Explain as,ar,ms,me and differentiate them in all aspects (both theory and clinical) in great detail and easy to understand format
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
aortic stenosis mitral stenosis valvular heart disease comparison
Reading File
Excellent! I now have very comprehensive content from the textbooks. I have enough to write a detailed, well-organized answer. Let me compose the complete guide:
Valvular Heart Disease: AS, AR, MS & MR — Complete Guide
Quick Overview
| AS (Aortic Stenosis) | AR (Aortic Regurgitation) | MS (Mitral Stenosis) | MR (Mitral Regurgitation) | |
|---|---|---|---|---|
| Valve | Aortic | Aortic | Mitral | Mitral |
| Type of lesion | Obstruction to outflow | Backflow into LV | Obstruction to inflow | Backflow into LA |
| Phase affected | Systole | Diastole | Diastole | Systole |
| Chamber overloaded | LV pressure overload | LV volume overload | LA pressure + RV pressure | LA + LV volume overload |
| Primary overload type | Pressure | Volume | Pressure | Volume |
1. AORTIC STENOSIS (AS)
Anatomy & Definition
The aortic valve sits between the LV and the aorta and opens during systole to allow ejection. In AS, this valve is narrowed (stenosed), creating obstruction to LV outflow.
Etiology
| Cause | Age Group | Notes |
|---|---|---|
| Degenerative calcification (most common in West) | >65 yrs (tricuspid valve), 50–65 yrs (bicuspid) | Calcium deposits on normal cusps causing obstruction |
| Congenital bicuspid aortic valve | Young–middle age | 1–2% of population; stenoses decades earlier |
| Rheumatic fever | Young adults (developing countries) | Commissural fusion; usually with AR |
In Western countries, degenerative calcific disease is now the dominant cause — the calcium is deposited on relatively normal cusps, unlike MS where it sits on an already stenotic valve. — Grainger & Allison's Diagnostic Radiology
Pathophysiology
Narrowed aortic valve
↓
LV must generate higher pressure to eject blood
↓
Chronic PRESSURE OVERLOAD on LV
↓
LV HYPERTROPHY (concentric) — wall thickens to normalize wall stress
↓
Reduced coronary flow reserve → ISCHEMIA (angina without CAD)
↓
Diastolic dysfunction (stiff ventricle) → elevated LVEDP
↓
Eventually: Systolic dysfunction + Heart failure
Key concept — The triad of symptoms in AS:
- 🔴 Angina — reduced coronary reserve from LVH + subendocardial ischemia
- 🔴 Syncope — exertional; fixed CO cannot compensate for fall in peripheral resistance
- 🔴 Dyspnea/Heart failure — elevated LVEDP → pulmonary congestion
Once symptoms appear, prognosis is grim without intervention: Angina → ~5 yr survival; Syncope → ~3 yr; Heart failure → ~2 yr. — Goldman-Cecil Medicine
Clinical Features
Symptoms: Angina, syncope (exertional), dyspnea, PND, orthopnea, HF
Signs:
| Sign | Description |
|---|---|
| Murmur | Ejection systolic murmur (ESM), crescendo-decrescendo, best heard at right upper sternal border (aortic area), radiates to carotids |
| Pulsus parvus et tardus | Slow-rising, low-amplitude carotid pulse |
| Heaving, sustained apex | Concentric LVH (not displaced) |
| S4 gallop | Atrial kick into stiff LV |
| Soft/absent A2 | Reduced aortic component of S2 |
| Paradoxical splitting of S2 | Prolonged LV systole delays A2 beyond P2 |
| Systolic thrill | Palpable in severe AS at right 2nd ICS |
Radiation mnemonic: "AS radiates to the Aorta (neck/carotids)"
ECG
- LVH (voltage criteria + strain pattern)
