23 year old male patient complaints of low libido, nor morning erections and lack of masculine mascular features, although has decent beard and pubic hair growth, explain testosterone physiology and the diagnosis and treatment for this condition
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching PubMed
"male hypogonadism" AND "testosterone" AND diagnosis treatment
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
testosterone biosynthesis pathway Leydig cells HPG axis
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive information to compile a detailed, well-structured answer. Let me synthesize everything.
Testosterone Physiology, Diagnosis & Treatment: Hypogonadotropic Hypogonadism in a Young Male
Clinical Interpretation
This 23-year-old male presents with low libido, absent morning erections, and poor muscular development — classic symptoms of androgen deficiency. The key clinical clue is that he has pubic and beard hair but lacks masculine muscle mass and erectile function. This pattern points to partial or secondary (hypogonadotropic) hypogonadism, where adrenal androgens (DHEA/androstenedione) are sufficient to produce axillary/pubic/facial hair (which are less androgen-dependent), but testicular testosterone is insufficient to drive libido, erections, and anabolic muscle growth (which require higher testosterone levels).
Part 1 — Testosterone Physiology
1.1 Synthesis
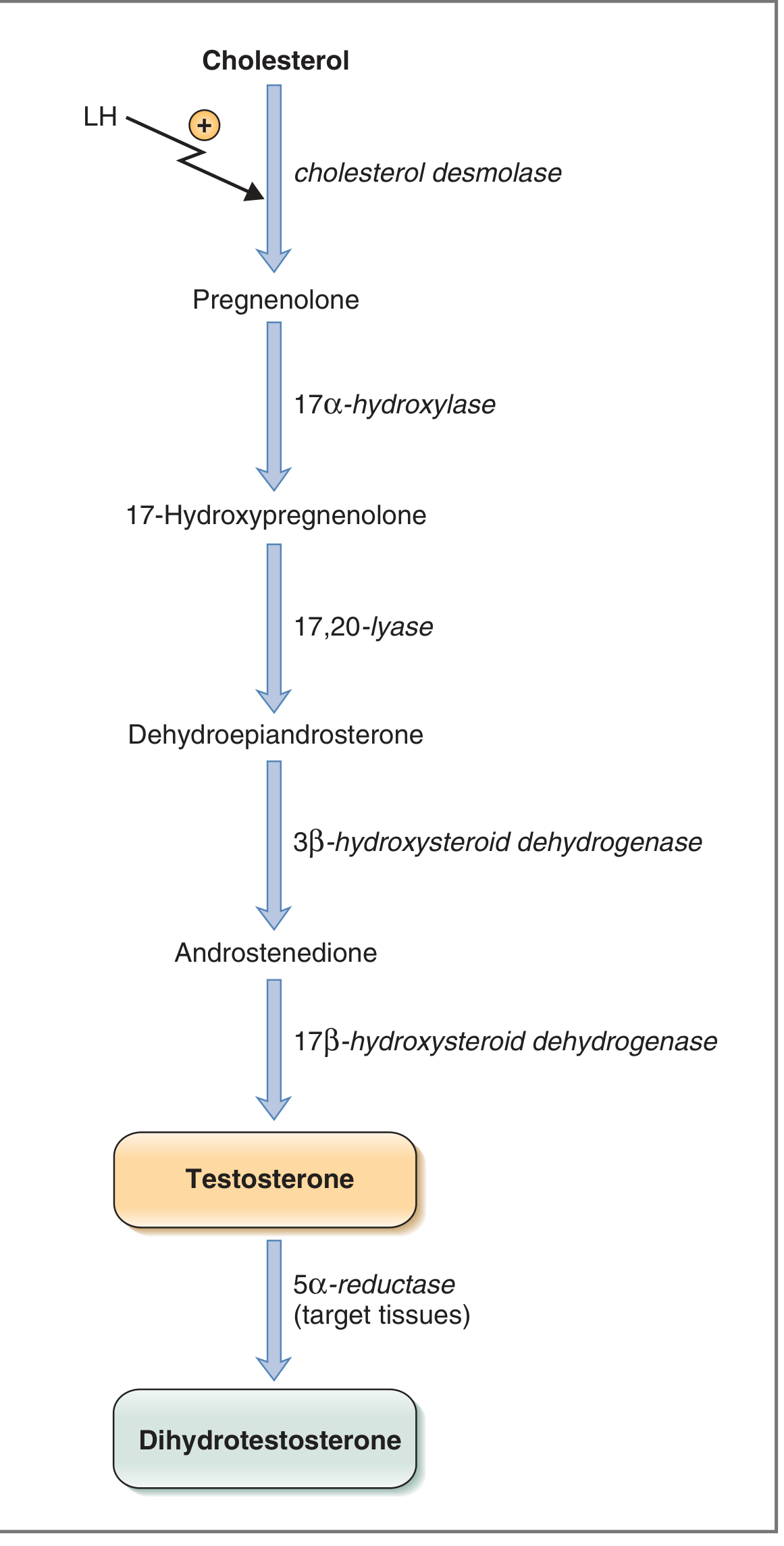
Testosterone is synthesized by the Leydig cells of the testes. The steroidogenic pathway proceeds from cholesterol:
Cholesterol → Pregnenolone → 17-Hydroxypregnenolone → DHEA → Androstenedione → Testosterone
Key enzymes:
- Cholesterol desmolase (rate-limiting; activated by LH)
- 17α-hydroxylase
- 17,20-lyase
- 3β-hydroxysteroid dehydrogenase
- 17β-hydroxysteroid dehydrogenase — unique to testes; converts androstenedione to testosterone
The testes lack 21β-hydroxylase and 11β-hydroxylase, so they cannot make glucocorticoids or mineralocorticoids. In target tissues (prostate, skin), testosterone is further converted to dihydrotestosterone (DHT) by 5α-reductase — DHT is ~3–5× more potent and mediates external genital virilization and male-pattern hair growth.
1.2 Transport
98% of circulating testosterone is bound to plasma proteins: ~44% tightly to sex hormone-binding globulin (SHBG) and ~54% loosely to albumin. Only free (unbound) testosterone (~2%) is biologically active. Estrogens stimulate SHBG synthesis; androgens inhibit it. — Costanzo Physiology, 7th Ed.
