Alep -TC
Reading File
search_medical_text
Aleppo boil cutaneous leishmaniasis diagnosis treatment
search_medical_image
cutaneous leishmaniasis Aleppo boil skin lesion
GradeResponse
GradeResponse
Aleppo Boil — Cutaneous Leishmaniasis (CL)
"Alep" is a traditional name for the Aleppo boil (also called Oriental sore, Delhi boil, Baghdad boil), the classic form of cutaneous leishmaniasis (CL) — a vector-borne parasitic infection caused by Leishmania protozoa transmitted by the bite of infected female sandflies (Phlebotomus spp. in the Old World).
Etiology & Epidemiology
| Feature | Detail |
|---|---|
| Causative organisms | L. major, L. tropica (Old World); L. braziliensis, L. mexicana (New World) |
| Vector | Female sandfly (Phlebotomus in Old World; Lutzomyia in New World) |
| Endemic regions | Middle East, Central Asia, North Africa, Mediterranean, Latin America |
| Classic name origin | "Aleppo boil" from Aleppo, Syria — a historically endemic region |
Clinical Presentation
- Incubation: weeks to months after sandfly bite
- Initial lesion: papule at the bite site → enlarges into a nodule → ulcerates
- Classic lesion: painless, well-demarcated ulcer with raised, indurated ("volcano sign") borders and central crust/exudate
- Healing: spontaneous healing over months, leaving a depressed scar
- Lymphatic spread may occur (L. braziliensis can cause mucosal leishmaniasis — ML)
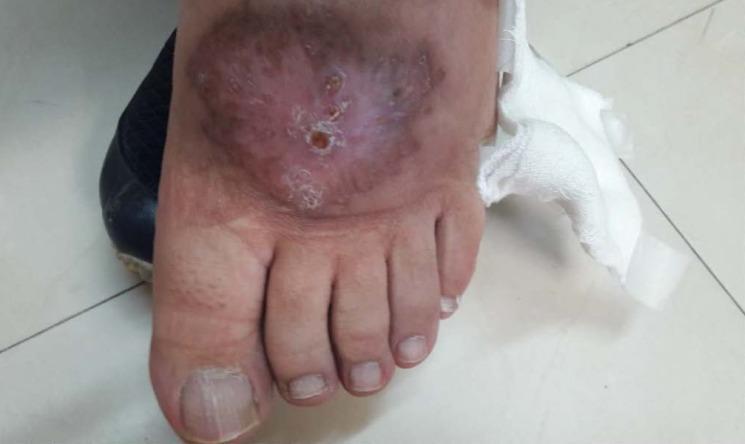
Diagnosis
- Slit-skin smear / biopsy: Giemsa stain showing amastigotes within macrophages (Leishman-Donovan bodies)
- Culture: NNN (Novy-MacNeal-Nicolle) medium
- PCR: most sensitive; allows species identification
- Montenegro skin test (leishmanin test): delayed hypersensitivity; positive in most CL cases
- Serology: less reliable in CL (more useful in visceral leishmaniasis)
Treatment
(Harrison's, p. 6501)
Indications for treatment (beyond watchful waiting):
- Lesions on face, hands, or joints
- Multiple lesions
- Large ulcers
- Lymphatic spread
- New World CL (risk of mucosal progression)
- HIV co-infection
Topical agents (for small, single, self-healing species lesions):
- Topical paromomycin ointment
- Intralesional sodium stibogluconate (SSG)
- Cryotherapy / thermotherapy
Systemic treatment:
| Drug | Dose / Notes |
|---|---|
| Meglumine antimoniate (Glucantime) | 20 mg/kg/day IM/IV × 20–28 days; first-line in most endemic areas |
| Sodium stibogluconate (Pentostam) | 20 mg/kg/day IV/IM × 20–28 days |
| Miltefosine (oral) | 2.5 mg/kg/day × 28 days; teratogenic, avoid in pregnancy |
| Fluconazole | 200 mg/day × 6 weeks; used for L. major |
| Liposomal amphotericin B | Reserved for refractory/HIV cases |
Complications
- Mucocutaneous leishmaniasis (ML): destructive lesions of nose/palate/pharynx — mainly L. braziliensis
- Disseminated CL: multiple non-ulcerating lesions — L. amazonensis
- Secondary bacterial superinfection
- Scarring (cosmetically significant especially on face)
Key Points
- The Aleppo boil is the prototypical Old World CL caused by L. major or L. tropica
- Many cases self-heal over 3–18 months, but treatment is warranted for high-risk locations or spread
- Species identification guides prognosis and treatment duration
- Always consider mucosal progression in New World CL patients
Alop TC
search_medical_text
alopecia totalis capitis treatment management
search_medical_image
alopecia totalis scalp hair loss
GradeResponse
GradeResponse
Alopecia Totalis (Alop TC)
Alopecia Totalis (AT) is a severe subtype of alopecia areata (AA) characterized by complete loss of all scalp hair, sparing the body. It sits on the spectrum between patchy AA and alopecia universalis (total body hair loss).
