management of traumatic perforation of tympanic membrane in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances
traumatic perforation tympanic membrane management treatment
tympanic membrane perforation spontaneous healing myringoplasty surgical repair
traumatic tympanic membrane perforation diagram management flowchart
traumatic tympanic membrane perforation etiology causes blast injury slap barotrauma
myringoplasty tympanoplasty technique graft underlay overlay fascia fat paper patch
tympanic membrane perforation types anatomy otoscopy
TRAUMATIC PERFORATION OF THE TYMPANIC MEMBRANE
Comprehensive Management — 50 Marks Answer
(Scott-Brown | Cummings | Stell & Maran | Zakir Hussain | Dhingra | Hazarika | Recent Advances)
1. INTRODUCTION
2. APPLIED ANATOMY
┌─────────────────────────────────────────────────────────────┐
│ TYMPANIC MEMBRANE — ANATOMY │
│ │
│ Pars Flaccida (Shrapnell's membrane) │
│ ↑ │
│ ┌─────┴──────────────────────────────────┐ │
│ │ PARS TENSA │ │
│ │ ┌──────────────────────────────────┐ │ │
│ │ │ Anterior │ Posterior │ │ │
│ │ │ Superior │ Superior │ │ │
│ │ ├────────────┼────────────────────-┤ │ │
│ │ │ Anterior │ Posterior │ │ │
│ │ │ Inferior │ Inferior │ │ │
│ │ └──────────────────────────────────┘ │ │
│ │ Umbo (center) │ │
│ │ Cone of Light │ │
│ └────────────────────────────────────────┘ │
│ │
│ Layers: Squamous epithelium → Fibrous layer → Mucosa │
│ Blood supply: Deep auricular artery (outer) │
│ Anterior tympanic artery (inner) │
│ Nerve supply: Auriculotemporal + Arnold's nerve │
└─────────────────────────────────────────────────────────────┘
- Pars tensa: 85% of TM — site of most traumatic perforations
- Pars flaccida: 15%, lacks fibrous layer, rarely traumatically perforated
- Annulus fibrosus: Peripheral fibrocartilaginous ring anchoring TM to tympanic sulcus
3. ETIOLOGY AND CLASSIFICATION OF TRAUMATIC PERFORATION
3.1 Causes (Dhingra — Diseases of ENT, 8th Ed.)
| Category | Mechanism | Examples |
|---|---|---|
| Direct (Mechanical) | Sharp/blunt object penetrating EAC | Cotton buds, hair pins, welding sparks, pencils, forceful syringing |
| Indirect (Barotrauma) | Sudden pressure change | Slap on ear (commonest), blast injury, explosion, diving, flying, kiss on ear |
| Thermal | Heat/chemical injury | Welding sparks (molten metal), chemical burns, thermal burns |
| Iatrogenic | Procedural | Syringing, myringotomy complications, instrumentation |
| Lightning strike | Combined blast + thermal | — |
| Penetrating head trauma | Basilar skull fracture | Associated with hemotympanum |
3.2 Pathomechanism
- Compression type (indirect/blast): Sudden increase in EAC pressure → TM unable to equilibrate → rupture, typically in anterior inferior quadrant (the thinnest area)
- Laceration type (direct): Sharp instrument perforates TM → ragged, irregular edges; may involve ossicles
3.3 Classification by Site (Scott-Brown's Otolaryngology, 8th Ed.)
┌───────────────────────────────────────────────┐
│ CLASSIFICATION OF PERFORATIONS │
│ │
│ By LOCATION: │
│ • Central — within pars tensa, rim of │
│ annulus intact (SAFE) │
│ • Marginal — reaches tympanic annulus │
│ (risk of cholesteatoma) │
│ • Attic (Pars flaccida) — dangerous │
│ │
│ By SIZE: │
│ • Small: <25% of TM area │
│ • Medium: 25–50% │
│ • Large: 50–75% │
│ • Subtotal: >75%, annulus intact │
│ • Total: entire TM lost │
│ │
│ By APPEARANCE (Acute): │
│ • Slit-like, stellate, or crescentic │
│ • Edges: fresh (red) vs. rolled (chronic) │
└───────────────────────────────────────────────┘
4. CLINICAL FEATURES
4.1 Symptoms
| Symptom | Mechanism |
|---|---|
| Sudden severe otalgia | TM disruption + mucosal exposure |
| Bleeding from ear | Tearing of vascular epithelial layer |
| Conductive hearing loss | Loss of acoustic coupling (magnitude ∝ size) |
| Tinnitus | Perilymphatic fistula / inner ear concussion |
| Vertigo/disequilibrium | Inner ear involvement, PLF, labyrinthine concussion |
| Sensation of fullness | Middle ear exposure |
| Facial palsy (rare) | Associated temporal bone fracture |
4.2 Signs
- Otoscopy: Irregular rent in TM (acute — red edges, fresh blood); rolled white epithelial edges in healing phase
- Rinne's test: Negative (BC > AC) — conductive hearing loss
- Weber's test: Lateralises to the affected ear
- Pure Tone Audiogram: Air-bone gap (typically 20–40 dB) in low frequencies
- Tuning fork tests: 256 Hz and 512 Hz
- Tympanometry: Type B (flat) curve
5. INVESTIGATIONS
┌────────────────────────────────────────────────────────────┐
│ INVESTIGATIONS — ALGORITHMIC APPROACH │
├────────────────────────────────────────────────────────────┤
│ 1. Otoscopy (diagnostic) │
│ ↓ │
│ 2. Pure Tone Audiogram (PTA) │
│ - Air-bone gap quantification │
│ - SNHL component → suspect inner ear injury │
│ ↓ │
│ 3. Tympanometry │
│ - Type B: flat (perforation/fluid) │
│ - Type Ad: ossicular discontinuity │
│ ↓ │
│ 4. HRCT Temporal Bone │
│ - Indicated if: SNHL, vertigo, facial palsy, │
│ suspected ossicular disruption, blast/fracture │
│ - Assess: ossicular chain, tegmen, labyrinth │
│ ↓ │
│ 5. Speech Audiometry / Impedance audiometry │
│ 6. Vestibular function tests (if vertigo present) │
│ - ENG/VNG, VEMP │
│ 7. MRI (if intracranial extension suspected) │
└────────────────────────────────────────────────────────────┘
6. MANAGEMENT
OVERVIEW FLOWCHART
┌────────────────────────────────────────────────────────────────────────┐
│ TRAUMATIC TM PERFORATION — MANAGEMENT ALGORITHM │
└────────────────────────────────────────────────────────────────────────┘
│
▼
┌───── INITIAL ASSESSMENT ─────┐
│ History + Otoscopy + PTA │
│ Check: size, site, infection │
│ vertigo, SNHL, facial nerve │
└───────────────┬───────────────┘
│
┌───────────────▼───────────────┐
│ EMERGENCY FEATURES? │
│ • Profound SNHL │
│ • Perilymph fistula │
│ • Facial nerve palsy │
│ • Temporal bone fracture │
└──────┬────────────────┬────────┘
│YES │NO
▼ ▼
┌──────────────┐ ┌─────────────────────┐
│ URGENT REFER │ │ CONSERVATIVE Rx │
│ to Neurotol. │ │ (Watchful waiting) │
│ + HRCT TB │ └──────────┬───────────┘
└──────────────┘ │
▼
┌──────────────────────────┐
│ REVIEW at 3 MONTHS │
│ Has it healed? │
└──────┬──────────┬─────────┘
│YES │NO
▼ ▼
┌──────────┐ ┌──────────────────────┐
│ DISCHARGE│ │ SURGICAL REPAIR │
│ with │ │ Myringoplasty/ │
│ PTA │ │ Tympanoplasty │
└──────────┘ └──────────────────────┘
6.1 CONSERVATIVE MANAGEMENT
A. Immediate First Aid
- Do NOT irrigate the ear — risk of introducing infection into middle ear
- Keep the ear dry — cotton wool plug lightly placed at meatus (NOT packed tightly)
- No ear drops unless infected — especially avoid aminoglycoside drops (ototoxic with open TM)
- Reassurance — most small traumatic perforations heal spontaneously
B. Medical Management
| Measure | Rationale | Details |
|---|---|---|
| Systemic antibiotics | Prevent secondary infection | Amoxicillin-clavulanate 625 mg TDS × 7 days OR Ciprofloxacin (oral) |
| Analgesics | Pain control | Paracetamol / NSAIDs |
| Ear drops | Only if infected | Ciprofloxacin ear drops (NOT aminoglycosides) |
| Dry ear precautions | Prevent water contamination | Petroleum jelly-coated cotton ball while bathing |
| Avoid nose blowing | Prevent Eustachian tube-mediated pressure | — |
| Avoid swimming/diving | Prevent infection | For minimum 4–6 weeks |
| Avoid aviation | Barotrauma prevention | Until healed |
C. Observation Period
- 85–90% of traumatic perforations <50% size heal spontaneously within 4–8 weeks (Scott-Brown, 8th Ed.)
- Bailey & Love (28th Ed., p. 777): "Traumatic perforations of the tympanic membrane usually heal spontaneously but explosive and welding injuries do not."
