Mantoux test full details for md exams 15marks
Reading File
search_medical_text
Mantoux test tuberculin skin test technique interpretation results
search_medical_text
Mantoux test false positive false negative BCG vaccination immunocompromised
search_medical_image
Mantoux test tuberculin skin test reading induration
GradeResponse
GradeResponse
GradeResponse
Here is a comprehensive, exam-ready answer on the Mantoux Test — structured for a 15-mark MD exam response.
MANTOUX TEST (Tuberculin Skin Test / PPD Test)
1. INTRODUCTION & HISTORICAL BACKGROUND
The Mantoux test (also called the Tuberculin Skin Test / TST / PPD test) is the standard intradermal test used for:
- Detection of latent tuberculosis infection (LTBI)
- Epidemiological surveys of TB prevalence
- Contact tracing of TB cases
- Screening high-risk populations
It was developed by Charles Mantoux in 1908, based on earlier work by Robert Koch with tuberculin.
2. PRINCIPLE
The Mantoux test is based on a Type IV (delayed-type) hypersensitivity reaction (cell-mediated immunity).
- When an individual has been previously exposed to Mycobacterium tuberculosis (or BCG), sensitized CD4+ T-lymphocytes are generated
- On intradermal injection of tuberculin (PPD), these T-cells recognize mycobacterial antigens and release cytokines (IFN-γ, IL-2)
- This recruits macrophages and other cells → induration at the site within 48–72 hours
3. ANTIGEN USED — TUBERCULIN / PPD
| Feature | Detail |
|---|---|
| Full name | Purified Protein Derivative (PPD) |
| Prepared from | Heat-sterilized, filtered, concentrated culture filtrate of M. tuberculosis |
| Standard preparation | PPD-S (Seibert's standard) — reference standard |
| Common formulation | PPD-RT23 (used in India and many countries) |
| Dose | 1 TU (tuberculin unit) or 2 TU depending on preparation |
| Volume injected | 0.1 mL |
4. TECHNIQUE
Materials Required
- 1 mL tuberculin syringe with short-bevel 26–27G needle
- PPD tuberculin (2 TU/0.1 mL of PPD-RT23 or 5 TU in some guidelines)
- 70% alcohol swab
Site
- Volar (flexor) surface of the left forearm — middle third (away from veins)
Method (Mantoux / Intradermal Injection)
- Clean the site with alcohol; allow to dry
- Hold the syringe at 5–15° angle to the skin, bevel upward
- Inject 0.1 mL intradermally (not subcutaneously)
- A pale wheal of 6–10 mm should appear immediately (confirms correct intradermal placement)
- Do NOT massage the site
Reading
- Read at 48–72 hours (optimal: 72 hours)
- Measure the transverse diameter of induration (not erythema) in millimeters, using a ruler or ballpoint pen technique
- Use fingertip palpation to delineate the indurated area
5. INTERPRETATION
Standard Cut-offs (ATS/CDC Criteria)
| Induration Size | Positive in These Groups |
|---|---|
| ≥ 5 mm | HIV-positive patients; Recent close contacts of active TB; Patients on immunosuppressants; CXR showing fibrotic changes consistent with old TB; Organ transplant recipients |
| ≥ 10 mm | Recent immigrants from high-prevalence countries; IV drug users; Healthcare workers; Residents/staff of high-risk congregate settings (prisons, nursing homes); Children <4 years; Lab workers working with mycobacteria; Patients with diabetes, CRF, silicosis, malnutrition, leukemia/lymphoma |
| ≥ 15 mm | All other persons with no risk factors |
In India (high-burden country), for children: ≥10 mm is considered positive regardless of BCG vaccination.
WHO / Indian Guidelines (Children)
| Induration | Interpretation |
|---|---|
| < 6 mm | Negative |
| 6–9 mm | Doubtful / Inconclusive |
| ≥ 10 mm | Positive |
6. POSITIVE MANTOUX TEST — SIGNIFICANCE
A positive test indicates prior exposure/sensitization to mycobacterial antigens. It does NOT by itself diagnose:
- Active tuberculosis
- Disease vs. latency
Further workup (CXR, sputum AFB, CBNAAT/GeneXpert, culture) is needed to confirm active disease.
