Causes of jaundice in neonates based on day of life
neonatal jaundice causes by day of life classification
neonatal hyperbilirubinemia etiology physiological pathological
neonatal jaundice day 1 hemolytic disease ABO Rh incompatibility
prolonged neonatal jaundice breast milk jaundice hypothyroidism causes
Neonatal Jaundice: Causes by Day of Life
Key Principle
Day-by-Day Classification
🔴 Day 1 (< 24 hours of life) — Always Pathological
| Cause | Mechanism |
|---|---|
| Rh incompatibility (Rh hemolytic disease) | Maternal anti-D IgG crosses placenta → fetal RBC destruction |
| ABO incompatibility | Maternal anti-A or anti-B IgG → hemolysis |
| Other blood group incompatibility (Kell, Duffy, Kidd) | Alloimmune hemolysis |
| Congenital infections (TORCH) | CMV, rubella, toxoplasmosis, syphilis → hemolysis + hepatitis |
| G6PD deficiency | Enzyme deficiency → oxidative hemolysis (can present day 1) |
🟡 Day 2–3 — Physiological + Pathological
| Cause | Type | Notes |
|---|---|---|
| Physiological jaundice | Unconjugated | Most common; due to immature UGT1A1, high RBC turnover, enterohepatic circulation. Peaks day 3–4 in term infants |
| Prematurity | Unconjugated | More profound due to greater hepatic immaturity |
| Polycythemia | Unconjugated | Increased RBC breakdown → more bilirubin load |
| Cephalohematoma / bruising | Unconjugated | Extravascular blood breakdown |
| ABO/Rh incompatibility (ongoing) | Unconjugated | If not treated from day 1 |
| G6PD deficiency | Unconjugated | Can manifest on day 2–3, especially with oxidant exposure |
🟡 Day 4–7 — Pathological Causes More Prominent
| Cause | Type | Notes |
|---|---|---|
| Breast milk jaundice (early / breastfeeding jaundice) | Unconjugated | Due to inadequate intake → increased enterohepatic circulation |
| Sepsis / infection | Conjugated or unconjugated | E. coli, Staph → hemolysis or hepatic dysfunction |
| Crigler-Najjar syndrome (Type I & II) | Unconjugated | Complete or partial UGT1A1 deficiency |
| Gilbert syndrome | Unconjugated | Mild; often incidental |
| Hypothyroidism / hypopituitarism | Unconjugated | Impaired hepatic maturation |
| Pyloric stenosis | Unconjugated | Increased enterohepatic circulation |
| Hemolytic anemias (hereditary spherocytosis, elliptocytosis) | Unconjugated | RBC membrane defects |
🟠 > Day 7 (Prolonged Jaundice) — Beyond 2 Weeks (Term) / 3 Weeks (Preterm)
Unconjugated Causes
| Cause | Notes |
|---|---|
| Breast milk jaundice (late) | Most common cause of prolonged jaundice; benign; mediated by β-glucuronidase in breast milk increasing enterohepatic circulation |
| Hypothyroidism | Screen with TSH/T4; neonatal screening programs |
| Hemolytic disease (ongoing) | G6PD, spherocytosis, etc. |
| Crigler-Najjar | Severe, persistent unconjugated hyperbilirubinemia |
Conjugated Causes (Direct bilirubin > 1 mg/dL or > 20% of total) — Always Pathological
| Cause | Notes |
|---|---|
| Biliary atresia | Most urgent — requires Kasai procedure before 8 weeks; hallmark is acholic stools + dark urine |
| Neonatal hepatitis (idiopathic or viral) | CMV, hepatitis B, EBV |
| Choledochal cyst | Biliary dilation |
| Alagille syndrome | Bile duct paucity + dysmorphic features |
| Total parenteral nutrition (TPN) cholestasis | Common in premature infants on prolonged TPN |
| Alpha-1 antitrypsin deficiency | Metabolic liver disease |
| Galactosemia / tyrosinemia | Metabolic disorders |
| Progressive familial intrahepatic cholestasis (PFIC) | Genetic bile transport defects |
| Sepsis-associated cholestasis | E. coli UTI particularly implicated |
Summary Table
| Timing | Pattern | Common Causes |
|---|---|---|
| < 24 hours | Always pathological | Rh/ABO incompatibility, TORCH, G6PD |
| Day 2–3 | Physiological peak | Physiological jaundice, prematurity, cephalohematoma |
| Day 4–7 | Mixed | Breastfeeding jaundice, sepsis, hemolytic anemias, metabolic |
| > 2 weeks (term) / > 3 weeks (preterm) | Prolonged | Breast milk jaundice (unconj.), biliary atresia (conj.) — must split bilirubin |
Key Clinical Pearls
- Any jaundice in the first 24 hours = hemolysis until proven otherwise; send urgent bilirubin + Coombs test.