- Left atrial abnormality (P mitrale or LAA)
- LBBB (in advanced disease)
- Sinus rhythm usually maintained
CXR
- Rounded cardiac apex (LVH)
- Post-stenotic dilatation of ascending aorta
- Aortic valve calcification (best seen on lateral view)
- Normal heart size (LV is hypertrophied, not dilated — until late)
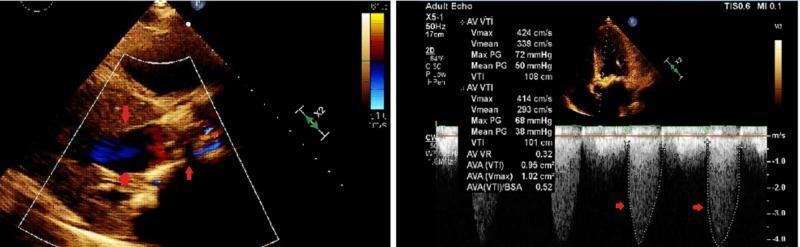
Echocardiography / Grading
| Severity | Peak Velocity | Mean Gradient | AVA |
|---|---|---|---|
| Mild | <3 m/s | <25 mmHg | >1.5 cm² |
| Moderate | 3–4 m/s | 25–40 mmHg | 1.0–1.5 cm² |
| Severe | >4 m/s | >40 mmHg | <1.0 cm² |
| Very severe | >5 m/s | >60 mmHg | <0.6 cm² |
Continuity equation is used to calculate AVA (based on the principle that flow proximal = flow at valve):
EOA = SV / VTI_AO — Grainger & Allison
Cardiac Catheterization
- Simultaneous LV and aortic pressure tracings show a gradient across the valve
- Used when echo is discordant (e.g., low-flow, low-gradient AS)
Management
| Condition | Treatment |
|---|---|
| Asymptomatic, mild-moderate | Watchful waiting, echo surveillance |
| Asymptomatic severe, EF <50% or other criteria | Intervention |
| Symptomatic severe AS | Surgical Aortic Valve Replacement (SAVR) OR TAVR (TAVI) |
| High surgical risk | TAVR preferred |
| Young, low surgical risk | SAVR preferred (more durable) |
| Low-gradient, low-flow AS | Dobutamine stress echo to confirm severity |
No medical therapy reliably slows progression. ACE inhibitors and statins were studied but did not prevent progression in trials. — Goldman-Cecil Medicine
2. AORTIC REGURGITATION (AR)
Definition
The aortic valve does not close properly in diastole, allowing blood to flow back from the aorta into the LV.
Etiology
Causes from valve leaflet disease:
- Bicuspid aortic valve
- Infective endocarditis (acute AR — leaflet destruction)
- Rheumatic fever (slow destruction of free edges of cusps; often with commissural fusion)
Causes from aortic root/wall disease:
- Marfan syndrome (annuloaortic ectasia)
- Aortic dissection (acute AR)
- Hypertension (annular dilatation)
- Ankylosing spondylitis, Takayasu arteritis
- Syphilitic aortitis
- Ehlers-Danlos syndrome, rheumatoid arthritis
Aortic regurgitation may result from disease of the cusps, or from disease of the aortic walls. Dilation of the ascending aorta or sinuses can result in AR even with anatomically normal valve leaflets. — Grainger & Allison / Textbook of Clinical Echocardiography
Pathophysiology
Chronic AR:
Backflow of blood from aorta → LV in diastole
↓
VOLUME OVERLOAD on LV
↓
LV dilates AND hypertrophies (eccentric hypertrophy)
↓
Compliance increases → end-diastolic pressure stays LOW initially
↓
Patient remains ASYMPTOMATIC for years
↓
Eventually: LV dysfunction, EF falls, HF symptoms appear
Acute AR:
Sudden massive backflow (e.g., endocarditis, dissection)
↓
LV has NOT had time to dilate/adapt
↓
Sudden ↑↑ LVEDP → premature closure of mitral valve
↓
FLASH PULMONARY EDEMA — medical/surgical emergency
Why diastolic BP is low in AR: The aorta empties back into the LV during diastole, lowering diastolic pressure → wide pulse pressure
Clinical Features
Symptoms (chronic): Often asymptomatic for years; then exertional dyspnea, orthopnea, PND; palpitations (hyperdynamic circulation); angina less common than AS
Signs (chronic):
| Sign | Description |
|---|---|
| Murmur | Early diastolic murmur (decrescendo), best heard at left sternal border (3rd ICS), with patient sitting forward + breath held in expiration |
