Necrotizing enterocolitis in pediatrics
Reading File
Finding Sources
Searching PubMed
"necrotizing enterocolitis"[MeSH Terms] AND infant
Searching Images
necrotizing enterocolitis NEC pediatric radiology pathology
Reading File
Reading File
Reading File
Searching PubMed
"necrotizing enterocolitis"[MeSH Terms] AND "infant, premature"[MeSH Terms]
Reading File
Reading File
Searching PubMed
"necrotizing enterocolitis" treatment prevention
Loading Image
Loading Image
Loading Image
I now have comprehensive content from multiple authoritative sources. Here is the complete, integrated review:
Necrotizing Enterocolitis (NEC) in Pediatrics
Overview
Necrotizing enterocolitis is the most common and lethal gastrointestinal emergency in the neonatal period. It is a severe inflammatory disease of the small and large intestine, occurring in approximately 1 in 1,000 live births overall and in 4–13% of very-low-birth-weight (VLBW) infants (<1,500 g). It affects 1–5% of all NICU admissions. Mortality ranges from 10–30% overall and approaches 50% in surgical cases. — Creasy & Resnik's Maternal-Fetal Medicine, Current Surgical Therapy 14e, Schwartz's Principles of Surgery 11e
Epidemiology & Risk Factors
- Prematurity and enteral feeding are the only consistent epidemiologic precursors
- Peak incidence at ~32 weeks corrected gestational age; onset has an inverse relationship with gestational age
- Higher risk in infants <1,500 g
- Other associated risk factors: birth asphyxia, umbilical artery catheterization, patent ductus arteriosus (PDA), cyanotic congenital heart disease, bacterial sepsis, maternal cocaine abuse
- NEC occurs in episodic cluster waves abrogated by infection control measures, and almost exclusively in enterally fed infants
- Antenatal corticosteroids are protective; antenatal indomethacin tocolysis may be associated with early-onset NEC in some studies (evidence mixed) — Creasy & Resnik's
Pathogenesis
The exact mechanism is multifactorial and incompletely understood, involving a triad of:
-
Intestinal immaturity — The premature intestinal epithelium over-expresses Toll-like receptor 4 (TLR4), which plays a role in normal gut development. When colonizing bacteria activate TLR4 in the NICU, an exaggerated proinflammatory response is triggered, increasing mucosal permeability via enterocyte apoptosis and tight junction disruption.
-
Microbial dysbiosis — No single pathogen is causative. Common isolates from blood, peritoneal fluid, and stool in advanced NEC include E. coli, Enterobacter, Klebsiella, and coagulase-negative Staphylococcus. Outbreaks have been linked to Enterobacter sakazakii-contaminated formula. Antibiotic exposure (antenatal and postnatal) increases risk by disrupting the gut microbiome.
-
Ischemia–inflammation cascade — Mucosal barrier failure allows bacterial translumination, triggering a vicious cycle: inflammation → necrosis → further bacterial entry → systemic sepsis and shock. Circulating proinflammatory cytokines drive multiorgan dysfunction.
