Dengue fever, PSM.
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Loading Image
I now have comprehensive information to deliver a thorough PSM-focused answer on dengue fever. Let me compile it.
Dengue Fever — PSM Perspective
1. Agent (Etiological Agent)
Dengue is caused by four distinct but closely related RNA viruses — DENV-1, DENV-2, DENV-3, and DENV-4 — belonging to the genus Flavivirus, family Flaviviridae.
- Each serotype can cause the full spectrum of disease
- Infection with one serotype confers lifelong homotypic immunity but only 1–3 years of cross-protection against the other three serotypes
- Secondary infection with a heterologous serotype predisposes to more severe disease (antibody-dependent enhancement)
- A person can be infected up to 4 times in a lifetime
2. Epidemiology
Global Burden
- Dengue is the most common arboviral disease worldwide, with ~400 million infections per year (estimated); ~100 million symptomatic cases
- Approximately 75% of infections are asymptomatic
- Endemic in >100 countries across tropical and subtropical regions
- Accounts for the leading cause of fever in returning travelers from most tropical/subtropical destinations outside sub-Saharan Africa (where malaria predominates)
Distribution
- Primarily endemic across the tropical belt: South/Southeast Asia, Latin America, the Caribbean, Pacific Islands, and parts of Africa
- The geographic distribution largely overlaps with Aedes mosquito distribution
- US territories (Puerto Rico, Guam, American Samoa, USVI, Northern Mariana Islands) experience repeated outbreaks
- Climate change is driving expansion of endemic zones
Re-emergence Pattern
Despite having only a human→mosquito→human transmission cycle (no direct human-to-human spread), dengue re-emergences can be explosively rapid — during simultaneous 1977 dengue and influenza outbreaks in Puerto Rico, household clustering was greater for dengue than for influenza. — Harrison's Principles of Internal Medicine, 22E