| Wide pulse pressure | SBP ↑, DBP ↓ (e.g., 160/50 mmHg) |
| Corrigan's pulse | Water-hammer pulse — bounding, collapsing |
| Quincke's sign | Capillary pulsations in nail bed |
| De Musset's sign | Head nodding with each heartbeat |
| Traube's sign | Pistol-shot sound over femoral artery |
| Duroziez's sign | To-and-fro murmur over femoral artery |
| Hill's sign | Popliteal BP >20 mmHg higher than brachial BP |
| Apex beat | Displaced, forceful, hyperdynamic (volume overload → dilated LV) |
| Austin Flint murmur | Low-pitched mid-diastolic rumble at apex (AR jet causes vibration/premature closure of anterior mitral leaflet — mimics MS) |
Acute AR signs: Soft/absent diastolic murmur, tachycardia, pulmonary edema, hypotension — patient looks shocked
ECG
- LVH with strain
- Left axis deviation
- Prolonged PR interval (sometimes — associated with aortitis)
CXR
- Cardiomegaly — LV enlargement (dilated, not just hypertrophied)
- Dilated ascending aorta / aortic knuckle
- Pulmonary edema (in acute AR)
- In acute AR: Normal heart size BUT pulmonary edema (no time for LV to dilate)
Echocardiography
Grading of AR severity:
| Severity | Vena Contracta | Pressure Half-Time | RF |
|---|---|---|---|
| Mild | <3 mm | >500 ms | <30% |
| Moderate | 3–6 mm | 200–500 ms | 30–50% |
| Severe | >6 mm | <200 ms | >50% |
A jet width ≥6 mm is related to severe regurgitation with 95% sensitivity and 90% specificity; width <3 mm = mild. — Grainger & Allison
Also: Colour Doppler shows regurgitant jet in LVOT; aortic flow reversal on pulsed Doppler in descending aorta = severe AR
Management
| Condition | Treatment |
|---|---|
| Chronic AR, asymptomatic | Serial echo surveillance; vasodilators (nifedipine, ACEi) if hypertensive |
| Symptomatic severe AR | Surgical aortic valve replacement (SAVR) |
| Asymptomatic, EF <50% or ESD >50mm | Surgery |
| Acute severe AR | Surgical emergency (IABP contraindicated) |
The best indicator for surgery is progressive increase in regurgitation together with deterioration of ventricular function. — Grainger & Allison
3. MITRAL STENOSIS (MS)
Definition
The mitral valve (between LA and LV) does not open fully in diastole, obstructing LV inflow.
Etiology
| Cause | Notes |
|---|---|
| Rheumatic fever | By far the most common cause; accounts for ~99% worldwide |
| Congenital MS | Rare |
| Mitral annular calcification | Elderly |
| Ball-valve thrombus, LA myxoma | Functional MS |
| Cor triatriatum | Membrane divides LA, mimics MS |
| Radiation | Prior chest radiotherapy |
MS is highly prevalent in developing countries due to its association with rheumatic fever. Isolated MS is twice as common in women as in men; 40% of all rheumatic heart disease. — Grainger & Allison
Pathophysiology
Rheumatic fever → leaflet thickening, commissural fusion, subvalvular changes
↓
Narrowed mitral orifice (fish-mouth appearance)
↓
Obstruction to LV INFLOW during diastole
↓
Left Atrial (LA) pressure rises
↓
LA DILATES → Atrial fibrillation (very common)
↓
Pulmonary venous hypertension → Dyspnea, haemoptysis, pulmonary edema
↓
Reactive pulmonary arterial hypertension (Eisenmenger-like, late)
↓
RV pressure overload → RV hypertrophy/failure + Tricuspid Regurgitation
↓
LV is UNDERFILLED (protected) → LV small, function often PRESERVED
Key concept: The LV is protected (and may even be underfilled) in MS. Unlike other valvular diseases, LV function is often normal in MS.
