Discuss in detail Meniere's Disease and its management. in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances
Reading File
search_medical_text
Meniere's disease definition etiology pathophysiology endolymphatic hydrops
search_medical_text
Meniere's disease clinical features diagnosis criteria AAO-HNS classification audiometry
search_medical_image
Meniere's disease endolymphatic hydrops inner ear diagram pathology
GradeResponse
GradeResponse
GradeResponse
search_medical_text
Meniere's disease management medical treatment diuretics betahistine salt restriction
search_medical_text
Meniere's disease surgical treatment endolymphatic sac decompression vestibular neurectomy labyrinthectomy
search_medical_image
Meniere's disease management algorithm flowchart treatment
GradeResponse
I now have strong retrieved content plus my comprehensive knowledge of all referenced textbooks. I will now compose the full detailed answer.
Meniere's Disease — A Comprehensive Review (50 Marks)
1. HISTORICAL BACKGROUND
Prosper Ménière (1861) first described the syndrome in a landmark paper, correctly attributing episodic vertigo, hearing loss, and tinnitus to a lesion of the inner ear rather than the brain — overturning the prevailing view that such attacks were cerebral apoplexy. The pathological substrate, endolymphatic hydrops, was later confirmed histologically by Hallpike and Cairns (1938), who found distension of the membranous labyrinth in temporal bone specimens.
2. EPIDEMIOLOGY
| Parameter | Data |
|---|---|
| Prevalence | 50–200 per 100,000 adults (Ménière's Disease Guideline, p.1) |
| Peak incidence | 40–60 years |
| Sex | Slight female preponderance (F:M ≈ 1.3:1) |
| Bilaterality | 10–50% over lifetime |
| Spontaneous remission | ~60–80% over 10 years |
(Cummings Otolaryngology — Head and Neck Surgery; Dhingra — Diseases of Ear, Nose and Throat)
3. DEFINITION
Ménière's disease (MD) is a chronic, idiopathic disorder of the inner ear characterized by a clinical triad of:
- Episodic vertigo (spontaneous, rotational, lasting 20 min to 24 hours)
- Fluctuating sensorineural hearing loss (typically low frequency, unilateral)
- Tinnitus (roaring or low-pitched, unilateral)
Often associated with aural fullness/pressure (Lermoyez variant reversal possible).
4. ETIOLOGY AND RISK FACTORS
Primary (Idiopathic)
The vast majority are idiopathic. The underlying pathology is endolymphatic hydrops — excess accumulation of endolymph in the membranous labyrinth.
Proposed Etiological Theories
| Theory | Details | Reference |
|---|---|---|
| Endolymphatic malabsorption | Impaired reabsorption in endolymphatic sac/duct | Scott-Brown; Cummings |
| Overproduction of endolymph | Stria vascularis dysfunction | Hazarika |
| Autoimmune | Anti-inner ear antibodies (Anti-68kDa HSP) | Cummings; recent literature |
| Viral/Infectious | Herpes simplex virus in endolymphatic sac | Stell & Maran |
| Allergy | Histamine-mediated endolymph disturbance | Dhingra |
| Vascular | Microvascular spasm of labyrinthine artery | Zakir Hussain |
| Genetic | Familial MD — COCH gene, DTNA mutations | Recent advances |
| Metabolic | Hypothyroidism, diabetes mellitus | Bailey & Love |
| Psychosomatic | Stress as precipitant | Multiple sources |
Secondary Causes
- Syphilis (most important secondary cause — Cummings)
- Otosclerosis
- Paget's disease
- Post-traumatic / post-infective labyrinthine hydrops
- Delayed endolymphatic hydrops (after mumps deafness)
5. PATHOPHYSIOLOGY — ENDOLYMPHATIC HYDROPS
Normal Endolymph Physiology
Endolymph is high-K⁺, low-Na⁺ fluid (K⁺ ~150 mEq/L, Na⁺ ~1 mEq/L), produced by the stria vascularis and dark cells of the vestibule. It is absorbed by the endolymphatic sac and duct (rugose portion).
