Q. How will u evaluate and manage a case of glomus jugulare tumor in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances according to rguhs university
Reading File
search_medical_text
glomus jugulare tumor evaluation classification clinical features diagnosis
search_medical_text
glomus jugulare tumor management surgery radiotherapy treatment
search_medical_image
glomus jugulare tumor imaging CT MRI angiography anatomy jugular foramen
GradeResponse
GradeResponse
GradeResponse
Excellent results obtained. Now let me synthesize a complete, comprehensive 50-mark answer.
GLOMUS JUGULARE TUMOR — EVALUATION AND MANAGEMENT
(50 Marks — RGUHS Standard Answer)
References: Scott Brown's Otorhinolaryngology, Cummings Otolaryngology, Stell & Maran's Head & Neck Surgery, Dhingra's Diseases of Ear Nose & Throat, Hazarika's Textbook of ENT, Zakir Hussain's ENT, and Current Literature
1. INTRODUCTION & DEFINITION
A glomus jugulare tumor (also called jugular paraganglioma) is a benign, highly vascular, locally aggressive tumor arising from the paraganglionic tissue (glomus bodies) located in the adventitia of the jugular bulb within the jugular foramen at the skull base.
- Synonyms: Chemodectoma, Non-chromaffin paraganglioma, Jugular body tumor
- Part of the paraganglioma family — same cell origin as carotid body tumor, glomus tympanicum, glomus vagale
- Histologically identical to glomus tympanicum but arises from the jugular fossa, making it far more extensive and surgically challenging
- Most common tumor of the jugular foramen
- Second most common tumor of the temporal bone (after acoustic neuroma)
(Cummings Otolaryngology, 7th Ed.; Scott Brown's 8th Ed., Vol. 3)
2. APPLIED ANATOMY — JUGULAR FORAMEN
┌─────────────────────────────────────────────────────────┐
│ JUGULAR FORAMEN ANATOMY │
│ │
│ Divided by the intrajugular process into: │
│ │
│ ┌──────────────────┐ ┌──────────────────────────┐ │
│ │ PARS NERVOSA │ │ PARS VASCULARIS │ │
│ │ (anteromedial) │ │ (posterolateral) │ │
│ │ │ │ │ │
│ │ CN IX │ │ CN X (Vagus) │ │
│ │ (Glossopharyn.) │ │ CN XI (Accessory) │ │
│ │ Inferior petrosal│ │ Jugular Bulb │ │
│ │ sinus │ │ Sigmoid sinus │ │
│ └──────────────────┘ └──────────────────────────┘ │
│ │
│ CN XII exits via hypoglossal canal (nearby) │
│ Internal carotid artery passes anteromedially │
└─────────────────────────────────────────────────────────┘
Key relations:
- Superior: Middle ear, floor of middle cranial fossa
- Medial: Internal carotid artery (ICA), CN IX, X, XI, XII
- Posterior: Sigmoid sinus
- Inferior: Upper neck — CN X, XI, XII, internal jugular vein, ICA
(Dhingra, 7th Ed.; Hazarika ENT)
3. EPIDEMIOLOGY
| Parameter | Details |
|---|---|
| Incidence | 1 per 1.3 million population/year |
| Sex | F:M = 4–6:1 (most common head & neck tumor in women) |
| Age | 4th–6th decade |
| Bilateral | 10% of sporadic; up to 30–78% of familial cases |
| Functional (secreting catecholamines) | 1–3% |
| Malignant | 3–4% (SDHB mutation highest risk) |
| Hereditary | ~30–35% (autosomal dominant, PGL syndromes) |
(Cummings 7th Ed., p. 1735; Scott Brown 8th Ed.)
4. ETIOLOGY & GENETICS
Paraganglioma Syndromes (PGL)
| Syndrome | Gene Mutation | Features |
|---|---|---|
| PGL-1 | SDHD | Multiple HN paragangliomas; paternal imprinting |
| PGL-2 | SDHAF2 | Head & neck PGL |
| PGL-3 | SDHC | Head & neck PGL |
| PGL-4 | SDHB | Higher risk of malignancy; abdominal PGL |
| PGL-5 | SDHA | — |
| MEN-2A/2B | RET proto-oncogene | Pheochromocytoma + medullary thyroid ca |
| VHL | VHL gene | Hemangioblastoma + PGL |
| NF-1 | NF1 gene | Multiple neurofibromas + PGL |
SDHB mutations carry the highest risk of malignancy (~30–40%).
