Details (1) Malformations of the musculoskeletal system in children. muscular torticollis. 2.Hernias: hernias of the anterior abdominal wall, inguinal hernias-D
muscular torticollis children congenital deformity diagnosis treatment
inguinal hernia anterior abdominal wall hernia children diagnosis management
congenital muscular torticollis sternocleidomastoid fibrosis etiology classification
inguinal hernia types direct indirect femoral anatomy repair hernioplasty
1. Malformations of the Musculoskeletal System in Children: Muscular Torticollis
Definition
Etiology & Pathogenesis
| Mechanism | Details |
|---|---|
| Intrauterine moulding | Abnormal fetal position → compartment syndrome → ischemia of SCM |
| Birth trauma | Difficult delivery (forceps, breech) → rupture of SCM → hematoma → fibrosis |
| Venous occlusion | Compression of SCM vasculature in utero → ischemic fibrosis |
| Intrinsic myopathy | Abnormal mesenchymal development of SCM |
Classification
- Postural torticollis – No palpable mass; only positional preference; most benign
- Muscular torticollis (SCM tightness) – Tight SCM band without mass
- SCM mass (sternocleidomastoid tumor/pseudotumor) – Palpable fibrotic mass within SCM; most common form; identified in first few weeks of life
Clinical Features
- Head tilt to the affected side, chin rotated to opposite side
- Palpable firm, non-tender mass within SCM (in the mass type), usually 1–3 cm, appearing at 2–4 weeks of age and typically regressing by 6 months
- Limited range of motion of the neck
- Facial asymmetry (plagiocephaly): if untreated, facial flattening on the ipsilateral side (due to sleeping on same side), orbital and zygomatic asymmetry
- Associated anomalies: developmental dysplasia of the hip (DDH) (2–3%), foot deformities (metatarsus adductus), obstetric brachial plexus palsy
Diagnosis
- Ultrasound (USG) of neck: gold standard for soft tissue — confirms SCM thickening/fibrosis vs. other masses
- X-ray cervical spine / AP skull: to exclude osseous anomalies (Klippel-Feil syndrome, atlanto-axial instability, cervical hemivertebra)
- MRI: reserved for atypical or non-resolving cases
Differential diagnoses: Klippel-Feil syndrome (bony fusion), atlantoaxial rotatory subluxation, ocular torticollis (squint), Sandifer syndrome (GERD), cervical lymphadenitis, osseous tumors
Treatment
Conservative (First-line, <1 year of age)
- Passive stretching exercises by physiotherapist and trained parents: lateral tilt and rotation stretches, 3–4 sessions/day
- Active repositioning: encourage the child to turn toward the affected side
- Botulinum toxin A injections: adjunct in resistant cases to temporarily relax SCM
- Success rate: ~90% if started within first 3 months of life
Surgical (Indicated when conservative fails or age > 1 year with significant restriction)
- Distal release (most common): SCM release from clavicular and sternal heads
- Bipolar release: origin (mastoid) + insertion for severe cases
- Endoscopic release: minimally invasive option
- Post-operative: collar/brace + physiotherapy
Complications of Untreated CMT
- Permanent facial asymmetry / cranial plagiocephaly
- Scoliosis (compensatory)
- Cervical osteoarthritis in adulthood
- Psychosocial effects
2. Hernias of the Anterior Abdominal Wall & Inguinal Hernias
Definition
Anatomy of the Anterior Abdominal Wall
- Skin
- Subcutaneous fat (Camper's fascia)
- Scarpa's fascia (deep membranous layer)
- External oblique aponeurosis
- Internal oblique muscle
- Transversus abdominis
- Transversalis fascia
- Extraperitoneal fat
- Peritoneum
- Inguinal canal
- Umbilicus
- Linea alba (epigastric region)
- Femoral canal
- Incisional scars
- Spigelian line
General Hernia Anatomy
| Component | Description |
|---|---|
| Sac | Peritoneal diverticulum: mouth → neck → body → fundus |
| Contents | Omentum (omentocele), small bowel (enterocele), large bowel, bladder |
| Coverings | Layers of the abdominal wall through which the hernia passes |
Types of Anterior Abdominal Wall Hernias
1. Umbilical Hernia
- In infants: due to failure of umbilical ring to close; very common; most close spontaneously by age 3–4 years; repair if still present at 4–5 years
- In adults: due to raised intra-abdominal pressure (obesity, ascites, multiparity)
- Repair: Mayo repair (vest-over-pants technique) or mesh repair
2. Paraumbilical Hernia
- Through the linea alba just above/below umbilicus
- Common in obese, multiparous women
- Does NOT spontaneously resolve → surgical repair advised
- High strangulation risk
3. Epigastric Hernia
- Through defects in linea alba between xiphoid and umbilicus
- Often contains extraperitoneal fat (no true sac)
- Small but painful; surgical repair
4. Incisional / Ventral Hernia
- Through previous surgical scars
- Risk factors: wound infection, obesity, poor technique, malnutrition, steroids
- Management: mesh repair (open or laparoscopic); recurrence rate higher than primary hernias
5. Spigelian Hernia
- Through semilunar line (lateral border of rectus sheath) at level of arcuate line
- Interparietal hernia (lies between layers) — clinically occult; diagnosed by USS or CT
- Surgical repair required
6. Lumbar Hernia
- Petit's triangle (inferior lumbar) or Grynfelt's triangle (superior lumbar)
- Rare; repair needed
Management Principles (Abdominal Wall Hernias)
- Not all hernias need immediate repair
- Surgery is recommended when:
- Complications are likely (strangulation, obstruction) — especially narrow-necked hernias
- All femoral hernias should be repaired
- Symptomatic or irreducible hernias
- Increasing difficulty in reduction or increasing size
- Younger adult patients (lifetime complication risk)
- Surgery may be deferred only if coexisting medical factors create prohibitive surgical risk
Inguinal Hernias — Detailed
Anatomy of the Inguinal Canal
| Feature | Details |
|---|---|
| Length | ~4 cm; runs obliquely superomedially |
| Direction | From deep (internal) inguinal ring → superficial (external) inguinal ring |
| Anterior wall | External oblique aponeurosis (+ internal oblique laterally) |
| Posterior wall | Transversalis fascia (+ conjoint tendon medially) |
| Floor | Inguinal (Poupart's) ligament |
| Roof | Arched fibers of internal oblique + transversus abdominis |
| Contents | Spermatic cord (male) / round ligament (female) + ilioinguinal nerve |
Hesselbach's Triangle (Direct Hernia site)
- Laterally: inferior epigastric vessels
- Medially: lateral border of rectus abdominis
- Inferiorly: inguinal ligament
Classification: Direct vs. Indirect vs. Femoral
| Feature | Indirect Inguinal | Direct Inguinal | Femoral |
|---|---|---|---|
| Pathway | Through deep ring, along inguinal canal | Directly through posterior wall (Hesselbach's triangle) | Through femoral canal (below inguinal ligament) |
| Relationship to inferior epigastric vessels | Lateral | Medial | Below & medial to femoral vessels |
| Sac | Patent processus vaginalis | Acquired weakness of transversalis fascia | Peritoneal protrusion into femoral canal |
| Age/Sex | All ages, M > F; common in children | Middle-aged/elderly men | More common in women (wider pelvis) |
| Congenital basis | Yes (patent processus vaginalis) | No (acquired) | No (acquired) |
| Strangulation risk | Moderate | Low | High (narrow femoral ring) |
| Reducibility | Usually reducible | Usually reducible | Often irreducible |
| Scrotal descent | Can reach scrotum | Rarely | Never |
Indirect Inguinal Hernia — Special Considerations in Children
- Most common hernia in children and infants
- Due to patent processus vaginalis (PPV): failure of obliteration after testicular descent
- Right > left (right testis descends later)
- Bilateral in ~10% (higher in premature infants)
- Associated conditions: undescended testis, hydrocele
- Risk of incarceration/strangulation is highest in infancy (<1 year)
- Treatment: elective herniotomy (high ligation of sac) — NOT herniorrhaphy (no repair of floor needed in children)
- In infants: surgery within days of diagnosis due to strangulation risk
Clinical Features of Inguinal Hernia
- Reducible: lump in groin that disappears on lying down or manual pressure; expansile cough impulse
- Irreducible (incarcerated): lump that cannot be reduced; tender; no cough impulse
- Strangulated: irreducible + signs of bowel obstruction + ischemia (severe pain, vomiting, absent bowel sounds, peritonism) — surgical emergency
Diagnosis
- Primarily clinical
- Ultrasound: for occult or uncertain cases
- CT/MRI: for complex, recurrent, or bilateral cases
Surgical Repair (Hernioplasty)
Open Techniques
| Technique | Key Features |
|---|---|
| Lichtenstein (tension-free mesh) | Gold standard for adults; mesh placed on posterior wall; low recurrence (<1%) |
| Shouldice | Multi-layer transversalis fascia repair; best non-mesh option |
| Bassini | Classic; conjoint tendon to inguinal ligament; higher recurrence |
| Herniotomy (children) | Ligation of sac at deep ring; no floor repair needed |
Laparoscopic Techniques
| Technique | Approach |
|---|---|
| TAPP (Trans-Abdominal Pre-Peritoneal) | Enters peritoneal cavity; mesh behind posterior wall |
| TEP (Totally Extra-Peritoneal) | Stays in extraperitoneal space; avoids peritoneal entry; preferred |
Complications of Inguinal Hernia Repair
| Complication | Notes |
|---|---|
| Hematoma/seroma | Common; most resolve spontaneously |
| Wound infection | 1–2%; higher with mesh |
| Chronic groin pain (inguinodynia) | Nerve entrapment (ilioinguinal, genitofemoral) |
| Recurrence | 0.5–5% with mesh; higher with tissue repairs |
| Testicular atrophy | Damage to testicular vessels; rare |
| Vas deferens injury | Risk in pediatric herniotomy |
Summary Table: Key Points
| Topic | Key Facts |
|---|---|
| CMT | SCM fibrosis; head tilt ipsilateral, chin contralateral; treat with physio < 1 yr; surgery if fails |
| Umbilical hernia (children) | Closes spontaneously by age 4; repair if persistent |
| Indirect inguinal hernia | Patent processus vaginalis; most common pediatric hernia; herniotomy |
| Direct inguinal hernia | Acquired; adults; medial to epigastric vessels; mesh repair |
| Femoral hernia | Women > men; high strangulation risk; always repair |
| Strangulation | Surgical emergency; compromised blood supply to hernia contents |
Treatment Acute hematogenous osteomyelitis in children
acute hematogenous osteomyelitis children treatment antibiotics surgery
acute hematogenous osteomyelitis children pathology imaging
acute hematogenous osteomyelitis empirical antibiotic choice MRSA Staphylococcus duration IV to oral switch
osteomyelitis surgical drainage debridement indications
Treatment of Acute Hematogenous Osteomyelitis (AHO) in Children
Based on PIDS/IDSA Clinical Practice Guidelines, Harrison's Principles of Internal Medicine (21st ed., p. 3948), and Bailey & Love's Surgery.