1.3 Mechanism of Action
Testosterone (and DHT) enter target cells and bind androgen receptors (AR) in the cytoplasm. The hormone-receptor complex translocates to the nucleus and binds nuclear DNA, activating DNA-RNA transcription. This increases RNA polymerase activity, protein synthesis, and ultimately cell growth — the anabolic basis for muscle and bone effects. — Guyton & Hall Medical Physiology
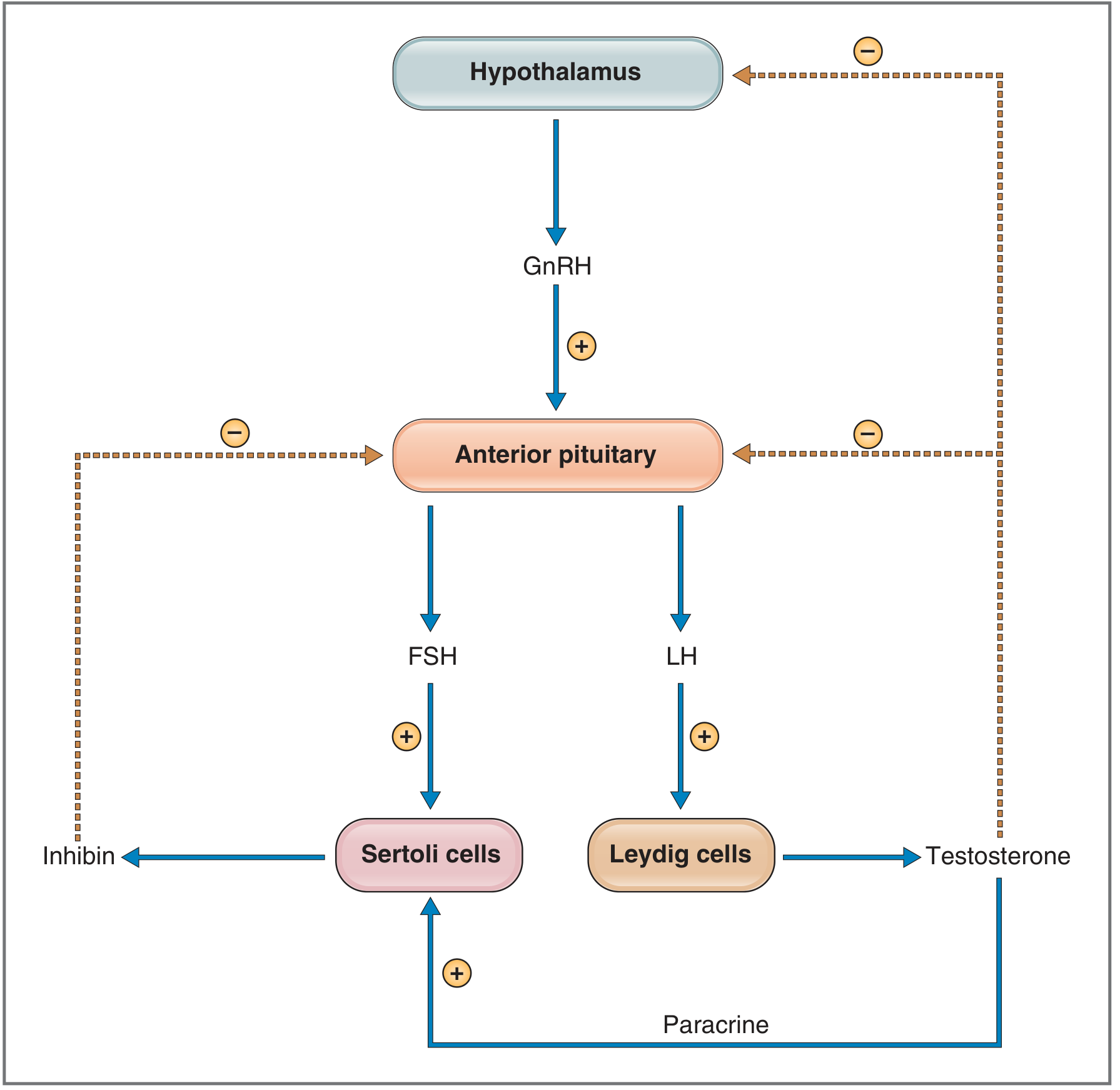
1.4 The HPG Axis (Hypothalamic-Pituitary-Gonadal Axis)
The entire system is regulated by a hierarchical feedback loop:
| Level | Hormone | Action |
|---|---|---|
| Hypothalamus | GnRH (pulsatile decapeptide, arcuate nucleus) | Stimulates anterior pituitary |
| Anterior pituitary | LH | Stimulates Leydig cells → testosterone |
| Anterior pituitary | FSH | Stimulates Sertoli cells → spermatogenesis |
| Testes — Leydig | Testosterone | Negative feedback on hypothalamus & pituitary (↓GnRH, ↓LH) |
| Testes — Sertoli | Inhibin | Negative feedback on pituitary (↓FSH) |
Critical point: GnRH must be secreted pulsatily — continuous GnRH downregulates its own receptor and paradoxically suppresses LH/FSH (the principle behind GnRH agonists in prostate cancer). — Costanzo Physiology 7th Ed.