7. GRADING OF REACTION
| Grade | Induration |
|---|---|
| Negative | < 5 mm |
| Weakly positive | 5–9 mm |
| Positive | 10–14 mm |
| Strongly positive | ≥ 15 mm |
| Vesiculation/necrosis | Highly significant — indicates high degree of sensitization |
8. FALSE POSITIVE RESULTS
Causes of false positivity (positive test in absence of TB infection):
| Cause | Mechanism |
|---|---|
| BCG vaccination | Cross-reactive antigens in PPD (most common cause in India) |
| NTM infection | M. kansasii, M. marinum, M. szulgai may cross-react |
| Improper test reading | Measuring erythema instead of induration |
| Repeated TST (booster effect) | Sensitization from prior testing |
As noted in Harrison's (p. 5252), PPD does not differentiate well between M. tuberculosis and some non-tuberculous mycobacteria (NTM), and BCG can cause significant cross-reactivity.
9. FALSE NEGATIVE RESULTS
Causes of false negativity (negative test despite TB infection):
Technical Causes
- Incorrect injection (subcutaneous instead of intradermal)
- Improper storage/handling of PPD (exposure to light, heat)
- Reading before 48 hours or after 72 hours
- Insufficient dose
Patient-Related Causes (Anergy)
| Cause | Examples |
|---|---|
| Immunosuppression | HIV/AIDS (most important), corticosteroids, cytotoxics |
| Severe/disseminated TB | Miliary TB — exhaustion of immune response |
| Malnutrition | Protein-energy malnutrition |
| Viral infections | Measles, EBV, varicella, influenza |
| Hematological malignancies | Leukemia, lymphoma, Hodgkin's disease |
| Sarcoidosis | Immunological anergy |
| Very old age / very young age | Immature or waning immunity |
| Recent TB (< 8–10 weeks) | Pre-sensitization window period |
| Live virus vaccines | MMR, oral polio (within 4–6 weeks) |
Key MD exam point: Advanced immunosuppression causes false-negative results in all immunologically-based TB tests including TST and IGRA (Primary Care Guidance for HIV, p. 14).
10. BOOSTER PHENOMENON
- Some individuals with remote TB infection may show a negative TST initially (waned immunity)
- Repeat testing 1–3 weeks later may show a positive result — this is called the booster effect
- This is NOT a new infection — it represents recall/boosting of previously sensitized T-cells
- Important in serial testing programs (e.g., healthcare workers) to establish baseline
11. TUBERCULIN CONVERSION
- A person previously negative who becomes positive on repeat testing
- Defined as: increase of ≥10 mm in induration size within a 2-year period
- Indicates recent TB infection
12. COMPARISON: MANTOUX TEST vs. IGRA
| Feature | Mantoux (TST) | IGRA (QuantiFERON-TB Gold / T-SPOT) |
|---|---|---|
| Antigen | PPD (mixed mycobacterial antigens) | ESAT-6, CFP-10, TB7.7 (TB-specific) |
| BCG cross-reactivity | Yes | No (preferred post-BCG) |
| Visit required | 2 (injection + reading) | 1 (blood draw) |
| Booster effect | Yes | No |
| Cost | Cheap | Expensive |
| Use in children <5 yrs | Preferred | Limited data; not routinely recommended |
| False negative in HIV | Yes | Yes (similar sensitivity) |
| Preferred in | Resource-limited settings, children | BCG-vaccinated, low return probability |
IGRA is preferred over TST in BCG-vaccinated patients and those unlikely to return for reading (Primary Care Guidance for HIV, p. 14).
13. CONTRAINDICATIONS
- Absolute: Previous severe reaction to tuberculin (vesiculation, anaphylaxis)
- Relative: Active skin disease at the injection site; severe immunosuppression (result unreliable)
Note: BCG vaccination is NOT a contraindication to TST.