(Cummings 7th Ed.; Ivan et al., J Neurosurg 2011)
5. PATHOLOGY
Gross:
- Reddish-purple, highly vascular, lobulated
- "Sunrise" or "setting sun" appearance on otoscopy (pulsatile red mass behind intact TM)
Histology:
- Zellballen pattern — clusters (balls) of chief cells (type I) surrounded by sustentacular cells (type II) in a highly vascular stroma
- Chief cells: round to polygonal, vesicular nuclei, granular cytoplasm
- Rich capillary network
- Chromogranin A and synaptophysin positive (neuroendocrine markers)
- S-100 positive in sustentacular cells
Malignancy Criteria (Thompson):
Malignancy is defined only by presence of metastasis (regional lymph nodes or distant), NOT by histological features alone.
(Stell & Maran's; Dhingra 7th Ed.)
6. CLINICAL FEATURES
Symptoms (in order of frequency):
SYMPTOM PROGRESSION WITH TUMOR GROWTH
───────────────────────────────────────────────────────────
EARLY:
▶ Pulsatile tinnitus (most common — 80%)
▶ Conductive hearing loss
▶ Aural fullness / pressure
AS TUMOR GROWS (middle ear invasion):
▶ Otorrhagia (bleeding from ear)
▶ "Pulsatile red mass" visible behind TM
ADVANCED (CN involvement at jugular foramen):
▶ CN IX → Dysphagia, loss of gag reflex
▶ CN X → Hoarseness, aspiration, palatal palsy
▶ CN XI → Ipsilateral trapezius/SCM weakness
▶ CN XII → Ipsilateral tongue paralysis/atrophy
= VERNET'S SYNDROME (IX, X, XI)
= COLLET-SICARD SYNDROME (IX, X, XI, XII)
INTRACRANIAL EXTENSION:
▶ Headache, raised ICP
▶ CN V, VI, VII palsy
▶ Cerebellar signs
Signs:
- Pulsatile red mass behind intact tympanic membrane
- Brown's sign: Mass blanches with pneumatic otoscopy (pathognomonic)
- Aquino's sign: Pulsatile bruit audible over mastoid
- Conductive hearing loss on tuning fork tests
- Lower cranial nerve palsies (CN IX–XII)
- Cervical lymphadenopathy (if malignant)
- Horner's syndrome (sympathetic chain involvement)
(Hazarika ENT, 4th Ed.; Dhingra; Zakir Hussain ENT)
7. CLASSIFICATION
A. FISCH CLASSIFICATION (Most widely used)
(Fisch U, 1982 — Standard for surgical planning)
| Type | Description |
|---|---|
| Type A | Limited to middle ear cleft (tympanic paraganglioma) |
| Type B | Limited to tympanomastoid compartment; no infralabyrinthine extension |
| Type C | Involves infralabyrinthine compartment & extends into petrous apex |
| C1 | Erodes jugular foramen, but no intracranial extension |
| C2 | Invades vertical carotid canal |
| C3 | Invades horizontal carotid canal |
| C4 | Reaches foramen lacerum and cavernous sinus |
| Type D | Intracranial extension |
| De1 | Intracranial displacement <2 cm |
| De2 | Intracranial displacement >2 cm |
B. GLASSCOCK-JACKSON CLASSIFICATION
| Type | Description |
|---|---|
| I | Small mass limited to jugular bulb, middle ear, mastoid |
| II | Extends beneath IAC; may have intracranial extension |
| III | Extends into petrous apex; may have intracranial extension |
| IV | Extends beyond petrous apex into clivus or infratemporal fossa; may extend intracranially |
(Scott Brown's 8th Ed.; Cummings 7th Ed.)