- Exceptions with poor spontaneous healing:
- Blast/explosion injuries
- Welding spark injuries (thermal coagulation of edges prevents epithelial migration)
- Large perforations (>50%)
- Infections complicating perforation
- Marginal perforations
6.2 OFFICE PROCEDURES (NON-SURGICAL REPAIR)
A. Chemical Cauterisation / Edge Freshening
- Applied to rolled, epithelialised edges to stimulate healing
- Agent: 50% Trichloroacetic acid (TCA) applied with fine applicator to edge only
- Mechanism: Destroys epithelium migrating across perforation, stimulates fibrous regeneration
- Indication: Small perforations with epithelialised edges after 6 weeks; perforations not healing spontaneously
B. Paper Patch Myringoplasty (Fat Plug / Cigarette Paper Patch)
- Historical technique (Derlacki, 1953; later popularised by Rizer)
- Indication: Small, dry, central perforations in clinic setting
- Procedure:
- Edges freshened with a fine needle (de-epithelialisation)
- Phenol/TCA applied to edges
- Cigarette paper / Steri-strip / Gelfoam patch placed over defect
- Acts as scaffold for epithelial migration
- Success rate: 70–90% for perforations <30%
C. Fat Plug Myringoplasty (Ringenberg Technique)
- Indication: Small central perforations (<4 mm), office setting or minor OT
- Graft: Lobular fat from earlobe
- Technique: Fat plug placed through perforation using "dumbbell" technique — fat sits on both medial and lateral surfaces
- Advantages: Simple, quick, no hospital admission, high success rate (80–92%) for small perforations
- Hazarika (Textbook of ENT and Head & Neck Surgery): Advocates fat graft myringoplasty as first-line for small traumatic perforations
6.3 SURGICAL MANAGEMENT — MYRINGOPLASTY / TYMPANOPLASTY
When to Operate?
┌─────────────────────────────────────────────────────────────┐
│ INDICATIONS FOR SURGICAL REPAIR │
├─────────────────────────────────────────────────────────────┤
│ 1. Perforation not healed after 3 months observation │
│ 2. Large perforation (>50% TM area) │
│ 3. Blast/welding injuries (unlikely to heal spontaneously) │
│ 4. Marginal perforation (cholesteatoma risk) │
│ 5. Associated ossicular disruption / discontinuity │
│ 6. Patient preference / professional needs (swimming etc.) │
│ 7. Recurrent infections │
│ 8. Persistent conductive hearing loss │
├─────────────────────────────────────────────────────────────┤
│ CONTRAINDICATIONS │
├─────────────────────────────────────────────────────────────┤
│ 1. Active infection (operate after infection controlled) │
│ 2. Only hearing ear (relative contraindication) │
│ 3. Poor Eustachian tube function │
│ 4. Unfit for anaesthesia │
│ 5. Children <4–5 years (Eustachian tube immature) │
└─────────────────────────────────────────────────────────────┘
Wullstein Classification of Tympanoplasty (1956)
| Type | Procedure | Indication |
|---|---|---|
| Type I (Myringoplasty) | TM repair only, ossicular chain intact | Traumatic TM perforation with intact ossicles |
| Type II | TM repair + graft to incus | Malleus eroded |
| Type III | TM graft placed directly on stapes head (myringostapediopexy) | Malleus + incus absent |
| Type IV | Graft to mobile stapes footplate | Stapes suprastructure absent |
| Type V | Fenestration of lateral semicircular canal | Fixed stapes footplate |
Surgical Approaches
┌─────────────────────────────────────────────────────┐
│ SURGICAL APPROACH DECISION TREE │
├─────────────────────────────────────────────────────┤
│ │
│ Perforation size? │
│ │ │
│ Small/Medium Large/Total │
│ │ │ │
│ Transcanal approach Postauricular / Endaural │
│ (if good EAC access) approach │
│ │
│ Microscope vs Endoscope? │
│ • Endoscopic: better visualisation, no incision │
│ • Microscopic: traditional, better bimanual │
│ │
└─────────────────────────────────────────────────────┘
| Approach | Indication | Advantages |
|---|---|---|
| Transcanal | Anterior/small perforations, wide EAC | Minimal dissection, day care |
| Endaural (Lempert) | Medium perforations, anterior visibility needed | Good access, small scar |
| Postauricular (Wilde's incision) | Large/subtotal, narrow EAC, ossicular work | Best exposure, graft harvest nearby |
Graft Materials
┌────────────────────────────────────────────────────────────────┐
│ GRAFT MATERIALS COMPARISON │
├───────────────────┬────────────────┬───────────────────────────┤
│ GRAFT │ TAKE RATE │ NOTES │
├───────────────────┼────────────────┼───────────────────────────┤
│ Temporalis fascia │ 85–95% │ GOLD STANDARD — fibrous, │
│ (TF) │ │ strong, easily harvested │
├───────────────────┼────────────────┼───────────────────────────┤
│ Perichondrium │ 85–92% │ Good for atelectatic TM, │
│ (Tragal/conchal) │ │ stiffer, no retraction │
├───────────────────┼────────────────┼───────────────────────────┤
│ Cartilage + │ 90–97% │ Best for revision, tube │
│ Perichondrium │ │ otitis, poor ET function │
├───────────────────┼────────────────┼───────────────────────────┤
│ Fat (lobule) │ 75–90%* │ Office use, small defects │
├───────────────────┼────────────────┼───────────────────────────┤
│ Vein graft │ 70–85% │ Historical, rarely used │
├───────────────────┼────────────────┼───────────────────────────┤
│ Allografts │ Variable │ Donor TM — largely │
│ │ │ abandoned (infection risk) │
├───────────────────┼────────────────┼───────────────────────────┤
│ Synthetic │ 80–85% │ Biodesign, AlloDerm — │
│ (acellular) │ │ RECENT ADVANCE │
└───────────────────┴────────────────┴───────────────────────────┘
*For small perforations only
Graft Placement Techniques
┌───────────────────────────────────────────────────────────────────┐
│ UNDERLAY vs OVERLAY TECHNIQUE │
├──────────────────────────┬────────────────────────────────────────┤
│ UNDERLAY │ OVERLAY │
├──────────────────────────┼────────────────────────────────────────┤
│ Graft placed MEDIAL to │ Graft placed LATERAL to TM remnant │
│ TM remnant and handle │ and handle of malleus │
│ of malleus │ │
├──────────────────────────┼────────────────────────────────────────┤
│ Advantages: │ Advantages: │
│ • Technically easier │ • Better for anterior perforations │
│ • Less blunting │ • Good visualisation of graft │
│ • Less lateralization │ │
├──────────────────────────┼────────────────────────────────────────┤
│ Disadvantages: │ Disadvantages: │
│ • Anterior visibility ↓ │ • Lateralization risk │
│ • Medialisation possible │ • Blunting of anterior sulcus │
├──────────────────────────┼────────────────────────────────────────┤
│ Used by: Most surgeons │ Used by: Selected cases │
│ (Scott-Brown, Cummings) │ (large anterior perforations) │
└──────────────────────────┴────────────────────────────────────────┘
Step-by-Step Operative Technique: Postauricular Underlay Myringoplasty
STEP 1: Anaesthesia + Patient Positioning
└── GA (preferred) or LA with sedation
└── Head turned to opposite side, ear up
└── Subperiosteal infiltration (1:100,000 adrenaline)
└── Operating microscope / Endoscope setup
STEP 2: Incision
└── Postauricular incision (Wilde's) 5 mm behind post. auricular fold
└── Down to temporalis fascia
STEP 3: Graft Harvest
└── Temporalis fascia harvested (2×2 cm)
└── Dried on Teflon block (10–15 min)
└── Pressed thin — semitransparent sheet
STEP 4: Exposure of EAC
└── Postauricular flap elevated
└── Soft tissue from EAC posterior wall cleared
└── Ear speculum placed / posterior meatal skin elevated
STEP 5: Perforation Management
└── Edges of perforation freshened / de-epithelialised
└── 360° marginal strip removed with Rosen's needle
└── Tympanomeatal flap elevated (if needed)
└── Middle ear inspected — assess ossicles, mucosa
STEP 6: Graft Placement (Underlay technique)
└── Graft slid MEDIAL to TM remnant
└── Supported medially with Gelfoam packing
└── Graft draped over handle of malleus
└── Anterior flap repositioned
└── Graft edges checked — no folding
STEP 7: Closure
└── Gelfoam in EAC over graft
└── Pope wick / ribbon gauze
└── Postauricular wound closed in layers
└── Mastoid dressing applied
6.4 MANAGEMENT OF ASSOCIATED INJURIES
A. Ossicular Chain Disruption
- Commonest: Incudostapedial joint dislocation (Stell & Maran, 5th Ed.)