- Conjugated (direct) hyperbilirubinemia at any age = always pathological; never physiological.
- Physiological jaundice peaks at day 3–4 in term infants, day 5–7 in preterm; resolves by 2 weeks (term) and 3 weeks (preterm). (Harrison's, p. 9341)
- Breast milk jaundice (late form) peaks at week 2–3 and can persist up to 12 weeks; it is a diagnosis of exclusion.
- Biliary atresia is a surgical emergency — the window for Kasai hepatoportoenterostomy closes by ~8 weeks of age.
Reticulocyte count
reticulocyte count corrected reticulocyte index interpretation anemia
reticulocyte production index hypoproliferative hyperproliferative anemia classification
reticulocyte count formula correction hematocrit maturation factor calculation
reticulocyte count anemia classification algorithm
Reticulocyte Count
What Are Reticulocytes?
- Normal reticulocyte count: 1–2%
- Reflects daily replacement of 0.8–1.0% of the circulating RBC population
Calculations
1. Raw (Uncorrected) Reticulocyte Percentage
2. Corrected Reticulocyte Count (CRC)
- Normal CRC: 1–2%
- CRC > 2% → adequate marrow response (hemolysis/blood loss)
- CRC < 2% → inadequate marrow response
3. Reticulocyte Production Index (RPI)
| Hematocrit | Maturation Factor |
|---|---|
| 45% | 1.0 |
| 35% | 1.5 |
| 25% | 2.0 |
| 15% | 2.5 |
- RPI > 2.5 → Hyperproliferative (adequate marrow response) → think hemolysis or blood loss
- RPI < 2.0 → Hypoproliferative or maturation defect
4. Absolute Reticulocyte Count (ARC)
- Normal: 25,000–75,000 cells/µL (some sources: 50,000–100,000/µL)
- Most straightforward and increasingly preferred in modern automated analyzers
Clinical Interpretation: Anemia Classification
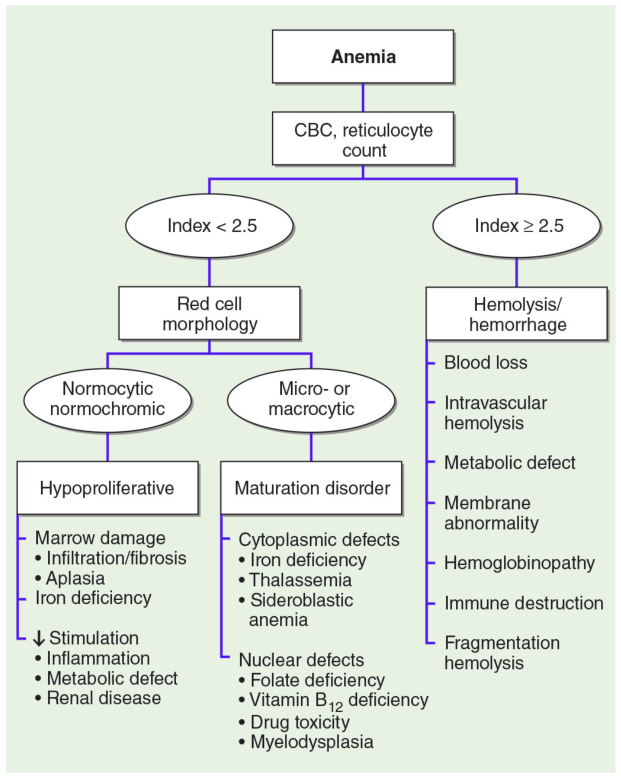
RPI ≥ 2.5 — Hyperproliferative (Marrow Responding)
| Category | Examples |
|---|---|
| Blood loss | Acute hemorrhage (GI bleed, trauma) |
| Hemolysis — immune | AIHA, transfusion reaction, drug-induced |
| Hemolysis — intrinsic RBC defect | G6PD deficiency, hereditary spherocytosis, sickle cell |
| Hemolysis — intravascular | TTP/HUS, DIC, mechanical heart valves |
| Hemolysis — hemoglobinopathy | Sickle cell crisis, thalassemia |
RPI < 2.0 — Hypoproliferative or Maturation Disorder (Marrow Failing to Respond)
A. Normocytic Normochromic → Hypoproliferative
| Cause | Notes |
|---|---|
| Iron deficiency (early) | MCV normal early on |
| Anemia of chronic disease | Most common cause in hospitalized patients |
| Renal failure | ↓ EPO production |
| Bone marrow aplasia | Aplastic anemia |
| Bone marrow infiltration | Leukemia, myeloma, myelofibrosis |
| Hypothyroidism / hypopituitarism | ↓ EPO stimulus |
B. Microcytic Hypochromic → Maturation Disorder (Iron/Heme defect)