3. Host
- All ages susceptible; however children bear the greatest burden of severe disease (DHF/DSS), particularly in hyperendemic areas
- Pre-existing immunity from a prior serotype is the key host determinant of severe disease
- Immunocompromised individuals, pregnant women, and infants are at higher risk of complications
- Vertical transmission rate ~20% (higher near delivery)
4. Vector (Epidemiological Triad — Environment)
| Feature | Details |
|---|---|
| Primary vector | Aedes aegypti |
| Secondary vectors | Aedes albopictus, Aedes polynesiensis |
| Breeding habit | Small collections of stagnant, clean water — peridomestic containers (flower pots, tyres, water storage vessels, even toilet bolt-holes) |
| Biting time | Daytime biter (peak early morning and late afternoon) |
| Flight range | Short (~100 metres) |
| Extrinsic incubation period (EIP) | 8–12 days (temperature-dependent; ~7 days at >32°C, ≥12 days at 30°C) |
| Intrinsic incubation period | 3–14 days (typically 4–7 days) in humans |
| Viremia in human host | 4–5 days |
Temperature & Vector Biology
The graph below (from Harrison's) illustrates how temperature profoundly affects Ae. aegypti biology and dengue transmission:
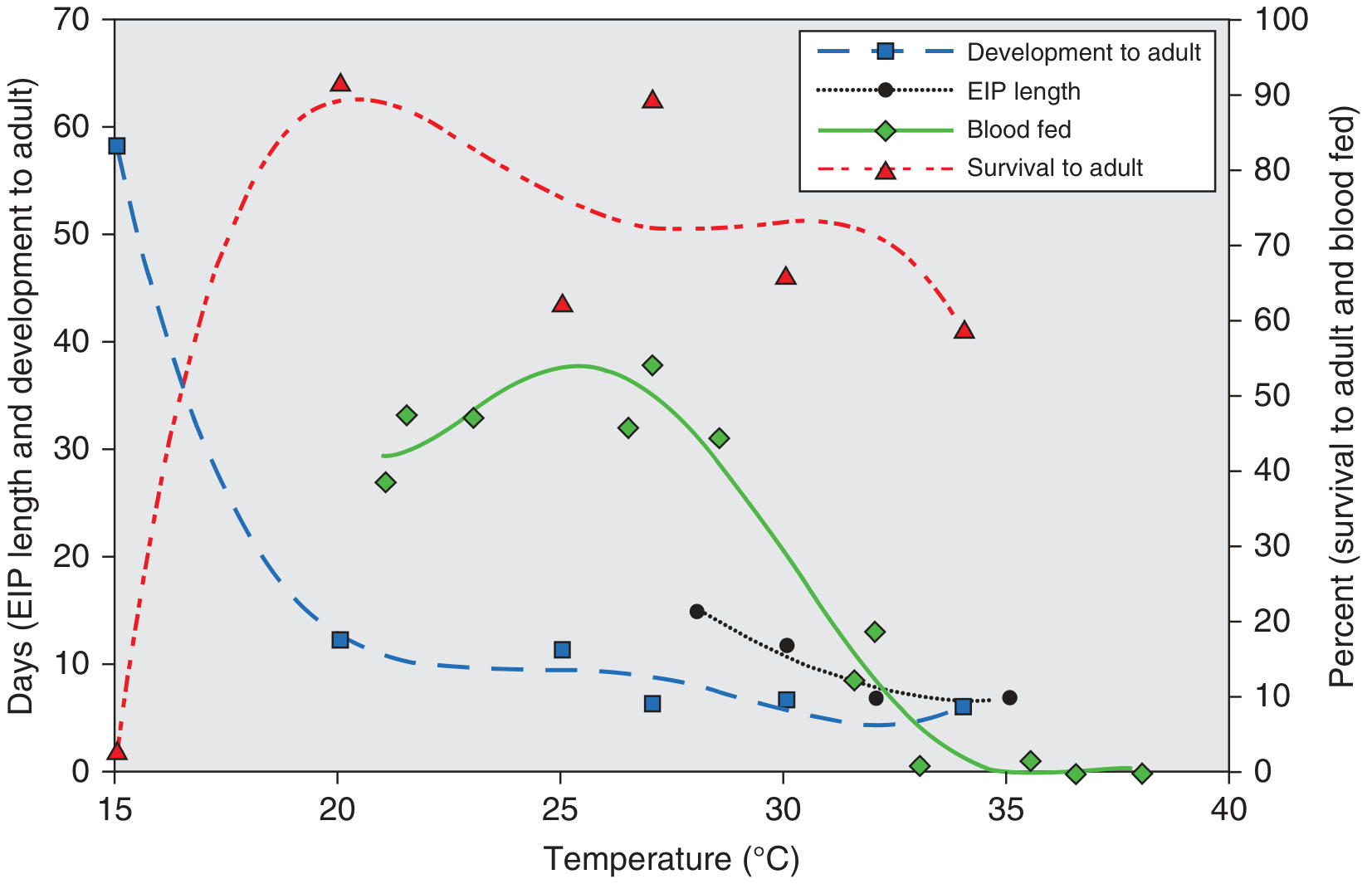
- <15°C or >36°C: mosquito feeding substantially reduced
- Optimal transmission: ~32°C (shorter EIP + higher feeding frequency + faster larval development)
- Peak relative humidity is a strong co-predictor of outbreaks — Harrison's Principles of Internal Medicine, 22E
5. Modes of Transmission
| Route | Notes |
|---|---|
| Mosquito bite (primary) | Aedes aegypti bite — accounts for virtually all cases |
| Vertical (maternal→fetal) | ~20%; higher risk peripartum |
| Blood/organ donation | Rare but documented |
| Breastfeeding | Very rare |
| Needlestick/mucocutaneous | Healthcare-associated, rare |
| Sexual | Possible but extremely rare |
No direct person-to-person respiratory transmission.