Overview of Treatment Approach
- Antibiotic therapy — to eradicate the causative organism
- Surgical intervention — when indicated (abscess, non-response, complications)
Step 1: Initial Assessment Before Treatment
| Investigation | Purpose |
|---|---|
| Blood cultures (×2) | Positive in ~30–50% of AHO in children |
| CRP, ESR, WBC, Procalcitonin | Baseline inflammatory markers; guide response |
| Bone/joint aspiration | Direct microbiological diagnosis; done if surgical drainage planned |
| Wound/sinus swab | Only if open wound present |
Key principle: Obtain cultures before starting antibiotics whenever possible, but do not delay antibiotics if the child is septic.
Step 2: Empirical Antibiotic Therapy
Most Common Causative Organisms by Age
| Age Group | Primary Pathogens |
|---|---|
| Neonates (<3 months) | S. aureus, Group B Streptococcus, Gram-negative rods (E. coli, Klebsiella) |
| Infants & children (3 mo – 5 yr) | S. aureus, Streptococcus pyogenes, S. pneumoniae |
| School-age children (5–12 yr) | S. aureus (dominant), Streptococcus spp. |
| Adolescents | S. aureus, consider Neisseria gonorrhoeae (sexually active) |
| Sickle cell disease | Salmonella spp. + S. aureus |
| Immunocompromised | Pseudomonas, fungi, atypical organisms |
Empirical Antibiotic Selection
If MRSA prevalence is LOW (<10–15%) or community MSSA suspected:
| Drug | Dose | Route |
|---|---|---|
| Oxacillin / Nafcillin | 150–200 mg/kg/day ÷ q4–6h | IV |
| Cefazolin (alternative) | 100–150 mg/kg/day ÷ q8h | IV |
| Flucloxacillin (UK/Europe) | 50 mg/kg/dose q6h | IV |
If MRSA prevalence is HIGH (>10–15%) or MRSA suspected:
| Drug | Dose | Route | Notes |
|---|---|---|---|
| Vancomycin | 15–20 mg/kg/dose q6h (target AUC/MIC 400–600) | IV | First-line for MRSA |
| Clindamycin | 10–13 mg/kg/dose q6–8h | IV/PO | Excellent bone penetration; use if local susceptibility allows |
| Linezolid | 10 mg/kg/dose q8h (<12 yr); 600 mg q12h (≥12 yr) | IV/PO | Reserve for vancomycin intolerance; excellent oral bioavailability |
| Daptomycin | 6–10 mg/kg/day | IV | Alternative; not for pulmonary co-infection |
Neonates: Broad-spectrum coverage
- Oxacillin + Gentamicin or Ampicillin-sulbactam + Gentamicin
- Add anti-MRSA coverage if community MRSA risk
Sickle cell disease:
- Cover both Salmonella and S. aureus: Ceftriaxone + Oxacillin (or Vancomycin if MRSA concern)
Step 3: IV-to-Oral (Sequential) Therapy
Criteria for Oral Switch (PIDS/IDSA Guidelines):
- Clinical improvement (afebrile, pain decreasing, improved mobility)
- CRP trending down significantly
- Organism identified and susceptible to oral agent with high bioavailability
- No surgical drainage required / procedure completed
- Child can tolerate oral medications
- Reliable follow-up assured
Timing of Switch:
- Typically after 2–4 days of IV therapy in uncomplicated AHO
- Some centers switch as early as 48–72 hours if criteria are met
Oral Antibiotic Options:
| Organism | Preferred Oral Agent | Dose |
|---|---|---|
| MSSA | Cephalexin / Dicloxacillin / Amoxicillin-clavulanate | 75–100 mg/kg/day ÷ q6–8h |
| MRSA | Clindamycin (if susceptible) | 10–13 mg/kg/dose q6–8h |
| MRSA (clinda-R) | TMP-SMX | 8–12 mg/kg/day TMP ÷ q12h |
| Salmonella | Ciprofloxacin | 20–30 mg/kg/day ÷ q12h |
| Streptococcus | Amoxicillin | 80–90 mg/kg/day ÷ q8–12h |
Step 4: Total Duration of Antibiotic Therapy
| Clinical Scenario | Recommended Duration |
|---|---|
| Uncomplicated AHO, good response | 3–4 weeks total |
| Complicated AHO (subperiosteal/soft tissue abscess, delayed presentation) | 4–6 weeks |
| AHO with concurrent septic arthritis | 4–6 weeks |
| Chronic or recurrent osteomyelitis | 6+ weeks (guided by surgical debridement) |
| Neonatal osteomyelitis | 4–6 weeks |
CRP normalization is a useful guide — therapy can often be stopped safely once CRP returns to normal and the child is clinically well.
Step 5: Surgical Treatment
Indications for Surgery
| Indication | Urgency |
|---|---|
| Subperiosteal or intraosseous abscess on imaging | Urgent |
| Septic arthritis (adjacent joint involvement) | Emergency |
| Failed medical therapy (no improvement after 48–72 h of appropriate antibiotics) | Urgent |
| Sequestrum formation (necrotic bone) | Elective/semi-urgent |
| Neurological compromise (spine) | Emergency |
| Neonatal AHO (high risk of spread to growth plate) | Often early surgical drainage |
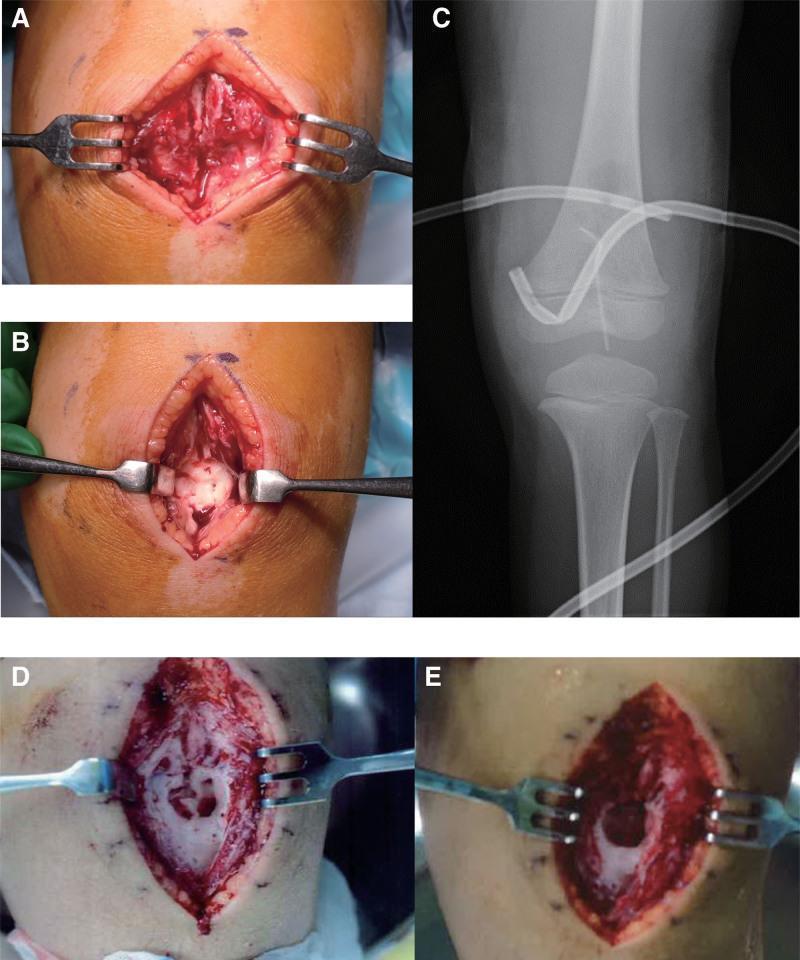
Surgical Procedures
-
Needle aspiration / bone aspiration
- Diagnostic + therapeutic
- Performed under imaging guidance or in theatre
- Useful for subperiosteal collections
-
Surgical drainage and debridement
- Cortical windowing (drilling or creating a bone window to drain pus)
- Thorough curettage of necrotic tissue
- Irrigation of the cavity
- Closed suction drainage may be placed
-
Sequestrectomy
- Removal of dead bone (sequestrum) in subacute/chronic cases
- Only after involucrum is sufficiently formed
-
Joint washout / arthrotomy
- If concurrent septic arthritis: emergency surgical washout of the joint
- Hip joint septic arthritis in children = surgical emergency (risk of avascular necrosis of femoral head)
Step 6: Monitoring Response to Treatment
| Parameter | Frequency | Expected Trend |
|---|---|---|
| CRP | Every 2–3 days initially | Should fall 30–50% within 48–72h of treatment |
| ESR | Weekly | Lags behind CRP; may take weeks to normalize |
| WBC | Every 2–3 days | Should normalize within days |
| Temperature / clinical exam | Daily | Afebrile within 48–72h expected |
| Imaging (MRI) | Only if clinical deterioration or no improvement | Not routinely repeated |
Failure to improve within 48–72 hours = reassess: wrong antibiotic? abscess requiring drainage? resistant organism? wrong diagnosis?