8. INVESTIGATIONS
FLOWCHART: DIAGNOSTIC ALGORITHM
SUSPECTED GLOMUS JUGULARE TUMOR
(Pulsatile tinnitus + Pulsatile red mass + CN palsies)
│
▼
┌─────────────────────────────────┐
│ OTOSCOPY + AUDIOMETRY │
│ Pulsatile red mass (Brown's │
│ sign +ve); Conductive HL │
└──────────────┬──────────────────┘
│
▼
┌──────────────────────────────────────────────────────┐
│ IMAGING STUDIES │
│ │
│ ┌──────────────┐ ┌───────────────┐ ┌──────────┐ │
│ │ HRCT │ │ MRI + MRA │ │ DSA/4- │ │
│ │ TEMPORAL │ │ BRAIN + │ │ VESSEL │ │
│ │ BONE │ │ SKULL BASE │ │ ANGIO │ │
│ │ │ │ │ │ │ │
│ │ Bony erosion │ │ Salt & pepper │ │ Tumor │ │
│ │ Jugular fossa│ │ sign │ │ blush │ │
│ │ enlargement │ │ Assess extent │ │ Dominant │ │
│ │ "Moth-eaten" │ │ ICA encasement│ │ supply │ │
│ │ appearance │ │ Venous sinuses│ │ Pre-op │ │
│ └──────────────┘ └───────────────┘ │ emboliz. │ │
│ └──────────┘ │
└──────────────────────────────────────────────────────┘
│
▼
┌─────────────────────────────────┐
│ FUNCTIONAL/NUCLEAR IMAGING │
│ │
│ • 68Ga-DOTATATE PET/CT │
│ (Best sensitivity — SSR) │
│ • 123I/131I-MIBG scintigraphy │
│ • 18F-FDG PET (SDHB mutations) │
│ • Octreotide scintigraphy │
└──────────────────┬──────────────┘
│
▼
┌──────────────────────────────────┐
│ BIOCHEMICAL TESTS │
│ │
│ • 24h Urine catecholamines │
│ • Plasma metanephrines │
│ • Chromogranin A (serum) │
│ • VMA (vanillylmandelic acid) │
│ (Rule out functional tumor) │
└──────────────────┬───────────────┘
│
▼
┌─────────────────────────────────────┐
│ GENETIC TESTING │
│ │
│ • SDHB, SDHC, SDHD, SDHA mutation │
│ • If familial or bilateral │
│ • RET, VHL, NF1 if MEN/VHL/NF susp.│
└─────────────────────────────────────┘
A. AUDIOLOGICAL ASSESSMENT
- Pure Tone Audiometry: Conductive hearing loss (air-bone gap)
- Tympanometry: Type B or As pattern (reduced compliance)
- BERA/ABR: Sensorineural component if cochlear involvement
B. IMAGING IN DETAIL
i. High-Resolution CT (HRCT) Temporal Bone:
- Gold standard for bony detail
- Findings:
- Irregular/permeative erosion of jugular foramen margins ("moth-eaten" appearance)
- Enlargement of jugular fossa
- Erosion of caroticojugular spine (separating jugular bulb from carotid canal)
- Destruction of petrous apex
- Air cell involvement
ii. MRI with Gadolinium (MRI Brain + Skull Base):
- Gold standard for soft tissue extent
- Classic "Salt and Pepper" Sign on T1WI:
- Salt = Hyperintense foci (hemorrhage/slow-flow vessels)
- Pepper = Hypointense signal voids (rapid-flow vessels/flow voids)
- Intense enhancement with gadolinium
- Assesses: Intracranial extension, dural involvement, brainstem compression, ICA encasement, sigmoid/jugular vein patency
(See multimodal imaging below)
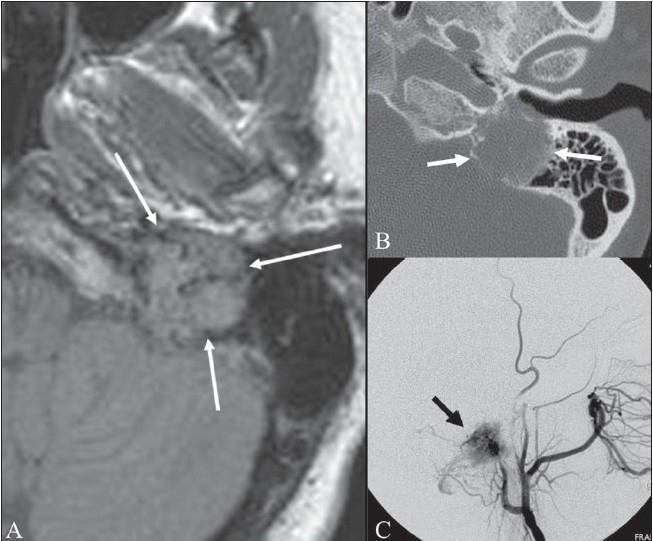
iii. Digital Subtraction Angiography (DSA) / 4-Vessel Angiography:
