Risk factors of arterial hypertension detail explanation
risk factors arterial hypertension modifiable non-modifiable
hypertension pathophysiology secondary causes epidemiology
hypertension age obesity sodium alcohol lifestyle risk
secondary hypertension renal renovascular sleep apnea endocrine causes
Risk Factors of Arterial Hypertension
A. Non-Modifiable Risk Factors
1. Age
- BP rises progressively with age due to arterial stiffening (arteriosclerosis), reduced vascular compliance, and loss of elasticity in large vessels.
- Systolic hypertension becomes the dominant pattern after age 50 as pulse pressure widens.
- Prevalence rises from ~5% in those aged 20–40 to >65% in those over 65.
2. Sex
- Before age 50, hypertension is more common in men.
- After menopause, prevalence in women rises sharply and eventually surpasses men, due to loss of estrogen's vasodilatory and natriuretic effects.
3. Race / Ethnicity
- Black/African-American individuals have the highest prevalence of hypertension globally — it develops earlier, is more severe, and carries higher target-organ damage risk (stroke, CKD, LVH).
- Genetic differences in renin-angiotensin-aldosterone system (RAAS) activity, sodium retention, and endothelial function are implicated.
4. Family History / Genetic Factors
- Having a first-degree relative with hypertension approximately doubles the risk.
- Hypertension is polygenic; key genes involve RAAS regulation, sodium handling, and aldosterone synthesis.
- Rare monogenic forms include Liddle syndrome, Gordon syndrome, and glucocorticoid-remediable aldosteronism.
5. History of Preeclampsia / Gestational Hypertension
- Women with a history of preeclampsia carry a 2–4× higher lifetime risk of chronic hypertension, heart disease, and stroke (Living Kidney Donors Guidelines, p. 59).
B. Modifiable (Lifestyle) Risk Factors
1. Obesity and Overweight (BMI ≥25 kg/m²)
- Every 10 kg increase in body weight raises systolic BP by ~3–5 mmHg.
- Mechanisms: increased cardiac output, sympathetic nervous system (SNS) activation, hyperinsulinemia, increased renal sodium reabsorption, RAAS activation, and adipokine dysregulation (leptin resistance).
- Visceral/abdominal obesity (waist circumference >102 cm in men, >88 cm in women) is particularly strongly associated.
2. High Dietary Sodium Intake
- Strong dose-response relationship: each 2.3 g/day increase in sodium raises SBP by ~2–3 mmHg in normotensives and up to 4–8 mmHg in hypertensives ("salt-sensitive" individuals).
- Salt sensitivity is more common in older adults, Black individuals, those with CKD, and diabetics.
- Mechanism: volume expansion, increased preload, and impaired pressure natriuresis.
3. Physical Inactivity / Sedentary Behavior
- Physically inactive individuals have a 30–50% greater risk of developing hypertension.
- Regular aerobic exercise lowers SBP by 5–8 mmHg on average.
- Exercise improves endothelial function, reduces SNS activity, and promotes natriuresis.
4. Excess Alcohol Consumption
- Chronic heavy alcohol use (>2 standard drinks/day) raises BP directly.
- Alcohol raises BP via SNS activation, suppression of baroreceptors, increased cortisol, and endothelial dysfunction.
- Reducing intake from heavy to moderate lowers SBP by ~5–7 mmHg.
- Paradoxically, very low intake may have mild protective effects (debated).
5. Smoking and Tobacco Use
- Nicotine causes acute BP elevation via catecholamine release and vasoconstriction.
- Chronic smoking accelerates atherosclerosis, reduces arterial compliance, and promotes endothelial dysfunction — all amplifying hypertension risk.
- Smoking cessation is essential for cardiovascular risk reduction.
6. Dietary Patterns — Low Potassium, Low Calcium, Low Magnesium
- Potassium deficiency promotes sodium retention and vasoconstriction; increasing dietary potassium lowers BP by 3–5 mmHg.
- Low dietary calcium and magnesium are independently associated with elevated BP.
- The DASH (Dietary Approaches to Stop Hypertension) diet — rich in fruits, vegetables, whole grains, and low-fat dairy — lowers SBP by 8–14 mmHg.
7. Psychological Stress / Mental Health
- Chronic stress activates the hypothalamic-pituitary-adrenal axis and SNS, elevating catecholamines and cortisol.
- Depression and anxiety are independently associated with incident hypertension.
- Work-related stress and low socioeconomic status are also significant contributors.
8. Diabetes Mellitus and Insulin Resistance
- Insulin resistance leads to SNS activation, renal sodium retention, and RAAS upregulation.
- ~75% of patients with type 2 diabetes have hypertension.
- Hyperinsulinemia promotes vascular smooth muscle proliferation and endothelial dysfunction.
9. Dyslipidemia
- Elevated LDL-cholesterol promotes endothelial dysfunction and atherosclerosis, which reduces arterial compliance and raises blood pressure.
- Acts synergistically with hypertension to amplify cardiovascular risk.