Special Situations
Neonatal AHO
- Often presents subtly (pseudoparalysis, irritability, swelling)
- High risk of spreading to adjacent growth plate and joint
- Multi-focal in up to 40%
- Requires aggressive IV therapy + early surgical drainage
- Prolonged course: 4–6 weeks
Pelvic Osteomyelitis
- Uncommon; involves ilium, ischium, pubis
- Often misdiagnosed (mimics septic hip, appendicitis)
- MRI is key for diagnosis
- Usually responds to antibiotics alone (less often requires surgery)
Vertebral Osteomyelitis (Discitis in Children)
- Most common at lumbar spine
- S. aureus most common; often hematogenous
- Presents with back pain, refusal to walk
- Usually treated with antibiotics alone; surgery if epidural abscess
MRSA-associated AHO (Severe)
- Community-acquired MRSA (especially USA300 strain) can cause deep vein thrombosis, pulmonary septic emboli, multi-focal disease
- Consider Vancomycin + Clindamycin (for anti-toxin effect) in severe presentations
- Higher surgical rate
Imaging Modalities for Monitoring
| Modality | Role |
|---|---|
| MRI | Gold standard — best soft tissue, marrow, abscess delineation |
| X-ray | First-line but insensitive early (changes appear at 10–21 days) |
| Ultrasound | Detects subperiosteal collections; guides aspiration |
| Bone scintigraphy | Sensitive early; useful for multi-focal disease; limited specificity |
| CT scan | Best for cortical destruction, sequestrum; radiation concern in children |
Treatment Algorithm Summary
Suspected AHO in Child
↓
Blood cultures + CRP/ESR/WBC + CXR
↓
MRI (preferred) or bone scan
↓
Abscess / collection identified?
YES → Surgical drainage + antibiotics
NO → IV antibiotics empirically
↓
48–72 h: Clinical & CRP response?
NO → Reassess; consider surgery / change antibiotics
YES → Continue; plan IV→oral switch
↓
Switch to oral when criteria met (2–4 days)
↓
Total duration: 3–6 weeks guided by CRP + clinical response
Key Take-Home Points
| Point | Detail |
|---|---|
| S. aureus dominates | Empirical therapy must always cover it |
| MRSA prevalence guides initial choice | Vancomycin or Clindamycin for high-MRSA settings |
| Early oral switch is safe | After 2–4 days IV if clinically improving |
| Total duration 3–6 weeks | Shorter for uncomplicated; longer for abscesses/neonates |
| Surgery for abscess or failure | Indispensable when pus is present or medical therapy fails |
| CRP is the best monitoring marker | Falls rapidly with effective treatment |
| Hip joint AHO/septic arthritis = emergency | Risk of femoral head AVN requires immediate washout |
Acute hematogenous osteomyelitis in children
acute hematogenous osteomyelitis children pathogenesis classification clinical features diagnosis
acute hematogenous osteomyelitis children X-ray MRI bone pathology
osteomyelitis children metaphysis blood supply sinusoidal vessels mechanism spread subperiosteal abscess
osteomyelitis bone anatomy metaphysis periosteum abscess formation stages
Acute Hematogenous Osteomyelitis (AHO) in Children
Sources: PIDS/IDSA Guidelines; Bailey & Love's Surgery (28th ed., p. 656); Harrison's Principles of Internal Medicine (21st ed., p. 3948); Guide to Utilization of the Microbiology Laboratory (p. 80)
Definition
Epidemiology
- Incidence: ~8 per 100,000 children per year
- Peak age: 2–12 years (prepubertal); neonates form a separate high-risk group
- Sex: Boys > Girls (2:1), likely due to greater trauma exposure
- Most common sites: Distal femur > Proximal tibia > Proximal humerus > Proximal femur
- Majority (~85%) affect a single bone; multi-focal disease seen in neonates and immunocompromised
Etiology / Causative Organisms
| Age Group | Primary Organisms |
|---|---|
| Neonates (<3 months) | S. aureus, Group B Streptococcus (S. agalactiae), E. coli, Klebsiella |
| Infants & children <4 years | S. aureus, Kingella kingae (very common <4 yr), S. pneumoniae |
| Children 4–12 years | S. aureus (dominant), Streptococcus pyogenes |
| Adolescents | S. aureus; consider N. gonorrhoeae if sexually active |
| Sickle cell disease | Salmonella spp. + S. aureus |
| Immunocompromised / IV drug use | Pseudomonas aeruginosa, Candida spp. |
S. aureus is the single most common causative pathogen at all pediatric ages. MRSA strains (especially community-acquired USA300) are increasingly prevalent and associated with more severe disease.
Pathogenesis
Why Does Infection Localize to the Metaphysis?
- Terminal capillary loops in the metaphysis form sharp hairpin turns → sluggish blood flow → bacterial lodgment
- These sinusoidal vessels lack phagocytic lining cells → bacteria evade early clearance
- High metabolic activity of growth plate provides favorable environment for bacterial proliferation
- Trauma (even minor) may cause microhematomas in metaphysis → bacterial colonization during transient bacteremia
Stages of Progression
Bacteremia → Metaphyseal seeding
↓
Acute inflammation (hyperemia, edema, WBC infiltration)
↓
Pus formation within medullary cavity (INTRAMEDULLARY ABSCESS)
↓
Rising intraosseous pressure → pus tracks through Volkmann's canals
↓
SUBPERIOSTEAL ABSCESS (periosteum stripped from bone)
↓
Periosteum ruptures → SOFT TISSUE ABSCESS
↓
Interruption of periosteal blood supply → BONE NECROSIS (SEQUESTRUM)
↓
Periosteum lays down new bone → INVOLUCRUM (reactive new bone shell)
↓
CHRONIC OSTEOMYELITIS
Special Anatomical Considerations (Bailey & Love, p. 656)
- Intracapsular metaphyses: The proximal femur, proximal humerus, proximal radius, and distal fibula have their metaphyses within the joint capsule → metaphyseal infection can directly spread into the adjacent joint → septic arthritis
- A sympathetic (sterile) joint effusion may occur adjacent to metaphyseal osteomyelitis — requires differentiation from true septic arthritis
- In neonates: the epiphyseal cartilage is crossed by transphyseal vessels → infection readily spreads to epiphysis and joint → growth plate destruction and joint sepsis are common
Clinical Features
Symptoms
| Feature | Details |
|---|---|
| Fever | High-grade (>38.5°C), often with chills; may be absent in neonates |
| Bone pain | Severe, localized to metaphysis; worsens with movement or palpation |
| Refusal to use limb | Classic in toddlers — "pseudoparalysis" |
| Limp or inability to bear weight | Lower limb involvement |
| Swelling & redness | Develops as infection tracks to periosteum/soft tissue |
| Systemic toxicity | Irritability, malaise, anorexia |
Signs
- Point tenderness over metaphysis — hallmark sign
- Local warmth, erythema, swelling (later finding)
- Restricted active and passive movement of adjacent joint
- Neonates: often subtle — irritability, reduced limb movement, swelling only
Investigations
Laboratory Tests
| Test | Findings / Notes |
|---|---|
| CRP | Most sensitive early marker; rises within hours; best for monitoring |
| ESR | Elevated but lags; peaks at ~3–5 days; slow to normalize |
| WBC | Elevated with neutrophilia; may be normal early |
| Procalcitonin | Elevated in bacterial infection; useful adjunct |
| Blood cultures | Positive in ~30–50%; obtained before antibiotics |
| Bone/abscess aspirate | Most definitive; culture + Gram stain + sensitivity |
Imaging
1. Plain X-ray
- First-line but insensitive early — changes appear only after 10–21 days
- Early: soft tissue swelling, loss of fat planes
- Late: lytic lesion in metaphysis, periosteal reaction, cortical erosion
- Role: exclude fracture, tumor; baseline for comparison
2. Ultrasound
- Detects subperiosteal fluid/abscess early (within days)
- Guides needle aspiration
- Cannot assess bone marrow or cortex
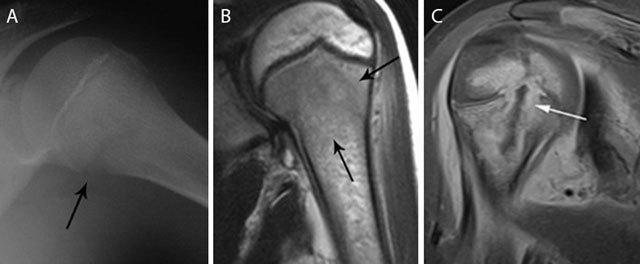
3. MRI — Gold Standard
- Most sensitive and specific modality for early AHO
- Detects bone marrow edema, intramedullary abscess, subperiosteal collection, soft tissue extension and transphyseal spread
- T1: low signal marrow; T2/STIR: high signal edema; Gd-contrast: rim-enhancing abscess
4. Bone Scintigraphy (Tc-99m)
- Sensitive early (within 24–48 h)
- Useful for multi-focal disease (whole-body scan)
- Less specific than MRI; involves radiation
- May be falsely negative in neonates (due to vascular compromise)
5. CT Scan
- Best for cortical destruction, sequestrum, involucrum detail
- Guides surgical planning in chronic/complicated cases
- Radiation concern limits use in children
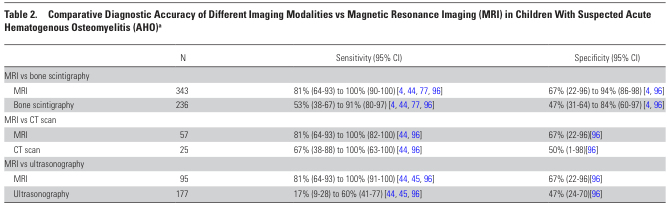
Imaging Accuracy Comparison (PIDS/IDSA Guidelines, p. 13):
| Modality vs. MRI | Sensitivity | Specificity |
|---|---|---|
| Bone scintigraphy | 53–91% | 47–84% |
| CT scan | 67–100% | 50–67% |
| Ultrasonography | 17–60% | ~47% |
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Septic arthritis | Maximal tenderness over joint; all movements painful; joint effusion on USS |
| Cellulitis | Superficial; no bone tenderness; no bone changes on imaging |
| Ewing's sarcoma | Chronic course; "onion skin" periosteal reaction; biopsy differentiates |
| Langerhans cell histiocytosis | Lytic lesion; more indolent course |
| Leukemia | Bone pain, cytopenias; leukemic lines on X-ray; bone marrow biopsy |
| Transient synovitis | Afebrile / low-grade fever; CRP normal/mildly raised; self-limiting |
| Trauma / fracture | History; fracture line on X-ray |
| Sickle cell bone crisis | Known SCD; multi-focal; normal CRP possible |
Kocher criteria help differentiate septic arthritis from transient synovitis of the hip: fever, non-weight-bearing, ESR >40, WBC >12,000 — 4/4 criteria = ~99% probability of septic arthritis.
