitors, ARBs (fetal renal toxicity)
- Low-dose aspirin 81 mg/day from 12–16 weeks to reduce preeclampsia risk
2. Gestational Hypertension
- De novo BP ≥140/90 mmHg at ≥20 weeks without proteinuria or end-organ dysfunction
- Resolves within 12 weeks postpartum (if persistent → reclassified as chronic hypertension)
- ~25–50% progress to preeclampsia — requires close monitoring
- Management:
- Mild (140–159/90–109): Outpatient monitoring; antihypertensives if BP sustained
- Severe (≥160/110): Hospitalize; treat urgently; deliver at 37 weeks
- Fetal surveillance and growth scans from 32 weeks
3. Preeclampsia
Diagnostic Criteria (ACOG)
| Criterion | Threshold |
|---|---|
| Proteinuria | ≥300 mg/24h OR protein:creatinine ratio ≥0.3 OR dipstick ≥2+ |
| Thrombocytopenia | Platelets <100,000/μL |
| Renal insufficiency | Creatinine >1.1 mg/dL or doubling of baseline |
| Impaired liver function | LFTs >2× upper limit; severe RUQ/epigastric pain |
| Pulmonary edema | New onset |
| Neurological symptoms | New-onset headache unresponsive to analgesia, visual disturbances |
Severe Features
| Feature | Criterion |
|---|---|
| Severe hypertension | BP ≥160/110 mmHg on 2 occasions ≥4 hours apart |
| Thrombocytopenia | Platelets <100,000/μL |
| Renal impairment | Creatinine >1.1 mg/dL |
| Hepatic dysfunction | Transaminases ≥2× normal; severe RUQ pain |
| Pulmonary edema | — |
| New-onset headache/visual disturbances | Unresponsive to medication |
Pathophysiology (Brief)
Failed trophoblast invasion → uteroplacental ischemia → oxidative stress → excess sFlt-1/sEng → endothelial dysfunction → hypertension + proteinuria + end-organ damage
Management of Preeclampsia
| Aspect | Detail |
|---|---|
| Definitive treatment | Delivery (only cure) |
| Seizure prophylaxis | Magnesium sulfate IV (loading 4–6 g, then 1–2 g/hr infusion) |
| Antihypertensives | IV labetalol, IV hydralazine, oral nifedipine (for acute severe HTN) |
| Corticosteroids | Betamethasone/dexamethasone if <34 weeks (fetal lung maturity) |
| Aspirin prophylaxis | 81 mg/day from 12–16 weeks (high-risk women) |
Delivery Timing
| Scenario | Timing |
|---|---|
| Preeclampsia without severe features | 37 weeks |
| Preeclampsia with severe features | 34 weeks (after steroids) |
| Uncontrolled severe HTN, deteriorating status | Immediate delivery |
4. HELLP Syndrome
| Letter | Finding | Diagnostic Threshold |
|---|---|---|
| H | Hemolysis | Microangiopathic hemolytic anemia on smear; elevated LDH, low haptoglobin |
| EL | Elevated Liver enzymes | AST/ALT >70 IU/L |
| LP | Low Platelets | <100,000/μL |
- May present without hypertension or proteinuria in ~15–20%
- Presents with: epigastric/RUQ pain, nausea/vomiting, malaise
- Management: Stabilize, magnesium sulfate, urgent delivery (≥34 weeks); transfuse if platelets <50,000 before cesarean
- Complications: DIC, liver hematoma/rupture, acute renal failure, pulmonary edema
5. Eclampsia
- Definition: New-onset generalized tonic-clonic seizures in a preeclamptic patient — the most severe neurological complication
- Can occur antepartum (>50%), intrapartum, or postpartum (up to 48 hrs after delivery)
- 1 in 200 preeclampsia cases progress to eclampsia without treatment (Harrison's, p. 13632)
Management of Eclamptic Seizure
- Airway, Breathing, Circulation (ABC) — lateral decubitus position, oxygen
- Magnesium sulfate — 4–6 g IV over 15–20 min (first-line anticonvulsant and prophylaxis)
- Control BP — IV labetalol or hydralazine if ≥160/110 mmHg
- Deliver after stabilization — mode based on obstetric factors
- Continue MgSO₄ for 24 hours postpartum
Note: Diazepam/phenytoin are second-line only; magnesium sulfate is superior for eclampsia prevention and treatment.
6. Superimposed Preeclampsia
- Occurs in 20–50% of women with chronic hypertension (ACC/AHA Guidelines, p. 67)
- Diagnosed when a woman with chronic hypertension develops:
- Sudden worsening of BP
- New or worsening proteinuria
- New end-organ dysfunction (thrombocytopenia, elevated LFTs, renal impairment, pulmonary edema, neurological symptoms)
- Carries the highest risk of all HDPs for maternal and fetal complications
Maternal & Fetal Complications Overview
| Complication | Condition |
|---|---|
| Stroke | Severe preeclampsia, eclampsia |
| Pulmonary edema | Preeclampsia with severe features, HELLP |
| Acute kidney injury | Preeclampsia, HELLP |
| DIC | HELLP, severe preeclampsia |
| Liver rupture | HELLP |
| Placental abruption | All HDPs, especially chronic HTN |
| Preterm birth | All HDPs |
| IUGR / stillbirth | Chronic HTN, preeclampsia |
| Neonatal ICU admission | Preterm delivery in all HDPs |
Prevention Summary
| Intervention | Indication |
|---|---|
| Low-dose aspirin 81 mg/day (from 12–16 weeks) | High-risk women (chronic HTN, prior preeclampsia, multifetal pregnancy, diabetes, renal disease, autoimmune disease) |
| Calcium supplementation (1.5–2 g/day) | Women with low dietary calcium intake |
| Weight optimization preconception | Obesity is a major modifiable risk factor |
| Antihypertensive optimization preconception | Stop ACEi/ARBs; switch to safe agents |
Key Points
- HDPs are classified into 4 main types: chronic hypertension, gestational hypertension, preeclampsia, and superimposed preeclampsia
- Magnesium sulfate + antihypertensives are the mainstay of preeclampsia/eclampsia treatment (ACC/AHA Guidelines, p. 67)
- Delivery is the only cure — timing is individualized based on severity and gestational age
- Postpartum surveillance is essential — HDPs (especially eclampsia) can occur up to 6 weeks postpartum