- Pre-operative must: Defines tumor vascularity and feeding vessels
- Typical feeding vessels:
- Ascending pharyngeal artery (most common)
- Posterior auricular artery
- Occipital artery
- ICA and vertebral artery branches (large tumors)
- Balloon Occlusion Test (BOT): Assess contralateral ICA adequacy before sacrifice
- Pre-operative embolization: Performed 24–72 hours before surgery to reduce intraoperative bleeding by 50–70%
iv. 68Ga-DOTATATE PET/CT:
- Highest sensitivity (>95%) for paragangliomas
- Detects multifocal/bilateral disease, metastases, recurrence
- Janssen et al. (J Nucl Med 2016): Superior to MIBG and FDG-PET for HN paragangliomas
(Cummings 7th Ed.; Janssen et al., J Nucl Med 2016, p. 186–191)
C. BIOCHEMICAL TESTS
- 24-hour urine: Catecholamines (epinephrine, norepinephrine, dopamine), VMA, metanephrines
- Plasma: Free metanephrines, normetanephrine
- Serum: Chromogranin A (tumor bulk marker)
- Functional tumors (~1–3%): Elevated; requires alpha-blockade before surgery
9. DIFFERENTIAL DIAGNOSIS
| Condition | Distinguishing Features |
|---|---|
| Glomus tympanicum | Confined to middle ear; no jugular fossa erosion on CT |
| High jugular bulb | No bony erosion; venous structure on MRV |
| Aberrant ICA | Pulsatile; CT shows absent caroticojugular spine; no bony erosion |
| Persistent stapedial artery | Absent foramen spinosum on CT |
| Cholesterol granuloma | T1 hyperintense without salt-pepper; no flow voids |
| Endolymphatic sac tumor | Posterior petrous; associated with VHL |
| Meningioma | Dural tail; calcification; no salt-pepper sign |
| Schwannoma | Lower signal enhancement; no flow voids |
| Rhabdomyosarcoma | Children; aggressive destruction; histology |
10. MANAGEMENT
FLOWCHART: MANAGEMENT ALGORITHM
CONFIRMED GLOMUS JUGULARE TUMOR
│
▼
┌───────────────────────────────┐
│ MULTIDISCIPLINARY TEAM (MDT) │
│ ENT/Skull base surgeon + │
│ Neurosurgeon + Radiologist + │
│ Endocrinologist + Oncologist │
└───────────────┬───────────────┘
│
┌───────────┴──────────┐
▼ ▼
FUNCTIONAL? ASSESS EXTENT
(biochemistry) (Fisch/GJ Grade)
│ │
Preop alpha- ┌─────┴────────────────┐
blockade │ │
(Phenoxybenzamine) ▼ ▼
SURGICAL NON-SURGICAL
CANDIDATE? (Radiosurgery/RT)
│
┌─────────┴──────────┐
▼ ▼
PREOPERATIVE OBSERVE
EMBOLIZATION (Watch & Wait)
(24-72h before) (Elderly/Infirm/
│ Small tumors)
▼
SURGERY
(Approach based
on Fisch grade)
│
┌──────┴───────────┐
▼ ▼
COMPLETE SUBTOTAL
RESECTION RESECTION
+ Adjuvant RT
A. WATCH AND WAIT (Active Surveillance)
Indications:
- Elderly patients (>70 years)
- Asymptomatic, small tumor
- Serious medical comorbidities
- Contralateral lower CN deficits (surgery would cause bilateral palsy)
- Patient preference
Evidence: Many glomus jugulare tumors grow very slowly (~1 mm/year). Annual MRI surveillance is recommended.