C. Medication-Induced (Iatrogenic) Risk Factors
| Drug Class | Mechanism |
|---|---|
| NSAIDs (ibuprofen, naproxen) | Renal sodium retention, inhibit vasodilatory prostaglandins |
| Oral contraceptives (estrogen-containing) | RAAS activation, sodium retention |
| Decongestants (pseudoephedrine, phenylephrine) | Alpha-adrenergic vasoconstriction |
| Stimulants (amphetamines, cocaine, methylphenidate) | Massive catecholamine release |
| Corticosteroids | Mineralocorticoid effect → sodium/water retention |
| Cyclosporine / Tacrolimus | Vasoconstriction, renal toxicity |
| Erythropoietin | Increased red cell mass, vasoconstriction |
| Venlafaxine / SNRIs | Noradrenergic activation |
| Dietary supplements (licorice root, ephedra, ginseng) | Various vasopressor mechanisms |
D. Secondary Causes of Hypertension
| Cause | Key Features |
|---|---|
| Primary aldosteronism | Most common secondary cause; hypokalemia, adrenal adenoma or hyperplasia |
| Renovascular HTN (renal artery stenosis) | Atherosclerotic (elderly men) or fibromuscular dysplasia (young women); abdominal bruit |
| Chronic kidney disease (CKD) | Volume overload + RAAS activation |
| Obstructive sleep apnea (OSA) | SNS activation, hypoxia-induced vasoconstriction; very common in obese patients |
| Pheochromocytoma | Paroxysmal HTN + headaches + sweating + palpitations (the "5 Ps") |
| Cushing's syndrome | Hypercortisolism → mineralocorticoid effects |
| Hypothyroidism | Increased peripheral vascular resistance |
| Hyperthyroidism | Increased cardiac output → elevated systolic BP |
| Coarctation of the aorta | Upper extremity HTN + weak femoral pulses; presents in young patients |
| Hyperparathyroidism | Hypercalcemia → vasoconstriction |
E. Special Population Considerations
| Population | Key Points |
|---|---|
| Children/Adolescents | Obesity is the dominant driver; secondary causes more common than in adults |
| Pregnant women | Gestational HTN, preeclampsia; ACE inhibitors/ARBs contraindicated |
| Elderly | Isolated systolic HTN predominates; orthostatic hypotension must be considered |
| CKD patients | Bidirectional relationship — HTN causes CKD, and CKD worsens HTN |
| Diabetics | Lower BP target (<130/80); RAAS blockade preferred |
Summary: Risk Factor Classification at a Glance
NON-MODIFIABLE MODIFIABLE (Lifestyle) SECONDARY CAUSES
───────────────── ────────────────────── ─────────────────────
Age Obesity Primary aldosteronism
Male sex (<50) High sodium diet Renovascular disease
Female sex (>50) Physical inactivity CKD
Race (Black) Alcohol excess OSA
Family history Smoking Pheochromocytoma
Genetic variants Low K/Ca/Mg diet Cushing's syndrome
Preeclampsia hx Stress/depression Thyroid disorders
Diabetes/insulin resistance Coarctation of aorta
Medications (NSAIDs, OCP…)
- Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults (ACC/AHA 2017 Guidelines), p. 22
- Living Kidney Donors Guidelines, p. 59
- Harrison's Principles of Internal Medicine (background knowledge on pathophysiology)
- JNC 8 / ESC/ESH 2018 Hypertension Guidelines (background knowledge)
Arterial hypertension detail explanation
arterial hypertension definition classification diagnosis management
arterial hypertension blood pressure classification chart
hypertension blood pressure classification stages grades ESC ESH 2024
hypertension target organ damage complications heart kidney brain
Arterial Hypertension — Detailed Explanation

1. Definition
| Guideline | Hypertension Threshold |
|---|---|
| ESC/ESH 2018 & 2023 | SBP ≥140 mmHg and/or DBP ≥90 mmHg |
| ACC/AHA 2017 | SBP ≥130 mmHg and/or DBP ≥80 mmHg |
| JNC 8 | SBP ≥140/90 mmHg (general); ≥150/90 mmHg (>60 yrs) |
Diagnosis requires confirmation on at least 2–3 separate occasions or validated by ambulatory/home monitoring.
2. Classification
ESC/ESH 2023 BP Classification
| Category | SBP (mmHg) | DBP (mmHg) | |
|---|---|---|---|
| Optimal | <120 | and | <80 |
| Normal | 120–129 | and/or | 80–84 |
| High-Normal | 130–139 | and/or | 85–89 |
| Grade 1 Hypertension | 140–159 | and/or | 90–99 |
| Grade 2 Hypertension | 160–179 | and/or | 100–109 |
| Grade 3 Hypertension | ≥180 | and/or | ≥110 |
| Isolated Systolic HTN | ≥140 | and | <90 |
3. Epidemiology
- Affects approximately 1.28 billion adults worldwide (WHO, 2021).
- Prevalence is higher in low- and middle-income countries (~75% of cases).
- Rates increase sharply with age — over 65% prevalence in adults >65 years.
- Often called the "silent killer" — most patients are asymptomatic until complications develop.
- Responsible for ~10.4 million deaths per year globally, primarily from cardiovascular and cerebrovascular events.
4. Pathophysiology
BP = Cardiac Output (CO) × Total Peripheral Resistance (TPR)
A. Increased Cardiac Output
- Volume expansion (excess sodium/water retention)
- Sympathetic nervous system (SNS) hyperactivation → increased heart rate and contractility
- Insulin resistance → hyperinsulinemia → renal sodium retention
B. Increased Total Peripheral Resistance
- Endothelial dysfunction → reduced nitric oxide (NO) production → vasoconstriction
- Renin-Angiotensin-Aldosterone System (RAAS) overactivation → angiotensin II causes potent vasoconstriction + aldosterone-mediated sodium retention
- Vascular smooth muscle hypertrophy and remodeling
- Arterial stiffness (arteriosclerosis) with aging
Key Pathophysiological Pathways:
↑ Sodium intake / Obesity / Genetics
↓
RAAS activation → Angiotensin II ↑
↓
Vasoconstriction + Aldosterone ↑
↓
Na⁺/H₂O retention → ↑ Blood Volume
↓
↑ Cardiac Output + ↑ TPR
↓
HYPERTENSION
↓
Endothelial damage → Target Organ Damage
5. Types / Etiology
A. Primary (Essential) Hypertension (~90–95% of cases)
- No single identifiable cause
- Results from a complex interaction of genetic, environmental, and lifestyle factors
- Onset typically in the 4th–5th decade of life
- Diagnosis of exclusion
B. Secondary Hypertension (~5–10% of cases)
| Cause | Prevalence | Key Features |
|---|---|---|
| Primary Aldosteronism | Most common (5–10%) | Hypokalemia, high aldosterone, low renin |