14. SPECIAL SITUATIONS
| Situation | Recommendation |
|---|---|
| BCG vaccinated | Use IGRA; if TST done, ≥15 mm more likely true positive |
| HIV positive | Lower threshold (≥5 mm = positive); CD4 <200 may give false negative |
| Children (<5 years) | TST preferred over IGRA |
| Miliary TB | May be negative (anergy); do not rule out TB |
| Pregnancy | Safe to perform TST |
| Previous positive TST | Re-testing generally not recommended |
15. CLINICAL PHOTOGRAPH
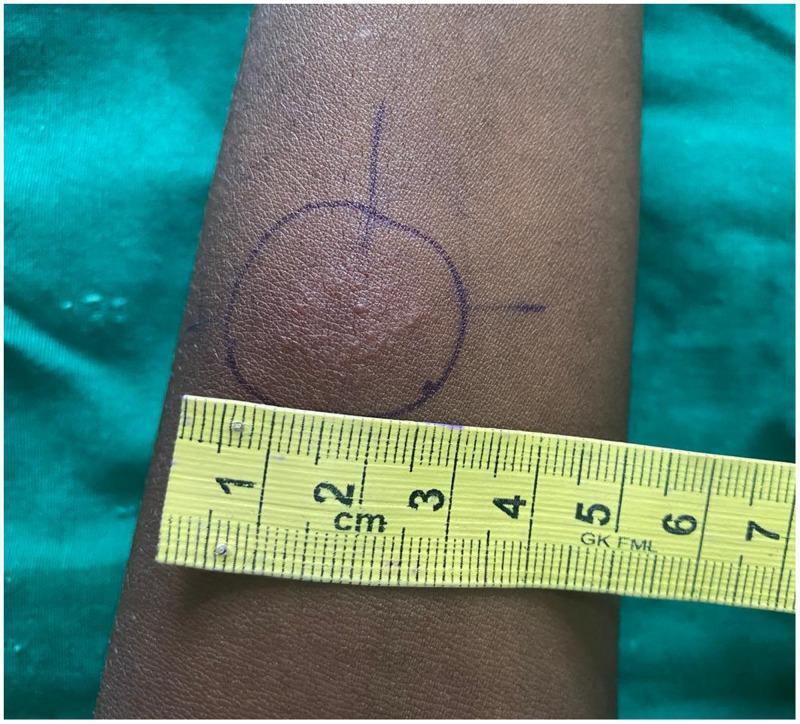
A positive Mantoux test on the volar forearm showing approximately 20 mm of induration and erythema, demarcated with ink for measurement.
16. USES OF MANTOUX TEST — SUMMARY
- Diagnosis of LTBI (most important use)
- Screening of contacts of active TB cases
- Epidemiological surveys (prevalence of TB infection)
- Diagnosis of TB in children (especially primary complex)
- Pre-treatment screening before starting anti-TNF therapy, transplant immunosuppression
- Assessment of cell-mediated immunity (e.g., in suspected immunodeficiency)
17. EXAM HIGH-YIELD POINTS
| Point | Fact |
|---|---|
| Antigen used | PPD (Purified Protein Derivative) |
| Volume injected | 0.1 mL intradermally |
| Standard dose | 2 TU (PPD-RT23) or 5 TU (PPD-S) |
| Site | Volar surface, left forearm |
| Reading time | 48–72 hours |
| What is measured | Induration (not erythema) |
| Standard positive | ≥10 mm (general); ≥5 mm (HIV/immunosuppressed) |
| Reaction type | Type IV hypersensitivity |
| Most common false positive | BCG vaccination |
| Most common false negative | HIV/AIDS (anergy) |
| Booster effect | Recall of waned immunity on repeat testing |
| Conversion | ≥10 mm increase within 2 years |
References:
- Harrison's Principles of Internal Medicine, 21st Edition, p. 5252
- Primary Care Guidance for Persons with HIV (CDC/GLGCA), p. 14
- ATS/CDC/IDSA Guidelines on LTBI Diagnosis and Treatment
- Park's Textbook of Preventive and Social Medicine
This is a shared conversation. Sign in to Orris to start your own chat.