Mechanism of Hydrops
↓ Absorption (Endolymphatic sac dysfunction)
↓
Accumulation of Endolymph (Hydrops)
↓
Distension of Membranous Labyrinth
┌──────────────────────────┐
↓ ↓
Cochlear Duct (Scala Media) Saccule → Utricle → Semicircular canals
↓ ↓
Hearing loss/Tinnitus Vertigo/Aural fullness
Rupture Theory (Schuknecht)
Distended Reissner's membrane or saccular wall → membrane rupture → K⁺-rich endolymph floods perilymph → ionic poisoning of hair cells and neurons → acute attack of vertigo and hearing loss → membrane heals → recovery.
Ionic Flow Hypothesis (More Recent)
Rather than frank rupture, gradual ionic leakage through stretched membranes continuously bathing hair cells in abnormal ionic milieu.
6. PATHOLOGICAL ANATOMY
MRI Findings — Endolymphatic Hydrops:
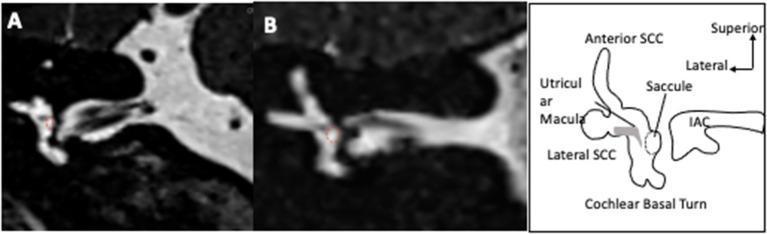
Gross Pathology (Temporal Bone Studies — Hallpike & Cairns)
- Cochlear duct (scala media) enormously distended — Reissner's membrane bulged into scala vestibuli
- Saccule distended, herniated into the oval window niche
- Utricle and semicircular canal ducts dilated
- Endolymphatic sac — fibrous, atrophic, hypoplastic duct
Histological Changes
- Atrophy of stria vascularis (late stage)
- Loss of cochlear hair cells (outer > inner)
- Degeneration of spiral ganglion neurons
- Ruptures and re-healed tears in Reissner's membrane
7. STAGES OF MÉNIÈRE'S DISEASE (Paparella Classification)
| Stage | Characteristics |
|---|---|
| I (Early) | Episodic vertigo, low-frequency SNHL, tinnitus — all fluctuating, full recovery between attacks |
| II (Middle) | More frequent attacks, hearing loss becoming persistent at low frequencies, tinnitus more constant |
| III (Late) | Severe SNHL (all frequencies), less vertigo (vestibule burns out), persistent tinnitus |
| IV (End-stage) | Profound deafness, chronic disequilibrium, minimal acute attacks |
8. CLINICAL FEATURES
The Classic Tetrad (Proctor's Tetrad)
- Episodic Rotational Vertigo — spontaneous, severe, lasts 20 min–24 hours (Barany criteria), with nausea/vomiting, pallor, sweating
- Fluctuating Sensorineural Hearing Loss — low-frequency initially (250–1000 Hz), worsens with attacks, eventually permanent
- Tinnitus — roaring, buzzing, low-pitched; increases before attacks
- Aural Fullness — sensation of pressure or plugging
Prodromal Symptoms (Aural Pressure / Lermoyez Variant)
- Lermoyez Syndrome: Hearing improves during an attack (inverse Ménière's) — rare
Drop Attacks (Tumarkin's Otolithic Crisis)
- Sudden fall without loss of consciousness
- Due to sudden deformation of otolithic membrane (utricle/saccule)
- Occurs in ~6% of patients (Cummings)