| Renovascular HTN (renal artery stenosis) | ~1–2% | Abdominal bruit, flash pulmonary edema |
| Chronic Kidney Disease | Common | eGFR ↓, proteinuria, RAAS activation |
| Obstructive Sleep Apnea | ~30–50% in obese HTN | Snoring, daytime somnolence, resistant HTN |
| Pheochromocytoma | Rare (<1%) | Paroxysmal HTN, diaphoresis, headache, palpitations |
| Cushing's Syndrome | Rare | Central obesity, moon face, striae, hyperglycemia |
| Hypothyroidism | Uncommon | Bradycardia, fatigue, diastolic HTN |
| Hyperthyroidism | Uncommon | Tachycardia, systolic HTN, weight loss |
| Coarctation of the Aorta | Young patients | Weak femoral pulses, rib notching on X-ray |
| Drug/Substance-Induced | Common | NSAIDs, OCP, steroids, stimulants, decongestants |
6. Clinical Features
A. Symptoms
- Headache (classically occipital, worse in the morning)
- Dizziness / lightheadedness
- Blurred vision
- Palpitations
- Epistaxis (nosebleeds)
- Tinnitus
B. Hypertensive Urgency vs. Emergency
| Feature | Urgency | Emergency |
|---|---|---|
| BP Level | Severely elevated (≥180/120 mmHg) | Severely elevated (≥180/120 mmHg) |
| Target Organ Damage (TOD) | Absent | Present |
| Symptoms | Minimal | Severe (chest pain, dyspnea, neurological Sx) |
| Management | Oral agents, reduce BP over 24–48 hrs | IV agents, ICU, reduce BP by 25% in 1st hour |
7. Diagnosis
Measurement Methods
- Office BP (OBP): Standard sphygmomanometer; average of 2 readings, 2 separate visits
- Ambulatory BP Monitoring (ABPM): Gold standard — 24-hour recording; detects white-coat and masked hypertension
- Home BP Monitoring (HBPM): 7-day morning and evening readings
Diagnostic Thresholds by Method
| Method | Hypertension Threshold |
|---|---|
| Office BP | ≥140/90 mmHg |
| ABPM (24-hour average) | ≥130/80 mmHg |
| ABPM (daytime average) | ≥135/85 mmHg |
| ABPM (nighttime average) | ≥120/70 mmHg |
| Home BP | ≥135/85 mmHg |
Evaluation Workup
- Urinalysis + urine albumin-to-creatinine ratio (renal damage)
- Serum creatinine + eGFR (CKD)
- Fasting glucose + HbA1c (diabetes)
- Lipid panel (cardiovascular risk)
- Serum electrolytes (K⁺ — hypokalemia suggests aldosteronism)
- ECG (LVH, arrhythmias)
- Thyroid function tests (secondary causes)
- Fundoscopy (hypertensive retinopathy)
8. Target Organ Damage (TOD)
Heart
- Left Ventricular Hypertrophy (LVH) → diastolic dysfunction → heart failure with preserved EF (HFpEF)
- Coronary artery disease → angina, myocardial infarction
- Atrial fibrillation
- Heart failure
Brain
- Ischemic stroke (most common)
- Hemorrhagic stroke / intracerebral hemorrhage
- Transient ischemic attack (TIA)
- Hypertensive encephalopathy (headache, confusion, seizures — PRES syndrome)
- Vascular dementia / cognitive decline
Kidneys
- Hypertensive nephrosclerosis → proteinuria → CKD → ESRD
- Bidirectional relationship: CKD worsens hypertension
Eyes
- Hypertensive Retinopathy — graded by Keith-Wagener-Barker classification:
- Grade I: Arteriovenous nicking, silver-wiring
- Grade II: AV nicking + focal arteriolar narrowing
- Grade III: Flame hemorrhages, cotton-wool spots, hard exudates
- Grade IV: Papilledema (hypertensive emergency)
Large Vessels
- Aortic aneurysm (abdominal and thoracic)
- Aortic dissection
- Peripheral artery disease
9. Management
A. Lifestyle Modifications (All Patients)
| Intervention | Expected SBP Reduction |
|---|---|
| DASH diet | 8–14 mmHg |
| Sodium restriction (<2.3 g/day) | 2–8 mmHg |
| Weight loss (per 10 kg) | 5–10 mmHg |
| Regular aerobic exercise | 4–9 mmHg |
| Alcohol reduction | 2–4 mmHg |
| Smoking cessation | Vascular risk ↓ (indirect) |
B. Pharmacological Treatment
| Drug Class | Mechanism | Preferred In |
|---|---|---|
| ACE Inhibitors (e.g., ramipril, lisinopril) | Block angiotensin II production | Diabetes, CKD, HF, post-MI |
| ARBs (e.g., losartan, valsartan) | Block AT1 receptor | ACE inhibitor-intolerant; diabetes, CKD |
| Calcium Channel Blockers (CCBs) (e.g., amlodipine) | Peripheral vasodilation | Elderly, isolated systolic HTN, angina |
| Thiazide Diuretics (e.g., hydrochlorothiazide, indapamide) | Reduce volume, vasodilate | Black patients, elderly, heart failure |
| Beta-Blockers (e.g., metoprolol, bisoprolol) | Reduce HR and CO | Post-MI, HF, AF, angina |
Treatment Targets (ESC/ESH 2023):
| Population | Target BP |
|---|---|
| General adults (18–65 yrs) | <130/80 mmHg |
| Adults >65 yrs | 130–140/70–80 mmHg |
| Diabetes | <130/80 mmHg |
| CKD (with proteinuria) | <130/80 mmHg |
| Post-stroke | <130/80 mmHg |
C. Resistant Hypertension
- Add spironolactone (mineralocorticoid receptor antagonist) as 4th agent
- Screen for secondary causes (especially primary aldosteronism, OSA)
- Consider renal denervation (interventional option)
10. Prognosis and Cardiovascular Risk Stratification
| Risk Factors Present | Grade 1 | Grade 2 | Grade 3 |
|---|---|---|---|
| None | Low | Moderate | High |
| 1–2 risk factors | Moderate | Moderate-High | High |
| ≥3 risk factors or TOD | High | High | Very High |
| Established CVD/DM/CKD | Very High | Very High | Very High |
11. Special Populations
| Population | Key Considerations |
|---|---|
| Pregnancy | Use methyldopa, labetalol, nifedipine; ACEi/ARBs CONTRAINDICATED |
| Elderly (>65 yrs) | Start low, go slow; watch for orthostatic hypotension |
| CKD | ACEi/ARB preferred; monitor K⁺ and creatinine |
| Diabetes | ACEi/ARB preferred; target <130/80 mmHg |
| Heart Failure | ACEi/ARB + beta-blocker + mineralocorticoid antagonist |
| Atrial Fibrillation | Beta-blockers or non-dihydropyridine CCBs for rate control |
| Black Patients | CCBs + thiazide diuretics preferred (lower RAAS activity) |
Summary
Arterial hypertension is a multifactorial, chronic cardiovascular syndrome characterized by persistently elevated BP that silently damages the heart, brain, kidneys, and blood vessels over time. Early detection, comprehensive risk stratification, lifestyle modification, and individualized pharmacotherapy are the cornerstones of management.