Complications
Early
- Septic arthritis — especially hip, shoulder (intracapsular metaphyses)
- Pathological fracture — due to cortical weakening
- Septicemia / multi-organ failure — in severe cases
Late / If Inadequately Treated
- Chronic osteomyelitis — sequestrum formation, persistent sinus tracts, recurrent flares
- Growth disturbance — damage to physis → limb length discrepancy, angular deformity
- Avascular necrosis — particularly femoral head when hip joint involved
- Garre's sclerosing osteomyelitis — chronic non-suppurative form
- Brodie's abscess — walled-off chronic intraosseous abscess
Treatment
Principles
- Early empirical antibiotics based on likely organism and local MRSA prevalence
- IV-to-oral switch early once criteria met
- Surgical drainage when abscess present or medical failure
- Monitor with serial CRP
Antibiotic Therapy
Empirical Selection
| MRSA Risk | Agent | Route |
|---|---|---|
| Low MRSA | Oxacillin / Nafcillin / Cefazolin / Flucloxacillin | IV |
| High MRSA | Vancomycin (AUC/MIC guided) | IV |
| MRSA alternative | Clindamycin (if susceptible) | IV/PO |
| Neonates | Oxacillin + Gentamicin (± Vancomycin for MRSA) | IV |
| Sickle cell | Ceftriaxone + anti-staphylococcal cover | IV |
IV-to-Oral Switch (after 2–4 days if criteria met):
- Afebrile, CRP declining, tolerating oral intake
- Oral agents: Cephalexin (MSSA), Clindamycin / TMP-SMX (MRSA)
Total Duration:
- Uncomplicated AHO: 3–4 weeks
- Complicated (abscess, neonatal, delayed): 4–6 weeks
Surgical Indications
- Subperiosteal or intraosseous abscess
- No clinical improvement after 48–72 h of antibiotics
- Septic arthritis (emergency joint washout)
- Sequestrum formation
- Neonatal AHO (early drainage often needed)
Summary
| Feature | Key Point |
|---|---|
| Peak age | Prepubertal children (2–12 yr) |
| Most common site | Metaphysis of long bones (distal femur, proximal tibia) |
| Most common pathogen | S. aureus |
| Pathogenesis | Metaphyseal sinusoidal sluggish flow → bacterial seeding → abscess → sequestrum |
| Best early marker | CRP |
| Gold standard imaging | MRI |
| Treatment | IV antibiotics → early oral switch; surgery if abscess or failure |
| Duration | 3–6 weeks total |
| Key complication | Septic arthritis, growth disturbance, chronic osteomyelitis |
Introduction to clinical pharmacology: general issues, fundamentals of rational pharmacotherapy
clinical pharmacology definition goals pharmacokinetics pharmacodynamics drug action
rational pharmacotherapy principles drug selection prescribing efficacy safety individualization
pharmacokinetics absorption distribution metabolism elimination bioavailability half-life volume of distribution
pharmacodynamics dose-response relationship receptor drug efficacy potency therapeutic index
Introduction to Clinical Pharmacology: General Issues & Fundamentals of Rational Pharmacotherapy
Based on: Harrison's Principles of Internal Medicine, 21st Ed. (p. 1880); core pharmacology principles from Goodman & Gilman's, Rang & Dale's, and WHO rational use of medicines framework.
1. What Is Clinical Pharmacology?
- To describe the conditions under which drug actions vary among human subjects
- To determine the mechanisms underlying this variability, with the aim of improving therapy with available drugs and identifying new therapeutic targets
Relationship to Adjacent Disciplines
| Discipline | Focus |
|---|---|
| Pharmacology | Study of drug–biological system interactions (basic science) |
| Clinical pharmacology | Drug effects and variability in humans; bridges lab and bedside |
| Pharmacotherapy | Application of drugs to treat, prevent, or diagnose disease |
| Toxicology | Study of adverse/harmful drug and chemical effects |
| Pharmacoepidemiology | Drug effects in populations |
| Pharmacoeconomics | Cost-effectiveness and economic impact of drug use |
2. Core Subdivisions of Pharmacology
2.1 Pharmacokinetics (PK) — "What the body does to the drug"
A — Absorption
- Process by which a drug moves from its site of administration into the systemic circulation
- Bioavailability (F): fraction of administered dose that reaches systemic circulation unchanged
- IV administration: F = 100%
- Oral: reduced by first-pass hepatic metabolism, gut wall metabolism, incomplete absorption
- Factors affecting absorption: drug solubility, formulation, GI motility, food, pH, surface area
- Routes of administration and their implications:
| Route | Onset | Bioavailability | Notes |
|---|---|---|---|
| Intravenous (IV) | Fastest | 100% | No absorption step; immediate effect |
| Intramuscular (IM) | Fast | Variable (75–100%) | Depot effect possible |
| Subcutaneous (SC) | Moderate | Variable | Slow, sustained release |
| Oral (PO) | Slow–moderate | Variable | First-pass effect; most convenient |
| Sublingual | Fast | High | Bypasses first-pass |
| Transdermal | Slow | Variable | Sustained; bypasses GI |
| Inhalation | Fast | High (lung surface) | Ideal for respiratory drugs |
| Rectal | Moderate | Partial bypass of first-pass | Useful in vomiting/unconscious |
D — Distribution
- Movement of drug from blood into tissues
- Volume of Distribution (Vd): apparent volume into which drug is distributed
- Vd = Dose / Plasma concentration at time 0
- Small Vd (e.g., 5–10 L): drug confined to plasma (large MW, highly protein-bound)
- Large Vd (e.g., hundreds of litres): extensive tissue binding (lipophilic drugs)
- Plasma protein binding: albumin (acidic drugs), α1-acid glycoprotein (basic drugs)
- Only free (unbound) drug is pharmacologically active
- Blood-brain barrier (BBB): limits CNS entry; lipophilic, non-ionized drugs cross more readily
- Placental transfer: lipophilic, low MW, non-ionized drugs cross freely — critical in pregnancy
M — Metabolism (Biotransformation)
- Primarily hepatic; also gut wall, lung, kidney, plasma
- Goal: convert lipophilic drugs into more polar (water-soluble) metabolites for excretion
- Phase I reactions: Oxidation, reduction, hydrolysis — introduce/unmask functional groups
- Mainly via CYP450 enzymes (CYP3A4, CYP2D6, CYP2C9, CYP2C19, CYP1A2)
- May produce active metabolites (e.g., codeine → morphine via CYP2D6)
- May produce toxic metabolites (e.g., paracetamol → NAPQI)
- Phase II reactions: Conjugation (glucuronidation, sulfation, acetylation, methylation) → inactive, polar products
- First-pass effect: drugs absorbed orally pass through portal circulation → hepatic metabolism before reaching systemic circulation → reduced bioavailability (e.g., morphine, GTN, propranolol)
- Enzyme induction (e.g., rifampicin, carbamazepine, phenytoin): increases CYP activity → reduces drug levels → therapeutic failure
- Enzyme inhibition (e.g., fluconazole, erythromycin, grapefruit juice): reduces CYP activity → increases drug levels → toxicity
E — Elimination (Excretion)
- Primary route: renal (glomerular filtration, tubular secretion, tubular reabsorption)
- Also: biliary/fecal, pulmonary (volatile agents), sweat, saliva, breast milk
- Clearance (CL): volume of plasma cleared of drug per unit time (mL/min)
- Total clearance = Hepatic clearance + Renal clearance + Other
- Half-life (t½): time for plasma drug concentration to halve
- t½ = 0.693 × Vd / CL
- Clinical importance: determines dosing interval; ~5 half-lives to reach steady state; ~5 half-lives to eliminate drug after stopping
- Steady state: achieved when rate of drug input = rate of elimination; reached after ~5 × t½
- Accumulation: drugs with long t½ or renal/hepatic impairment → dose adjustment required
2.2 Pharmacodynamics (PD) — "What the drug does to the body"
Drug Receptors and Mechanisms of Action
| Drug Target | Examples | Mechanism |
|---|---|---|
| Ion channels | Local anesthetics, antiepileptics | Block or modulate ion conductance |
| G-protein coupled receptors (GPCRs) | β-blockers, opioids, muscarinic agonists | Activate/inhibit second messenger cascades |
| Enzyme inhibition | ACE inhibitors, statins, NSAIDs | Block enzyme active site |
| Nuclear receptors | Corticosteroids, thyroid hormones | Alter gene transcription |
| Carrier/transporter proteins | SSRIs, loop diuretics | Block reuptake or transport |
| Structural proteins | Colchicine (tubulin), taxanes | Disrupt cytoskeletal function |
| Nucleic acids | Alkylating agents, antibiotics | Intercalation, DNA damage |
Dose-Response Relationships
- Graded dose-response curve: increasing dose → increasing effect (up to maximum = Emax)
- Quantal dose-response: proportion of population showing a defined effect at each dose
- Key parameters:
- Emax (Efficacy): maximum effect a drug can produce regardless of dose
- EC50: concentration producing 50% of Emax — measure of potency
- Potency: dose required to produce a given effect (lower dose = more potent)
- Efficacy ≠ Potency: a drug can be highly potent but have low efficacy
Therapeutic Index (TI) / Therapeutic Window
- TD50: dose toxic in 50% of population
- ED50: dose effective in 50% of population
- Wide TI (e.g., penicillin): large safety margin
- Narrow TI (e.g., digoxin, warfarin, lithium, aminoglycosides, phenytoin): small margin between therapeutic and toxic → requires therapeutic drug monitoring (TDM)
Agonists and Antagonists
| Type | Definition | Example |
|---|---|---|
| Full agonist | Binds receptor → maximal response (Emax) | Morphine |
| Partial agonist | Binds receptor → submaximal response even at full occupancy | Buprenorphine |
| Antagonist (competitive) | Binds receptor; blocks agonist; reversible by increasing agonist dose | Naloxone, β-blockers |
| Antagonist (non-competitive) | Binds receptor irreversibly or allosterically; cannot be overcome by agonist | Phenoxybenzamine |