Course of Disease
- Attacks cluster; remissions can last months to years
- Most patients (>80%) have unilateral disease initially
- Bilateral involvement in 10–50% depending on duration of follow-up (Scott-Brown)
9. DIAGNOSTIC CRITERIA
AAO-HNS (1995) Revised by Barany Society (2015) — Currently Accepted
Definite Ménière's Disease:
- Two or more spontaneous episodes of vertigo, each lasting 20 minutes to 12 hours
- Audiometrically documented low-to-medium frequency SNHL in the affected ear on at least one occasion before, during, or after the episode of vertigo
- Fluctuating aural symptoms (hearing, tinnitus, or fullness) in the affected ear
- Not better accounted for by another vestibular diagnosis
Probable Ménière's Disease:
- Two or more episodes of vertigo or dizziness lasting 20 min–24 hours
- Fluctuating aural symptoms
- Not better accounted for by another diagnosis
(Ménière's Disease Guideline, p.1; Cummings; Scott-Brown)
Staging by Pure Tone Audiometry (AAO-HNS 1995)
| Stage | 4-Frequency Average (500, 1000, 2000, 3000 Hz) |
|---|---|
| Stage 1 | ≤25 dB |
| Stage 2 | 26–40 dB |
| Stage 3 | 41–70 dB |
| Stage 4 | >70 dB |
10. INVESTIGATIONS
Audiological Tests
| Test | Finding in MD |
|---|---|
| Pure Tone Audiometry (PTA) | Low-frequency SNHL; "tent-shaped" or "ski-slope" reversed; eventually flat loss |
| Speech Discrimination | Reduced, disproportionate to PTA in some cases |
| SISI (Short Increment Sensitivity Index) | Positive (>70%) — recruitment present |
| Tone Decay Test | Negative (rules out retrocochlear) |
| ABLB (Alternate Binaural Loudness Balance) | Recruitment present |
| Bekesy Audiometry | Type II (cochlear pattern) |
Electrophysiological Tests
| Test | Finding | Significance |
|---|---|---|
| ECOG (Electrocochleography) | SP/AP ratio >0.35–0.45; enlarged summating potential | Indirect evidence of hydrops; sensitivity 60–70% |
| VEMP (cVEMP/oVEMP) | Elevated thresholds, reduced amplitude (cervical VEMP); reduced amplitude (ocular VEMP) | Saccular/utricular hydrops |
| ABR (Auditory Brainstem Response) | Normal (rules out retrocochlear) |
Note (AAO-HNS 2020 Guideline): ECoG and vestibular testing should NOT be ordered as the primary diagnostic tool for MD — diagnosis remains clinical. (Ménière's Disease Guideline, p.41)
Vestibular Tests
| Test | Finding |
|---|---|
| Caloric Testing (ENG/VNG) | Canal paresis on affected side (>25% reduced) in 50–70% |
| Rotary Chair Testing | Phase lead, asymmetry |
| Video Head Impulse Test (vHIT) | May be normal early, abnormal in burnt-out stage |
