Pcos according to bereks and Novak gyncelogy
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content from Berek & Novak's Gynecology. Let me compile a thorough, well-organized summary.
Polycystic Ovary Syndrome (PCOS)
— Berek & Novak's Gynecology
Definition & Epidemiology
PCOS is one of the most common endocrine disorders in women of reproductive age, affecting 5–10% of women worldwide. It is familial, inherited as a complex polygenic/multifactorial trait. The syndrome is characterized by a combination of:
- Hyperandrogenism (HA) — clinical or biochemical
- Chronic anovulation
- Polycystic ovarian (PCO) morphology
- Frequent association with insulin resistance (IR) and obesity
It is the most common cause of HA, hirsutism, and anovulatory infertility in developed countries. The classic description of amenorrhea with bilateral polycystic ovaries and obesity was first made by Stein and Leventhal in 1935.
Diagnostic Criteria
NIH 1990 Criteria (both required):
- Clinical and/or biochemical hyperandrogenism
- Chronic anovulation (Exclusion of other disorders)
Rotterdam 2003 Criteria (2 of 3 required):
- Menstrual cycle anomalies (amenorrhea, oligomenorrhea)
- Clinical and/or biochemical hyperandrogenism
- Polycystic ovaries on ultrasound
This gives rise to 4 phenotypes:
- HA + ovarian dysfunction + PCO morphology
- HA + ovarian dysfunction
- HA + PCO morphology
- Ovarian dysfunction + PCO morphology
The Rotterdam criteria are ratified by the Endocrine Society (2013) for adult women. In adolescents, diagnosis requires persistent anovulation + clinical/biochemical HA.
Exclusions required: Non-classic adrenal hyperplasia, adrenal/ovarian neoplasm, Cushing syndrome, hypo/hypergonadotropic disorders, hyperprolactinemia, thyroid disease.
Clinical Features
Hyperandrogenism
- Hirsutism: ~70% of PCOS patients in the US (10–20% in Japan — due to genetic differences in skin 5α-reductase activity)
- Acne
- Male-pattern alopecia
Menstrual Dysfunction
- Ranges from amenorrhea to oligomenorrhea (from anovulation/oligo-ovulation)
- Classically lifelong, with abnormal menses from puberty
- May emerge in adulthood with onset of obesity
Ultrasound (PCO Morphology)
- ≥20 follicles in either ovary, each measuring 2–9 mm in diameter and/or ovarian volume >10 mL
- A single ovary meeting criteria is sufficient
- PCO appearance occurs in ~23% of reproductive-age women, but only 5–10% have clinical PCOS
Obesity
- Present in >50% of PCOS patients
- Usually central (android) obesity; higher waist-to-hip ratio signals increased CV risk
- Geographic variation: 20% (Spain) to 69% (US)
Insulin Resistance & Metabolic Features
- IR and compensatory hyperinsulinemia are hallmarks
- ~1/3 of obese PCOS patients have impaired glucose tolerance (IGT)
- 7.5–10% have type 2 diabetes mellitus
- Rates are mildly increased even in nonobese PCOS women
- PCOS confers a 4–7× increased risk of type 2 DM vs. age-matched controls
- Screening: fasting glucose 100–125 mg/dL = IFG; 2-h 75g OGTT ≥200 mg/dL = DM
Pathophysiology
Ovarian Compartment
- Excess ovarian androgen production (androgens → estrogens via aromatization in adipose tissue)
- Increased LH activity stimulates theca cell androgen production