- Incus dislocation: Most common ossicular injury in temporal bone trauma (Bailey & Love, 28th Ed., p. 777: "With severe head trauma the incus can be displaced, which leads to a conductive hearing loss")
- Assessment: PTA showing >40 dB ABG, tympanometry Type Ad (Jerger)
- Management:
- Type II tympanoplasty: Ossiculoplasty — PORP / TORP (partial/total ossicular replacement prosthesis)
- Materials: Hydroxyapatite, titanium, Plastipore
- Timing: Can be staged — TM repair first, ossiculoplasty 6–12 months later
┌────────────────────────────────────────────────────────────┐
│ OSSICULAR INJURIES IN TEMPORAL BONE TRAUMA │
├───────────────────────┬────────────────────────────────────┤
│ INJURY │ TREATMENT │
├───────────────────────┼────────────────────────────────────┤
│ Incudostapedial │ Reposition / PORP │
│ joint dislocation │ │
├───────────────────────┼────────────────────────────────────┤
│ Incus dislocation │ Reposition or remove; PORP/TORP │
├───────────────────────┼────────────────────────────────────┤
│ Malleus fracture │ Ossiculoplasty │
├───────────────────────┼────────────────────────────────────┤
│ Stapes fracture/ │ Stapedectomy / TORP │
│ luxation │ │
├───────────────────────┼────────────────────────────────────┤
│ Stapedial tendon │ No specific treatment │
│ disruption │ │
└───────────────────────┴────────────────────────────────────┘
B. Perilymphatic Fistula (PLF)
- Mechanism: Blast overpressure → rupture of round/oval window membranes
- Features: Sensorineural hearing loss + vertigo after barotrauma
- Management:
- Conservative: Strict bed rest, head elevation 30°, stool softeners, avoid Valsalva (4–6 weeks)
- Surgical: Exploratory tympanotomy + fat/fascia patch to oval/round window if no improvement in 4–6 weeks
C. Inner Ear Concussion
- No membrane rupture, but cochlear/labyrinthine injury from pressure wave
- Features: SNHL, tinnitus, vertigo
- Management: Systemic corticosteroids (Prednisolone 1 mg/kg/day × 10 days), hyperbaric oxygen (emerging evidence)
D. Temporal Bone Fracture
- Longitudinal: 80% — follows EAC line; TM tear, blood in EAC, conductive HL; rare facial nerve palsy
- Transverse: 20% — crosses cochlea/labyrinth; profound SNHL, vertigo, facial palsy (50%)
- Management: HRCT temporal bone; neurosurgical consult; facial nerve decompression if paralysis complete + electrical evidence of degeneration (ENoG <10% at 72 hours — Cummings Otolaryngology, 7th Ed.)
6.5 SPECIAL SCENARIOS
Blast Injury / Explosive Injury
┌──────────────────────────────────────────────────────────────┐
│ BLAST INJURY MANAGEMENT PROTOCOL │
├──────────────────────────────────────────────────────────────┤
│ Primary blast wave → TM perforation + ossicular injury │
│ Secondary → shrapnel injury, penetrating trauma │
│ Tertiary → impact injury from body displacement │
│ │
│ Management: │
│ • All blast victims → PTA assessment │
│ • NO spontaneous healing expected │
│ • HRCT temporal bone (both sides) │
│ • High-dose steroids if SNHL component │
│ • Surgical repair at 3 months if not healed │
│ • Monitor for delayed perilymph fistula │
│ • Tympanoplasty results good (80–90% closure rate) │
└──────────────────────────────────────────────────────────────┘
Welding Spark Injuries
- Molten metal droplet burns through TM
- Edges are thermally cauterised → will not heal spontaneously
- Metal fragment may lodge in middle ear → HRCT mandatory
- Surgical removal of metal + myringoplasty required
- Risk of chronic suppurative otitis media if untreated
Paediatric Traumatic Perforation
- Zakir Hussain (ENT for Undergraduates): Most heal spontaneously even without treatment
- Management similar; surgical repair deferred until age 5–7 years due to:
- Immature Eustachian tube function
- Frequent upper respiratory infections
- Compliance issues
- Success rates of myringoplasty in children: 80–85% (comparable to adults when >7 years)
7. POSTOPERATIVE CARE AND FOLLOW-UP
IMMEDIATE (0–2 weeks):
• Mastoid dressing removed at 48 hours
• Systemic antibiotics × 5–7 days
• Analgesics
• Strict dry ear precautions
• No nose blowing, no sneezing with mouth closed
• Pope wick removed at 2 weeks
SHORT-TERM (2–6 weeks):
• Ear canal packing/Gelfoam dissolves
• Otoscopy — graft take assessment
• PTA at 6 weeks
MEDIUM-TERM (3–6 months):
• PTA at 3 months — ABG closure
• Graft viability assessment
• Evaluate for residual perforation
LONG-TERM:
• Annual audiometry if associated SNHL
• Monitor for cholesteatoma (marginal perforations)
8. COMPLICATIONS
8.1 Complications of Traumatic Perforation (Untreated)
| Complication | Mechanism |
|---|---|
| Chronic suppurative otitis media | Persistent infection through perforation |
| Cholesteatoma | Squamous epithelium migration (especially marginal perforations) |
| Persistent conductive HL | Ongoing ABG |
| Meningitis / brain abscess | Ascending infection (rare) |
| Tinnitus | Cochlear damage |
8.2 Complications of Myringoplasty
INTRAOPERATIVE:
• Chorda tympani nerve injury → taste disturbance (ageusia)
• Ossicular injury
• Carotid artery injury (rare, aberrant)
• Perilymph fistula (iatrogenic)
• Facial nerve injury (rare)
EARLY POSTOPERATIVE:
• Graft failure / displacement
• Haematoma
• Wound infection
• SNHL (rare)
LATE POSTOPERATIVE:
• Residual/recurrent perforation (5–15%)
• Graft lateralisation
• Blunting of anterior sulcus
• Tympanosclerosis
• Retraction pocket
• Cholesteatoma
• Adhesive otitis media
9. PROGNOSIS AND OUTCOMES
| Factor | Better Outcome | Worse Outcome |
|---|---|---|
| Perforation size | Small (<25%) | Large (>50%) |
| Cause | Mechanical/slap | Blast, welding |
| Duration | Acute (<3 months) | Chronic (>12 months) |
| Status | Dry, no infection | Wet, infected |
| Eustachian tube | Normal function | Dysfunctional |
| Graft type | Cartilage (for revision) | — |
| Surgeon experience | High volume | — |
| Age | Adults | Very young children |
10. RECENT ADVANCES
A. Endoscopic Ear Surgery (EES)
- Endoscopic myringoplasty: Introduced widely since 2010s
- Single-hand technique using 3 mm 0°/30°/45° Hopkins rod endoscopes
- Advantages: Panoramic view, no postauricular incision, day care, better anterior angle visualisation, less morbidity
- Outcomes: Comparable to microscopic (graft take 85–92%) — Tarabichi, Preyer studies
- Learning curve: Initially longer; now standard of care in many centres
B. Novel Scaffold Materials
- Acellular collagen matrix (Biodesign, Cook Medical): Xenograft scaffold — promotes regenerative healing
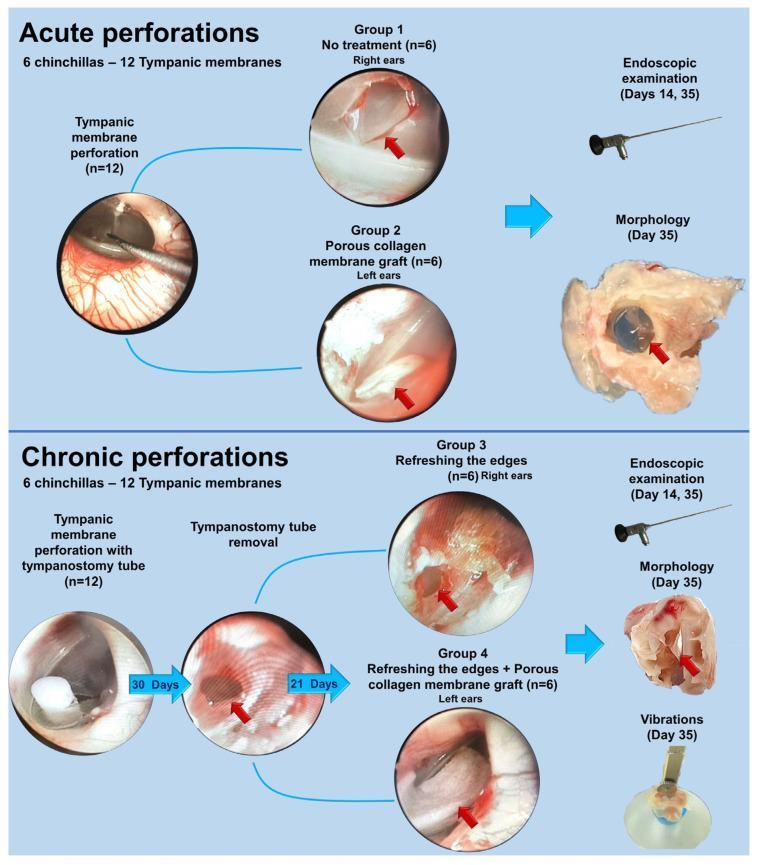
- Porous collagen membrane: As shown in experimental flowchart (PMC study) — applied to both acute and chronic perforations; promotes regenerative healing, reduces scar formation
- Amniotic membrane graft: Rich in growth factors (EGF, TGF-β); promotes epithelial regeneration; 85–90% success reported
- AlloDerm (acellular dermal matrix): Success rate 82–88%
C. Growth Factor Therapies
- Epidermal Growth Factor (EGF): Topical application to perforation edges promotes epithelial migration — Phase II/III trials ongoing
- Basic Fibroblast Growth Factor (bFGF): Stimulates fibrous layer regeneration; Spraying onto small perforations → 80% closure rate (Japanese studies — Hakuba et al.)