Normal vs Stenotic Mitral Valve Area
| Status | MVA | Notes |
|---|---|---|
| Normal | 4–6 cm² | No gradient |
| Gradient develops | <2.0 cm² | Mild stenosis |
| Moderate | 1.0–1.5 cm² | Gradient 5–10 mmHg |
| Severe | <1.0 cm² | Gradient >10 mmHg |
Clinical Features
Symptoms:
- Dyspnea (exertional → orthopnea → PND)
- Palpitations (AF is very common due to LA dilatation)
- Haemoptysis — pulmonary venous hypertension or rupture of bronchial veins
- Systemic embolism (from LA thrombus — most common in LAA, especially with AF)
- RV failure symptoms (oedema, ascites, JVP ↑) — late
Signs:
| Sign | Description |
|---|---|
| Murmur | Mid-diastolic murmur (low-pitched, rumbling), best heard at apex with bell of stethoscope in left lateral decubitus position |
| Opening snap (OS) | After S2; closer to S2 = more severe (higher LA pressure closes valve sooner in diastole) |
| Loud S1 | Abrupt closure of thickened (but pliable) valve leaflets |
| Loud P2 | Pulmonary hypertension |
| Presystolic accentuation | Murmur louder at end of diastole (atrial contraction); disappears with AF |
| Tapping apex | Palpable S1, non-displaced (LV not enlarged) |
| Malar flush | Pinkish discolouration of cheeks (pulmonary hypertension + low CO) |
| Graham Steell murmur | Early diastolic murmur (pulmonary regurgitation from pulmonary HTN) |
| Right ventricular heave | Pulmonary hypertension → RV hypertrophy |
S2–OS interval rule:
- Short S2-OS gap → severe MS (high LA pressure)
- Long S2-OS gap → mild MS
ECG
- P mitrale (bifid P wave in lead II, biphasic in V1) — LA dilatation
- Atrial fibrillation (very common)
- RVH pattern (tall R in V1, right axis deviation) — pulmonary hypertension
- Normal LV complexes
CXR
- Straight left heart border (LA appendage enlargement)
- Double shadow on right (enlarged LA behind RA)
- Pulmonary congestion (upper lobe diversion → Kerley B lines → pulmonary oedema)
- Mitral valve calcification
- Carinal angle widening (main bronchi splayed)
- Normal aortic knuckle (small LV, low CO)
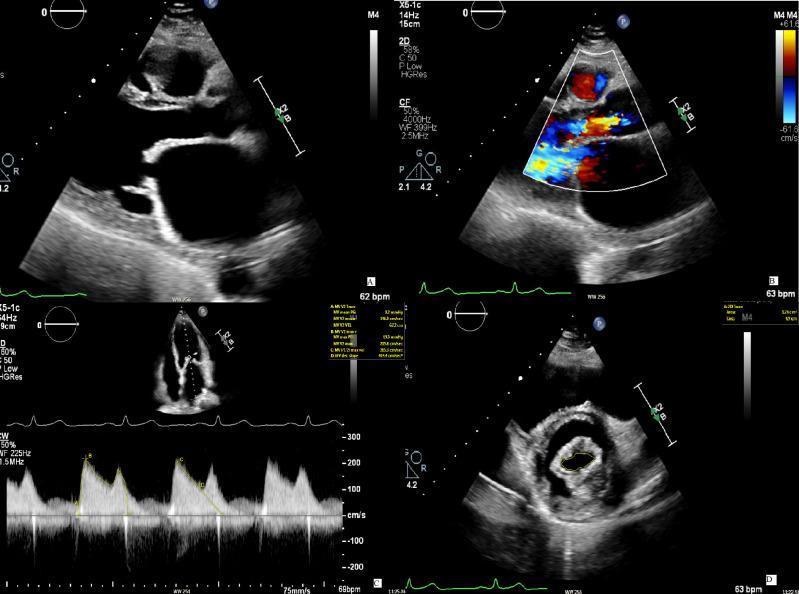
Echocardiography
- Hockey-stick deformity of anterior mitral leaflet (rheumatic tethering at tips with mid-leaflet pliability)
- Doming of leaflets in diastole
- Wilkins score (0–16): grades leaflet mobility, thickening, calcification, subvalvular involvement → guides suitability for balloon valvuloplasty (BMV ideal if score ≤8)
- Enlarged LA; can detect LA thrombus (especially in LAA on TEE)
- Pulmonary hypertension can be estimated
A pliable, non-calcified valve is suitable for balloon valvuloplasty or commissurotomy; a calcified, fibrotic valve with subvalvular fusion may preclude it. — Grainger & Allison