- FSH levels are relatively low — insufficient for follicle maturation and ovulation
Estrogen Metabolism
- Elevated E1 (estrone) from peripheral aromatization of androstenedione
- E2 (estradiol) remains at follicular phase levels
- In obesity: decreased 2-hydroxylation → more active 16-hydroxyestrogens (estriol)
- Chronic hyperestrogenic state, with reversed E1:E2 ratio, unopposed by progesterone
Hypothalamic-Pituitary Axis
- Increased GnRH pulse frequency → increased LH pulse frequency
- Elevated LH and elevated LH:FSH ratio
- FSH not elevated — suppressed by elevated estrogens and follicular inhibin
- ~25% of PCOS patients have mildly elevated prolactin (abnormal estrogen feedback to pituitary)
Genetics
- Complex multigenic disorder (gene-environment interaction)
- GWAS candidate genes relate to: insulin signaling, FSH receptor, insulin receptor, steroid hormone function, type 2 DM, IL-6, GABA-A receptors, and endocytosis
- Confirmed loci include: YAP1 (11q22.1), THADA (2p21), FSHB (11p14.1), ERBB4 (2q34)
- Other candidates: CYP11A, Calpain 10, IRS-1, IRS-2, SHBG, TCF7L2, FBN3 (D19S884)
Long-Term Risks
| System | Risk |
|---|---|
| Metabolic | Type 2 DM, metabolic syndrome, dyslipidemia |
| Cardiovascular | Hypertension, CAD risk |
| Reproductive | Anovulatory infertility, endometrial hyperplasia/carcinoma (from chronic unopposed estrogen) |
| Psychological | Depression (35–40% vs. 10.7% in controls), anxiety, eating disorders |
| Hepatic | Non-alcoholic steatohepatitis (NASH) |
Treatment
1. Lifestyle Modification (First-Line for Obese/Overweight)
- Weight loss of 5–10% recommended as initial goal
- Reduces free/total testosterone, adrenal androgens; improves SHBG, fasting insulin, glucose tolerance, lipids, and menstrual function
- A 5–7% weight loss over 6 months can restore ovulation in >75% of women
- Structured exercise reduces IR significantly
- Components: dietary management, structured exercise, behavior modification, stress management
- Dietary target: <30% calories from fat, <10% from saturated fat, increased fiber and whole grains; caloric reduction of 500–1,000 kcal/day
2. Oral Contraceptives (OCs)
Combination OCs reduce hair growth in nearly two-thirds of hirsute patients by:
- Progestin → suppresses LH → reduces ovarian androgen production
- Estrogen → increases SHBG → reduces free androgen levels
- Progestin → reduces adrenal androgen precursors
- Reduces LH-driven thecal androgen secretion
Preferred formulations: Progestins with low androgenicity (desogestrel, norgestimate, gestodene) or antiandrogenic activity (drospirenone, cyproterone acetate)
3. Antiandrogens
| Drug | Mechanism | Notes |
|---|---|---|
| Spironolactone | Androgen receptor blocker; also inhibits 5α-reductase | 50–200 mg/day; first-line antiandrogen in US |
| Cyproterone acetate | Potent progestogenic antiandrogen | Used with ethinyl estradiol in Europe; not FDA-approved |
| Flutamide | Pure nonsteroidal antiandrogen; inhibits nuclear androgen binding | Rare but severe hepatotoxicity — not first-line per Endocrine Society 2008 |
| Finasteride | Specific 5α-reductase type 2 inhibitor | 5 mg/day; comparable efficacy to spironolactone |
All antiandrogens must be combined with contraception due to risk of feminization of a male fetus.