| Inverse agonist | Binds receptor → opposite effect to agonist | Some benzodiazepines |
Tolerance and Tachyphylaxis
- Tolerance: decreased response to drug over time (requires dose escalation)
- Mechanisms: receptor downregulation, enzyme induction, physiological adaptation
- Tachyphylaxis: rapid tolerance developing within minutes-hours (e.g., nitrates, ephedrine)
- Desensitization: receptor becomes unresponsive despite continued agonist presence
- Physical dependence: physiological adaptation → withdrawal syndrome on cessation (e.g., opioids, benzodiazepines, alcohol)
2.3 Pharmacogenomics
- Study of how genetic variation affects drug response
- Key examples:
- CYP2D6 polymorphism: poor metabolizers → codeine toxicity; ultra-rapid metabolizers → inadequate analgesia
- CYP2C19: clopidogrel activation — poor metabolizers get inadequate antiplatelet effect
- TPMT deficiency: azathioprine toxicity (bone marrow suppression)
- G6PD deficiency: hemolysis with primaquine, nitrofurantoin
- HLA-B*5701: abacavir hypersensitivity (test before prescribing)
- Guides personalized/precision medicine
3. Drug Variability — Sources of Interindividual Differences
| Factor | Effect on Drug Response |
|---|---|
| Age | Neonates: immature CYP enzymes, higher Vd for water-soluble drugs; Elderly: reduced renal/hepatic clearance, more sensitive CNS |
| Body weight/composition | Obese patients: altered Vd for lipophilic drugs; lean vs. total body weight for dosing |
| Sex | Women: generally lower CYP3A4 activity; hormonal effects on drug metabolism |
| Genetics | CYP450 polymorphisms, transporter variants, receptor variants |
| Renal function | Reduced GFR → drug accumulation (renally cleared drugs); use eGFR for dose adjustment |
| Hepatic function | Cirrhosis → reduced first-pass, reduced protein synthesis (lower albumin → more free drug) |
| Drug interactions | Enzyme induction/inhibition, protein-binding displacement, pharmacodynamic synergy/antagonism |
| Disease states | HF → reduced hepatic blood flow; thyroid disease → altered metabolism |
| Pregnancy | Increased Vd, altered CYP activity, renal clearance changes; placental transfer |
4. Drug Interactions
Pharmacokinetic Interactions
| Mechanism | Example |
|---|---|
| Enzyme induction | Rifampicin reduces warfarin, OCP, antiretrovirals |
| Enzyme inhibition | Fluconazole increases warfarin → bleeding; erythromycin + statins → myopathy |
| Absorption interference | Antacids reduce quinolone/tetracycline absorption; cholestyramine reduces many drugs |
| Protein binding displacement | Rare clinically significant effect (usually transient) |
| Renal excretion | Probenecid blocks penicillin tubular secretion → increases penicillin levels |
Pharmacodynamic Interactions
| Type | Effect | Example |
|---|---|---|
| Synergism | Combined effect > sum | Alcohol + benzodiazepines → CNS depression |
| Antagonism | One drug reduces effect of another | Naloxone reverses opioids |
| Additive | Combined effect = sum | Two analgesics with different mechanisms |
| Potentiation | One drug enhances another's effect | Clavulanate potentiates amoxicillin |
5. Adverse Drug Reactions (ADRs)
Classification (Rawlins & Thompson — ABC System)
| Type | Description | Mechanism | Example |
|---|---|---|---|
| Type A (Augmented) | Dose-dependent, predictable, common (~80% of ADRs) | Exaggerated pharmacological effect | Bleeding with warfarin, hypoglycemia with insulin |
| Type B (Bizarre) | Dose-independent, unpredictable, rare | Immunological or idiosyncratic | Penicillin anaphylaxis, halothane hepatitis |
| Type C (Chronic) | Related to cumulative dose / long-term use | Adaptation | Adrenal suppression with prolonged steroids |
| Type D (Delayed) | Appear after prolonged latency | Carcinogenesis, teratogenesis | Thalidomide embryopathy |
| Type E (End of use) | Withdrawal reactions | Rebound phenomenon | Beta-blocker withdrawal → rebound tachycardia |
| Type F (Failure) | Unexpected failure of therapy | Drug interactions, non-adherence | OCP failure with rifampicin |
WHO-UMC Causality Assessment
6. Fundamentals of Rational Pharmacotherapy
Definition
The WHO 5-Step Prescribing Model (P-Drug Concept)
| Step | Action | Key Question |
|---|---|---|
| 1. Define the patient's problem | Accurate diagnosis | What is wrong? |
| 2. Specify the therapeutic objective | What outcome is desired? | Cure, palliation, prevention? |
| 3. Verify suitability of your P-drug | Is the standard drug suitable for THIS patient? | Any contraindications? Interactions? |
| 4. Start the treatment | Prescribe correctly: drug, dose, route, duration, instructions | Right drug, right patient, right dose, right time |
| 5. Give information, instructions, warnings | Counsel the patient | Adherence, side effects, what to do if problems arise |
| 6. Monitor (and stop?) treatment | Assess response; adjust or stop | Is it working? Any ADRs? |
Core Principles of Rational Prescribing
1. Correct Diagnosis
- Drug therapy must be guided by an accurate diagnosis
- Empirical treatment is appropriate only when diagnosis is uncertain and delay is harmful (e.g., sepsis, meningitis)
2. Indication
- A drug should only be prescribed when there is a clear clinical indication
- Avoid polypharmacy — each drug must have a justified indication
3. Drug Selection Criteria (STEP approach)
- Safe — acceptable risk-benefit ratio for this patient
- Tolerable — acceptable adverse effect profile
- Effective — proven efficacy for the condition
- Price — affordable; least costly among equally effective options
- Evidence base: prefer drugs with RCT-level evidence; use guidelines
- Formulation: appropriate for the patient (age, swallowing ability, route)
- Spectrum: narrowest spectrum antibiotic; most specific drug
4. Correct Dosing
- Right dose: based on body weight (mg/kg), age, renal/hepatic function, pharmacogenomics
- Right interval: based on t½; too frequent → toxicity; too infrequent → subtherapeutic
- Loading dose: used when rapid steady state is needed (e.g., digoxin, amiodarone, phenytoin)
- Loading dose = Target concentration × Vd
- Maintenance dose: sustains steady state
- Maintenance dose = Target concentration × CL × dosing interval
- Dose reduction: required in renal/hepatic impairment, elderly, neonates
5. Route of Administration
- Determined by: urgency, drug properties (bioavailability, stability), patient factors (vomiting, unconscious), site of action
- "Start IV, switch oral as soon as possible" — reduces hospital stay, cost, line infections
6. Duration of Therapy
- Must be defined at the time of prescribing
- Too short: therapeutic failure, relapse, resistance (antibiotics)
- Too long: ADRs, drug dependence, unnecessary cost
- Chronic conditions: review regularly; step-down when possible
7. Monitoring
- Therapeutic Drug Monitoring (TDM): mandatory for narrow TI drugs
| Drug | Monitoring Parameter | Target Range |
|---|---|---|
| Digoxin | Serum level | 0.5–2 ng/mL |
| Lithium | Serum level | 0.6–1.2 mmol/L (therapeutic); >1.5 toxic |
| Phenytoin | Serum level | 10–20 mg/L |
| Vancomycin | AUC/MIC or trough | AUC 400–600 |
| Aminoglycosides | Peak and trough | Drug-specific |
| Warfarin | INR | Indication-dependent (usually 2–3) |
| Cyclosporine | Trough level | Transplant protocol-specific |
- Clinical monitoring: efficacy endpoints (BP, glucose, pain score)
- Safety monitoring: LFTs (statins, anti-TB), renal function (ACEi, metformin), CBC (azathioprine, methotrexate)
8. Patient Adherence (Compliance)
- Non-adherence is the leading cause of therapeutic failure
- Causes: complex regimens, side effects, cost, poor understanding, cognitive impairment
- Strategies to improve adherence:
- Simplify regimens (once-daily dosing; fixed-dose combinations)
- Clear verbal and written instructions
- Patient education about purpose and expected effects
- Blister packs, pill organizers
- Regular follow-up
9. Special Populations
| Population | Key Considerations |
|---|---|
| Neonates/Infants | Immature drug metabolism (CYP enzymes develop postnatally); higher water content → larger Vd for hydrophilic drugs; dose per kg; avoid chloramphenicol (grey baby syndrome), sulfonamides |
| Children | Weight-based dosing (mg/kg); avoid tetracyclines (<8 yr), quinolones (<18 yr in most contexts); age-appropriate formulations |
| Elderly | Reduced renal clearance, hepatic mass, albumin; increased CNS sensitivity; polypharmacy risk; use START/STOPP criteria; Beers criteria for inappropriate drugs |
| Pregnancy | FDA/ADEC categories (A/B/C/D/X); avoid ACEi/ARBs (2nd/3rd trimester), NSAIDs (3rd trimester), warfarin (1st trimester), tetracyclines, methotrexate, thalidomide |
| Breastfeeding | Assess infant dose (relative infant dose <10% = usually safe); avoid amiodarone, cytotoxics, radioiodine, lithium |
| Renal impairment | Reduce dose or increase interval for renally-cleared drugs; use eGFR; avoid nephrotoxins (NSAIDs, aminoglycosides, contrast) |
| Hepatic impairment | Reduce dose of drugs with high first-pass metabolism; avoid hepatotoxins; check Child-Pugh score |
7. Prescription Writing — Elements of a Valid Prescription
- Patient details: name, age, weight (pediatrics), date
- Drug name: preferably generic (INN) name
- Dose: amount per administration
- Route: oral, IV, topical, etc.
- Frequency: once daily, q8h, PRN, etc.
- Duration: number of days/weeks; total quantity
- Instructions: with food/without, special precautions
- Prescriber signature & details
Generic prescribing is preferred: reduces cost, avoids brand confusion, allows pharmacist substitution with bioequivalent products.