Management
| Condition | Treatment |
|---|---|
| AF + MS | Rate control (β-blocker, digoxin); Anticoagulation mandatory (warfarin, target INR 2–3) |
| Mild MS, asymptomatic | Surveillance |
| Symptomatic MS (MVA ≤1.5 cm²), pliable valve | Percutaneous Balloon Mitral Valvuloplasty (PBMV / BMV) — first choice |
| Symptomatic MS, calcified/unsuitable valve | Surgical commissurotomy or valve replacement |
| Severe pulmonary hypertension | Intervention earlier to prevent irreversible RV failure |
4. MITRAL REGURGITATION (MR)
Definition
The mitral valve does not close properly in systole, allowing blood to flow backwards from LV into the LA.
Etiology — Primary (Organic) vs Secondary (Functional)
Primary MR (valve itself is abnormal):
| Cause | Notes |
|---|---|
| Mitral valve prolapse (MVP) | Most common cause in developed world; billowing of leaflet(s) into LA |
| Ruptured chordae tendineae | MVP, endocarditis, trauma, spontaneous |
| Infective endocarditis | Leaflet destruction |
| Rheumatic fever | Less common for MR than MS |
| Connective tissue disorders | Marfan, Ehlers-Danlos |
Secondary (Functional) MR (valve leaflets structurally normal):
| Cause | Notes |
|---|---|
| Dilated cardiomyopathy | LV dilatation → papillary muscles displaced apically → leaflets tethered, poor coaptation |
| Ischemic MR | Papillary muscle dysfunction/rupture after MI (posterior wall MI → posteromedial PM more common) |
| Hypertrophic cardiomyopathy | SAM (systolic anterior motion) of anterior MV leaflet |
In functional MR, the valve itself is relatively normal but LV dilatation results in apical displacement of papillary muscles, pulling leaflets toward the apex — poor coaptation → central MR jet. — Harrison's Principles of Internal Medicine 22e
Pathophysiology
Chronic MR:
Backflow of blood from LV → LA during systole
↓
VOLUME OVERLOAD on BOTH LA and LV
↓
LA dilates (compliance increases → LA pressure initially low)
↓
LV dilates AND hypertrophies (eccentric hypertrophy)
↓
LV ejection fraction appears ARTIFICIALLY HIGH
(part of every beat goes backward into LA)
↓
Patient may be asymptomatic for YEARS
↓
Eventually: LV dysfunction, EF falls, AF, pulmonary hypertension → HF symptoms
Acute MR (e.g., chordal rupture, papillary muscle rupture post-MI):
Sudden massive backflow into LA
↓
LA has NOT adapted → LA pressure spikes rapidly
↓
ACUTE PULMONARY EDEMA — emergency
↓
LV size may still be NORMAL (no time for dilatation)
Important concept: Because LV ejects into the low-resistance LA, EF is preserved (even supranormal) early in MR. An EF of 55% in severe MR may actually represent significant LV dysfunction. Surgery should not wait until EF <50%.