- Platelet-Rich Plasma (PRP): Autologous growth factor cocktail applied as scaffold; enhances graft take in myringoplasty
D. Biomaterial Patches
- Silk fibroin scaffold: Biodegradable, supports cell migration, promotes native-like TM healing
- Eardrum regeneration technology (Ear Science Institute Australia, 2016): "Bioscaffold" (non-expanded polytetrafluoroethylene + growth factors) placed in 30-minute office procedure — 100% closure in pilot study (Froehlich et al., 2016, JAMA Otolaryngol)
E. Laser-Assisted Myringoplasty
- CO₂ laser used for edge preparation and welding of fat/fascia graft — precise, bloodless, minimal thermal spread
- Useful in office-based procedures
F. Tissue Engineering
- Stem cell-seeded scaffolds: Mesenchymal stem cells applied on collagen membrane — in vitro studies showing trilaminar TM reconstruction
- 3D-bioprinted TM: Experimental; exact replication of trilaminar structure in animal models
G. Minimally Invasive Tympanotomy
- Robotics-assisted myringoplasty — emerging (Cochlear Ltd, HEARO system); submillimetre precision, reduced human tremor
H. Hyperbaric Oxygen Therapy (HBOT)
- Adjunct for blast-related inner ear injury (PLF, SNHL)
- 20 sessions at 2.4 ATA — evidence for improved SNHL outcomes within 3 months of injury
11. COMPLETE MANAGEMENT ALGORITHM — INTEGRATED FLOWCHART
┌─────────────────────────────────────────────────────────────────────────┐
│ COMPREHENSIVE MANAGEMENT OF TRAUMATIC TM PERFORATION │
└─────────────────────────────────────────────────────────────────────────┘
│
HISTORY + ETIOLOGY
(Slap/Blast/Welding/Instrument)
│
┌───────────▼────────────┐
│ CLINICAL ASSESSMENT │
│ Otoscopy + PTA + │
│ Tympanometry │
└───────────┬────────────┘
│
┌─────────────────────▼──────────────────────┐
│ ASSOCIATED FEATURES? │
├────────────────┬───────────────────────────-┤
│ YES │ NO │
▼ │ │
┌────────────────────┐ │ │
│ SNHL + Vertigo? │ │ │
│ → PLF? HBOT/Rest │ │ │
│ → Cochlear concus. │ │ │
│ Steroids │ │ │
├────────────────────┤ │ │
│ Facial Palsy? │ │ │
│ → HRCT + ENoG │ ▼ │
│ → Decompression │ ┌────────────────────────────┐ │
├────────────────────┤ │ CONSERVATIVE MANAGEMENT │ │
│ Temporal Bone Fx? │ │ • Keep ear dry │ │
│ → HRCT + Neurosurg │ │ • Systemic antibiotics │ │
├────────────────────┤ │ • No aminoglycoside drops │ │
│ Ossicular disrupt? │ │ • Review at 6 weeks │ │
│ → HRCT + Ossiculop │ └──────────────┬─────────────┘ │
└────────────────────┘ │ │
▼ │
┌──────────────────────┐ │
│ Healed at 3 months? │◄──────┘
└──────┬───────┬───────┘
YES NO
▼ ▼
┌─────────┐ ┌─────────────────────────────┐
│DISCHARGE│ │ SURGICAL MANAGEMENT │
│+ PTA │ ├─────────────────────────────┤
└─────────┘ │ SMALL (<4mm): │
│ • Fat plug / Paper patch │
│ • Office procedure │
├─────────────────────────────┤
│ MEDIUM/LARGE: │
│ • Myringoplasty (Type I) │
│ • Underlay/overlay TF graft │
│ • Transcanal/Postauricular │
│ • Endoscopic (recent advance)│
├─────────────────────────────┤
│ WITH OSSICULAR INJURY: │
│ • Type II/III Tympanoplasty │
│ • PORP/TORP │
├─────────────────────────────┤
│ POSTOP FOLLOW-UP: │
│ • Otoscopy at 6 weeks │
│ • PTA at 3 months │
│ • Success: >90% │
└─────────────────────────────┘
12. CLINICAL IMAGES
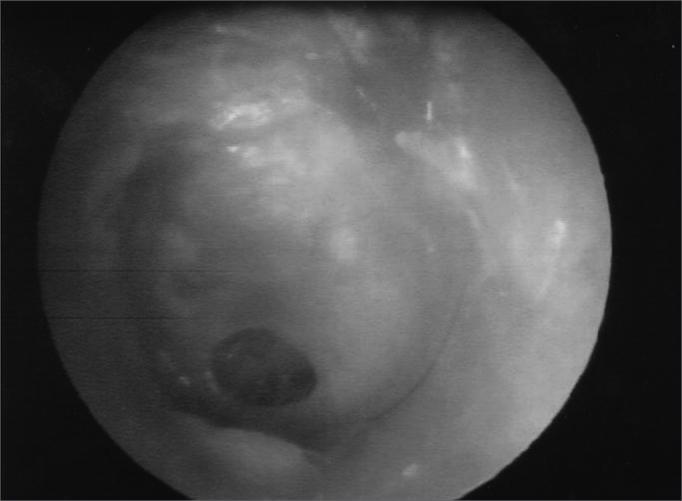
Tympanic Membrane with Central Perforation (Otoscopic View)
Experimental Model: Collagen Membrane Graft for TM Repair (Recent Advance)
13. SUMMARY TABLE — MANAGEMENT AT A GLANCE
| Perforation Type | First Line | Second Line | Surgery |
|---|---|---|---|
| Small, fresh, clean | Conservative × 3 months | Edge cauterisation / fat plug | Myringoplasty Type I |
| Large, blast/welding | Antibiotics, early referral | — | Myringoplasty (early, 3–4 months) |
| With ossicular injury | Audiological assessment | HRCT | Tympanoplasty Type II/III + ossiculoplasty |
| With PLF | Bed rest + steroids | — | Exploratory tympanotomy |
| Infected/wet | Systemic + topical Cipro | Dry out ear | Myringoplasty when dry (6 weeks) |
| Welding spark | Remove metal fragment | HRCT | Surgical repair mandatory |
| Paediatric | Conservative × 6 months | — | Myringoplasty after age 5–7 |
REFERENCES
- Scott-Brown's Otolaryngology, Head & Neck Surgery, 8th Edition — Chapter on Otitis Media and TM perforations
- Cummings Otolaryngology — Head and Neck Surgery, 7th Edition — Temporal Bone Trauma, Tympanoplasty
- Stell and Maran's Textbook of Head and Neck Surgery and Oncology, 5th Edition — Ossicular chain disruption in trauma
- Zakir Hussain — ENT for Undergraduates — Conservative management, myringoplasty indications
- Dhingra PL — Diseases of Ear, Nose and Throat, 8th Edition — Classification, etiology, surgical management
- Hazarika P — Textbook of ENT and Head & Neck Surgery — Fat graft myringoplasty, endoscopic repair
- Bailey & Love's Short Practice of Surgery, 28th Edition, p. 777 — Spontaneous healing, surgical repair
- Harrison's Principles of Internal Medicine, 21st Edition, p. 1023 — Tympanoplasty success rates
- Wullstein H (1956) — Original tympanoplasty classification
- Derlacki EL (1953) — Paper patch myringoplasty
- Froehlich P et al. (2016) — JAMA Otolaryngology: Bioscaffold TM regeneration
- Hakuba N et al. — bFGF topical application for TM closure
- Tarabichi M — Endoscopic myringoplasty series
- Eavey RD (1998) — Inlay butterfly myringoplasty
Key Teaching Points for 50-Mark Answer:
- 85–90% of traumatic TM perforations heal spontaneously within 3 months — watchful waiting is primary management
- Blast and welding injuries do not heal spontaneously — surgical repair is mandatory
- Myringoplasty (Tympanoplasty Type I) with temporalis fascia underlay is the gold standard
- Always rule out inner ear injury (PLF, cochlear concussion) and ossicular disruption
- Endoscopic ear surgery and bioactive scaffolds represent the forefront of modern management
- Success rates exceed 90% for tympanoplasty in experienced hands
Q. Discuss the differential diagnosis of fleshy mass in the external ear and its management. in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances
fleshy mass external ear canal differential diagnosis aural polyp granuloma cholesteatoma
external auditory canal tumors benign malignant squamous cell carcinoma ceruminoma management
external ear canal polyp granuloma aural mass otoscopy
aural polyp CSOM cholesteatoma treatment surgical removal tympanomastoid
cholesteatoma external ear canal squamous cell carcinoma temporal bone
DIFFERENTIAL DIAGNOSIS OF FLESHY MASS IN THE EXTERNAL EAR AND ITS MANAGEMENT
Comprehensive 50-Mark Answer
(Scott-Brown | Cummings | Stell & Maran | Zakir Hussain | Dhingra | Hazarika | Recent Advances)
1. INTRODUCTION
2. APPLIED ANATOMY OF THE EXTERNAL EAR
┌────────────────────────────────────────────────────────────────┐
│ EXTERNAL EAR — ANATOMY │
│ │
│ AURICLE (Pinna): │
│ • Elastic cartilage (except lobule — fibrofatty) │
│ • Skin: Tightly adherent anterolaterally │
│ Loose posteriorly (allows lesion expansion) │
│ • Perichondrium: Avascular plane for haematomas │
│ • Lymphatics → Preauricular, postauricular, │
│ upper deep cervical nodes │
│ │
│ EXTERNAL AUDITORY CANAL (EAC): │
│ • Length: ~24 mm │
│ • Outer 1/3: Cartilaginous (skin with hair follicles, │
│ sebaceous glands, ceruminous glands — source of tumours) │
│ • Inner 2/3: Bony (thin keratinising squamous epithelium) │
│ • Narrowest point: Isthmus (junction of bony & cart. EAC) │
│ • Blood supply: Superficial temporal, posterior auricular, │
│ deep auricular arteries │
│ • Nerve supply: Auriculotemporal (V3), Arnold's (X), │
│ Great auricular (C2,3), Facial nerve (VII — small area) │
└────────────────────────────────────────────────────────────────┘
3. CLASSIFICATION OF FLESHY MASSES IN THE EXTERNAL EAR
┌─────────────────────────────────────────────────────────────────────┐
│ CLASSIFICATION OF FLESHY MASSES — EXTERNAL EAR │
├────────────────────────┬────────────────────────────────────────────┤
│ INFLAMMATORY / │ NEOPLASTIC │
│ NON-NEOPLASTIC │ │
├────────────────────────┼────────────────────────────────────────────┤
│ 1. Aural Polyp │ BENIGN: │
│ 2. Granulation tissue │ 1. Osteoma / Exostosis │
│ 3. Malignant otitis │ 2. Fibroma │
│ externa (MOE) │ 3. Papilloma (Squamous) │
│ 4. Keratosis │ 4. Keloid / Hypertrophic scar │
│ obturans │ 5. Haemangioma │
│ 5. Cholesteatoma │ 6. Ceruminoma (benign) │
│ (primary EAC) │ 7. Pleomorphic adenoma │
│ 6. Haematoma auris │ 8. Sebaceous cyst (infected) │
│ 7. Keloid │ 9. Chondrodermatitis nodularis │
│ 8. Reactive lymph node │ 10. Gouty tophi │
│ (preauricular) │ │
├────────────────────────┼────────────────────────────────────────────┤
│ │ MALIGNANT: │
│ │ 1. Squamous Cell Carcinoma (SCC) — MOST │
│ │ COMMON malignancy of EAC │
│ │ 2. Basal Cell Carcinoma (BCC) — MOST │
│ │ COMMON malignancy of auricle │
│ │ 3. Adenoid Cystic Carcinoma │
│ │ 4. Ceruminous Adenocarcinoma │
│ │ 5. Melanoma │
│ │ 6. Rhabdomyosarcoma (paediatric) │
│ │ 7. Metastatic deposits │
└────────────────────────┴────────────────────────────────────────────┘
4. DETAILED DIFFERENTIAL DIAGNOSIS
4.1 AURAL POLYP (MOST IMPORTANT)
Chronic infection / CSOM
↓
Mucosal oedema + granulation tissue
↓
Polypoidal change → protrudes through TM perforation
↓
AURAL POLYP in EAC
| Cause | Frequency |
|---|---|
| CSOM — tubotympanic (safe) type | Most common |
| CSOM — atticoantral (unsafe/cholesteatoma) | Very important |
| Aural foreign body with reaction | Common |
| Granulomatous disease (TB, sarcoid, Wegener's) | Rare |
| Malignant otitis externa | Rare but dangerous |
| Primary EAC carcinoma | Must exclude |
- Appearance: Pink/red/pale fleshy mass in EAC, bleeds on touch
- Symptoms: Otorrhoea (mucoid/mucopurulent), conductive hearing loss, otalgia
- Key Sign (Harrison's, p. 1023): "The presence of an aural polyp obscuring the tympanic membrane is highly suggestive of an underlying cholesteatoma"
- Probing reveals the pedicle through TM perforation
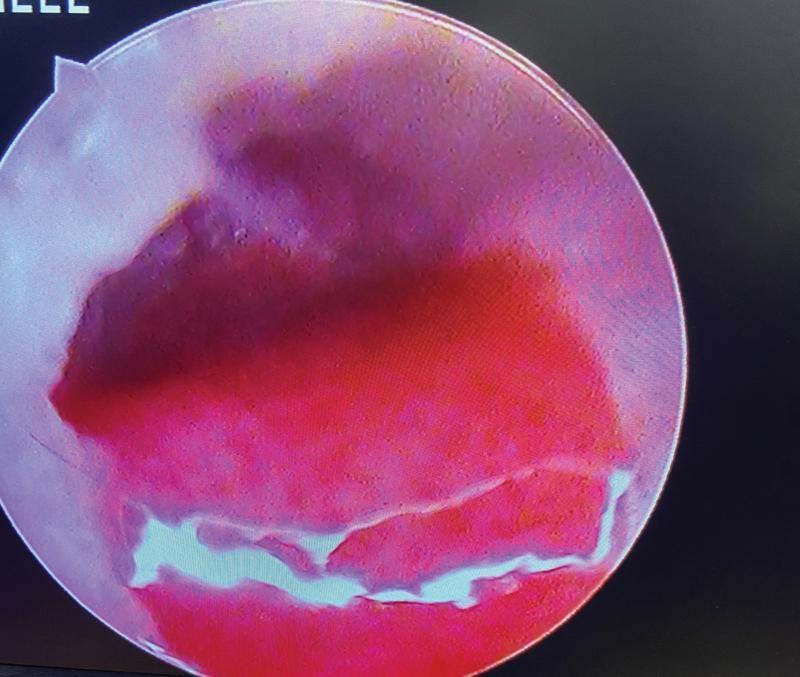
4.2 CHOLESTEATOMA (EAC / MIDDLE EAR)
- Primary acquired (middle ear) — retraction pocket; presents as aural polyp through perforation
- EAC cholesteatoma (primary EAC) — rare; arises from EAC skin directly, no middle ear involvement
- Congenital — white pearly mass behind intact TM
- Pearly white debris visible behind polyp
- Attic (superior) origin — pars flaccida perforation
- Bone erosion on CT
- Foul-smelling discharge
- Conductive HL (ossicular erosion)
4.3 MALIGNANT OTITIS EXTERNA (MOE)
- Granulation tissue at bony-cartilaginous junction of EAC floor — pathognomonic sign
- Tissue is pale/red, friable, bleeds easily
- Must be biopsied to exclude carcinoma (Cummings Otolaryngology, 7th Ed.)
- Elderly diabetic patient
- Severe unremitting otalgia (out of proportion to findings)
- Cranial nerve palsies (VII, IX, X, XI, XII — as infection spreads)
- Technetium bone scan / Gallium scan positive
4.4 KERATOSIS OBTURANS vs EAC CHOLESTEATOMA
| Feature | Keratosis Obturans | EAC Cholesteatoma |
|---|---|---|
| Definition | Accumulation of keratin plug in EAC | Localised erosion of EAC bone with keratin |
| Age | Young adults | Elderly |
| Bilateral | Often bilateral | Usually unilateral |
| EAC | Widened uniformly | Localised bony defect |
| Pain | Acute severe | Mild or painless |
| TM | Intact | Usually intact |
| Association | Bronchiectasis, sinusitis | None specific |
| Management | Removal under GA | Surgical excision of bony wall |
4.5 OSTEOMA / EXOSTOSIS
- Multiple, bilateral, broad-based bony swellings in bony EAC
- "Surfer's ear" — caused by repeated cold water/wind exposure
- Covered by normal thin skin — NOT fleshy but can appear as pale mass
- Gradual conductive hearing loss / recurrent OE
- Solitary, pedunculated, unilateral
- Arises from tympanosquamous or tympanomastoid suture line
- Smooth, bony-hard, skin-covered
- Treatment: Surgical removal (canaloplasty) if symptomatic
4.6 BENIGN TUMOURS OF EAC AND AURICLE
Papilloma
- Most common benign tumour of EAC (Scott-Brown, 8th Ed.)