8. Polypharmacy and Deprescribing
- Polypharmacy: use of ≥5 drugs simultaneously (problematic polypharmacy = inappropriate drugs)
- Risks: ADRs, drug interactions, non-adherence, falls (elderly), hospitalization
- Deprescribing: systematic, supervised process of tapering/stopping drugs that are no longer needed or causing harm
- Tools: STOPP criteria (drugs to stop), START criteria (drugs to start that may be omitted), Beers criteria (potentially inappropriate medications in elderly)
9. Pharmacovigilance and Drug Safety Monitoring
- Ongoing surveillance of drug safety after market approval (post-marketing surveillance)
- Methods: spontaneous reporting (yellow card system), prescription event monitoring, database studies, case-control studies
- WHO Programme for International Drug Monitoring — global ADR reporting
- Goal: detect rare, delayed, or long-term adverse effects not captured in pre-marketing trials
Summary Table: Key Concepts
| Concept | Key Points |
|---|---|
| Clinical pharmacology | Study of drug effects in humans; explains variability in drug response |
| Pharmacokinetics (PK) | ADME — what body does to drug; Vd, CL, t½, bioavailability |
| Pharmacodynamics (PD) | Receptor mechanisms, dose-response, efficacy, potency, TI |
| Therapeutic index | TI = TD50/ED50; narrow TI drugs require TDM |
| Rational pharmacotherapy | Right drug, right patient, right dose, right route, right duration |
| P-Drug model | 6-step WHO approach to prescribing |
| Drug selection | STEP: Safe, Tolerable, Effective, Price |
| Monitoring | TDM for narrow TI; clinical and safety endpoints |
| Special populations | Neonates, elderly, pregnancy — significant PK/PD alterations |
| ADR classification | Type A (predictable) to Type F (failure); most are Type A |
| Pharmacogenomics | Genetic basis of drug variability; foundation of precision medicine |
| Adherence | Most common cause of therapeutic failure; simplify and educate |
Which drug contraindications in pregnancy
drug contraindications pregnancy teratogenic medications fetal harm
pregnancy drug safety categories FDA teratology ACE inhibitors warfarin NSAIDs tetracycline
teratogenic drugs list methotrexate thalidomide isotretinoin warfarin valproate pregnancy trimester effects
ACE inhibitors ARBs statins fluoroquinolones aminoglycosides contraindicated pregnancy fetal effects
Drug Contraindications in Pregnancy
Based on: FDA Pregnancy and Lactation Labeling Rule (2015); Harrison's Principles of Internal Medicine (21st ed.); Goodman & Gilman's Pharmacology; Management of Arrhythmias During Pregnancy (p. 12); clinical teratology references.
Background: FDA Pregnancy Classification
Old System (pre-2015): Letter Categories
| Category | Meaning |
|---|---|
| A | Controlled studies show no risk to fetus |
| B | Animal studies show no risk; no adequate human studies |
| C | Animal studies show adverse effects; no adequate human studies |
| D | Evidence of human fetal risk; benefits may justify use in serious situations |
| X | Fetal risk outweighs any benefit — absolutely contraindicated |
Key Principle: Trimester Sensitivity
| Trimester | Period | Risk |
|---|---|---|
| 1st trimester (weeks 1–12) | Organogenesis | Teratogenesis — structural malformations |
| 2nd trimester (weeks 13–26) | Growth & maturation | Functional defects, growth restriction |
| 3rd trimester (weeks 27–40) | Final maturation | Neonatal toxicity, premature closure of ductus arteriosus, neonatal withdrawal |
Absolutely Contraindicated Drugs (Former FDA Category X)
1. Thalidomide
- Effect: Phocomelia (limb reduction defects), internal organ malformations, ear/eye defects
- Mechanism: Inhibits angiogenesis, TNF-α, affects cereblon protein in limb development
- Trimester: 1st (days 20–36 of gestation = most critical)
- Historical significance: 1950s–60s disaster leading to modern drug safety regulation
- Current use: Multiple myeloma, leprosy — requires mandatory pregnancy prevention program (REMS)
2. Isotretinoin (13-cis-retinoic acid)
- Effect: Retinoic acid embryopathy — craniofacial defects, CNS malformations (hydrocephalus, microcephaly), cardiac defects (conotruncal), thymic aplasia
- Risk: Even brief exposure in 1st trimester causes severe teratogenicity
- Contraception: Requires two forms of contraception; mandatory iPLEDGE program in USA
- Avoidance: Stop ≥1 month before conception
3. Methotrexate
- Effect: Folic acid antagonist → neural tube defects, skeletal abnormalities, spontaneous abortion, fetal death
- Trimester: All (abort at any stage)
- Uses: RA, psoriasis, ectopic pregnancy, cancer
- Note: Also used therapeutically to terminate ectopic pregnancy
- Washout: Stop ≥3 months before conception; supplement folic acid
4. Warfarin
- Effects by trimester:
- 1st trimester: Warfarin embryopathy — nasal hypoplasia, stippled epiphyses, chondrodysplasia punctata
- 2nd–3rd trimester: CNS defects (Dandy-Walker, agenesis of corpus callosum), optic atrophy
- Near delivery: Fetal/neonatal hemorrhage (fetus cannot reverse anticoagulation)
- Safe alternative: Low-molecular-weight heparin (LMWH) — does not cross placenta
- Exception: Mechanical heart valves may require warfarin in 2nd trimester under specialist guidance (risk-benefit decision)
5. Valproic Acid (Sodium Valproate)
- Effects:
- Neural tube defects (spina bifida, anencephaly) — 1–2% risk (10× background)
- Fetal valproate syndrome: midface hypoplasia, long philtrum, thin lips, digital anomalies
- Cognitive impairment: children exposed in utero have significantly lower IQ scores; autism spectrum disorder
- Mechanism: Inhibits histone deacetylase; folate antagonism
- Trimester: 1st most critical; cognitive effects across all trimesters
- UK MHRA: Valproate is banned in women of childbearing age unless Pregnancy Prevention Programme is in place
6. Carbamazepine
- Effects: Neural tube defects (spina bifida ~1%), craniofacial defects, fingernail hypoplasia, developmental delay
- Safer than valproate but still teratogenic
- Alternative: Lamotrigine or levetiracetam (relatively safer in pregnancy)
7. Phenytoin
- Effects: Fetal hydantoin syndrome — hypertelorism, broad nasal bridge, digital/nail hypoplasia, cleft lip/palate, cardiac defects, cognitive impairment
- Mechanism: Folate antagonism, toxic epoxide metabolites
Cardiovascular Drugs
8. ACE Inhibitors & ARBs
- Safe in 1st trimester (debated, but generally used if needed)
- Contraindicated from 2nd trimester onward:
- ACE inhibitor fetopathy: oligohydramnios, renal tubular dysgenesis, anuria, neonatal renal failure, skull ossification defects (calvaria hypoplasia), limb contractures, pulmonary hypoplasia
- Mechanism: Fetal RAA system blockade → fetal hypotension → renal ischemia
- Drugs: Enalapril, lisinopril, ramipril; Losartan, valsartan, candesartan (ARBs)
- Alternative for hypertension: Methyldopa, labetalol, nifedipine (preferred in pregnancy)
9. Statins (HMG-CoA reductase inhibitors)
- Effect: Cholesterol essential for fetal development; statins may cause CNS and limb defects
- Historically FDA Category X; evidence is still evolving (some newer data suggest lower risk)
- Current recommendation: Discontinue during pregnancy
- Drugs: Atorvastatin, simvastatin, rosuvastatin, lovastatin
10. Amiodarone
- Effects: Neonatal hypothyroidism/hyperthyroidism (contains 37% iodine by weight), neonatal bradycardia, QT prolongation, IUGR, premature birth
- Use only when no alternative for life-threatening arrhythmia
- Monitor neonatal thyroid function
11. Aldosterone Antagonists (Spironolactone)
- Effect: Anti-androgenic → feminization of male fetus; genital ambiguity
- Contraindicated throughout pregnancy
Antimicrobials
12. Tetracyclines
- Effect: Chelate calcium → deposited in developing teeth and bones
- Yellow-brown tooth discoloration (deciduous teeth) — permanent
- Bone growth inhibition
- Maternal hepatotoxicity at high doses (IV)
- Contraindicated after week 14 (tooth bud development); avoid throughout pregnancy as precaution
- Drugs: Tetracycline, doxycycline, minocycline
- Alternative: Amoxicillin, azithromycin, erythromycin (base)
13. Fluoroquinolones
- Effect: Arthropathy in weight-bearing joints of developing animals (cartilage damage); theoretical risk of tendinopathy in fetus
- Human data relatively reassuring but avoid as precaution
- Drugs: Ciprofloxacin, levofloxacin, moxifloxacin
- Alternative: Beta-lactams, azithromycin
14. Aminoglycosides
- Effect: Ototoxicity — 8th cranial nerve damage → irreversible sensorineural hearing loss in neonate
- Drugs: Streptomycin (highest risk), gentamicin, tobramycin, amikacin
- Use only if essential (life-threatening infection with no alternative); monitor levels
15. Chloramphenicol (near term)
- Effect: Gray baby syndrome — cardiovascular collapse, cyanosis, abdominal distension, death
- Mechanism: Immature neonatal hepatic glucuronyl transferase → drug accumulation
- Avoid especially in 3rd trimester and near delivery
16. Sulfonamides (near term)
- Effect: Neonatal hyperbilirubinemia / kernicterus — compete with bilirubin for albumin binding → free bilirubin crosses BBB
- Avoid in 3rd trimester and near delivery
17. Metronidazole
- Avoid in 1st trimester (some evidence of mutagenicity in bacteria; human teratogenicity not established but precautionary)