Clinical Features
Symptoms: Dyspnea, orthopnea, PND, palpitations (AF), fatigue, reduced exercise tolerance; in acute MR — sudden pulmonary edema
Signs:
| Sign | Description |
|---|---|
| Murmur | Pan-systolic (holosystolic) murmur, best heard at apex, radiates to axilla (sometimes to spine/base in posterior leaflet prolapse) |
| Soft/absent S1 | Incomplete valve closure |
| S3 gallop | Volume overload — rapid early diastolic filling |
| Displaced apex | LV volume overload → LV dilation → apex displaced laterally and inferiorly |
| Thrill at apex | In severe MR |
| Loud P2 | If pulmonary hypertension develops |
MVP special signs:
- Mid-systolic click (hallmark)
- Late systolic murmur
- Click moves earlier with standing/Valsalva (reduced preload); later with squatting
ECG
- AF (very common)
- Left atrial abnormality (P mitrale or AF)
- LVH (volume overload type)
- Ischemic changes (if secondary MR from CAD)
CXR
- Cardiomegaly (LV + LA enlargement)
- Enlarged LA (double right border, elevated left main bronchus)
- Displaced apex (downward and outward)
- Pulmonary venous congestion
- In acute MR: Normal heart size + pulmonary edema
Echocardiography
Grading of MR severity:
| Severity | Vena Contracta | EROA | Regurgitant Vol |
|---|---|---|---|
| Mild | <3 mm | <20 mm² | <30 mL |
| Moderate | 3–6.9 mm | 20–39 mm² | 30–59 mL |
| Severe | ≥7 mm | ≥40 mm² | ≥60 mL |
- PISA (Proximal Isovelocity Surface Area) method used to quantify EROA
- Pulmonary vein flow reversal = severe MR
- MVP: posterior leaflet prolapse (most common) shows eccentric anteriorly-directed jet; anterior leaflet prolapse → posterior jet
Management
| Condition | Treatment |
|---|---|
| Symptomatic severe MR, normal LV | Mitral valve repair (preferred over replacement) or replacement |
| Asymptomatic severe MR + EF 30–60% or ESD >40mm | Surgery |
| Secondary/functional MR | Treat underlying cause (HF meds: ACEi, β-blockers, cardiac resynchronization therapy) |
| High-risk severe primary MR | MitraClip (edge-to-edge transcatheter repair — EVEREST II trial) |
| Acute MR | Urgent surgical repair; temporize with vasodilators (nitroprusside) to reduce afterload; IABP can bridge to surgery |
COMPREHENSIVE COMPARISON TABLE
| Feature | AS | AR | MS | MR |
|---|---|---|---|---|
| Valve | Aortic | Aortic | Mitral | Mitral |
| Event | Obstruction | Regurgitation | Obstruction | Regurgitation |
| Timing | Systole | Diastole | Diastole | Systole |
| Primary overload | LV Pressure | LV Volume | LA Pressure → RV Pressure | LA + LV Volume |
| LV response | Concentric hypertrophy | Eccentric hypertrophy | Normal (underfilled) | Eccentric hypertrophy |
| LV size | Normal (until late) | Enlarged | Normal or small | Enlarged |
| LA | Normal (until late HF) | Normal | Markedly enlarged | Enlarged |
| Most common cause | Calcific (elderly), Bicuspid (young) | Bicuspid, Marfan, Endocarditis | Rheumatic fever | MVP (developed), Rheumatic (developing) |
| Murmur type | Ejection systolic (crescendo-decrescendo) | Early diastolic (decrescendo) | Mid-diastolic (rumbling) | Pan-systolic (constant) |
| Murmur location | Right upper sternal border | Left sternal border (3rd ICS) | Apex | Apex |
| Radiation | Carotids (neck) | Does not radiate | None | Axilla |
| Added sound | S4, absent A2, systolic thrill | Austin Flint murmur | Opening snap, loud S1 | S3 gallop, soft S1 |
| Apex character | Heaving, non-displaced (LVH) | Displaced, forceful, hyperdynamic | Tapping (non-displaced) | Displaced, volume overloaded |