Classification of Alopecia Areata Spectrum
| Type | Extent of Hair Loss |
|---|---|
| Alopecia Areata (AA) | Patchy scalp hair loss |
| Alopecia Totalis (AT) | Complete scalp hair loss |
| Alopecia Universalis (AU) | Complete scalp + body hair loss |
Pathophysiology
- Autoimmune T-cell mediated attack on hair follicles
- Loss of immune privilege of the hair follicle
- CD8⁺ NKG2D⁺ T cells infiltrate the follicle bulb
- Strong genetic association: HLA-DR, HLA-DQ alleles
- Associated with other autoimmune conditions: thyroid disease, vitiligo, atopic dermatitis (AD)
- Atopic dermatitis is a known comorbidity — patients with AD have higher likelihood of progressing to AT/AU
Clinical Features
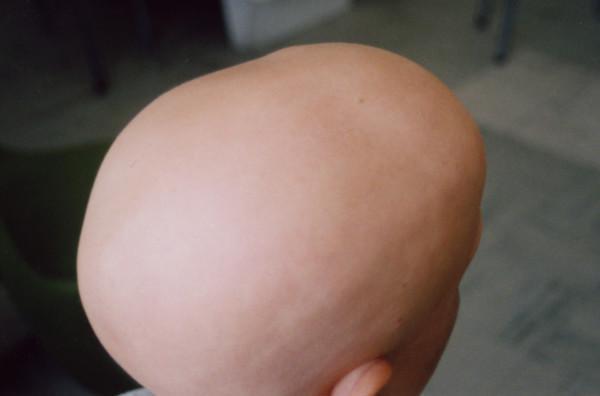
- Complete loss of scalp hair (eyebrows and eyelashes may be spared)
- Non-scarring alopecia — follicles preserved, regrowth possible
- Scalp appears smooth, slightly glossy, non-inflammatory
- Exclamation mark hairs at patch margins (tapering proximally) — characteristic of AA spectrum
- Nail changes: pitting, trachyonychia in ~10–20% of cases
- Onset: any age, including children
Diagnosis
- Primarily clinical
- Dermoscopy: yellow dots, black dots, broken hairs, exclamation mark hairs, vellus hairs
- Scalp biopsy: peribulbar lymphocytic infiltrate ("swarm of bees" pattern) — confirms when in doubt
- Pull test: positive at margins
- Labs: TSH, anti-TPO (rule out thyroid disease), CBC, ferritin, ANA
Treatment
Treatment is challenging; spontaneous remission is less likely in AT than in patchy AA.
First-Line
| Treatment | Notes |
|---|---|
| Potent topical corticosteroids | Clobetasol propionate; limited efficacy in AT |
| Intralesional corticosteroids | Triamcinolone acetonide 5–10 mg/mL; impractical for full scalp |
| Systemic corticosteroids | Short-term pulse therapy; high relapse rate on discontinuation |
Second-Line
| Treatment | Notes |
|---|---|
| Contact immunotherapy (DPCP/SADBE) | Diphencyprone applied weekly; most established second-line for extensive AA/AT |
| Minoxidil | Adjunct to stimulate regrowth; not curative alone |
| Methotrexate | ± systemic corticosteroids; used in refractory cases |
| Cyclosporine | Effective but high relapse rate; use limited by toxicity |
Emerging / Targeted Therapy (First-in-Class)
| Drug | Mechanism | Status |
|---|---|---|
| Baricitinib (Olumiant) | JAK1/2 inhibitor | FDA-approved for severe AA (2022) |
| Ritlecitinib (Litfulo) | JAK3/TEC inhibitor | FDA-approved for severe AA ≥12 yrs (2023) |
| Deuruxolitinib | JAK1/2 inhibitor | Phase III; promising results |
JAK inhibitors are the most significant therapeutic advance in AA/AT — response rates of 30–50% for complete/near-complete regrowth in clinical trials.
Prognosis
- AT carries a poorer prognosis than patchy AA
- Spontaneous remission occurs in ~10–20%
- Prognostic factors for poor outcome: early onset (childhood), long duration, atopic history, nail involvement, family history
- Even with treatment, relapse is common after discontinuation
Key Points
- AT = complete scalp hair loss, non-scarring, autoimmune etiology
- Rule out thyroid disease and other autoimmune comorbidities
- JAK inhibitors (baricitinib, ritlecitinib) represent the current standard of care for severe/refractory cases
- Psychological impact is significant — counseling and support groups are an important part of management
This is a shared conversation. Sign in to Orris to start your own chat.