- Generally safe in 2nd–3rd trimester when clearly indicated (e.g., bacterial vaginosis, Trichomonas)
NSAIDs and Analgesics
18. NSAIDs (especially 3rd trimester)
- Effect:
- Premature closure of ductus arteriosus → pulmonary hypertension of the newborn (PPHN)
- Oligohydramnios (renal vasoconstriction → reduced fetal urine output)
- Inhibit platelet function → fetal/neonatal bleeding
- Contraindicated from 30 weeks gestation onward (FDA 2020 warning extended to ≥20 weeks for oligohydramnios risk)
- Drugs: Ibuprofen, naproxen, diclofenac, indomethacin
- Safe alternative: Paracetamol (acetaminophen) — drug of choice for analgesia/antipyresis
19. Aspirin (high dose)
- High-dose aspirin: similar effects to NSAIDs (ductus arteriosus closure, bleeding)
- Low-dose aspirin (75–150 mg/day): safe and actually recommended in pre-eclampsia prevention from 12 weeks
- Avoid high-dose aspirin, especially in 3rd trimester
Hormonal Drugs
20. Combined Oral Contraceptive Pill (COCP)
- Theoretical risk of virilization of female fetus (progestogen component)
- Generally associated with low risk in practice, but discontinue if pregnancy confirmed
21. Danazol / Androgenic Progestogens
- Effect: Virilization of female fetus — clitoral hypertrophy, labial fusion
- Absolutely avoid
22. Diethylstilbestrol (DES)
- Historical use (1940s–1970s) for miscarriage prevention
- Effect: Vaginal clear cell adenocarcinoma in female offspring (DES daughters); structural reproductive anomalies
- No longer used
23. Misoprostol (in 1st trimester)
- Prostaglandin E1 analogue; used for cervical ripening, post-partum hemorrhage, and medical abortion
- If used in 1st trimester without completing abortion: Möbius sequence (facial nerve palsy, limb defects)
- Contraindicated as anti-ulcer drug in 1st trimester
CNS / Psychiatric Drugs
24. Lithium
- Effect: Ebstein's anomaly (tricuspid valve malformation, right heart defects) — risk ~2× background but absolute risk small
- Also: neonatal hypotonia, cyanosis, bradycardia ("floppy baby"), neonatal diabetes insipidus
- Use with caution; monitor levels; avoid if possible in 1st trimester
- Trimester: 1st (cardiac); all trimesters (neonatal toxicity)
25. Benzodiazepines
- 1st trimester: Possible association with oral cleft (controversial, small risk)
- 3rd trimester / near delivery: Neonatal withdrawal syndrome (jitteriness, hypotonia, respiratory depression) and floppy infant syndrome
- Use the lowest effective dose for the shortest possible time
- Drugs: Diazepam, lorazepam, clonazepam, alprazolam
26. MAO Inhibitors (MAOIs)
- Hypertensive crisis potential; fetal growth restriction; neonatal toxicity
- Generally avoid; switch to safer antidepressants (SSRIs relatively safer)
27. Sodium Valproate (repeated for emphasis)
- Highest teratogenic risk among antiepileptics — never use in women of childbearing potential without strict pregnancy prevention
Cytotoxic / Immunosuppressive Drugs
28. Cyclophosphamide
- Effect: Fetal growth restriction, limb defects, eye abnormalities, cleft palate; miscarriage
- Alkylating agent — DNA damage in developing fetus
- Avoid especially in 1st trimester
29. Mycophenolate Mofetil (MMF)
- Effect: Miscarriage, ear abnormalities (microtia, atresia), facial clefts, limb/heart/kidney defects
- FDA Category D/X level risk; mandatory pregnancy prevention with this drug
- Stop ≥6 weeks before planned conception
30. JAK Inhibitors (Tofacitinib, Upadacitinib, Filgotinib)
- Per Management of Pregnancy in IBD guidelines (p. 28): animal data show teratogenicity and fetal death; limited human data
- Relatively contraindicated; discontinue ≥4 weeks before planned conception (tofacitinib, upadacitinib) or ≥1 week (filgotinib)
31. Leflunomide
- Effect: Teratogenic in animals; active metabolite (teriflunomide) persists for years
- Requires washout procedure with cholestyramine before conception can be attempted
- Absolute contraindication during pregnancy
Miscellaneous
32. Radioactive Iodine (¹³¹I)
- Effect: Fetal thyroid ablation (thyroid begins concentrating iodine after 10–12 weeks) → permanent neonatal hypothyroidism
- Absolutely contraindicated from 2nd trimester onward
33. Retinoids (Acitretin, Tretinoin systemic)
- Similar to isotretinoin — vitamin A derivatives
- Acitretin: contraception required for 3 years after stopping (converts back to etretinate, which persists)
34. Ergotamine
- Effect: Uterotonic → placental vasoconstriction, fetal hypoxia, spontaneous abortion, premature labor
- Contraindicated throughout pregnancy
- Alternative for migraine: Paracetamol ± metoclopramide; sumatriptan (use with caution, limited data)
35. Quinine (high dose)
- High doses: uterotonic, auditory nerve damage, thrombocytopenia
- Low therapeutic doses for malaria: relatively safe when benefit outweighs risk
Summary Reference Table
| Drug/Class | Trimester of Greatest Risk | Fetal/Neonatal Effect |
|---|---|---|
| Thalidomide | 1st | Phocomelia, visceral defects |
| Isotretinoin | 1st | CNS, cardiac, craniofacial defects |
| Methotrexate | All | Neural tube defects, abortion |
| Warfarin | 1st, 3rd | Embryopathy, CNS defects, hemorrhage |
| Valproate | All (esp. 1st) | NTDs, cognitive impairment, autism |
| Carbamazepine | 1st | NTDs, craniofacial defects |
| Phenytoin | 1st | Fetal hydantoin syndrome |
| ACE inhibitors/ARBs | 2nd–3rd | Renal failure, oligohydramnios, skull defects |
| Statins | All | CNS and limb defects (theoretical) |
| Amiodarone | All | Neonatal thyroid dysfunction |
| Spironolactone | All | Feminization of male fetus |
| Tetracyclines | 2nd–3rd | Tooth discoloration, bone growth inhibition |
| Aminoglycosides | All | Sensorineural hearing loss |
| Chloramphenicol | 3rd | Gray baby syndrome |
| Sulfonamides | 3rd | Neonatal kernicterus |
| NSAIDs | 3rd (≥20 wk) | Ductus arteriosus closure, oligohydramnios |
| Lithium | 1st | Ebstein's anomaly |
| Benzodiazepines | 3rd | Neonatal withdrawal, floppy infant |
| Cyclophosphamide | 1st | Limb defects, growth restriction |
| Mycophenolate | All | Ear, facial, limb, cardiac defects |
| Leflunomide | All | Teratogenic; long washout required |
| Radioactive iodine | 2nd–3rd | Permanent fetal hypothyroidism |
| Ergotamine | All | Placental ischemia, abortion |
| Danazol | All | Virilization of female fetus |
| JAK inhibitors | All | Teratogenicity (animal data) |
Safe Alternatives for Common Conditions
| Condition | Avoid | Use Instead |
|---|---|---|
| Hypertension | ACE inhibitors, ARBs, statins | Methyldopa, labetalol, nifedipine |
| Pain/fever | NSAIDs (3rd trimester) | Paracetamol |
| Epilepsy | Valproate, phenytoin | Lamotrigine, levetiracetam (specialist guidance) |
| Anticoagulation | Warfarin | LMWH (enoxaparin) |
| Infection | Tetracyclines, fluoroquinolones | Amoxicillin, azithromycin, cephalosporins |
| Migraine | Ergotamine, NSAIDs (3rd trim) | Paracetamol, metoclopramide |
| Hyperthyroidism | Radioactive iodine | Propylthiouracil (1st trim), carbimazole (2nd–3rd) |
| RA/IBD | Methotrexate, leflunomide, JAKi | Sulfasalazine, hydroxychloroquine, biologics (case-by-case) |
Acute hematogenous osteomyelitis in children
acute hematogenous osteomyelitis children etiology pathogenesis clinical features diagnosis
acute hematogenous osteomyelitis children X-ray MRI bone metaphysis abscess
osteomyelitis children metaphysis pathology subperiosteal abscess sequestrum involucrum chronic complications
acute osteomyelitis children treatment antibiotics surgery drainage indications duration
Acute Hematogenous Osteomyelitis (AHO) in Children
Sources: Bailey & Love's Short Practice of Surgery, 28th ed. (pp. 656–657); Guide to Utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases (p. 80); Harrison's Principles of Internal Medicine, 21st ed. (p. 3948); PIDS/IDSA Guidelines.
Definition
Epidemiology
| Feature | Details |
|---|---|
| Incidence | ~8 per 100,000 children per year |
| Peak age | Prepubertal children (2–12 years); neonates form a separate high-risk group |
| Sex | Boys > Girls (2:1) — greater exposure to minor trauma |
| Laterality | Usually unilateral; multi-focal in neonates and immunocompromised |
| Most common bones | Distal femur > Proximal tibia > Proximal humerus > Proximal femur |
Etiology
Causative Organisms by Age
| Age Group | Primary Pathogens |
|---|---|
| Neonates (<3 months) | S. aureus, Group B Streptococcus (S. agalactiae), E. coli, Klebsiella spp. |
| Infants & toddlers (<4 years) | S. aureus, Kingella kingae (very common <4 yr), S. pneumoniae |
| School-age children (4–12 yr) | S. aureus (dominant), Streptococcus pyogenes |
| Adolescents | S. aureus; consider N. gonorrhoeae if sexually active |
| Sickle cell disease | Salmonella spp. + S. aureus |
| Immunocompromised / IV drug use | Pseudomonas aeruginosa, Candida spp. |
S. aureus is the leading pathogen across all pediatric age groups. MRSA strains (especially community-acquired USA300) produce more severe, complicated disease with higher rates of deep vein thrombosis, multi-focal involvement, and surgical need.
Kingella kingae is a fastidious gram-negative rod that colonizes the upper respiratory tract in young children. It is frequently missed on routine blood cultures — requires specialized media (blood culture bottles inoculated with bone aspirate).
Pathogenesis
Why the Metaphysis?