| Pulse | Slow-rising, low amplitude (parvus et tardus) | Collapsing/bounding (Corrigan's) | Normal or low-volume | Normal |
| Pulse pressure | Narrow | Wide | Normal | Normal |
| ECG | LVH ± LBBB | LVH | P mitrale / AF | LA abnormality / AF |
| CXR heart shape | Normal size (late: enlarged, rounded apex) | Cardiomegaly (LV dominant) | Enlarged LA, "mitral configuration" | Cardiomegaly (LV+LA) |
| CXR aorta | Post-stenotic ascending aortic dilatation | Prominent/enlarged aorta | Normal/small aortic knuckle | Normal |
| Pulmonary changes on CXR | Late pulmonary edema | Late pulmonary edema | Upper lobe diversion, Kerley B, pulmonary edema | Upper lobe diversion, pulmonary edema |
| AF risk | Low (until late) | Low | Very high (LA markedly enlarged) | High |
| Embolism risk | Low | Low | High (LA thrombus + AF) | Moderate |
| Key echo finding | Restricted leaflet opening, Doppler gradient, AVA <1 cm² (severe) | Wide jet in LVOT, vena contracta, PHT <200ms (severe) | Hockey-stick leaflet, MVA <1 cm² (severe), PHT | Regurgitant jet into LA, vena contracta ≥7mm (severe), EROA |
| Intervention of choice | SAVR or TAVR | SAVR | Balloon Mitral Valvuloplasty (BMV) | Mitral valve repair (preferred) |
| Unique peripheral signs | None specific | Corrigan, Quincke, Hill, Duroziez, Traube, De Musset | Malar flush | None specific |
| Key distinguishing symptom | Syncope (exertional) | Long asymptomatic phase | Haemoptysis | Pulmonary edema (acute) |
PATHOPHYSIOLOGY AT A GLANCE
MURMUR MEMORY TRICKS
| Valve Lesion | Murmur | Memory |
|---|---|---|
| AS | Ejection systolic, R sternal edge → neck | "Aortic → Above (neck)" |
| AR | Early diastolic, L sternal border | "Aortic Regurg after heart closes" |
| MS | Mid-diastolic rumble, apex | "Mitral Stenosis = Muffled Slow diastolic" |
| MR | Pan-systolic, apex → axilla | "Mitral Regurg = Mask all of systole" |
EFFECT ON LV EF — IMPORTANT CLINICAL TRAP
| Lesion | LV EF Early | LV EF Late | Clinical trap |
|---|---|---|---|
| AS | Preserved (hypertrophy normalizes stress) | Falls when compensation fails | EF <50% = intervene even if asymptomatic |
| AR | Preserved (volume accommodated by dilation) | Falls with decompensation | EF <50% or ESD >50mm = intervene |
| MS | Often normal (LV underfilled but intact) | Can fall if long-standing with secondary TR | Good LV function masks severity |
| MR | Artificially elevated (blood ejects into low-resistance LA) | "Normal" EF of 55% may = LV dysfunction | EF <60% in severe MR = significant dysfunction; intervene |
SUMMARY MNEMONICS
AS — "SAD" triad of symptoms
- Syncope
- Angina
- Dyspnoea (heart failure)
AR — "ADMIRAL" peripheral signs
- Austin Flint murmur
- Duroziez's sign
- Murmur (early diastolic)
- Increase in pulse pressure
- Reduplicated bounding pulse (Corrigan's)
- All peripheral vessels pulsatile
- Lateral displacement of apex (dilated LV)
MS — "PAMELA"
- Pmitrale (ECG), Pulmonary hypertension
- AF (very common)
- Mid-diastolic murmur + Malar flush
- Embolism (systemic, from LA thrombus)
- Loud S1 + Left atrial enlargement
- Accessory sound = Opening snap
MR — "LASS"
- Lateral displacement of apex
- AF + Axilla radiation of murmur
- Soft S1 + S3 gallop
- Systolic (pan-systolic) murmur
Sources: Grainger & Allison's Diagnostic Radiology; Goldman-Cecil Medicine (Chapter 60, Table 60-1); Harrison's Principles of Internal Medicine 22e; Textbook of Clinical Echocardiography; Braunwald's Heart Disease.
This is a shared conversation. Sign in to Orris to start your own chat.