(Scott Brown's 8th Ed.; Suárez et al., Head Neck 2013)
B. PRE-OPERATIVE PREPARATION
-
Embolization (24–72 hours preop):
- Selective catheterization and embolization of feeding vessels
- Embolic agents: PVA particles, Onyx, Gelfoam, coils
- Reduces intraoperative blood loss significantly
- Risk: Stroke, cranial nerve palsy if perforators embolized
-
Functional tumor: Alpha-adrenergic blockade for minimum 2 weeks:
- Phenoxybenzamine 10 mg BD (dose titrated)
- Beta-blocker added after adequate alpha-blockade
- Goal: BP <130/80, orthostatic drop <20 mmHg
- Avoid anesthetic agents that precipitate catecholamine release
-
Tracheostomy: Pre-operative (if significant CN X palsy — aspiration risk)
-
Lumbar drain: For large tumors with intracranial extension (to relax brain)
-
Blood cross-matching: 4–6 units packed RBCs
-
Neurophysiological monitoring: Intraoperative CN monitoring (CN VII, IX, X, XI, XII)
C. SURGICAL MANAGEMENT
Surgical Approaches Based on Fisch Classification:
SURGICAL APPROACH SELECTION
═══════════════════════════════════════════════════════
Fisch A → TRANSCANAL / TRANSMEATAL approach
(limited middle ear)
Fisch B → EXTENDED MASTOIDECTOMY + TYMPANOTOMY
(tympanomastoid compartment)
Fisch C1 → INFRATEMPORAL FOSSA APPROACH TYPE A
(Fisch Type A IFA)
- Blind sac closure of EAC
- Anterior rerouting of facial nerve
- Neck dissection (control of great vessels)
Fisch C2-C4 → INFRATEMPORAL FOSSA APPROACH TYPE A
+ Petrous apex + ICA control
Fisch D (intracranial) → COMBINED:
- Infratemporal Fossa Type A
+ Posterior Fossa Craniotomy
(Neurosurgical collaboration)
═══════════════════════════════════════════════════════
Infratemporal Fossa Approach Type A (ITFA-A) — Fisch's Approach (Gold Standard):
Steps:
- Positioning: Lateral/supine with head turned
- Incision: C-shaped postauricular + cervical extension
- Neck dissection: Identify and control ICA, IJV, CN IX, X, XI, XII in neck
- Mastoidectomy: Canal wall down mastoidectomy
- EAC closure: Blind sac closure (oversew meatus)
- Facial nerve: Anterior rerouting (permanent conductive HL accepted)
- Sigmoid sinus: Ligation and packed (if tumor invades)
- Jugular vein: Ligation in neck
- Tumor removal: En-bloc where possible; meticulous hemostasis
- Reconstruction: Fat obliteration of cavity; temporalis muscle flap
Operative risks:
- Facial nerve palsy (temporary/permanent)
- Lower CN palsies (IX, X, XI, XII)
- Sensorineural hearing loss
- CSF leak
- Stroke (ICA injury/sacrifice)
- Meningitis
- Death
(Fisch U, 1982; Cummings 7th Ed.; Scott Brown's 8th Ed.)
Other Approaches:
| Approach | Indication |
|---|---|
| Transcochlear approach | Anterior extension to petrous apex/clivus |
| Combined petrosal approach | Large tumors with CPA and middle fossa extension |
| Retrosigmoid/posterior fossa | Primarily intracranial, posterior fossa extension |
| Transmastoid approach | Type A/B, small tumors |
D. RADIATION THERAPY
1. Stereotactic Radiosurgery (SRS) — Gamma Knife / CyberKnife:
- Single fraction: 12–15 Gy to tumor margin
- Tumor control rate: 90–95% at 5 years
- Mechanism: Vascular endothelial damage → tumor growth arrest (not ablation)
- Best for: Fisch C–D (inoperable), elderly, residual/recurrent disease, good CN function
- Ivan et al. meta-analysis (J Neurosurg 2011): Comparable tumor control to surgery with lower morbidity
2. Fractionated Stereotactic Radiotherapy (FSRT):
- 45–54 Gy in 25–30 fractions
- For larger tumors where single-fraction SRS unsafe
- Chun et al. (Stereotact Funct Neurosurg 2014): 5-fraction FSRT effective with volumetric response
3. Conventional External Beam Radiotherapy (EBRT):
- 45–50 Gy in 25 fractions
- Historical; Hinerman et al. (Head Neck 2008): 93% local control at 35-year follow-up
- Used when SRS not available; Van Hulsteijn et al. meta-analysis (Radiother Oncol 2013): confirms efficacy
Comparison:
| Modality | Tumor Control | CN Morbidity | Hearing | Use |
|---|---|---|---|---|
| Surgery | 80–90% | High | May lose | Young, operable, Fisch A–C |
| Gamma Knife SRS | 90–95% | Low | Preserved | Elderly, inoperable, residual |
| FSRT | 88–93% | Low | Preserved | Large, near critical structures |
| EBRT | 85–93% | Moderate | Preserved | When SRS unavailable |
(Ivan et al. J Neurosurg 2011; Suárez et al. Head Neck 2013; Scott Brown's 8th Ed.)