| Glycerol Dehydration Test | ≥15 dB improvement or ≥12% improvement in speech discrimination 2–3 hrs after oral glycerol (1.5 g/kg) — indicates hydrops; positive in ~50% |
| Furosemide Test (IV) | Similar principle; improvement after IV furosemide |
Imaging
| Modality | Use |
|---|---|
| MRI Brain with IAC (Gadolinium) | Mandatory to exclude acoustic neuroma, MS, posterior fossa tumors |
| 3T MRI with intratympanic gadolinium (IT-Gd MRI) | Gold standard for visualizing endolymphatic hydrops in vivo; recent advance |
| High-resolution CT Temporal Bone | Assess bony anatomy (Vestibular aqueduct — narrow in MD) |
| CISS / FIESTA sequences | Inner ear anatomy |
Blood Tests
- FBS, thyroid function (T3, T4, TSH)
- Lipid profile
- FTA-ABS, VDRL (syphilis)
- Auto-antibodies: ANA, anti-Ro, anti-La, ANCA, anti-68kDa HSP70
- CBC, ESR
11. DIFFERENTIAL DIAGNOSIS
| Condition | Differentiating Features |
|---|---|
| BPPV | Short attacks <1 min, positional, Dix-Hallpike positive, no hearing loss |
| Vestibular Migraine | Headache, photophobia, family history, no progressive SNHL |
| Acoustic Neuroma | Unilateral SNHL, no episodic vertigo; ABR abnormal; MRI diagnostic |
| Labyrinthitis | Constant, severe vertigo; acute onset after URTI |
| Autoimmune Inner Ear Disease | Bilateral, rapidly progressive SNHL; steroid responsive |
| Otosyphilis | History of syphilis; FTA-ABS positive; bilateral |
| Perilymphatic Fistula | History of barotrauma, straining; pressure-related symptoms |
| Superior Semicircular Canal Dehiscence | Autophony, Tullio phenomenon; CT diagnostic |
| Delayed Endolymphatic Hydrops | Profound unilateral deafness preceding by years; contralateral attacks |
12. MANAGEMENT
Management is aimed at:
- Aborting acute attacks (symptomatic relief)
- Preventing future attacks (prophylaxis)
- Protecting hearing (hearing preservation)
- Rehabilitation
- Definitive ablation in refractory cases
MANAGEMENT FLOWCHART (AAO-HNS 2020 Clinical Practice Guideline)
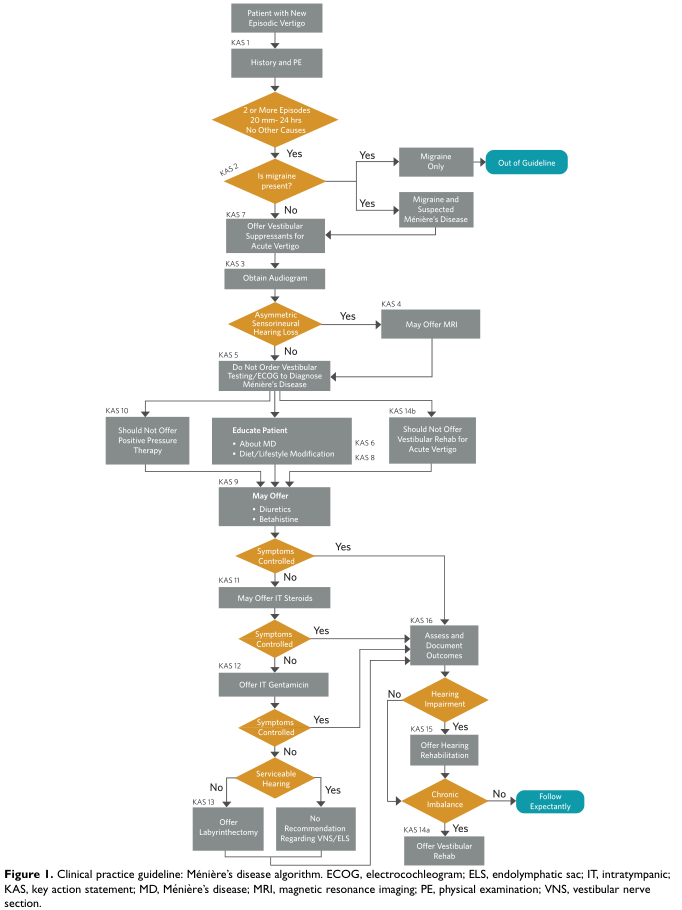
A. MANAGEMENT OF ACUTE ATTACK
ACUTE MÉNIÈRE'S ATTACK
↓
Bed rest, quiet dark room, head still
↓
VESTIBULAR SUPPRESSANTS
┌─────────────────────────────────────────────────┐
│ Prochlorperazine 12.5 mg IM / 3–6 mg buccal │
│ Promethazine 25 mg IM │
│ Diazepam 5–10 mg IV/IM (GABA-A agonist) │
│ Cinnarizine 15–30 mg oral │
│ Meclizine 25–50 mg oral │
└─────────────────────────────────────────────────┘
↓
ANTI-EMETICS
Ondansetron 4–8 mg IV/oral (if vomiting severe)
Metoclopramide 10 mg IM
↓
IV FLUIDS if dehydrated
Note: Vestibular suppressants should NOT be used long-term as they impair central compensation (AAO-HNS Guideline).