4. Insulin Sensitizers
- Metformin: reduces IR; may restore ovulatory function; also used for hirsutism
- Combination of metformin + flutamide + drospirenone OC effective in hyperinsulinemic adolescent PCOS
5. GnRH Agonists
- Suppress ovarian androgen production
- Used for severe cases; typically add-back estrogen to prevent bone loss
6. Glucocorticoids
- Used for adrenal component of androgen excess
7. Pharmacologic Anti-obesity Agents
- Orlistat, sibutramine, rimonabant — adjuncts when lifestyle fails
- Bariatric surgery: indicated when BMI >40 kg/m² or >35 kg/m² with high-risk comorbidities, after failure of other treatments
8. Endometrial Protection
- Progestational interruption of unopposed estrogen is mandatory in all women with significant ovulatory dysfunction — via OCs, cyclic progestins, or ovulation induction
Diagnostic Algorithm
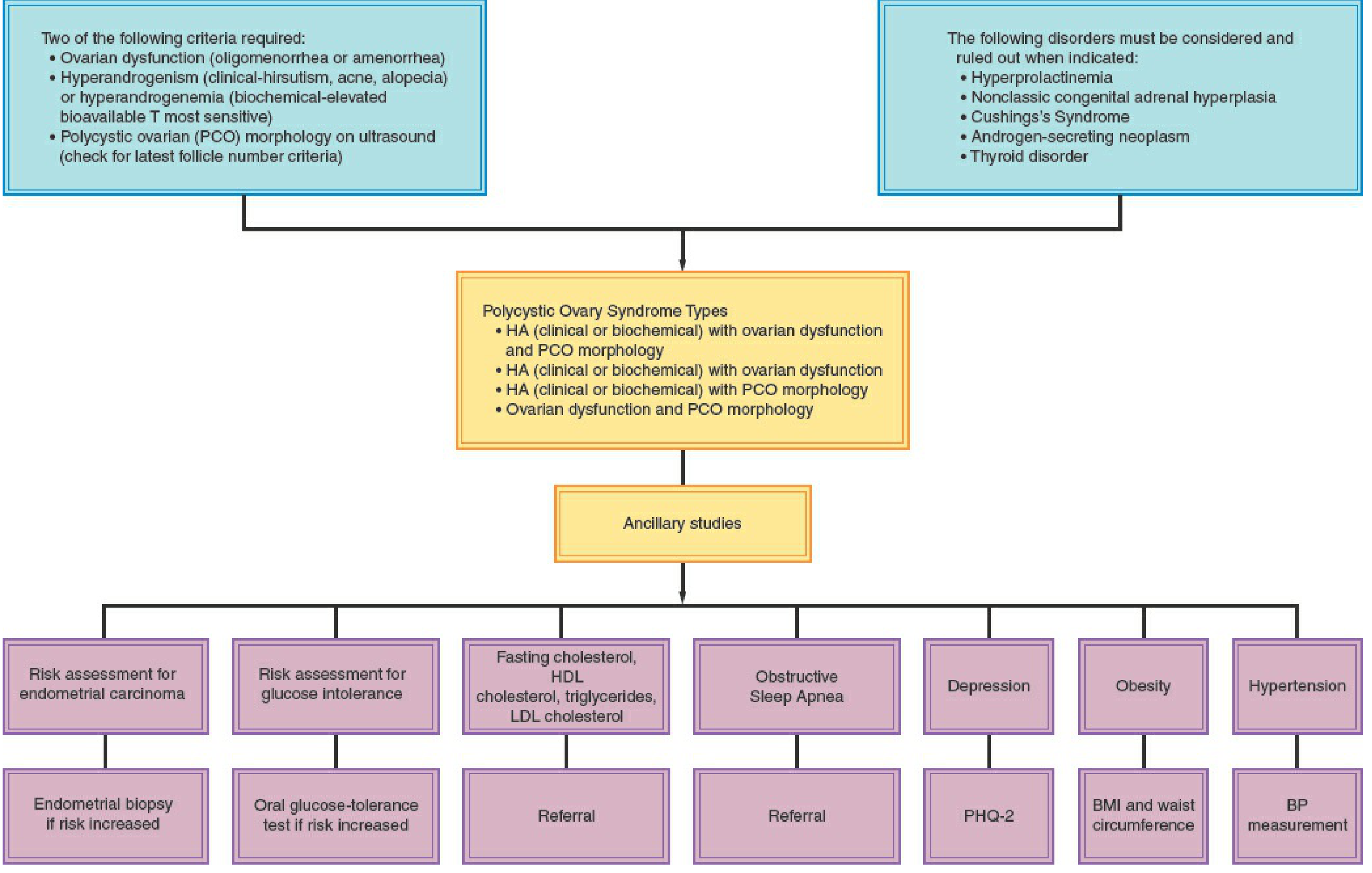
Figure 35-4 — Diagnostic algorithm for PCOS (Berek & Novak's Gynecology)
Psychological Assessment
The PHQ-2 questionnaire is recommended as a simple initial screen:
- Little interest or pleasure in doing things (0–3)
- Feeling down, depressed, or hopeless (0–3)
Lifestyle management improves quality of life and reduces depression in obese/overweight PCOS patients.
Source: Berek & Novak's Gynecology (ISBN 9781496380357), Chapter 35, pp. 1919–1940
This is a shared conversation. Sign in to Orris to start your own chat.