| Cause | Notes |
|---|---|
| Iron deficiency anemia | Most common anemia worldwide |
| Thalassemia | Ineffective erythropoiesis |
| Sideroblastic anemia | Ringed sideroblasts on marrow |
| Anemia of chronic disease | Can be microcytic |
C. Macrocytic → Maturation Disorder (Nuclear/DNA defect)
| Cause | Notes |
|---|---|
| Vitamin B12 deficiency | Megaloblastic; hypersegmented neutrophils |
| Folate deficiency | Megaloblastic |
| Myelodysplastic syndrome | Dysplastic cells on smear |
| Drug-induced (hydroxyurea, methotrexate) | Impaired DNA synthesis |
| Liver disease / alcohol | Non-megaloblastic macrocytosis |
Quick Reference Summary
| Measure | Formula | Normal | Interpretation |
|---|---|---|---|
| Raw reticulocyte % | Retics/Total RBC × 100 | 1–2% | Unreliable alone in anemia |
| Corrected retic count | Retic% × (Pt Hct / 45) | 1–2% | Adjusts for anemia |
| RPI | CRC / Maturation factor | ≥ 2.0 (normal response) | < 2 = marrow failure; > 2.5 = hemolysis/loss |
| Absolute retic count | Retic% × RBC count | 25,000–75,000/µL | Most direct measure of output |
Clinical Pearls
- Always use RPI or ARC rather than raw reticulocyte percentage — the raw percentage is misleading in anemia.
- A high RPI with a falling Hb means the marrow is responding but can't keep up — look for ongoing hemolysis or blood loss.
- A low RPI despite severe anemia is a marrow failure signal — consider bone marrow examination.
- In post-treatment monitoring (e.g., iron, B12 therapy), a rising reticulocyte count ("reticulocyte crisis") at 5–10 days confirms the diagnosis and treatment response.
- Reticulocytosis is also expected after EPO therapy, recovery from aplastic anemia, or after stopping a marrow-suppressive agent.
What to see in newborn if child is having ctev
congenital talipes equinovarus clubfoot clinical examination findings newborn
clubfoot CTEV assessment Pirani score Dimeglio classification neonatal
CTEV clubfoot deformity components cavus adductus varus equinus
congenital talipes equinovarus clubfoot newborn examination
Clinical Examination of a Newborn with CTEV
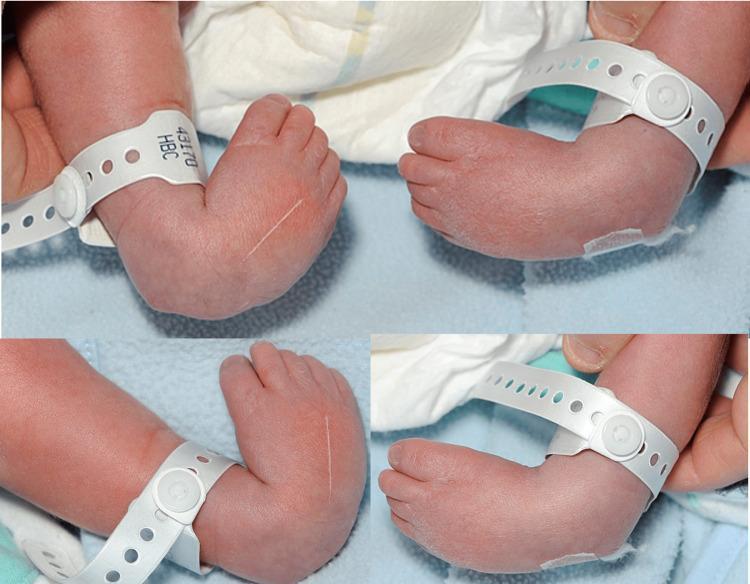
The Four Core Deformity Components (CAVE mnemonic)
| Component | Description | Location |
|---|---|---|
| C — Cavus | High medial longitudinal arch; plantar flexion of 1st ray | Forefoot/midfoot |
| A — Adductus | Forefoot deviated medially (inward) | Midfoot |
| V — Varus | Heel tilted inward | Hindfoot |
| E — Equinus | Foot pointed downward (plantar flexion at ankle) | Hindfoot/ankle |
All four components are present in true CTEV. The deformity is fixed, unlike postural talipes which is fully correctable.