E. EMBOLIZATION ALONE (Palliative)
- For elderly/medically unfit patients
- Controls pulsatile tinnitus and hemorrhage
- Not curative; re-growth occurs
F. MEDICAL/SYSTEMIC THERAPY
1. PRRT — Peptide Receptor Radionuclide Therapy:
- 177Lu-DOTATATE (Lutathera): For somatostatin receptor-positive, progressive/metastatic disease
- Delivers targeted radiation to tumor cells
- Used for unresectable, metastatic, or recurrent cases
2. Somatostatin Analogues:
- Octreotide LAR, Lanreotide: Symptomatic control (catecholamine secretion)
- Limited anti-proliferative effect
3. Chemotherapy (Malignant Paraganglioma):
- CVD regimen: Cyclophosphamide + Vincristine + Dacarbazine
- Partial response in ~50% of malignant cases
- Temozolomide (alkylating agent)
4. Targeted/Immunotherapy (Recent Advances):
- Pembrolizumab (anti-PD-1): Phase II trial (Jimenez et al., Cancers 2020) — modest activity in progressive metastatic paraganglioma
- Sunitinib, Axitinib (anti-VEGF/TKI): Promising in SDHB-mutated metastatic disease
- Hypoxia-inducible factor (HIF) pathway inhibitors: Under investigation
(Janssen et al. J Nucl Med 2016; Caplin et al. NEJM 2014; Jimenez et al. Cancers 2020)
11. RECENT ADVANCES
| Area | Advance |
|---|---|
| Genetics | Next-generation sequencing (NGS) panels for SDHx, MAX, TMEM127, FH mutations; germline testing recommended for ALL paragangliomas |
| Imaging | 68Ga-DOTATATE PET/CT — superior sensitivity (>95%) vs. MIBG, now first-line nuclear imaging |
| Radiosurgery | Proton beam radiosurgery for skull base paragangliomas — conformality advantage |
| Embolization | Liquid embolic agents (Onyx 18) — improved devascularization, lower recanalization |
| PRRT | 177Lu-DOTATATE FDA-approved; trials ongoing for HN paragangliomas |
| Immunotherapy | Checkpoint inhibitors (PD-1/PD-L1) under investigation |
| Endoscopic surgery | Endoscopic-assisted approaches for selected small glomus jugulare (limited role) |
| Intraoperative monitoring | Continuous CN monitoring + neuronavigation for safer skull base surgery |
| AI/ML | Radiomics from MRI for predicting tumor aggressiveness and SDH mutation status |
12. POSTOPERATIVE MANAGEMENT & REHABILITATION
Immediate Postoperative:
- ICU monitoring (BP, CN deficits, CSF leak)
- Wound care; lumbar drain management
- NG tube feeding (if swallowing unsafe)
- Chest physiotherapy (aspiration prevention)
Rehabilitation:
- Speech therapy: Dysphagia rehabilitation; voice therapy for CN X palsy
- Laryngeal medialization (thyroplasty): For persistent hoarseness/aspiration
- Vocal fold injection: Injection laryngoplasty for early phase
- Hearing rehabilitation: Bone-anchored hearing aid (BAHA) post-surgery
Follow-Up:
FOLLOW-UP PROTOCOL
─────────────────────────────────────────
• MRI skull base at 6 weeks postop (baseline)
• MRI at 6 months, then annually ×5 years
• 68Ga-DOTATATE PET/CT at 1–2 years
• Audiometry annually
• Endocrine review if functional tumor
• Genetic counseling + family screening
─────────────────────────────────────────
13. COMPLICATIONS OF SURGERY
| Complication | Frequency | Management |
|---|---|---|
| Facial nerve palsy | 15–30% | CN monitoring, decompression, eye care |
| Lower CN palsies (IX–XII) | 20–40% | Aspiration precautions, rehab |
| Hearing loss | Common (ITFA) | BAHA |
| CSF leak | 5–10% | Lumbar drain, repair |
| Wound infection/meningitis | Rare | Antibiotics |
| Stroke (ICA injury) | <5% | Prevention: BOT preop |
| Recurrence | 5–15% | Adjuvant RT, re-operation |
| Death | <2% | — |
14. PROGNOSIS
- Surgery: 80–90% long-term tumor control in experienced centers