1.5 Actions of Testosterone
| Target | Effect |
|---|---|
| Muscle | ↑ protein synthesis → muscle mass and strength |
| Bone | ↑ bone density; epiphyseal closure at puberty |
| Larynx | Voice deepening |
| Skin/hair | DHT → male pattern body/facial hair, sebum |
| Genitalia | External virilization (DHT), libido, erections |
| Erythropoiesis | ↑ RBC count ~15–20% (explains higher hematocrit in men) |
| Metabolic | ↑ BMR ~5–15%; sodium and water retention |
| CNS | Libido, sexual motivation, mood |
Part 2 — Diagnosis
2.1 Clinical Classification
Male hypogonadism is divided by the anatomical level of the defect:
| Type | Level | LH/FSH | Testosterone | Pattern |
|---|---|---|---|---|
| Primary (Hypergonadotropic) | Testis | ↑ HIGH | ↓ Low | Testis fails; pituitary compensates |
| Secondary (Hypogonadotropic) | Hypothalamus/Pituitary | Low or inappropriately normal | ↓ Low | Signal fails; testis is not stimulated |
This patient's preserved beard and pubic hair growth (driven partly by adrenal androgens and less testosterone-dependent DHT pathways) alongside markedly impaired libido, erections, and muscle mass in a 23-year-old strongly favors secondary/hypogonadotropic hypogonadism — the most likely diagnosis being Congenital Hypogonadotropic Hypogonadism (CHH) with or without anosmia (Kallmann syndrome if anosmia is present).
2.2 Most Likely Diagnoses
A. Kallmann Syndrome / Congenital Hypogonadotropic Hypogonadism (CHH)
The hallmark diagnosis for a young adult male with absent puberty/partial virilization + low testosterone + low/normal LH/FSH.
- Caused by GnRH neuron migration failure during embryogenesis — GnRH neurons arise from the olfactory placode and fail to migrate to the hypothalamus
- Associated with anosmia or hyposmia (Kallmann) or normal smell (normosmic CHH)
- Genetic causes include mutations in: ANOS1 (KAL1), FGFR1, FGF8, PROKR2, CHD7, and many others (Harrison's 22E)
- Phenotype: small testes, absent/reduced secondary sexual characteristics, eunuchoid proportions, cryptorchidism possible; beard and pubic hair may be partially present from adrenal androgens
B. Klinefelter Syndrome (47,XXY) — Primary Hypogonadism
- Most common chromosomal cause of male hypogonadism (1 in 600 males)
- Small firm testes, gynecomastia, tall stature, azoospermia
- LH and FSH are elevated (distinguishes it from CHH)
- Less likely here given no gynecomastia is mentioned and normal beard, but karyotype should be checked
2.3 Diagnostic Workup
Step 1 — Confirm hypogonadism:
- Total testosterone (morning fasting, 8–10 AM; confirm on two separate occasions if borderline)
- Normal adult male range: ~300–1000 ng/dL (10.4–34.7 nmol/L)
- If low, proceed to Step 2
Step 2 — Localize the defect:
- LH and FSH:
- Low/normal LH + low T → Secondary (hypogonadotropic) hypogonadism
- High LH + low T → Primary (hypergonadotropic) hypogonadism
- SHBG and free testosterone if SHBG suspected to be elevated
Step 3 — Identify cause:
| Test | Reason |
|---|---|
| Karyotype | Rule out Klinefelter (47,XXY); if LH elevated + small testes |
| MRI pituitary (sellar) | Rule out pituitary/hypothalamic tumor, Rathke cyst, hemochromatosis |
| Smell test (olfactometry) | Anosmia → Kallmann syndrome |
| Prolactin | Hyperprolactinemia suppresses GnRH |
| FSH, inhibin B | Sertoli cell function / spermatogenic reserve |
| Testicular ultrasound | Volume, cryptorchidism, tumor |
| GnRH stimulation test | Differentiates hypothalamic from pituitary origin |
| Ferritin / transferrin | Rule out hemochromatosis (iron deposits in pituitary) |
As per Harrison's 22E: "A karyotype should be performed in men with low testosterone and elevated LH to diagnose Klinefelter syndrome. Men who have a low testosterone but 'inappropriately normal' or low LH levels have secondary hypogonadism; their defect resides at the hypothalamic-pituitary level."
Part 3 — Treatment
Treatment depends on whether fertility is desired or only virilization/symptom relief.