- ESC 2024 / ESH 2023 Guidelines on Management of Arterial Hypertension (p. 8)
- Management of Primary Aldosteronism — Case Detection, Diagnosis, and Treatment (p. 10)
- ACC/AHA 2017 Guidelines on Prevention, Detection, Evaluation, and Management of High Blood Pressure
- Harrison's Principles of Internal Medicine; Braunwald's Heart Disease
man, 45 years old, complained of regular headaches, dizziness and pulsation in the temples, especially in the evening. He also notices the increase in hearts of crushing and a feeling of heaviness in the chest area after physical activity. The patient complains of sleep disturbance, frequent mood swings and fatigue. These symptoms bother him over the past few months, and gradually become more pronounced. NEW: risk factor: risk factor: hereditary predisposition to hypertension (the patient's father suffers from hypertension), overweight (body mass index 32), sedentary lifestyle, high C Salt and alcohol removal, comorbidities: hypercholesterolemia, no signs of diabetes. Drug history does not take permanent medications. Periodically uses painkillers for headaches. Objectively: general condition: the patient is conscious, active, not restless. Skin and slimy normal color. no signs of anemia. Blood pressure: 160/100 mm Hg. st. empty 88 beats / min. Rhythmic. Heart sounds: enhanced, normal. Breathing: rapid, without signs of shortness of breath at rest. Belly: soft, painless. Neurological status: without pathology Laboratory and instrumental data: 1 emoglobin: 14 1/L, erythrocytes: 5.2 10% ml. Necoocytes: 6.2 x 10% (within normal limits), platelets: 220 - 10% (norm), C-reactive protein: 3 within the normal range). Biochemistry: total cholesterol 6.5 mmol/l, linip 4.5 mmol/l, HDL 1.1 mmol of risicerides 2.0 mmol/l. ECG: sinus rhythm, signs of left ventricular hypertrophy X-ray of a difficult cell: without pathologies. Echocardiography: left ventricular hypertrophy, hemodynamic disorders. Ultrasound of the abdominal organs cavities: No change. Tasks: 1. What diagnosis can be made on the basis of clinical data? 2. What additional examinations can help clarify the diagnosis? What diseases should be excluded in differential diagnosis? What is the management strategy
arterial hypertension grade 2 diagnosis criteria left ventricular hypertrophy cardiovascular risk stratification
hypertension management strategy treatment antihypertensive drugs obese hypercholesterolemia
secondary hypertension differential diagnosis exclusion renal pheochromocytoma aldosteronism workup
hypertension additional investigations ABPM urine albumin creatinine renal function lipid statin
Clinical Case Analysis — Male, 45 Years Old
TASK 1 — Diagnosis
Primary Diagnosis:
Primary (Essential) Arterial Hypertension, Grade 2, Stage II High cardiovascular risk Complication: Hypertension-Mediated Organ Damage (HMOD) — Left Ventricular Hypertrophy (LVH)
Justification:
| Criterion | Patient's Data | Interpretation |
|---|---|---|
| BP level | 160/100 mmHg | Grade 2 hypertension (ESH 2023: SBP 160–179 and/or DBP 100–109) |
| Stage | LVH on ECG + Echo | Stage II (HMOD present) |
| CV Risk | LVH + multiple risk factors | High cardiovascular risk |
| Symptoms | Headaches, temple pulsation, dizziness, heaviness in chest after exertion, fatigue, sleep disturbance, mood swings | Classic hypertension + early cardiac involvement |
| ECG | Signs of LVH | Hypertension-mediated organ damage confirmed |
| Echo | LVH, no hemodynamic disorders (compensated) | Concentric LVH — typical of pressure overload |
| Chest X-ray | No pathology | No pulmonary congestion — no overt heart failure yet |
Risk Factor Burden:
| Risk Factor | Present? |
|---|---|
| Age ≥45 (male) | ✅ |
| Hereditary predisposition (father with HTN) | ✅ |
| Overweight — BMI 32 (obesity class I) | ✅ |
| Sedentary lifestyle | ✅ |
| High salt intake | ✅ |
| Alcohol consumption | ✅ |
| Hypercholesterolemia (total cholesterol 6.5 mmol/L, LDL 4.5 mmol/L, low HDL 1.1 mmol/L) | ✅ |
| Hypertriglyceridemia (TG 2.0 mmol/L) | ✅ |
| Diabetes mellitus | ❌ |
→ Multiple risk factors + HMOD (LVH) = HIGH cardiovascular risk category
Accompanying Diagnosis:
- Hypercholesterolemia / Mixed Dyslipidemia (total cholesterol ↑, LDL ↑, HDL low-normal, TG ↑)
- Obesity class I (BMI 32)
TASK 2 — Additional Examinations
- Confirm sustained hypertension
- Detect further target organ damage
- Exclude secondary causes
- Refine cardiovascular risk
A. To Confirm and Characterize Hypertension
| Investigation | Purpose |
|---|---|
| 24-Hour Ambulatory BP Monitoring (ABPM) | Gold standard — confirms sustained HTN, detects white-coat or masked HTN, assesses nocturnal dipping |
| Home BP monitoring (HBPM) | 7-day morning + evening readings as alternative to ABPM |
B. To Detect Further Target Organ Damage (HMOD)
| Investigation | Purpose |
|---|---|
| Urine albumin-to-creatinine ratio (ACR) | Microalbuminuria — earliest sign of hypertensive nephropathy |
| Serum creatinine + eGFR | Renal function assessment |
| NT-proBNP or BNP | Subclinical heart failure / diastolic dysfunction |
| High-sensitivity cardiac troponin T (hs-cTnT) | Subclinical myocardial injury |
| Fundoscopy (ophthalmoscopy) | Hypertensive retinopathy grade assessment |
| Carotid ultrasound (intima-media thickness) | Preclinical atherosclerosis, carotid plaques |
| Pulse wave velocity (PWV) | Arterial stiffness — marker of vascular HMOD |
| Ankle-brachial index (ABI) | Peripheral artery disease screening |
C. To Screen for Secondary Hypertension
| Investigation | Suspected Cause |
|---|---|
| Serum aldosterone / plasma renin activity (aldosterone-to-renin ratio) | Primary aldosteronism (most common secondary cause) |
| 24-hour urine cortisol or dexamethasone suppression test | Cushing's syndrome |
| Plasma/urine metanephrines | Pheochromocytoma |
| Renal artery Doppler ultrasound | Renovascular hypertension (renal artery stenosis) |
| Polysomnography (sleep study) | Obstructive sleep apnea (BMI 32 + fatigue + sleep disturbance = high suspicion) |
| Thyroid function tests (TSH, fT4) | Hypothyroidism (diastolic HTN, fatigue) |
Clinical note: In this patient, OSA is a particularly important secondary contributor to consider, given BMI 32 + sleep disturbance + fatigue.
TASK 3 — Differential Diagnosis — Conditions to Exclude
| Condition | Distinguishing Features |
|---|---|
| Secondary HTN — Primary Aldosteronism | Hypokalemia, high aldosterone:renin ratio; resistant HTN |
| Secondary HTN — Renovascular | Young onset, abdominal bruit, asymmetric kidneys on ultrasound |
| Secondary HTN — Pheochromocytoma | Paroxysmal HTN, sweating, palpitations, headache triad |