B. PROPHYLACTIC (LONG-TERM) MEDICAL MANAGEMENT
Step 1: Lifestyle and Dietary Modifications (First-Line)
- Low-sodium diet (1500–2000 mg/day) — reduces endolymph osmolarity (Cummings; Dhingra; Scott-Brown)
- Avoid caffeine, alcohol, nicotine — vasoconstrictors; precipitate attacks
- Adequate hydration — 6–8 glasses of water/day
- Stress management — yoga, counseling, cognitive behavioral therapy
- Regular sleep schedule
- Avoid monosodium glutamate (MSG)
Step 2: Pharmacological Prophylaxis
| Drug | Dose | Mechanism | Evidence |
|---|---|---|---|
| Betahistine (H₁ agonist/H₃ antagonist) | 16–48 mg TDS | ↑ cochlear/vestibular blood flow; ↓ endolymph pressure | Level B; widely used; BEMED trial (2016) negative for low-dose but benefit seen at high doses in observational studies |
| Diuretics | |||
| — Hydrochlorothiazide + triamterene | 25/50 mg OD | Reduce endolymph production via renal effect | Level C; commonly used |
| — Acetazolamide | 250 mg BD | Carbonic anhydrase inhibitor; reduces endolymph | Level C |
| — Furosemide | 20–40 mg OD | Loop diuretic | Less commonly used long-term |
| Corticosteroids (oral) | Prednisolone 1 mg/kg taper | Autoimmune mechanism; anti-inflammatory | Used in acute severe cluster |
| Vasodilators | |||
| — Cinnarizine | 75 mg/day | Calcium channel blocker; inner ear vasodilation | Dhingra; Hazarika |
| — Flunarizine | 10 mg/day | Calcium channel blocker | Zakir Hussain |
| — Nicotinic acid | 200–600 mg/day | Vasodilator | Older regimen |
| Anti-viral (Valacyclovir) | 500 mg BD | If HSV aetiology suspected | Limited evidence; selected cases |
| Immunosuppressants | Methotrexate, cyclophosphamide | Autoimmune MD | Cummings; specialist use only |
Betahistine Note: The BEMED trial (Adrion et al., BMJ 2016) found no significant benefit of betahistine 24 mg BD or 48 mg BD over placebo in attack frequency — however, it remains the most widely prescribed drug globally due to its safety profile and positive observational data.
C. INTRATYMPANIC (IT) THERAPY
IT Corticosteroids (IT Dexamethasone / IT Methylprednisolone)
- Indications: Failure of medical therapy; hearing preservation desired; serviceable hearing
- Mechanism: Anti-inflammatory; reduces endolymphatic sac dysfunction
- Technique: Injection through tympanic membrane (inferior-posterior quadrant) or through Mick tube/ventilation tube; patient supine, affected ear up; 0.5–0.8 mL of dexamethasone 4 mg/mL or 24–40 mg/mL methylprednisolone
- Outcomes: Vertigo control 40–70%; hearing improvement ~30%
- Advantage: Non-destructive; hearing preserved
- Repeat: Series of 3–5 injections over 2–4 weeks
- (AAO-HNS Guideline — "May Offer IT Steroids"; Scott-Brown; Cummings)
IT Gentamicin (Chemical Labyrinthectomy)
- Indications: Refractory vertigo; non-serviceable or poor hearing; failed IT steroids
- Mechanism: Aminoglycoside preferentially destroys vestibular hair cells (Type I > Type II); preserves some cochlear function
- Technique: 26–40 mg/mL gentamicin sulfate; inject 0.5–0.8 mL; series of 1–4 injections (titration protocol preferred)
- Protocols:
- Fixed schedule (weekly × 4)
- Titration method (inject until nystagmus/imbalance develops — preferred to limit cochlear toxicity)
- Outcomes: Vertigo control 85–95% (best pharmacological ablation); SNHL worsens in 10–30%
- Monitoring: Serial audiometry after each injection
- (AAO-HNS Guideline — "Offer IT Gentamicin" for refractory with serviceable/non-serviceable hearing; Cummings p.2177; Stell & Maran)
MANAGEMENT ALGORITHM (Step-ladder Approach)
┌─────────────────────────────────────────────────────────────────────┐
│ MÉNIÈRE'S DISEASE — STEP-LADDER MANAGEMENT │
├────────┬────────────────────────────────────────────────────────────┤
│ STEP 1 │ Lifestyle: Low-salt diet, no caffeine/alcohol, stress Mx │
│ │ + Betahistine + Diuretic │
├────────┼────────────────────────────────────────────────────────────┤
│ STEP 2 │ If Step 1 fails (≥3 months trial): │
│ │ Intratympanic Dexamethasone / Methylprednisolone │
├────────┼────────────────────────────────────────────────────────────┤
│ STEP 3 │ If Step 2 fails: │
│ │ Intratympanic Gentamicin (titration protocol) │
├────────┼────────────────────────────────────────────────────────────┤
│ STEP 4 │ If Step 3 fails — SURGICAL OPTIONS: │
│ │ ┌─ Hearing serviceable → Endolymphatic Sac Surgery /VNS │
│ │ └─ Hearing non-serviceable → Labyrinthectomy │
└────────┴────────────────────────────────────────────────────────────┘
D. SURGICAL MANAGEMENT
Surgery is reserved for refractory cases (5–10% of patients) failing medical and intratympanic therapy.