Step-by-Step Examination
1. Look (Inspection)
- Overall posture of foot: plantarflexed, inverted, adducted
- Hindfoot: small, underdeveloped heel; varus position of calcaneus
- Calf: may show calf muscle hypoplasia (thinner calf compared to normal side — especially in unilateral cases)
- Skin creases:
- Deep medial crease at the midfoot (due to adductus/cavus)
- Deep posterior crease at the ankle (due to equinus)
- These creases indicate severity — deeper creases = more rigid deformity
- Foot size: the affected foot is often shorter and smaller
- Bilateral vs. unilateral: note if one or both feet are involved (bilateral in ~50%)
2. Feel (Palpation)
- Head of talus: prominently palpable on the dorsolateral aspect of the foot (as navicular is displaced medially)
- Calcaneus: small, difficult to palpate in severe cases; positioned in varus and equinus
- Medial structures: tight — tibialis posterior, flexor digitorum longus, flexor hallucis longus tendons
- Posterior structures: tight — tendo-Achilles, posterior capsule
- Muscle bulk: assess calf bulk bilaterally
3. Move (Mobility — Most Important Step)
- True CTEV: deformity is fixed/rigid — cannot be passively corrected to neutral
- Postural talipes: deformity is fully correctable to neutral or beyond — benign, resolves with physiotherapy
- Dorsiflexion at ankle (equinus correction)
- Eversion at subtalar joint (varus correction)
- Abduction of forefoot (adductus correction)
4. Severity Scoring
Pirani Scoring System (0–6)
| Sign | What to Assess |
|---|---|
| Medial crease | Depth of medial skin crease |
| Curvature of lateral border | Normally straight — curved = adductus |
| Talar head coverage | How much talar head is uncovered laterally |
| Posterior crease | Depth of posterior ankle crease |
| Rigid equinus | Degree of fixed plantarflexion |
| Empty heel | How empty/underfilled the heel feels |
- Score 0 = normal foot
- Score 6 = most severe
- Predicts number of Ponseti casts needed and likelihood of tenotomy
Diméglio Classification (0–20)
| Grade | Score | Severity |
|---|---|---|
| I | 1–5 | Benign (soft-soft) |
| II | 6–10 | Moderate (soft-stiff) |
| III | 11–15 | Severe (stiff-soft) |
| IV | 16–20 | Very severe (stiff-stiff) |
5. Associated Findings to Look For
| System | What to Look For |
|---|---|
| Spine | Neural tube defects, spina bifida (→ neurogenic clubfoot) |
| Hips | Developmental dysplasia of hip (DDH) — check Barlow/Ortolani |
| Knees | Knee contractures, popliteal webbing |
| Upper limbs | Arthrogryposis multiplex congenita (multiple joint contractures) |
| General dysmorphism | Chromosomal syndromes (trisomy 18, 13) |
| Neurological | Tone, reflexes — rule out myelomeningocele, spinal muscular atrophy |
6. Differentiate True CTEV from Postural Talipes
| Feature | True CTEV | Postural Talipes |
|---|---|---|
| Reducibility | Fixed, non-correctable | Fully correctable passively |
| Skin creases | Deep medial and posterior | Absent or shallow |
| Calf hypoplasia | Present | Absent |
| Foot size | Smaller than normal | Normal |
| Treatment | Ponseti casting + possible tenotomy | Physiotherapy/stretching |
Key Clinical Pearls
- First step in management after diagnosis: refer early for Ponseti method (serial casting) — ideally started within the first 1–2 weeks of life when ligaments are most pliable.
- The lateral border of a normal foot is straight — a curved lateral border on inspection is a reliable sign of forefoot adductus.
- Calf wasting in unilateral cases is a clue to an underlying neurological cause (e.g., spina bifida, polio).
- Always palpate the head of the talus — in CTEV it is uncovered laterally and is the key landmark for Ponseti casting correction.
- A deep posterior crease + rigid equinus = higher likelihood of needing Achilles tenotomy.