| Secondary HTN — Obstructive Sleep Apnea | BMI 32, fatigue, sleep disturbance, snoring — screen actively |
| Secondary HTN — Cushing's syndrome | Central obesity, moon face, striae, hyperglycemia |
| Hypertensive Heart Disease → HFpEF | LVH present; echo shows no hemodynamic disorders currently — monitor |
| Ischemic Heart Disease / Angina | Chest heaviness after exertion requires stress testing or coronary assessment if symptoms worsen |
| White-coat Hypertension | Rule out with ABPM (BP elevated only in office setting) |
| Hypothyroidism | Can cause diastolic HTN + fatigue; check TSH |
TASK 4 — Management Strategy
Step 1: Lifestyle Modification (MANDATORY — all patients, immediately)
| Intervention | Recommendation |
|---|---|
| Weight reduction | Target BMI <25; even 5–10 kg loss reduces SBP by 5–10 mmHg |
| DASH diet / Low sodium | Sodium <2 g/day; increase fruits, vegetables, low-fat dairy |
| Aerobic exercise | ≥30 min moderate intensity, 5 days/week (reduces SBP 4–9 mmHg) |
| Alcohol reduction | ≤2 standard drinks/day; total ≤14 units/week |
| Smoking cessation | If applicable |
| Sleep hygiene | Especially important given sleep disturbance; investigate OSA |
Step 2: Antihypertensive Pharmacotherapy
Recommended First-Line Combination:
ACE Inhibitor (or ARB) + Calcium Channel Blocker (CCB) (preferred initial dual combination per ESH 2023 single-pill combination strategy)
| Drug | Dose | Rationale |
|---|---|---|
| Ramipril 5–10 mg OD (ACEi) OR Perindopril 5–10 mg OD | Start 5 mg → titrate | Reduces LVH regression, renoprotective, improves endothelial function |
| Amlodipine 5–10 mg OD (CCB) | Add to ACEi | Potent vasodilator, proven CV outcome benefit, metabolically neutral |
Drugs to Avoid/Use Cautiously:
- Beta-blockers: Not first-line unless concurrent angina, AF, or HF with reduced EF
- Thiazide diuretics alone: May worsen dyslipidemia and glucose tolerance (use indapamide — more metabolically neutral)
Step 3: Manage Dyslipidemia (Cardiovascular Risk Reduction)
| Parameter | Patient Value | Target |
|---|---|---|
| Total cholesterol | 6.5 mmol/L | <5.0 mmol/L |
| LDL-C | 4.5 mmol/L | <1.8 mmol/L (high CV risk) |
| HDL-C | 1.1 mmol/L | >1.0 mmol/L ✅ |
| Triglycerides | 2.0 mmol/L | <1.7 mmol/L |
Start statin therapy — e.g., Rosuvastatin 10–20 mg OD or Atorvastatin 20–40 mg OD LDL target: <1.8 mmol/L (high CV risk per ESC guidelines) If TG remain elevated after LDL control: consider fenofibrate
Step 4: Address NSAID Use
- Patient uses NSAIDs periodically for headaches — advise to discontinue (NSAIDs raise BP by inhibiting vasodilatory prostaglandins and promoting sodium retention)
- Use paracetamol for headache management instead
Step 5: Follow-Up Plan
| Timeframe | Action |
|---|---|
| 2–4 weeks | Recheck BP, assess medication tolerance, reinforce lifestyle |
| 4–8 weeks | Evaluate BP response; titrate/add drugs if target not reached |
| 3 months | Repeat lipid panel, renal function, electrolytes; urine ACR |
| 6–12 months | Repeat ECG ± Echo to assess LVH regression |
| Annually | Full cardiovascular risk reassessment |
Summary Diagram
DIAGNOSIS
─────────────────────────────────────────────────────
Primary Arterial HTN, Grade 2, Stage II
(BP 160/100 + LVH on ECG/Echo)
HIGH Cardiovascular Risk
+ Dyslipidemia + Obesity Class I
MANAGEMENT
─────────────────────────────────────────────────────
1. Lifestyle: DASH diet, ↓ Na, ↓ alcohol,
exercise, weight loss, stop NSAIDs
2. Antihypertensive: ACEi (Ramipril) +
CCB (Amlodipine) → target <130/80 mmHg
3. Statin: Rosuvastatin/Atorvastatin
→ LDL target <1.8 mmol/L
4. Investigate: ABPM, urine ACR, eGFR,
fundoscopy, sleep study (OSA), screen
for secondary causes
5. Monitor: BP q4wk until target, then q6mo
Echo at 12 months for LVH regression
- ESC 2024 / ESH 2023 Guidelines on Management of Elevated Blood Pressure and Hypertension (p. 38)
- ACC/AHA 2017 Guidelines — Prevention, Detection, Evaluation, and Management of High Blood Pressure (p. 51)
- Management of Primary Aldosteronism — Case Detection, Diagnosis, and Treatment
- Harrison's Principles of Internal Medicine; Braunwald's Heart Disease
Dilagnostic criteria for ventricular fibrillation
ventricular fibrillation diagnostic criteria ECG features clinical presentation
ventricular fibrillation ECG pattern waveform
ventricular fibrillation pathophysiology causes management defibrillation ACLS
coarse fine ventricular fibrillation ECG comparison types
Diagnostic Criteria for Ventricular Fibrillation (VF)
1. Definition
2. ECG — The Gold Standard for Diagnosis
Classic ECG Criteria:
| ECG Feature | Finding in VF |
|---|---|
| P waves | Absent — no organized atrial activity |
| QRS complexes | Absent — no identifiable ventricular depolarization |
| T waves | Absent |
| Baseline | Completely chaotic, irregular, undulating oscillations |
| Rate | 150–500 electrical deflections/min (not true beats) |
| Rhythm | Totally irregular — no two deflections are alike |
| Amplitude | Variable — depends on type (coarse vs. fine) |
| Isoelectric line | None — baseline is never flat |
The hallmark is the complete absence of any recognizable P-QRS-T morphology, replaced by irregular, rapid, chaotic waveforms of varying shape, amplitude, and frequency.
ECG Example — Classic Ventricular Fibrillation:
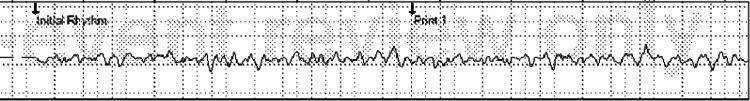
3. Types of VF — ECG Classification
| Type | Amplitude | Frequency | Clinical Significance |
|---|---|---|---|
| Coarse VF | >5 mm (often >10 mm) | High | Early VF; myocardium still metabolically active; better defibrillation success |
| Fine VF | <5 mm, near-isoelectric | Low | Later VF; ischemic, ATP-depleted myocardium; lower defibrillation success |
| Ultra-fine VF | Barely perceptible | Very low | May mimic asystole; can be mistaken for flat line — always confirm in 2 leads |
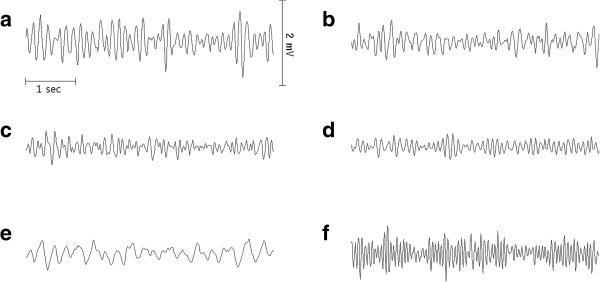
4. Clinical Diagnostic Criteria
Signs and Symptoms:
| Feature | Finding |
|---|---|
| Consciousness | Sudden loss of consciousness (within seconds of onset) |
| Pulse | Absent — no palpable carotid or femoral pulse |
| Breathing | Absent or agonal gasping |
| Blood pressure | Unmeasurable |
| Heart sounds | Absent |
| Skin | Cyanotic, ashen, diaphoretic |
| Pupils | Dilate within 1–2 minutes without treatment |
Clinical diagnosis of VF = Cardiac arrest + Shockable rhythm confirmed on cardiac monitor
5. Diagnostic Approach — Step by Step
STEP 1: Recognize cardiac arrest
↓ Unresponsive, no normal breathing, no pulse (< 10 sec check)
STEP 2: Activate emergency response + Start CPR immediately
STEP 3: Attach cardiac monitor / defibrillator AS SOON AS AVAILABLE
↓
STEP 4: Analyze rhythm on monitor
↓
Is it chaotic, irregular, no P-QRS-T?