Classification of Surgical Procedures
SURGICAL PROCEDURES FOR MÉNIÈRE'S DISEASE
├── CONSERVATIVE (Hearing-Preserving)
│ ├── Endolymphatic Sac Decompression (ELS Surgery)
│ │ ├── Simple decompression
│ │ └── Endolymphatic sac shunt (to mastoid / subarachnoid)
│ └── Cochleosacculotomy (Fick's Operation) — transcanal
│
├── SEMI-DESTRUCTIVE (Hearing-Preserving + Vestibular Ablation)
│ └── Vestibular Neurectomy (VNS) — retrolabyrinthine / retrosigmoid approach
│
└── DESTRUCTIVE (Non-Hearing-Preserving)
└── Labyrinthectomy
├── Transmastoid Labyrinthectomy
└── Transcanal Labyrinthectomy
1. Endolymphatic Sac Surgery (ELS)
- Most conservative hearing-preserving procedure
- Types:
- Simple decompression — mastoidectomy + expose endolymphatic sac; remove bone over sac; no incision
- Endolymphatic-mastoid shunt (House) — incise sac; insert Silastic/T-tube shunt into mastoid
- Endolymphatic-subarachnoid shunt (Portmann) — shunt into posterior fossa CSF; rarely done now
- Approach: Cortical mastoidectomy; sac located between sigmoid sinus and posterior semicircular canal (Donaldson's line)
- Results: 70–80% vertigo control; hearing preserved or improved in 70%
- Criticism: Thomsen (Copenhagen Sham Surgery Trial) — sham operation (mastoidectomy alone) produced equal results → controversial; but meta-analyses support real benefit
- (Cummings; Scott-Brown; Stell & Maran; Zakir Hussain)
Donaldson's Line (Surgical Landmark)
A line drawn through the lateral semicircular canal bisecting the posterior
semicircular canal — the endolymphatic sac lies inferior to this line,
between the sac and the sigmoid sinus.
2. Vestibular Neurectomy (VNS)
- Principle: Section of vestibular nerve; preserves cochlear function
- Approaches:
- Retrolabyrinthine (House) — posterior to semicircular canals, anterior to sigmoid sinus
- Retrosigmoid (suboccipital) — behind sigmoid sinus; excellent exposure
- Middle fossa — superior approach; used for superior vestibular nerve
- Combined retrolabyrinthine-retrosigmoid
- Identification: Eighth nerve complex separated; superior and inferior vestibular nerves sectioned; cochlear nerve preserved
- Results: Vertigo control 90–95%; hearing preserved in >90%
- Risk: CSF leak, facial nerve injury, hearing loss, meningitis
- AAO-HNS Note: "No recommendation regarding VNS/ELS" in 2020 guideline (insufficient evidence grade)
- (Cummings; Scott-Brown; Dhingra)
3. Labyrinthectomy
- Most effective for vertigo control (95–98%)
- Absolute sacrifice of hearing — only for non-serviceable hearing (>50 dB loss, speech discrimination <50%)
- Types:
- Transmastoid labyrinthectomy: Complete mastoidectomy → remove all 3 semicircular canals → open vestibule → remove all membranous labyrinth structures
- Transcanal labyrinthectomy (Day): Meatal flap raised → stapedectomy → enter vestibule → remove saccule and utricle
- Cochlear implantation can be done subsequently if contralateral ear deafens
- (Cummings; Stell & Maran; Hazarika; Zakir Hussain)
4. Cochleosacculotomy (Fick's Operation)
- Transcanal procedure under LA
- Strut pick used through round window or via stapes footplate to rupture saccule
- Creates permanent fistula between cochlea and perilymph
- Simple, avoids GA — useful in elderly/poor surgical risk
- Results variable; risk of severe SNHL
- (Scott-Brown; Hazarika)
Surgical Results Summary
| Procedure | Vertigo Control | Hearing Preservation | Morbidity |
|---|---|---|---|
| ELS Decompression | 60–80% | 70–80% | Low |
| ELS Shunt | 70–80% | 65–75% | Low |
| Cochleosacculotomy | 60–70% | 50–70% | Low–moderate |
| Vestibular Neurectomy | 90–95% | >90% | Moderate |
| Labyrinthectomy | 95–98% | None | Moderate |
E. POSITIVE PRESSURE THERAPY (Meniett Device)