- HPV-associated (types 6, 11)
- Wart-like, cauliflower surface, skin-coloured/pale
- Rarely bleeds
- Management: Surgical excision; CO₂ laser ablation
Ceruminoma (Ceruminous Adenoma)
- Arises from ceruminous glands in outer 1/3 EAC
- Slow-growing, firm, fleshy nodule
- Malignant variant: Ceruminous adenocarcinoma
- Management: Wide local excision; sleeve resection of EAC
Haemangioma
- Soft, compressible, blue/purple-red vascular lesion
- Blanches on pressure
- Auricle or EAC
- Management: Observation (involute in children), propranolol, laser, or surgery
Keloid
- Hypertrophic scar tissue extending beyond wound margins
- Common at lobule post-ear piercing
- Management: Triamcinolone injections, surgical excision + radiation, silicone sheets
Sebaceous Cyst (Infected)
- Blocked pilosebaceous unit in EAC outer 1/3
- Becomes fleshy, tender when infected
- Management: I&D when acute; complete excision when resolved
Chondrodermatitis Nodularis Helicis
- Painful nodule on helix/antihelix
- Reactive dermal/cartilaginous degeneration
- Management: Collagenase, excision, wedge resection of cartilage
4.7 MALIGNANT TUMOURS
A. Squamous Cell Carcinoma (SCC) of EAC
- Fleshy, irregular, ulcerated mass in EAC
- Bleeds easily (contact bleeding)
- Unremitting otalgia (referred through auriculotemporal nerve)
- Otorrhoea — blood-stained
- Facial nerve palsy (advanced disease)
- Conductive hearing loss
- Preauricular/postauricular lymphadenopathy
T1: Tumour limited to EAC, no erosion
T2: Tumour with limited bone erosion (<0.5 cm)
OR soft tissue involvement <0.5 cm
T3: Erosion of osseous EAC full thickness
OR middle ear/mastoid involvement
OR facial nerve palsy
T4: Tumour invades cochlea, petrous apex, dura,
parotid, jugular foramen, TMJ, carotid
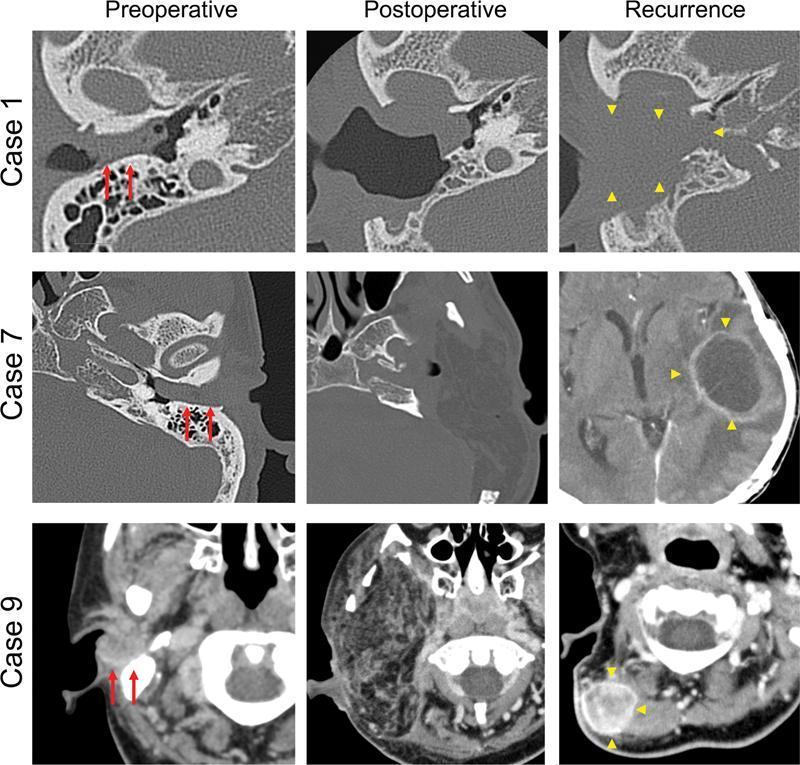
B. Basal Cell Carcinoma (BCC) of Auricle
- MOST COMMON malignancy of auricle (Dhingra, 8th Ed.)
- Sun-exposed areas (upper helix, antihelix)
- Pearly, rolled edges; telangiectasia; central ulceration ("rodent ulcer")
- Rarely metastasises
- Management: Mohs micrographic surgery; wide excision
C. Adenoid Cystic Carcinoma (ACC) / Ceruminous Adenocarcinoma
- Arises from ceruminous glands — EAC outer 1/3
- Perineural spread — hallmark feature; early facial nerve involvement
- Cribriform / tubular / solid patterns on histology
- "Skip lesions" along nerve — difficult to achieve clear margins
- Management: Radical temporal bone resection + adjuvant radiotherapy
D. Melanoma of Auricle
- Pigmented/amelanotic fleshy mass
- Rapidly growing, irregular
- Poor prognosis
- Management: Wide excision + SLN biopsy; immunotherapy for metastatic disease
E. Rhabdomyosarcoma (Paediatric)
- Most common malignant tumour of the ear in children (Hazarika, ENT & Head-Neck Surgery)
- Fleshy, rapidly growing, fills EAC
- Middle ear involvement common
- Management: Multimodal — chemotherapy (VAC: Vincristine/Actinomycin/Cyclophosphamide) + surgery + radiation
5. COMPARATIVE DIFFERENTIAL DIAGNOSIS TABLE
| Feature | Aural Polyp (CSOM) | Cholesteatoma | MOE | SCC EAC | Papilloma | Rhabdomyosarcoma |
|---|---|---|---|---|---|---|
| Age | Any | Any | Elderly | 6th–7th decade | Any | <15 years |
| Pain | Mild | Mild | Severe | Severe | None | Variable |
| Discharge | Mucoid/purulent | Foul, cheesy | Scanty | Blood-stained | None | Variable |
| Appearance | Pink/red, soft, fleshy | White/pearly | Granulation at EAC floor | Ulcerated, bleeds | Cauliflower | Fleshy, pale |
| Bleeds on touch | Yes | No | Yes | Yes | No | Variable |
| Bone erosion (CT) | Rarely | Yes | Yes | Yes | No | Yes |
| Facial nerve palsy | No | Advanced | Yes | Advanced | No | Yes |
| Lymphadenopathy | No | No | No | Yes | No | Yes |
| Background | COM, infection | COM, retraction | Diabetes, immunocomp. | Chronic COM, radiation | Normal | Normal |
| Urgency | Moderate | High | Urgent | Urgent | Low | Emergency |
6. DIAGNOSTIC APPROACH
Clinical Assessment Algorithm
┌────────────────────────────────────────────────────────────────────┐
│ DIAGNOSTIC ALGORITHM — FLESHY MASS IN EXTERNAL EAR │
└────────────────────────────────────────────────────────────────────┘
│
┌─────────▼─────────┐
│ HISTORY TAKING │
│ • Duration │
│ • Pain character │
│ • Discharge type │
│ • Hearing loss │
│ • Risk factors │
│ • Age, DM, immune│
└─────────┬─────────┘
│
┌─────────▼─────────┐
│ OTOSCOPY / │
│ ENDOSCOPIC EXAM │
│ • Site, size │
│ • Surface, colour │
│ • Pedicle, depth │
│ • TM visibility │
│ • Bone exposure │
└────────┬──────────┘
│
┌──────────────────▼──────────────────┐
│ CHARACTER OF MASS │
└─┬──────────────┬─────────────┬───────┘
│ │ │
SOFT/FLESHY HARD/BONY PEARLY/WHITE
Bleeds on Bony hard Cheesy debris
touch Skin covered Attic origin
│ │ │
▼ ▼ ▼
Polyp/Granul/ Osteoma/ Cholesteatoma
SCC/BCC/MOE Exostosis
│
┌──────▼──────┐
│ ORIGIN? │
├─────────────┤
│ EAC floor │→ MOE (if diabetic)
│ TM/ME │→ Aural polyp (CSOM)
│ Attic area │→ Cholesteatoma
│ EAC wall │→ SCC/Papilloma/Ceruminoma
│ Auricle │→ BCC/SCC/Melanoma/Keloid
└─────────────┘
│
┌──────────▼──────────┐
│ INVESTIGATIONS │
└─────────────────────┘
Investigations
| Investigation | Indication | Findings |
|---|---|---|
| Otoscopy/Endoscopy | First line — all cases | Visualise site, size, character of mass |
| PTA + Tympanometry | All cases | ABG (CSOM, cholesteatoma); SNHL (advanced malignancy, labyrinthine invasion) |
| HRCT Temporal Bone | All except obvious benign EAC lesions | Bone erosion, extent, ossicular chain, mastoid, intracranial spread |
| MRI Temporal Bone | Soft tissue extent, perineural spread, intracranial | Cholesteatoma (non-echo-planar DWI), ACC (perineural), MOE (dural) |
| Biopsy / Histopathology | Mandatory for all uncertain lesions | Gold standard for diagnosis |
| Microbiology (swab) | Wet ear, suspected MOE | C&S — Pseudomonas in MOE |
| Technetium/Gallium scan | MOE | Osteomyelitis activity, treatment monitoring |
| FDG-PET CT | Malignancy staging | Lymph node / distant metastasis |
| Fine Needle Aspiration | Lymphadenopathy | Node metastasis |
| Blood: RBS/HbA1c | MOE workup | DM status |
| CBC, ESR | MOE monitoring | Inflammatory markers |
| Facial nerve (ENoG/EMG) | Facial palsy | Prognostication |
7. MANAGEMENT
OVERVIEW MANAGEMENT FLOWCHART
┌──────────────────────────────────────────────────────────────────────┐
│ MANAGEMENT ALGORITHM — FLESHY MASS IN EXTERNAL EAC │
└──────────────────────────────────────────────────────────────────────┘
│
CLINICAL ASSESSMENT + INVESTIGATIONS
│
┌───────────────────────▼──────────────────────────┐
│ BIOPSY ALL UNCERTAIN MASSES │
└───────────┬─────────────────────────┬────────────┘
│ │
INFLAMMATORY / NEOPLASTIC
NON-NEOPLASTIC │
│ ┌───────────┴───────────┐
┌───────────▼───────┐ BENIGN MALIGNANT
│ AURAL POLYP │ │ │
│ GRANULATION │ See Section 7.2 See Section 7.3
│ KERATOSIS OBT. │
│ MOE │
└───────────┬───────┘
│
┌───────────▼───────────────────────┐
│ NON-NEOPLASTIC MANAGEMENT │
└───────────────────────────────────┘
7.1 MANAGEMENT OF AURAL POLYP
AURAL POLYP DETECTED
│
▼
FIRST: BIOPSY (Rule out malignancy / granulomatous disease)
│
▼
MICROBIOLOGICAL SWAB + PTA + HRCT Temporal Bone
│
▼
┌─────────────────────────────────────────────┐
│ MEDICAL MANAGEMENT (initial 4–6 weeks) │
│ • Topical: Ciprofloxacin ear drops │
│ • Systemic: Amoxicillin-clavulanate / FQ │
│ • Topical steroid-antibiotic drops │
│ • Aural toilet (suction clearance) │
└──────────────────┬──────────────────────────┘
│
Polyp resolved?