- Radiosurgery (Gamma Knife): 90–95% tumor control; neurological preservation superior
- Combined (subtotal resection + adjuvant RT): Equivalent to radical surgery with lower morbidity
- Malignant glomus jugulare: Poor; 5-year survival ~50%
- Recurrence: 5–15%; higher in Fisch D tumors
(Ivan et al., J Neurosurg 2011, p. 1299–1305; Suárez et al., Head Neck 2013, p. 1195–1204)
15. MASTER SUMMARY FLOWCHART
╔══════════════════════════════════════════════════════════════════╗
║ GLOMUS JUGULARE — COMPLETE MANAGEMENT SUMMARY ║
╚══════════════════════════════════════════════════════════════════╝
PRESENTATION
↓
Pulsatile tinnitus + Red pulsatile mass behind TM + CN IX–XII palsies
↓
OTOSCOPY → Brown's sign +ve
↓
AUDIOMETRY → Conductive HL ± SNHL
↓
IMAGING
HRCT: Bony destruction (moth-eaten jugular fossa)
MRI: Salt-and-pepper sign + extent + ICA/venous
DSA: Tumor blush + feeding vessels + BOT
68Ga-DOTATATE PET/CT: Multifocal/metastatic disease
↓
BIOCHEMISTRY
24h urine catecholamines, VMA, plasma metanephrines
↓
CLASSIFICATION (Fisch A–D)
↓
MDT DECISION
↓
┌─────────────────────────────────────────────────────────────┐
│ TREATMENT OPTIONS │
├─────────────┬────────────────────┬─────────────────────────┤
│ WATCH & │ SURGERY │ RADIOTHERAPY │
│ WAIT │ │ │
│ │ Preop embolization │ Gamma Knife SRS │
│ Elderly, │ Functional: Alpha │ FSRT / EBRT │
│ asymptomatic│ blockade │ │
│ small tumor │ ITFA-A (Fisch's │ Indications: │
│ │ approach) │ Elderly, inoperable, │
│ Annual MRI │ + Combined │ residual/recurrent, │
│ │ approaches │ Fisch C–D │
└─────────────┴────────────────────┴─────────────────────────┘
↓
POSTOP REHABILITATION
CN palsy rehab + Speech therapy + BAHA + Voice rehab
↓
FOLLOW-UP
MRI 6/52 → 6/12 → annually × 5 yrs
Genetic counseling + family screening
REFERENCES
- Cummings Otolaryngology Head & Neck Surgery, 7th Ed. — Flint et al., Chapter on Paragangliomas, pp. 1730–1755
- Scott Brown's Otorhinolaryngology, Head & Neck Surgery, 8th Ed. — Chapter on Skull Base Tumors, Vol. 3
- Stell & Maran's Head and Neck Surgery — Chapter on Chemodectomas
- Dhingra's Diseases of Ear, Nose & Throat, 7th Ed. — Chapter on Glomus Tumors
- Hazarika P — Textbook of ENT and Head & Neck Surgery, 4th Ed.
- Zakir Hussain — ENT Short Cases / Head & Neck — Glomus tumors section
- Ivan ME et al. — Meta-analysis of tumor control rates for glomus jugulare tumors. J Neurosurg. 2011;114:1299–1305
- Suárez C et al. — Jugular and vagal paragangliomas: systematic study of management. Head Neck. 2013;35:1195–1204
- Janssen I et al. — 68Ga-DOTATATE PET/CT in HN paragangliomas. J Nucl Med. 2016;57:186–191
- Hinerman RW et al. — Radiotherapy for paragangliomas: 35-year experience. Head Neck. 2008;30:1431–1438
- Van Hulsteijn LT et al. — Radiotherapy for jugulotympanic paragangliomas: meta-analysis. Radiother Oncol. 2013;106:161–168
- Jimenez C et al. — Pembrolizumab in paragangliomas: Phase II trial. Cancers. 2020;12:2307
- Chun SG et al. — Five-fraction FSRT for glomus tumors. Stereotact Funct Neurosurg. 2014;92:153–159
RGUHS Exam Tip: For 50 marks, ensure coverage of: Definition + Anatomy (5M) → Clinical Features + Brown's sign (8M) → Fisch Classification (5M) → Investigations with imaging diagram (10M) → Management flowchart with surgical approaches (15M) → Radiosurgery vs Surgery comparison (5M) → Recent advances + Prognosis (2M). Always draw the Jugular Foramen anatomy diagram, the Salt-and-Pepper MRI sign explanation, and the Management Algorithm flowchart for maximum marks.
This is a shared conversation. Sign in to Orris to start your own chat.