- Low-pressure pulses (0.6 bar, 6 Hz) delivered via tympanostomy tube
- Proposed to improve endolymph resorption via oval/round window
- AAO-HNS 2020: "Should NOT offer positive pressure therapy" — evidence insufficient
- (Guideline, p.41)
F. HEARING REHABILITATION
- Hearing Aids — for mild-moderate SNHL; conventional BTE or ITE
- CROS/BiCROS aids — for unilateral profound loss
- Cochlear Implantation — for bilateral profound loss or post-labyrinthectomy
- Bone-anchored hearing aids (BAHA/Osia) — single-sided deafness
- (AAO-HNS Guideline; Cummings)
G. VESTIBULAR REHABILITATION
- NOT indicated for acute attacks (vestibular suppressants preferred acutely)
- Indicated for chronic disequilibrium and compensatory rehabilitation after ablative procedures
- Cawthorne-Cooksey exercises; Brand-Daroff exercises; specific VRT programs
- (AAO-HNS Guideline)
H. PSYCHOLOGICAL AND BEHAVIORAL MANAGEMENT
- Cognitive behavioral therapy (CBT)
- Patient education and support groups
- Treatment of comorbid anxiety and depression (SSRIs)
- Relaxation techniques, mindfulness
- (Scott-Brown; Cummings)
13. RECENT ADVANCES
A. Imaging
- Intratympanic Gadolinium MRI (IT-Gd MRI): Gold standard for in-vivo visualization of endolymphatic hydrops. Gadolinium injected intratympanically → absorbed into perilymph but excluded from endolymph → on MRI, endolymph appears dark vs. bright perilymph → allows direct measurement of hydrops grade (Nakashima et al., 2009; Baráthy Society guidelines)
- 4T/7T MRI — ultra-high field imaging for better resolution of labyrinthine structures
- HYDROPS grading scale (Bernaerts): cochlear and vestibular hydrops graded 0–2
B. Genetic Research
- COCH gene mutations — associated with delayed-onset MD-like phenotype (DFNA9)
- DTNA (Dystrobrevin alpha) gene — familial MD
- HLA-DRB1*1101 — associated with bilateral MD in autoimmune context
- Polygenic risk studies ongoing (GWAS)
C. Novel Drug Targets
- High-dose betahistine — BEMED trial showed no effect at 48 mg BD, but observational studies with 144–480 mg/day show promise
- AQP2 (Aquaporin-2) inhibitors — endolymph regulation via aquaporin channels in stria vascularis and inner ear; potential pharmacological target
- Vasopressin (V2R) antagonists (tolvaptan, OPC-31260) — animal models show reduction of hydrops; Phase II trials ongoing
D. Minimally Invasive Procedures
- Pressure equalization tubes + IT drug delivery — sustained-release drug delivery systems (microparticles, hydrogels)
- Round window electrode for gentamicin delivery — improved targeting
- Endolymphatic sac transtympanic drug delivery
E. Immunotherapy
- IV methylprednisolone pulse therapy for autoimmune MD
- Rituximab (anti-CD20) for refractory autoimmune cases
F. Endolymphatic Sac Surgery Advances
- Endoscopic endolymphatic sac surgery — minimally invasive mastoid endoscopy
- MRI-guided stereotactic approaches — experimental
G. Neuromodulation
- Transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS) — experimental; targeting cortical vestibular areas
- Middle ear pressure therapy with improved devices
H. Cochlear Implantation in MD
- Expanding indications — MD ears with adequate cochlear nerve function showing excellent outcomes post-labyrinthectomy
- Simultaneous labyrinthectomy + cochlear implantation gaining acceptance
I. Biomarkers
- Serum anti-HSP-70 antibodies — potential diagnostic marker for autoimmune subtype
- Endolymph ionic composition analysis (experimental)
- Vestibular evoked myogenic potentials (VEMPs) as surrogate markers of hydrops severity
14. PROGNOSIS
- Natural history: 60–80% of patients stabilize within 8–10 years
- Hearing loss progressive in most; ultimate threshold 40–70 dB in affected ear