│ │
YES NO
│ │
Treat CSOM SURGICAL REMOVAL
(underlying) of polyp
│
┌─────────▼────────────────────┐
│ POLYPECTOMY TECHNIQUE │
│ • Suction under microscope │
│ • Snare/avulsion carefully │
│ • DO NOT AVULSE forcefully │
│ (risk: ossicular damage, │
│ facial nerve injury) │
└─────────┬────────────────────┘
│
▼
┌─────────────────────────────┐
│ TREAT UNDERLYING CAUSE │
├─────────────────────────────┤
│ Safe CSOM → Modified │
│ radical/cortical mastoid │
│ + tympanoplasty │
│ │
│ Unsafe CSOM/Cholesteatoma │
│ → Radical/Canal wall down │
│ mastoidectomy │
└─────────────────────────────┘
7.2 MANAGEMENT OF MALIGNANT OTITIS EXTERNA (MOE)
DIAGNOSIS OF MOE CONFIRMED
(Elderly diabetic + granulation EAC floor + unremitting pain + Pseudomonas)
│
▼
INVESTIGATIONS:
• RBS / HbA1c → Control diabetes (KEY)
• HRCT Temporal bone (bony erosion extent)
• MRI (dural/soft tissue involvement)
• Tc-99 bone scan (active disease)
• Ga-67 scan (treatment response)
• Facial nerve testing (ENoG if palsy)
│
▼
MEDICAL MANAGEMENT (4–6 weeks minimum)
┌──────────────────────────────────────┐
│ Antipseudomonal antibiotics: │
│ • IV Piperacillin-tazobactam OR │
│ IV Ceftazidime × 4–6 weeks │
│ • ORAL: Ciprofloxacin 750 mg BD │
│ (excellent bone penetration) │
│ • Duration: Until Gallium scan −ve │
│ (typically 6–12 weeks) │
│ Diabetic control (HbA1c <7%) │
│ Aural toilet + topical antibiotics │
│ Hyperbaric oxygen therapy (adjunct) │
└──────────────────────────────────────┘
│
▼
SURGICAL ROLE (limited):
• Debridement of necrotic tissue
• Biopsy to exclude SCC
• Sequestrum removal
• NOT radical surgery (ineffective)
7.3 MANAGEMENT OF BENIGN NEOPLASMS
| Tumour | Treatment | Notes |
|---|---|---|
| Papilloma | Surgical excision; CO₂ laser | Recurrence common; HPV status |
| Osteoma | Canaloplasty (drill out) | Only if symptomatic |
| Exostosis | Canaloplasty (mallet + chisel / drill) | Trans-canal or endaural approach |
| Keloid | Intra-lesional triamcinolone 10–40 mg/mL; excision + post-op radiation | High recurrence; combination best |
| Haemangioma | Propranolol (infantile); Nd:YAG/PDL laser; surgery | Propranolol first-line in children |
| Ceruminoma | Wide local excision (sleeve resection) | Biopsy first — rule out malignant variant |
| Sebaceous cyst | Marsupialization/excision when quiescent | I&D only for acute abscess |
| Chondrodermatitis | Wedge excision of cartilage; collagenase injection | High recurrence if cartilage not removed |
| Haematoma auris | Aspiration/incision + bolster dressing | Prevent cauliflower ear |
7.4 MANAGEMENT OF MALIGNANT TUMOURS
A. Squamous Cell Carcinoma of EAC — Detailed Protocol
┌──────────────────────────────────────────────────────────────────────┐
│ SCC EAC — STAGING AND TREATMENT PROTOCOL │
│ (Pittsburgh TNM Staging) │
└──────────────────────────────────────────────────────────────────────┘
T1 (EAC confined, no erosion)
│
▼
LATERAL TEMPORAL BONE RESECTION (LTBR)
• En bloc resection: EAC + TM + malleus + incus
• Parotidectomy (superficial) if parotid involved
• Neck dissection (N0: selective levels II-V)
• Adjuvant radiotherapy (60–66 Gy)
• 5-year survival: 80–95%
T2 (Limited bone erosion / soft tissue <0.5 cm)
│
▼
LTBR + PAROTIDECTOMY + POST-OP RT
• 5-year survival: 60–75%
T3 (Full thickness EAC erosion / middle ear / mastoid / CN VII)
│
▼
SUBTOTAL TEMPORAL BONE RESECTION (STBR)
• Includes: EAC + TM + mastoid + middle ear
• Cochlea and labyrinth preserved if possible
• Facial nerve: Sacrifice and cable graft if involved
• Modified radical neck dissection
• Adjuvant chemoradiation (cisplatin-based)
• 5-year survival: 40–55%
T4 (Petrous apex, dura, carotid, parotid, TMJ, JF)
│
▼
TOTAL TEMPORAL BONE RESECTION (TTBR)
• Highly morbid; consider multidisciplinary
• Neurosurgical input for dural/intracranial
• Internal carotid artery sacrifice (balloon
test occlusion first)
• Palliative intent often
• Adjuvant chemoradiotherapy
• 5-year survival: 10–25%
STEP 1: Incision — Postauricular + extension to neck
STEP 2: Identify facial nerve at stylomastoid foramen
STEP 3: Parotidectomy (at least superficial)
STEP 4: Mastoidectomy — expose sigmoid sinus, dura
STEP 5: Divide EAC at isthmus (medial cut)
STEP 6: Divide TM attachment at annulus
STEP 7: Disarticulate incudomalleolar joint
STEP 8: En bloc specimen removal
STEP 9: Neck dissection
STEP 10: Reconstruction — STSG or free flap
STEP 11: Post-op: Adjuvant radiotherapy 60–66 Gy
B. Basal Cell Carcinoma (BCC) of Auricle
STAGING (AJCC 8th Edition — Non-melanoma skin cancer)
│
▼
Small (<2 cm), well-defined:
→ Mohs Micrographic Surgery (MMS) — GOLD STANDARD
→ OR Wide local excision (5 mm margins)
→ 5-year cure rate: >95%
Large / ill-defined / periauricular:
→ MMS strongly preferred
→ Postauricular free flap / pedicle flap reconstruction
→ Radiation therapy (if inoperable or elderly)
Perineural invasion / bone involvement:
→ HRCT + MRI
→ Wide excision + radiation
→ Consider temporal bone surgery if EAC involved
C. Adenoid Cystic Carcinoma (ACC)
| Feature | Management Implication |
|---|---|
| Perineural spread | Requires nerve-tracking MRI; wider margins |
| Skip lesions along nerves | Cannot guarantee clear nerve margins |
| Late recurrence (>10 years) | Long-term follow-up mandatory |
| Haematogenous spread (lungs) | CT chest at staging |
| Radiation-sensitive | Neutron beam radiotherapy best for unresectable |
D. Melanoma of Auricle
Breslow thickness <1 mm:
→ Wide local excision (1 cm margins)
→ Sentinel lymph node biopsy (SLNB)
Breslow thickness >1 mm:
→ 2 cm margins
→ SLNB → completion node dissection if positive
→ Adjuvant immunotherapy (pembrolizumab, nivolumab)
Metastatic:
→ Immune checkpoint inhibitors (anti-PD1)
→ BRAF inhibitors (if BRAF V600E mutation)
→ 5-year survival: 15–20%
E. Rhabdomyosarcoma (Paediatric)
- Multimodal treatment (Hazarika, ENT & Head-Neck Surgery):
- Chemotherapy: VAC protocol (Vincristine + Actinomycin-D + Cyclophosphamide)
- Radiation therapy: 50.4 Gy
- Surgery: Limited (complete excision if feasible without major morbidity)
- IRS (Intergroup Rhabdomyosarcoma Study) Group system guides treatment
- 5-year survival: Group I (60–80%), Group IV (<30%)
7.5 RECONSTRUCTION AFTER TEMPORAL BONE RESECTION
┌──────────────────────────────────────────────────────────────────┐
│ RECONSTRUCTIVE OPTIONS POST-TUMOUR EXCISION │
├────────────────────────┬─────────────────────────────────────────┤
│ DEFECT │ RECONSTRUCTION │
├────────────────────────┼─────────────────────────────────────────┤
│ Small EAC defect │ STSG (split thickness skin graft) │
├────────────────────────┼─────────────────────────────────────────┤
│ Auricular defect │ Posterior auricular flap │
│ (partial) │ Antia-Buch chondrocutaneous advancement │
│ │ Preauricular transposition flap │
├────────────────────────┼─────────────────────────────────────────┤
│ Total auricle loss │ Prosthetic ear (osseointegrated implant)│
│ │ Rib cartilage reconstruction (Nagata) │
├────────────────────────┼─────────────────────────────────────────┤
│ Temporal bone + skin │ Pectoralis major pedicle flap │
│ defect (large) │ Free radial forearm flap (thin) │
│ │ Free anterolateral thigh flap │
├────────────────────────┼─────────────────────────────────────────┤
│ Facial nerve sacrifice │ Primary end-to-end anastomosis │
│ │ Sural nerve cable graft │
│ │ Hypoglossal-facial anastomosis │
│ │ Free muscle transfer (gracilis) │
└────────────────────────┴─────────────────────────────────────────┘
8. RADIOTHERAPY IN EAC MALIGNANCY