↓ YES
STEP 5: CONFIRM VF in ≥2 leads (rule out artifact or fine VF vs asystole)
↓
STEP 6: SHOCK — Immediate unsynchronized defibrillation
6. Differentiating VF from Other Rhythms
| Feature | VF | Pulseless VT | Asystole | Pulseless Electrical Activity (PEA) |
|---|---|---|---|---|
| ECG pattern | Chaotic, irregular, no QRS | Wide, monomorphic/polymorphic QRS | Flat line (± P waves) | Organized rhythm (any pattern) |
| Rate | 150–500/min (electrical) | 100–300/min | None | Any |
| P waves | Absent | Usually absent | May be present | May be present |
| QRS | Absent | Wide, regular/irregular | Absent | Present (any morphology) |
| Pulse | Absent | Absent | Absent | Absent |
| Shockable? | YES | YES | NO | NO |
| Management | Defibrillation | Defibrillation | Epinephrine + CPR | Treat reversible causes (H's & T's) |
7. Etiology (Causes to Identify)
Cardiac Causes (most common):
- Acute myocardial infarction (MI) — most frequent trigger
- Structural heart disease: dilated cardiomyopathy, hypertrophic cardiomyopathy
- Left ventricular hypertrophy / heart failure
- Valvular heart disease (aortic stenosis)
- Prior cardiac arrest / prior VF
- Congenital channelopathies: Brugada syndrome, Long QT syndrome, Short QT syndrome, Catecholaminergic Polymorphic VT (CPVT)
- Wolff-Parkinson-White syndrome with rapid AF
Non-Cardiac / Reversible Causes ("H's and T's"):
| H's | T's |
|---|---|
| Hypoxia | Tension pneumothorax |
| Hypovolemia | Tamponade (cardiac) |
| Hydrogen ion (acidosis) | Toxins (drug overdose) |
| Hypo/Hyperkalemia | Thrombosis — pulmonary (PE) |
| Hypothermia | Thrombosis — coronary (MI) |
8. Pathophysiology
Trigger (ischemia, ion channel mutation, electrolyte imbalance, etc.)
↓
Multiple re-entry circuits form in ventricular myocardium
↓
Electrical wavelets spiral chaotically through ventricles
↓
No uniform depolarization → No coordinated contraction
↓
Ventricular quivering → Zero cardiac output
↓
Cerebral perfusion ceases → LOC within seconds
↓
Biological death within 4–6 minutes without intervention
9. Immediate Management (ACLS Protocol)
Shockable Rhythm → Immediate Defibrillation is the definitive treatment
| Step | Action |
|---|---|
| 1 | Start high-quality CPR (100–120 compressions/min, depth 5–6 cm) |
| 2 | Charge defibrillator — 200 J biphasic (or 360 J monophasic) |
| 3 | Deliver shock → immediately resume CPR for 2 minutes |
| 4 | Re-analyze rhythm after 2-min CPR cycle |
| 5 | If VF persists: repeat shock + Epinephrine 1 mg IV every 3–5 min |
| 6 | If VF refractory (≥3 shocks): Amiodarone 300 mg IV bolus (or Lidocaine 1–1.5 mg/kg as alternative) |
| 7 | Identify and treat reversible causes (H's and T's) |
| 8 | Post-ROSC: targeted temperature management, coronary angiography, ICU care |
Refractory VF:
10. Prognosis
| Factor | Impact on Survival |
|---|---|
| Witnessed arrest | Survival 2–3× higher than unwitnessed |
| Bystander CPR | Doubles survival |
| Time to first shock | Every 1-minute delay → 10% decrease in survival |
| Coarse vs. fine VF | Coarse VF: higher defibrillation success |
| Shockable vs. non-shockable | VF/VT survival >3× higher than PEA/asystole |
| Post-arrest care (TTM, PCI) | Significantly improves neurological outcomes |
Overall survival to hospital discharge for out-of-hospital VF: ~25–40% with optimal response chain; drops to <5% without bystander CPR.
Summary
| Criterion | VF Finding |
|---|---|
| ECG | Chaotic, irregular waveforms; no P, QRS, or T waves |
| Rate | 150–500 irregular deflections/min |
| Clinical | Sudden LOC + absent pulse + absent breathing |
| Diagnosis confirmed by | Cardiac monitor (always check ≥2 leads) |
| Shockable? | YES — immediate defibrillation |
| Definitive treatment | Unsynchronized DC defibrillation |
- Optimal Post Cardiac Arrest and Refractory Cardiac Arrest Patient Care (p. 11) — DOSE VF trial data
- ECG images: PMC Clinical VQA Dataset — validated clinical ECG tracings
- AHA ACLS Guidelines 2020/2023; Harrison's Principles of Internal Medicine; Braunwald's Heart Disease
Principles of treatment of acute and chronic circulatory failure.
acute circulatory failure cardiogenic shock treatment principles management
chronic heart failure treatment principles pharmacotherapy management guidelines
acute heart failure classification Killip Forrester hemodynamic profile diuretics vasodilators inotropes
chronic heart failure HFrEF HFpEF device therapy ICD CRT NYHA classification stages
Principles of Treatment of Acute and Chronic Circulatory Failure
PART I — ACUTE CIRCULATORY FAILURE (ACF)
1. Definition and Overview
- Acute Heart Failure (AHF) — sudden onset or rapid worsening of HF symptoms
- Cardiogenic shock — severe end of the spectrum with critical hypoperfusion
- Distributive shock (septic, anaphylactic, neurogenic)
- Hypovolemic shock
- Obstructive shock (PE, cardiac tamponade, tension pneumothorax)
In clinical cardiology, "acute circulatory failure" most commonly refers to AHF and cardiogenic shock.
2. Classification of Acute Heart Failure
A. Killip Classification (Post-MI AHF)
| Class | Clinical Features | Mortality |
|---|---|---|
| I | No signs of HF | ~6% |
| II | S3 gallop, mild pulmonary rales, JVD | ~17% |
| III | Acute pulmonary edema | ~38% |
| IV | Cardiogenic shock (SBP <90 mmHg + hypoperfusion) | ~67% |
B. Forrester Hemodynamic Classification
| Profile | PCWP | CI | Clinical | Treatment Focus |
|---|---|---|---|---|
| I (Normal) | <18 mmHg | >2.2 L/min/m² | Compensated | Optimize outpatient therapy |
| II (Wet & Warm) | >18 mmHg | >2.2 L/min/m² | Congestion, adequate perfusion | Diuretics + Vasodilators |
| III (Dry & Cold) | <18 mmHg | <2.2 L/min/m² | Hypoperfusion, no congestion | IV fluids + Inotropes |
| IV (Wet & Cold) | >18 mmHg | <2.2 L/min/m² | Congestion + hypoperfusion | Inotropes + Vasopressors + Diuretics |
3. General Principles of Acute Circulatory Failure Treatment
Principle 1 — Stabilize and Support Vital Functions
- Secure airway, breathing, circulation (ABC)
- Supplemental oxygen if SpO₂ <90%; target SpO₂ 94–98%
- Non-invasive ventilation (NIV — CPAP/BiPAP) for acute pulmonary edema — reduces intubation rate, improves oxygenation, decreases preload
- Mechanical ventilation if NIV fails or GCS ↓
- Continuous cardiac monitoring, pulse oximetry, arterial line for BP in shock
Principle 2 — Reduce Preload (Decongest)
| Drug | Dose | Mechanism |
|---|---|---|
| Furosemide | 40–80 mg IV bolus (or continuous infusion) | Inhibits Na⁺/K⁺/2Cl⁻ in loop of Henle → rapid diuresis |
| Bumetanide | 1–2 mg IV | Same class, higher bioavailability |
| Torasemide | 10–20 mg IV | Longer acting |
If diuretic resistance: increase dose, add thiazide (metolazone), or switch to continuous infusion.