- Terminal capillary loops in the metaphysis make sharp hairpin bends → sluggish, turbulent blood flow → bacteria settle and adhere
- These sinusoidal capillaries lack phagocytic lining cells → bacteria evade early immune clearance
- High metabolic rate of the growth plate provides a favorable environment for bacterial growth
- Minor trauma (very common in active children) creates microhematomas in the metaphysis → bacteria colonize during episodes of transient bacteremia (e.g., from dental procedures, skin infections, upper respiratory tract infections)
Stages of Disease Progression
Bacteremia
↓
Metaphyseal seeding → Acute inflammation (hyperemia, edema, neutrophil infiltration)
↓
Pus formation within medullary cavity
(INTRAMEDULLARY / INTRAOSSEOUS ABSCESS)
↓
Rising intraosseous pressure → pus tracks through Volkmann's & Haversian canals
↓
Periosteum stripped from bone
(SUBPERIOSTEAL ABSCESS)
↓
Periosteum ruptures → pus enters soft tissues
(SOFT TISSUE ABSCESS)
↓
Disruption of periosteal blood supply → ischemic bone necrosis
(SEQUESTRUM — dead bone fragment)
↓
Periosteum lays down reactive new bone shell around sequestrum
(INVOLUCRUM) — Bailey & Love (p. 657)
↓
CHRONIC OSTEOMYELITIS (sinus tracts, recurrent flares)
Special Anatomical Considerations
| Anatomical Feature | Clinical Consequence |
|---|---|
| Intracapsular metaphyses (proximal femur, proximal humerus, proximal radius, distal fibula) | Metaphyseal infection can directly rupture into the joint → septic arthritis |
| Sympathetic joint effusion | Sterile fluid in adjacent joint — must distinguish from true septic arthritis (Bailey & Love, p. 656) |
| Neonatal transphyseal vessels | Infection crosses growth plate into epiphysis and joint → growth plate destruction, joint sepsis (as seen in MRI image below) |
Clinical Features
Symptoms
| Symptom | Details |
|---|---|
| Fever | High-grade (>38.5°C); may be absent or subtle in neonates |
| Severe bone pain | Localized to metaphysis; constant; worsened by movement or palpation |
| Refusal to use the limb | "Pseudoparalysis" — classic in toddlers |
| Limp / inability to weight-bear | Lower limb involvement |
| Systemic toxicity | Irritability, malaise, anorexia, tachycardia |
Signs
- Point tenderness directly over the metaphysis — the hallmark sign
- Local warmth, erythema, and swelling (later finding as infection reaches periosteum/soft tissue)
- Restricted active and passive movement of adjacent joint
- Muscle spasm and guarding
- Neonates: often subtle — irritability, reduced spontaneous limb movement, swelling, feeding difficulties
Investigations
Laboratory
| Investigation | Finding / Purpose |
|---|---|
| CRP | Most sensitive early marker; rises within hours of infection onset; best for monitoring response |
| ESR | Elevated; lags behind CRP (peaks 3–5 days); slow to normalize (weeks) |
| WBC + differential | Leukocytosis with neutrophilia; may be normal early |
| Procalcitonin | Elevated in bacterial infection; useful adjunct |
| Blood cultures (×2) | Positive in ~30–50% of pediatric AHO; draw before starting antibiotics |
| Bone/abscess aspirate | Most definitive microbiological diagnosis; culture + Gram stain + sensitivities |
Imaging
1. Plain X-ray
- First-line investigation — but insensitive in early disease
- Bone changes visible only after 10–21 days (requires 30–50% bone mineral loss)
- Early findings: soft tissue swelling, obliteration of fat planes
- Late findings: metaphyseal lytic lesion, periosteal reaction, cortical erosion, sequestrum
- Role: exclude fracture, tumor; establish baseline
2. Ultrasound
- Detects subperiosteal fluid and soft tissue abscess within days of onset
- Guides needle aspiration for culture
- Cannot assess bone marrow or cortex directly
3. MRI — Gold Standard
- Most sensitive and specific modality for early AHO
- Detects: bone marrow edema, intramedullary abscess, subperiosteal collection, soft tissue extension, transphyseal spread, joint involvement
- T1: low signal marrow (edema replaces fat)
- T2/STIR: hyperintense edema
- Gd-contrast T1 fat-sat: rim-enhancing abscess cavity

4. Bone Scintigraphy (Tc-99m)
- Sensitive within 24–48 hours of onset
- Useful for detecting multi-focal disease (whole-body survey)
- Less specific than MRI; involves ionizing radiation
- May be falsely negative in neonates (vascular compromise reduces uptake)
5. CT Scan
- Best for defining cortical destruction, sequestrum, and involucrum in subacute/chronic cases
- Guides surgical planning
- Not first-line due to radiation exposure in children
Comparative Imaging Accuracy vs. MRI (PIDS/IDSA Guidelines)
| Modality | Sensitivity | Specificity |
|---|---|---|
| Bone scintigraphy | 53–91% | 47–84% |
| CT scan | 67–100% | 50–67% |
| Ultrasonography | 17–60% | ~47% |
| MRI | Reference standard | Reference standard |
Differential Diagnosis
| Condition | Key Distinguishing Features |
|---|---|
| Septic arthritis | Pain/tenderness maximal over joint; all movements painful; joint effusion on USS |
| Cellulitis | Superficial; no bone tenderness; no deep imaging changes |
| Ewing's sarcoma | Insidious onset; "onion-skin" periosteal reaction on X-ray; requires biopsy |
| Langerhans cell histiocytosis | Lytic lesion; chronic course; "beveled edge" on X-ray |
| Leukemia | Diffuse bone pain; cytopenias; "leukemic lines" on X-ray; bone marrow biopsy |
| Transient synovitis (hip) | Afebrile or low-grade fever; CRP normal or mildly raised; self-limiting |
| Fracture | Clear trauma history; fracture line on X-ray |
| Sickle cell bone crisis | Known SCD; multi-focal; CRP may be normal; often indistinguishable without culture |
| Rheumatic fever | Migratory polyarthritis; elevated ASO titre; carditis |
Kocher criteria (hip): fever + non-weight-bearing + ESR >40 mm/hr + WBC >12,000/μL — 4/4 criteria gives ~99% probability of septic arthritis requiring emergency surgery.
Treatment
General Principles (Bailey & Love, p. 657)
- Rest and splintage of the affected limb — reduces pain and prevents pathological fracture
- Analgesia — adequate pain control
- Antibiotic therapy — parenteral initially, then oral
- Surgical drainage — when pus is present
- Treatment of underlying conditions — sickle cell disease, nutritional deficiency, immunodeficiency
Antibiotic Therapy
Empirical Selection Based on MRSA Prevalence
| Setting | Drug | Route | Dose |
|---|---|---|---|
| Low MRSA prevalence | Oxacillin / Nafcillin / Flucloxacillin | IV | 150–200 mg/kg/day ÷ q4–6h |
| Low MRSA (alternative) | Cefazolin | IV | 100–150 mg/kg/day ÷ q8h |
| High MRSA prevalence | Vancomycin | IV | 15–20 mg/kg/dose q6h (target AUC/MIC 400–600) |
| MRSA (alternative) | Clindamycin (if susceptible) | IV/PO | 10–13 mg/kg/dose q6–8h |
| Neonates | Oxacillin + Gentamicin | IV | Weight/age adjusted |
| Sickle cell disease | Ceftriaxone + Oxacillin (or Vancomycin) | IV | Standard dosing |
IV-to-Oral Sequential Therapy
- Child is afebrile and clinically improving
- CRP trending down significantly
- Tolerating oral intake
- Organism identified and susceptible to an oral agent with high bioavailability
| Organism | Oral Drug |
|---|---|
| MSSA | Cephalexin / Dicloxacillin / Amoxicillin-clavulanate |
| MRSA (clinda-S) | Clindamycin |
| MRSA (clinda-R) | TMP-SMX |
| Salmonella (sickle cell) | Ciprofloxacin |
| Kingella kingae | Amoxicillin / Amoxicillin-clavulanate |
Total Duration
| Scenario | Duration |
|---|---|
| Uncomplicated AHO, good response | 3–4 weeks |
| Complicated AHO (abscess, delayed presentation) | 4–6 weeks |
| AHO + concurrent septic arthritis | 4–6 weeks |
| Neonatal AHO | 4–6 weeks |
| Chronic / recurrent osteomyelitis | 6+ weeks (guided by surgery) |
Surgical Treatment
Indications for Surgery
| Indication | Urgency |
|---|---|
| Subperiosteal or intraosseous abscess on imaging | Urgent |
| No clinical improvement after 48–72 h of appropriate antibiotics | Urgent |
| Septic arthritis (adjacent joint involvement) | Emergency |
| Sequestrum formation (necrotic bone) | Semi-urgent/elective |
| Neurological compromise (spinal AHO) | Emergency |
| Neonatal AHO (high spread risk to growth plate) | Early surgical drainage |
Surgical Procedures
-
Needle aspiration / bone aspiration
- Diagnostic + therapeutic
- Drains subperiosteal collections
- Performed under ultrasound guidance or in theater
-
Surgical drainage and debridement
- Cortical windowing: drill holes / bone window to decompress and drain intramedullary pus
- Curettage of necrotic tissue
- Irrigation of the cavity
- Closed suction drain placement
-
Sequestrectomy
- Removal of dead bone (sequestrum) in subacute/chronic cases
- Only performed once sufficient involucrum has formed to maintain structural integrity
-
Joint washout / arthrotomy
- Emergency for concurrent septic arthritis
- Hip joint = surgical emergency — delay risks avascular necrosis of femoral head
Monitoring Response to Treatment
| Parameter | Frequency | Expected Response |
|---|---|---|
| CRP | Every 2–3 days initially | Falls 30–50% within 48–72 h of effective treatment |
| ESR | Weekly | Lags behind CRP; takes weeks to normalize |
| WBC | Every 2–3 days | Normalizes within days |
| Temperature / clinical exam | Daily | Afebrile within 48–72 h |
| MRI | Only if deterioration or no improvement | Not routinely repeated |
Failure to improve within 48–72 hours = reassess: wrong antibiotic? resistant organism? undrained abscess? wrong diagnosis?
Complications
Early
| Complication | Notes |
|---|---|
| Septic arthritis | Especially hip, shoulder (intracapsular metaphyses); emergency |
| Pathological fracture | Cortical weakening by lytic process |
| Septicemia / multi-organ failure | Severe MRSA or neonatal disease |
| Deep vein thrombosis | Particularly community-acquired MRSA |
Late (if inadequately treated)
| Complication | Notes |
|---|---|
| Chronic osteomyelitis | Sequestrum, persistent sinus tracts, recurrent flares |
| Growth disturbance | Physeal damage → limb length discrepancy, angular deformity |
| Avascular necrosis | Particularly femoral head in hip septic arthritis |
| Brodie's abscess | Walled-off chronic intraosseous abscess; sclerotic rim on X-ray |
| Garre's sclerosing osteomyelitis | Chronic non-suppurative form; dense cortical sclerosis |
Special Situations
| Situation | Key Points |
|---|---|
| Neonatal AHO | Subtle presentation; multi-focal in 40%; transphyseal spread common; early drainage; 4–6 weeks antibiotics |
| Pelvic AHO | Often misdiagnosed (mimics appendicitis, septic hip); MRI essential; usually responds to antibiotics alone |
| Vertebral AHO (discitis) | Back pain, refusal to walk; lumbar > thoracic; MRI key; usually antibiotics alone unless epidural abscess |
| Severe MRSA-AHO | Multi-focal, DVT, septic pulmonary emboli; consider Vancomycin + Clindamycin (anti-toxin effect) |
| Sickle cell AHO | Cover Salmonella + S. aureus; clinically indistinguishable from vaso-occlusive crisis |
Summary
| Feature | Key Point |
|---|---|
| Peak age | Prepubertal children; neonates separately |
| Most common site | Distal femur metaphysis > proximal tibia |
| Most common pathogen | S. aureus at all ages; Kingella kingae in <4 yr |
| Pathogenesis | Sluggish metaphyseal capillary flow → seeding → abscess → sequestrum |
| Hallmark sign | Point tenderness over metaphysis |
| Best early lab marker | CRP |
| Gold standard imaging | MRI |
| Antibiotic duration | 3–6 weeks; IV → oral switch after 2–4 days |
| Surgery | When abscess present or antibiotics fail in 48–72 h |
| Most feared complication | Hip septic arthritis → AVN of femoral head |
| Chronic sequela | Sequestrum + involucrum → chronic osteomyelitis |