6. Clinical Classification (WHO 2009)
WHO 2009 Classification:
A. Dengue without Warning Signs
Fever + ≥2 of: nausea/vomiting, rash, aches and pains, leukopenia, positive tourniquet test
B. Dengue with Warning Signs
Dengue (above) + any of:
- Abdominal pain/tenderness
- Persistent vomiting
- Clinical fluid accumulation (ascites, pleural effusion)
- Mucosal bleeding
- Lethargy/restlessness
- Liver enlargement >2 cm
- Rapid decline in platelets with rising haematocrit
C. Severe Dengue
Dengue + at least one of:
- Severe plasma leakage → shock or respiratory distress
- Severe bleeding (clinician-assessed)
- Severe organ involvement: AST/ALT ≥1000 IU/L, impaired consciousness, heart/organ failure
(Previously classified as DHF Grades I–IV and DSS)
7. Clinical Phases
| Phase | Timing | Key Features |
|---|---|---|
| Febrile | Days 1–3 | Sudden high fever, myalgia, arthralgia ("breakbone fever"), retro-orbital pain, facial erythema, maculopapular rash, leukopenia |
| Critical | Days 3–7 (defervescence) | Plasma leakage, haemoconcentration (rising Hct), thrombocytopenia; lasts 24–48 hrs; risk of DHF/DSS |
| Recovery/Convalescent | Days 7+ | Gradual reabsorption of leaked fluids, improvement; risk of fluid overload if over-hydrated during critical phase |
8. Diagnosis
| Test | Timing | Notes |
|---|---|---|
| NS1 antigen (ELISA) | Days 1–5 | Earliest marker |
| RT-PCR | Days 1–5 | Most sensitive early; quantitative |
| IgM ELISA | From day 5 | Diagnostic in secondary infection from day 1–2 |
| IgG | Later / paired sera | Confirms past exposure; useful epidemiologically |
| Tourniquet test | Clinical | ≥10 petechiae/inch² = positive |
| CBC | Throughout | Leukopenia, thrombocytopenia, rising Hct |
9. Prevention & Control (PSM Core)
A. Vector Control (Most Important)
Source reduction (anti-larval measures):
- Remove/empty/cover water-holding containers weekly
- Abate (temephos) — larvicide in water containers
- Biological control: Bacillus thuringiensis israelensis (Bti), larvivorous fish (Gambusia)
- Cover water storage containers
- Proper solid waste disposal (old tyres, cans)
Anti-adult measures:
- Indoor residual spraying (IRS) — less effective for Ae. aegypti (daytime/outdoor biter)
- Fogging/space spraying (malathion, pyrethroids) — for outbreak control
- Insecticide-treated curtains/screens
Personal protection:
- Repellents (DEET, picaridin)
- Protective clothing (long sleeves, light colours)
- Bed nets (less useful — daytime biter)
- Window/door screens, air conditioning
Novel approaches:
- Sterile insect technique (SIT)
- Wolbachia-infected mosquitoes — reduces dengue transmission by impairing virus replication in the vector; field trials ongoing with promising results
- Genetically modified mosquitoes (OX513A — self-limiting males)
B. Vaccine
| Vaccine | Type | Status |
|---|---|---|
| Dengvaxia (CYD-TDV) | Tetravalent live-attenuated (Sanofi Pasteur) | FDA-approved for seropositive individuals aged 9–16 in endemic regions; contraindicated in seronegative (risk of severe disease on first natural infection) |
| Qdenga (TAK-003) | Tetravalent live-attenuated (Takeda) | Approved in EU and several endemic countries for ≥4 years; useful in seronegative recipients |
Key PSM point: Dengvaxia requires pre-vaccination screening for prior dengue exposure — a critical public health policy lesson after the Philippines (2016–2017) controversy where seronegative children received the vaccine and experienced increased severe dengue risk.
C. Surveillance
- Passive surveillance: mandatory notification of dengue cases
- Active surveillance: sentinel sites, serosurveys
- Entomological indices for monitoring vector density:
- Breteau Index (BI) — number of positive containers per 100 houses (most informative)
- House Index (HI) — % houses with Ae. aegypti larvae
- Container Index (CI) — % water-holding containers positive
- Pupal/ovitrap index — sensitive early-warning tools
D. Case Management (PSM Level)
- Supportive treatment — no specific antiviral
- Fluid management is critical during the critical phase
- Avoid NSAIDs/aspirin (risk of bleeding)
- Platelet transfusion only for active severe bleeding
10. Key Epidemiological Facts (High-Yield)
| Feature | Value |
|---|---|
| Causative agent | DENV 1–4 (Flavivirus) |
| Vector | Aedes aegypti (primary) |
| Incubation period | 3–14 days (typically 4–7 days) |
| Infectious period (viremia) | 4–5 days |
| Reservoir | Humans (mainly); non-human primates (sylvatic cycle — Africa, SE Asia) |
| Season | Post-monsoon / rainy season (peak vector breeding) |
| % asymptomatic | ~75% |
| DHF pathogenesis | Antibody-dependent enhancement (ADE) in secondary heterotypic infection |
| Notification | Notifiable disease in most endemic countries |
11. Dengue vs. Malaria (Differential — PSM Perspective)
| Feature | Dengue | Malaria |
|---|---|---|
| Vector | Aedes (day-biting) | Anopheles (night-biting) |
| Pattern | Continuous high fever | Periodic/relapsing |
| Rash | Common | Rare |
| Leukopenia | Characteristic | May have leukocytosis |
| Thrombocytopenia | Marked | Present but milder |
| Liver enlargement | May occur | Common |
Sources: Harrison's Principles of Internal Medicine, 22E | Red Book 2021 (AAP Committee on Infectious Diseases)
This is a shared conversation. Sign in to Orris to start your own chat.