| Drug | Route | Key Use |
|---|---|---|
| Nitroglycerin | IV infusion / sublingual | Predominantly venodilator (preload ↓); AHF + ischemia |
| Isosorbide dinitrate | IV / sublingual | Similar to NTG |
| Sodium nitroprusside | IV infusion | Balanced arterio- and venodilator; hypertensive AHF |
| Nesiritide (BNP analog) | IV | Vasodilation + natriuresis |
⚠️ Vasodilators are contraindicated in hypotension (SBP <90 mmHg)
Principle 3 — Improve Perfusion (Support Cardiac Output)
| Drug | Mechanism | Dose | Notes |
|---|---|---|---|
| Dobutamine | β₁ agonist → ↑ contractility + mild vasodilation | 2–20 mcg/kg/min IV | First-line inotrope; avoid in hypotension alone |
| Dopamine | Dose-dependent: DA₁ (low), β₁ (mod), α₁ (high) | 2–20 mcg/kg/min IV | Vasopressor at higher doses |
| Norepinephrine | α₁ + β₁ → vasoconstriction + inotrope | 0.1–1 mcg/kg/min IV | Preferred vasopressor in cardiogenic shock |
| Milrinone | PDE-3 inhibitor → ↑ cAMP → inotrope + vasodilator | 0.25–0.75 mcg/kg/min | Useful in β-blocker use; lowers SVR |
| Levosimendan | Ca²⁺ sensitizer → inotrope + vasodilator | 0.1–0.2 mcg/kg/min | Does not increase O₂ demand; preferred in ischemic HF |
Principle 4 — Treat the Underlying Cause (Etiology-Directed)
| Cause | Specific Treatment |
|---|---|
| Acute MI | Emergency PCI / thrombolysis (revascularization) |
| Hypertensive emergency | IV antihypertensives (nitroprusside, labetalol) |
| Acute valvular failure (papillary muscle rupture, acute AR) | Emergency cardiac surgery |
| Pulmonary embolism | Thrombolysis or catheter embolectomy |
| Cardiac tamponade | Emergency pericardiocentesis |
| Tension pneumothorax | Immediate needle decompression |
| Arrhythmia-induced AHF | Cardioversion, rate control, antiarrhythmics |
Principle 5 — Mechanical Circulatory Support (MCS)
| Device | Mechanism | Use |
|---|---|---|
| Intra-Aortic Balloon Pump (IABP) | Counterpulsation → ↑ coronary perfusion, ↓ afterload | Cardiogenic shock, post-MI, bridge to surgery |
| Impella | Microaxial pump — unloads LV directly | Severe cardiogenic shock; bridges to PCI/transplant |
| VA-ECMO | Extracorporeal circulatory + oxygenation support | Refractory cardiogenic shock; as bridge to decision |
| LVAD | Long-term mechanical LV assist | Bridge to transplant or destination therapy |
Principle 6 — Monitoring and Titration
- Urine output: target >0.5 mL/kg/hr (response to diuretics)
- Serum electrolytes (K⁺, Na⁺, Mg²⁺): monitor q4–6h in active diuresis
- Serum creatinine + eGFR: cardiorenal syndrome risk
- BNP / NT-proBNP: guides diuresis intensity and therapy response
- Echocardiography: urgent bedside echo for LVEF, wall motion, effusion, valve function
- Arterial line + CVP / Swan-Ganz catheter: in cardiogenic shock for hemodynamic profiling
4. Summary Algorithm — AHF Treatment
ACUTE HEART FAILURE
↓
ABC stabilization + O₂ + Cardiac monitoring
↓
Assess hemodynamic profile (Forrester)
↓
┌──────────────────────────────────────────────┐
│ WET & WARM WET & COLD │
│ (congestion + (congestion + │
│ adequate perfusion) hypoperfusion) │
│ → IV diuretics → Inotropes + │
│ → Vasodilators Vasopressors + │
│ Diuretics │
└──────────────────────────────────────────────┘
↓
Treat underlying cause
↓
If refractory → Mechanical Circulatory Support
↓
Stabilize → Transition to chronic HF therapy
PART II — CHRONIC CIRCULATORY FAILURE (Chronic Heart Failure — CHF)
1. Definition and Classification
Classification by LVEF:
| Type | LVEF | Predominant Mechanism |
|---|---|---|
| HFrEF (reduced EF) | <40% | Systolic dysfunction — impaired contractility |
| HFmrEF (mildly reduced EF) | 40–49% | Mixed systolic/diastolic |
| HFpEF (preserved EF) | ≥50% | Diastolic dysfunction — impaired relaxation/filling |
NYHA Functional Classification:
| Class | Symptoms |
|---|---|
| I | No limitation; ordinary activity causes no symptoms |
| II | Slight limitation; comfortable at rest; ordinary activity → fatigue, dyspnea |
| III | Marked limitation; comfortable at rest; less than ordinary activity → symptoms |
| IV | Unable to carry out any activity without discomfort; symptoms at rest |
2. Principles of Chronic HF Treatment
Principle 1 — Lifestyle Modification (All Patients)
| Intervention | Recommendation |
|---|---|
| Fluid restriction | 1.5–2 L/day in severe HF or hyponatremia |
| Sodium restriction | <2–3 g/day |
| Weight monitoring | Daily; report >2 kg gain in 2 days → early decompensation |
| Exercise / Cardiac rehabilitation | Moderate aerobic exercise (30 min, 5×/week) — reduces hospitalizations, improves QoL |
| Alcohol restriction | ≤1–2 standard drinks/day; abstinence if alcoholic cardiomyopathy |
| Smoking cessation | Mandatory |
| Vaccination | Annual influenza; pneumococcal vaccine |
Principle 2 — Foundational "Quadruple Therapy" for HFrEF
| Drug Class | Example | Mechanism | Benefits |
|---|---|---|---|
| ACEi / ARNI | Ramipril / Sacubitril-Valsartan (Entresto) | Blocks RAAS; ARNI also blocks neprilysin → ↑ natriuretic peptides | ↓ Mortality, ↓ hospitalizations, ↓ LV remodeling |
| Beta-Blocker | Carvedilol, Bisoprolol, Metoprolol succinate | Blocks SNS activation → ↓ HR, ↓ remodeling | ↓ Mortality, ↓ sudden cardiac death, ↓ rehospitalization |
| MRA (Mineralocorticoid Receptor Antagonist) | Spironolactone, Eplerenone | Blocks aldosterone → ↓ fibrosis, ↓ Na retention | ↓ Mortality, ↓ hospitalizations; monitor K⁺ |
| SGLT2 Inhibitor | Dapagliflozin, Empagliflozin | Osmotic diuresis, ↓ preload/afterload, cardiorenal protection | ↓ CV death, ↓ HF hospitalizations (independent of diabetes) |
STRONG-HF Trial evidence: Rapid up-titration to 100% recommended doses within 2 weeks of AHF discharge → 34% relative risk reduction in 180-day HF re-admission or all-cause death vs. usual care (p. 46).