3.1 If Fertility Is NOT Currently Desired — Testosterone Replacement Therapy (TRT)
The goal is restoration of serum testosterone to mid-normal range (~400–700 ng/dL).
| Formulation | Dose | Notes |
|---|---|---|
| Testosterone enanthate or cypionate (IM/SC) | 150–200 mg IM every 2 weeks, or 75–100 mg weekly SC | Gold standard; cost-effective |
| Testosterone gel (1–2%) | 50–100 mg/day topically | Avoids peaks/troughs; risk of transference |
| Testosterone undecanoate (IM long-acting) | 750 mg IM; repeat at 4 wks then every 10 wks | Stable levels; clinic-administered |
| Testosterone patch | 2.5–5 mg/day | Mimics diurnal rhythm |
| Testosterone buccal | 30 mg twice daily | Less convenient |
Effects to expect with TRT:
- Libido restored within 3–6 weeks
- Morning erections return
- Muscle mass increases over 3–6 months
- Energy, mood improve
- Bone density increases over 12–24 months
Monitor:
- Testosterone levels (trough before injection; mid-cycle for gels)
- Hematocrit (target <54%; TRT raises RBC count)
- PSA (not relevant at 23, but baseline useful)
- Testicular size (may atrophy further — warn patient)
- Fertility: TRT suppresses FSH/LH and will impair spermatogenesis — must counsel before starting
3.2 If Fertility IS Desired — Gonadotropin Therapy
This is the preferred treatment for CHH/Kallmann if the patient wishes to have children. TRT will suppress pituitary output and cause testicular atrophy.
| Drug | Mechanism | Dose |
|---|---|---|
| hCG (human chorionic gonadotropin) | LH-mimetic → stimulates Leydig cells → testosterone production AND testicular growth | 1500–3000 IU SC 2–3×/week |
| rFSH or urinary FSH | Added after 6 months of hCG if sperm count remains low | 75–150 IU SC 3×/week |
| GnRH pump (pulsatile) | Gold standard for hypothalamic CHH; restores physiologic pulsatile GnRH | 5–20 ng/kg per pulse every 90–120 min via portable pump |
Harrison's 22E notes: "Optimization of future fertility potential may require initial treatment with hCG to induce development of secondary sex characteristics as well as spermatogenesis to enable harvesting and cryopreservation of sperm."
For normosmic CHH: some patients achieve spontaneous recovery when treatment is stopped — a therapeutic withdrawal trial every 12–18 months is recommended.
3.3 Novel/Emerging Options
A 2025 systematic review and meta-analysis (PMID: 41066380) evaluated clomiphene or enclomiphene citrate for male hypogonadism — these selective estrogen receptor modulators (SERMs) block estrogen negative feedback at the hypothalamus/pituitary, thereby increasing endogenous GnRH → LH → testosterone. They preserve fertility and testicular volume, making them an attractive option in secondary hypogonadism where the testes are intact.
Summary Algorithm
23-yr male: low libido, no morning erections, poor muscle mass
↓
Morning testosterone (×2 confirmed)
↓
LOW (<300 ng/dL)
↓
Check LH + FSH
↙ ↘
LH/FSH LOW or LH/FSH HIGH
inappropriately normal (>10 IU/L)
↓ ↓
Secondary Hypogonadism Primary Hypogonadism
(CHH / Kallmann / pituitary) (Klinefelter; orchitis)
↓ ↓
MRI pituitary Karyotype
Olfactory testing Testicular USS
Prolactin
↓
Fertility desired?
YES → hCG ± FSH / GnRH pump
NO → Testosterone replacement
Key References
- Costanzo Physiology, 7th Ed. — Testosterone synthesis, HPG axis, negative feedback
- Guyton & Hall Textbook of Medical Physiology — Testosterone actions, hypogonadism
- Harrison's Principles of Internal Medicine 22E (2025) — Hypogonadotropic hypogonadism, Kallmann syndrome, TRT
- Goldman-Cecil Medicine — Male hypogonadism classification and management
- Hohl A et al. (2025). Clomiphene or enclomiphene for male hypogonadism. Arch Endocrinol Metab. [PMID: 41066380]
This is a shared conversation. Sign in to Orris to start your own chat.