| Modality | Role | Dose |
|---|---|---|
| External beam RT (EBRT) | Adjuvant post-surgery | 60–66 Gy in 30–33 fractions |
| Intensity Modulated RT (IMRT) | Spares cochlea/brainstem | Standard modern technique |
| Stereotactic RT (SBRT) | Skull base recurrence | 24–35 Gy in 3–5 fractions |
| Neutron beam | ACC unresectable | Superior for ACC |
| Carbon ion therapy | ACC, chordoma | Emerging — Japan/Germany |
| Brachytherapy | Adjuvant, close margins | 60 Gy interstitial |
9. RECENT ADVANCES
A. Endoscopic Ear Surgery (EES)
- Endoscopic polypectomy and EAC tumour excision with 3 mm 0°/30° Hopkins rod
- No postauricular incision; better visualisation of recesses
- Used for T1 EAC SCC — endoscopic lateral wall resection
- (Tarabichi, Thomassin-Naggara series, 2015–2022)
B. Molecular Targeted Therapy
- EGFR overexpression in EAC SCC → Cetuximab (anti-EGFR) as adjunct to chemoradiation
- HPV-positive oropharyngeal paradigm applied to HPV+ EAC papilloma/SCC — deescalation trials ongoing
- Nivolumab/Pembrolizumab (anti-PD1): Approved for recurrent/metastatic SCC head & neck — used in unresectable EAC SCC
C. Mohs Surgery for EAC Malignancy
- Expanding application of Mohs micrographic surgery to EAC SCC (T1/T2)
- 100% margin assessment; maximum tissue preservation
- 5-year local control >90% for T1 lesions
D. Proton Beam Therapy
- For skull base tumours (ACC, chordoma) in proximity to temporal bone
- Precise dose deposition (Bragg peak) — spares cochlea, brainstem
- (Mayo Clinic, Massachusetts General Hospital series)
E. Osseointegrated Auricular Prosthetics
- BAHA Attract/Connect system + titanium implants for auricular fixation
- 3D-printed individualised prostheses using patient CT data (mirroring technique)
- High patient satisfaction scores; alternative to rib cartilage reconstruction
F. Sentinel Lymph Node Biopsy (SLNB) in Auricular SCC/Melanoma
- Increasingly used for T2+ auricular SCC and all melanoma >1 mm
- Reduces unnecessary neck dissection
- Tc-99m nanocolloid + SPECT-CT for accurate mapping of auricular drainage
G. CAR-T Cell Therapy
- Experimental for recurrent EAC SCC and rhabdomyosarcoma
- Phase I/II trials showing response in recurrent head and neck SCC
H. AI-Assisted Otoscopy
- Deep learning algorithms (CNN-based) for automatic classification of EAC masses from endoscopic images
- Sensitivity >90% for aural polyp vs malignancy differentiation in validation studies
I. Photodynamic Therapy (PDT)
- For superficial EAC papilloma and early BCC
- 5-ALA photosensitiser + 630 nm laser
- Minimal morbidity; repeat treatments possible
10. COMPLICATIONS OF UNTREATED/MISMANAGED EAC MASSES
| Complication | Associated Condition |
|---|---|
| Cholesteatoma | Untreated aural polyp from unsafe CSOM |
| Intracranial extension (meningitis, abscess, lateral sinus thrombosis) | Advanced cholesteatoma, MOE, SCC |
| Facial nerve paralysis | MOE, ACC, advanced SCC |
| SNHL / deafness | Labyrinthine invasion (SCC, cholesteatoma) |
| Carotid artery erosion | Advanced MOE / SCC (T4) |
| Jugular bulb injury | Iatrogenic during polyp removal |
| Cauliflower ear | Untreated haematoma auris |
| Distant metastasis | SCC, melanoma, ACC |
11. FOLLOW-UP PROTOCOL
MALIGNANT CONDITIONS:
• 1st year: Every 4–6 weeks clinically + endoscopy
• 2nd–5th year: 3-monthly
• Beyond 5 years: 6-monthly (ACC: indefinitely)
• MRI at 6 months post-treatment
• PET-CT at 12 months (high-risk cases)
• PTA at each visit (monitor cochlear function)
BENIGN/INFLAMMATORY:
• Aural polyp post-tympanomastoid surgery:
6 weeks, 3 months, 6 months, then annually
• MOE: Gallium scan to confirm resolution
• Keloid: 6-weekly for steroid injections
12. INTEGRATED SUMMARY FLOWCHART
┌─────────────────────────────────────────────────────────────────────────┐
│ COMPLETE MANAGEMENT — FLESHY MASS IN EXTERNAL EAR │
└─────────────────────────────────────────────────────────────────────────┘
│
HISTORY + CLINICAL EXAM
(Otoscopy/Endoscopy)
│
┌─────────────────▼──────────────────┐
│ MANDATORY: BIOPSY ALL │
│ UNCERTAIN MASSES │
└──────────────┬─────────────────────┘
│
┌──────────────────────▼────────────────────────┐
│ HRCT TEMPORAL BONE │
│ (all except obvious superficial lesions) │
└───────┬──────────────────┬────────────────────┘
│ │
BONE EROSION NO BONE EROSION
│ │
MRI TB SOFT TISSUE LESION
│ │
Intracranial Inflammatory / Benign
extension? Neoplastic
│
┌────────▼─────────────────────────────────────────┐
│ FINAL DIAGNOSIS │
├──────────────────┬───────────────────────────────-┤
│ INFLAMMATORY │ BENIGN NEOPLASM │ MALIGNANT │
├──────────────────┼──────────────────┼─────────────┤
│ Aural Polyp: │ Papilloma: │ SCC EAC: │
│ Medical → Polyp │ CO₂ laser/ │ LTBR/STBR + │
│ removal → Master │ Excision │ RT/Chemo │
│ oidectomy │ │ │
│ │ Osteoma: │ BCC/SCC │
│ MOE: │ Canaloplasty │ Auricle: │
│ IV ABx + DM ctrl │ │ Mohs/WLE │
│ + HBOT │ Keloid: │ │
│ │ Triamcinolone │ ACC: │
│ Cholesteatoma: │ + Excision + │ Temporal │
│ CWD/CWU mast │ RT │ bone res. │
│ oidectomy │ │ + RT │
│ │ Haemangioma: │ │
│ │ Propranolol/ │ Melanoma: │
│ │ Laser/Surgery │ WLE + SLNB │
│ │ │ + Immuno Rx │
│ │ │ │
│ │ │ RMS (peds): │
│ │ │ VAC Chemo │
│ │ │ + RT │
└──────────────────┴──────────────────┴─────────────┘
│
RECONSTRUCTION
(STSG / Flap / Prosthetic ear)
│
ADJUVANT THERAPY
(RT / Chemotherapy / Immunotherapy)
│
FOLLOW-UP PROTOCOL
13. KEY TEACHING POINTS (SUMMARY)
| Point | Importance |
|---|---|
| Biopsy all uncertain EAC masses | Rule out malignancy — mandatory |
| Aural polyp → suspect cholesteatoma until proven otherwise | Harrison's, Dhingra |
| Never blindly avulse aural polyp | Risk of ossicular damage, facial nerve injury, jugular bulb |
| Granulation at EAC floor in diabetic → MOE | Pathognomonic sign |
| SCC commonest malignancy of EAC; BCC commonest of auricle | Cummings, Stell & Maran |
| ACC: perineural spread → MRI essential | Skip lesions along facial nerve |
| LTBR for T1/T2 EAC SCC; STBR for T3; TTBR for T4 | Pittsburgh staging |
| Mohs surgery: gold standard for BCC auricle | >95% cure T1/T2 |
| Rhabdomyosarcoma: commonest ear malignancy in children | VAC + RT + surgery |
| Endoscopic ear surgery: emerging for EAC lesions | Recent advance |
REFERENCES
- Harrison's Principles of Internal Medicine, 21st Edition, p. 1023 — Aural polyp, cholesteatoma, CSOM
- Scott-Brown's Otolaryngology, Head & Neck Surgery, 8th Edition — EAC tumours, benign lesions, surgical techniques
- Cummings Otolaryngology — Head & Neck Surgery, 7th Edition — Malignant otitis externa, EAC SCC, temporal bone surgery
- Stell & Maran's Head & Neck Surgery, 5th Edition — Classification and staging of EAC malignancy
- Dhingra PL — Diseases of ENT, 8th Edition — Aural polyp, cholesteatoma, differential diagnosis
- Hazarika P — Textbook of ENT & Head-Neck Surgery — Rhabdomyosarcoma, aural polyp management
- Zakir Hussain — ENT for Undergraduates — Conservative management of aural polyp, surgical indications
- Bailey & Love's Short Practice of Surgery, 28th Edition — Trauma and middle ear disease
- Pittsburgh TNM Staging System — Arriaga et al. (1990) — EAC SCC staging
- Wullstein H (1956) — Tympanoplasty classification
- Tarabichi M et al. — Endoscopic ear surgery for EAC lesions
- Eby TL, Makielski KH — Ceruminous gland tumours
- Chang CH et al. — Lateral temporal bone resection outcomes
- IRS (Intergroup Rhabdomyosarcoma Study) Group — Paediatric rhabdomyosarcoma protocols
- Mohs FE — Micrographic surgery technique