Preferred Sequence by Patient Profile:
| Scenario | Preferred Start |
|---|---|
| SBP ≥100 mmHg, SR | ARNI + Beta-blocker + MRA + SGLT2i |
| ACEi intolerant (cough) | Switch to ARB (valsartan, losartan) |
| HR >70 bpm, sinus rhythm, on max beta-blocker | Add Ivabradine (If-channel blocker) |
| Persistent congestion | Add loop diuretic (furosemide, torasemide) |
| Hyponatremia + refractory congestion | Consider Tolvaptan (V2 receptor antagonist) |
Principle 3 — Diuretics (Symptom Relief)
| Drug | Class | Dose Range | Notes |
|---|---|---|---|
| Furosemide | Loop | 20–240 mg/day PO | Most used; titrate to target weight/euvolemia |
| Torasemide | Loop | 5–20 mg/day | Better oral bioavailability than furosemide |
| Hydrochlorothiazide | Thiazide | 12.5–25 mg/day | Add-on for diuretic resistance |
| Metolazone | Thiazide-like | 2.5–5 mg/day | Potent add-on for severe resistance |
| Spironolactone | MRA/K-sparing | 25–50 mg/day | Dual role: diuresis + mortality benefit |
Principle 4 — Treatment of HFpEF
| Intervention | Evidence |
|---|---|
| SGLT2 inhibitors (dapagliflozin, empagliflozin) | ↓ HF hospitalizations (EMPEROR-Preserved, DELIVER trials) — now first-line for HFpEF |
| Diuretics | Symptom relief from congestion |
| Control of comorbidities | Hypertension, AF, diabetes, obesity — treating these improves outcomes |
| Exercise training | Improves exercise capacity and QoL |
| MRAs (spironolactone) | Modest benefit, especially in EF 45–55% range |
| ARBs / ACEi | Reduce hospitalizations modestly |
Principle 5 — Device Therapy
| Device | Indication | Benefit |
|---|---|---|
| ICD (Implantable Cardioverter-Defibrillator) | HFrEF with LVEF ≤35% + NYHA II–III on optimal medical therapy ≥3 months | Prevents sudden cardiac death |
| CRT (Cardiac Resynchronization Therapy — biventricular pacemaker) | LVEF ≤35% + LBBB with QRS ≥150 ms + NYHA II–IV | ↓ Mortality, ↓ hospitalizations, improves LVEF + symptoms |
| CRT-D (CRT + ICD) | Combined indications | Dual benefit — resynchronization + defibrillation |
| LVAD (Left Ventricular Assist Device) | Advanced HF (NYHA IV), refractory to max therapy | Bridge to transplant or destination therapy |
Principle 6 — Advanced / End-Stage HF
- Heart transplantation — gold standard for end-stage HF; 1-year survival ~85%; 5-year ~75%
- Palliative care — for patients not candidates for transplant/LVAD: symptom control, opioids for dyspnea, psychological and social support
- Multidisciplinary HF clinics — nurse-led, structured follow-up; reduce readmissions significantly
3. Monitoring in Chronic HF
| Parameter | Frequency | Target/Action |
|---|---|---|
| Daily weight | Daily (patient) | Alert if >2 kg gain in 2 days |
| BP, HR | Every visit | SBP 90–120 mmHg; HR 50–60 bpm on beta-blocker |
| Serum K⁺ + creatinine | At initiation, dose change, q3–6 months | K⁺ 4.0–5.0 mmol/L; eGFR >30 for ACEi/ARNI |
| NT-proBNP / BNP | At hospitalization and follow-up | Trend downward = favorable response |
| Echocardiography | At diagnosis; q1–2 years or after therapy change | LVEF, wall motion, valves, filling pressures |
| 6-Minute Walk Test | Functional assessment | Improvement with therapy |
Comparison: Acute vs. Chronic Circulatory Failure Treatment
| Principle | Acute (AHF) | Chronic (CHF) |
|---|---|---|
| Priority | Stabilize hemodynamics, relieve congestion, maintain perfusion | Prevent disease progression, reduce mortality/rehospitalization |
| Diuretics | IV loop diuretics (high dose, fast) | Oral loop diuretics (titrated to euvolemia) |
| Vasodilators | IV nitroglycerin / nitroprusside | ACEi / ARNI / hydralazine-nitrate |
| Inotropes | IV dobutamine, milrinone, levosimendan | NOT used in chronic stable HF (increase mortality) |
| Vasopressors | Norepinephrine in cardiogenic shock | Not applicable |
| Beta-blockers | Withheld/cautious in acute decompensation | Cornerstone of HFrEF therapy |
| SGLT2 inhibitors | Can continue if on them; start early post-stabilization | Proven mortality/hospitalization benefit |
| Mechanical support | IABP, Impella, VA-ECMO (temporary) | LVAD (long-term bridge/destination) |
| Monitoring | ICU-level (arterial line, hourly UO, Swan-Ganz) | Outpatient (weight, BP, labs, echo) |
Summary
ACUTE CIRCULATORY FAILURE
─────────────────────────────────────────────
1. ABC + O₂ + NIV if pulmonary edema
2. IV loop diuretics → decongest
3. Vasodilators (if SBP >90) → ↓ preload/afterload
4. Inotropes/vasopressors (if shock) → ↑ CO
5. Treat underlying cause (MI → PCI, tamponade → tap)
6. Mechanical support if refractory (IABP, ECMO)
7. Monitor: UO, electrolytes, BNP, echo
CHRONIC CIRCULATORY FAILURE
─────────────────────────────────────────────
1. Lifestyle: ↓ salt/fluid, exercise, weight monitoring
2. Quadruple therapy (HFrEF):
ACEi/ARNI + Beta-blocker + MRA + SGLT2i
3. Diuretics: symptom/volume control (no mortality benefit)
4. HFpEF: SGLT2i + diuretics + comorbidity control
5. Devices: ICD (↓ SCD) / CRT (↓ death + resync)
6. Advanced: LVAD / Heart transplant
7. Monitor: BNP, echo, K⁺/creatinine, daily weight
- ESC 2021 Guidelines for Diagnosis and Treatment of Acute and Chronic Heart Failure
- Management of Cardiovascular Disease in Patients with Diabetes (p. 46) — Quadruple therapy, STRONG-HF trial
- Management of Cardiovascular Disease and Pregnancy (p. 37) — MCS in AHF
- Optimal Post Cardiac Arrest and Refractory Cardiac Arrest Patient Care (p. 11)
- Harrison's Principles of Internal Medicine; Braunwald's Heart Disease