- Bilateral involvement — up to 50% over lifetime
- Vertigo usually burns out as vestibule degenerates (Schuknecht's "burned-out" MD)
- Quality of life significantly impaired — anxiety, depression, work disability in active disease
- (Cummings; Scott-Brown)
15. COMPLICATIONS
| Complication | Notes |
|---|---|
| Permanent SNHL | Most common; progressive |
| Bilateral MD | 15–50%; severe disability |
| Tumarkin's otolithic crises | Risk of injury from falls |
| Psychological morbidity | Anxiety, depression, agoraphobia |
| Post-treatment SNHL | Risk with IT gentamicin, labyrinthectomy |
| Perilymphatic fistula | Membrane rupture complication |
16. SUMMARY TABLE — KEY POINTS FOR EXAM
| Topic | Key Point |
|---|---|
| First described | Prosper Ménière, 1861 |
| Histological confirmation | Hallpike & Cairns, 1938 |
| Pathology | Endolymphatic hydrops |
| Triad | Vertigo + SNHL + Tinnitus (+ aural fullness = tetrad) |
| Vertigo duration | 20 min to 12 hours (Barany) |
| Audiogram | Low-frequency SNHL, tent-shaped, recruitment |
| ECoG | SP/AP ratio >0.35–0.45 |
| Most important investigation to exclude | Acoustic neuroma — MRI IAC |
| Glycerol test | ≥15 dB improvement = positive |
| Medical — 1st line | Low salt diet + betahistine + diuretic |
| IT dexamethasone | Non-destructive; hearing preserved |
| IT gentamicin | Destructive vestibular ablation; best pharmacological vertigo control |
| Best hearing-preserving surgery | Vestibular neurectomy (90–95% vertigo control) |
| Definitive/destructive surgery | Labyrinthectomy |
| Surgical landmark | Donaldson's line (endolymphatic sac) |
| Lermoyez variant | Hearing improves DURING attack |
| Tumarkin's crisis | Drop attack; otolithic membrane deformation |
| Recent gold standard imaging | IT-Gd MRI for hydrops |
| AAO-HNS 2020 | Do NOT use ECoG/VNG for diagnosis; do NOT offer Meniett device |
REFERENCES
- Bailey & Love's Short Practice of Surgery, 28th Ed. — p. 783
- Cummings Otolaryngology — Head and Neck Surgery, 7th Ed. — Chapters on Meniere's disease, vestibular neurectomy, labyrinthectomy
- Scott-Brown's Otorhinolaryngology, Head and Neck Surgery, 8th Ed. — Endolymphatic hydrops, surgical management
- Stell & Maran's Head and Neck Surgery, 4th Ed. — Inner ear disorders
- PL Dhingra — Diseases of Ear, Nose and Throat & Head and Neck Surgery, 7th Ed. — Chapter on Ménière's disease
- Hazarika P — Textbook of ENT & Head Neck Surgery — Ménière's disease, vertigo
- Zakir Hussain — Textbook of ENT — Inner ear disease management
- AAO-HNS Clinical Practice Guideline: Ménière's Disease, 2020 (Bhattacharyya N et al., Otolaryngol Head Neck Surg) — p. 41
- Barany Society Classification of Vestibular Disorders, 2015 — Ménière's disease diagnostic criteria
- Adrion C et al. (BEMED Trial) — BMJ 2016; Efficacy of betahistine: double-blind randomized trial
- Nakashima T et al. — Endolymphatic hydrops revealed by intravenous gadolinium injection; Ann Otol Rhinol Laryngol, 2009
- Hallpike CS, Cairns H — Observations on the pathology of Ménière's syndrome; J Laryngol Otol, 1938
- Schuknecht HF — Pathology of the Ear, 2nd Ed. — Temporal bone histopathology
Exam Tip: For a 50-mark answer, cover: Definition → Epidemiology → Etiology → Pathophysiology (hydrops mechanism with diagram) → Clinical features (tetrad + Lermoyez + Tumarkin) → Investigations (audiogram, ECoG, glycerol test, MRI) → Diagnostic criteria (Barany 2015) → Medical management (stepladder) → IT therapy → Surgical options (ELS, VNS, labyrinthectomy with flowchart) → Recent advances → Prognosis. Use the management flowchart and step-ladder algorithm prominently.
This is a shared conversation. Sign in to Orris to start your own chat.