Gas-producing enteric bacteria invade the ischemic mucosa, producing the hallmark finding of pneumatosis intestinalis (intramural gas). — Schwartz's Principles of Surgery 11e, Robbins & Cotran Pathologic Basis of Disease
Anatomy of Involvement
NEC most commonly involves the terminal ileum, followed by the cecum and right colon, though any segment can be affected. Gross findings include bowel distension, patchy thinning, pneumatosis, gangrene, and frank perforation. Microscopically: coagulative (bland) infarct, full-thickness necrosis, bacterial colonization, submucosal gas bubbles, and early reparative granulation tissue/fibrosis. — Robbins & Cotran, Robbins & Kumar Basic Pathology
Gross pathology (postmortem exam):
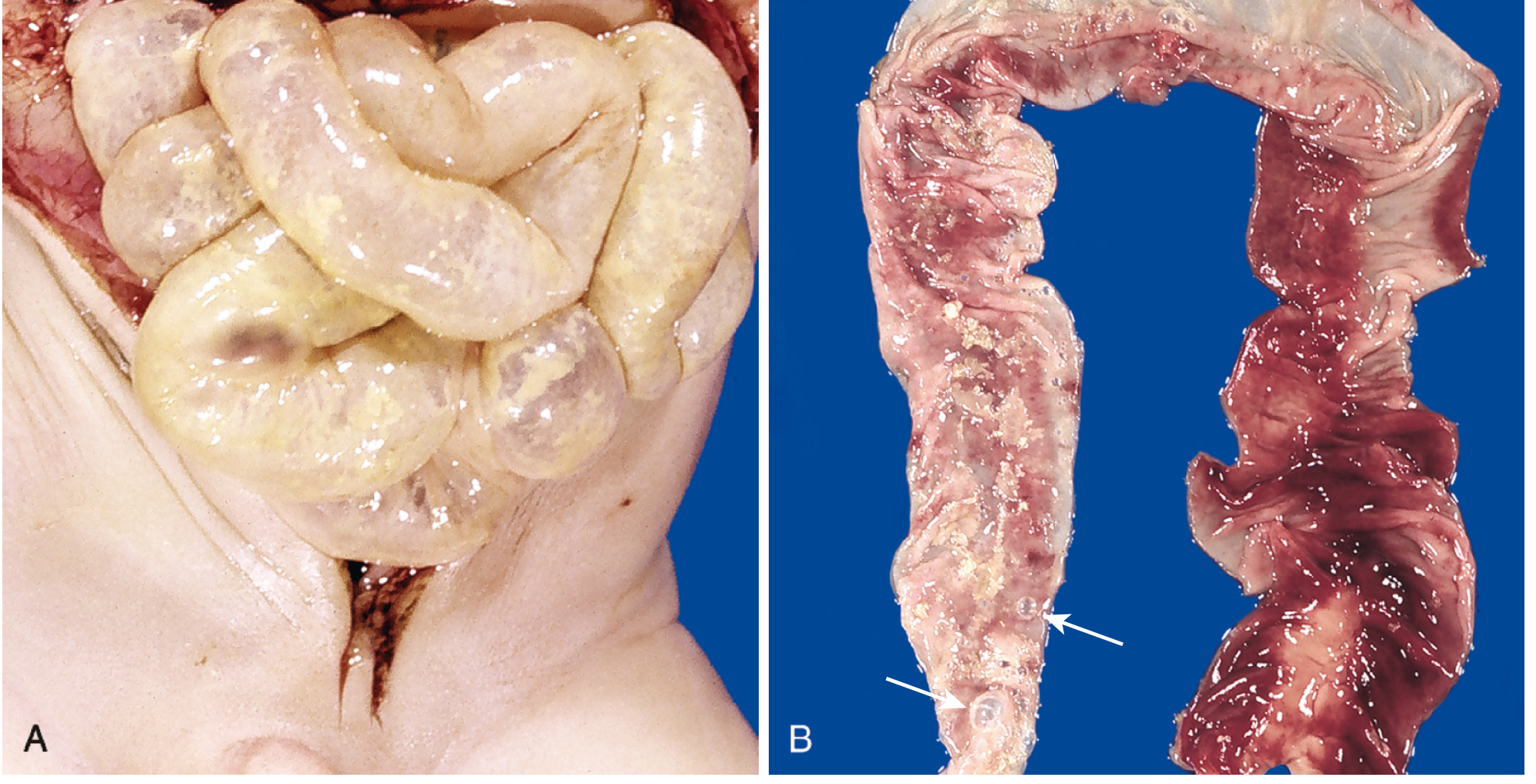
Fig. 10.8 — (A) Entire small bowel markedly distended with perilously thin wall suggesting impending perforation. (B) Congested ileum with hemorrhagic infarction, transmural necrosis, and submucosal gas bubbles (pneumatosis intestinalis, arrows). — Robbins & Cotran Pathologic Basis of Disease
Clinical Manifestations & Bell Staging
The clinical picture progresses through Bell's staging criteria:
| Stage | Category | Systemic Signs | GI Signs | Radiographic Findings | Treatment |
|---|---|---|---|---|---|
| IA | Suspected NEC | Temperature instability, apnea, bradycardia, lethargy | Gastric residuals, mild distension, emesis | Normal or intestinal dilation, mild ileus | NPO, bowel rest, rule out sepsis |
| IB | Suspected NEC | Same | Bright red blood per rectum | Same | Same |
| IIA | Definite NEC (mildly ill) | Same as above | Absent bowel sounds ± tenderness | Pneumatosis intestinalis | NPO, IV antibiotics 7–10 days |
| IIB | Definite NEC (moderately ill) | Mild metabolic acidosis, thrombocytopenia | ± Abdominal wall cellulitis, palpable mass | Same + portal venous gas, ascites | NPO, antibiotics 14 days |
| IIIA | Advanced NEC (severely ill, intact bowel) | Hypotension, acidosis, DIC, anuria | Peritonitis, marked distension | Persistent findings | IV fluids, vasopressors, antibiotics, surgical consult |
| IIIB | Advanced NEC (perforated bowel) | Same + deterioration | Same | Pneumoperitoneum | Surgery |
Source: Schwartz's Principles of Surgery 11e, Current Surgical Therapy 14e
Investigations
Laboratory
- CBC: leukocytosis or leukopenia, increased bands, thrombocytopenia (concerning finding)
- Blood gas: metabolic acidosis
- Electrolytes, BUN/Cr (renal dysfunction in advanced disease)
- Blood culture (bacteremia common in advanced stages)
- CRP, procalcitonin
Radiology
Plain abdominal X-ray (AP + left lateral decubitus) remains first-line:
- Pneumatosis intestinalis — pathognomonic; linear or bubbly intramural gas
- Bowel dilation / ileus
- Portal venous gas — branching lucencies in the liver; indicates severe disease
- Pneumoperitoneum — free air (football sign, Rigler's sign); indicates perforation and surgical emergency
- Fixed loop — persistent dilated loop on serial films; suggests localized necrosis/perforation
Abdominal ultrasound is more sensitive than plain films for pneumatosis and portal venous gas but its role in surgical decision-making remains under evaluation.
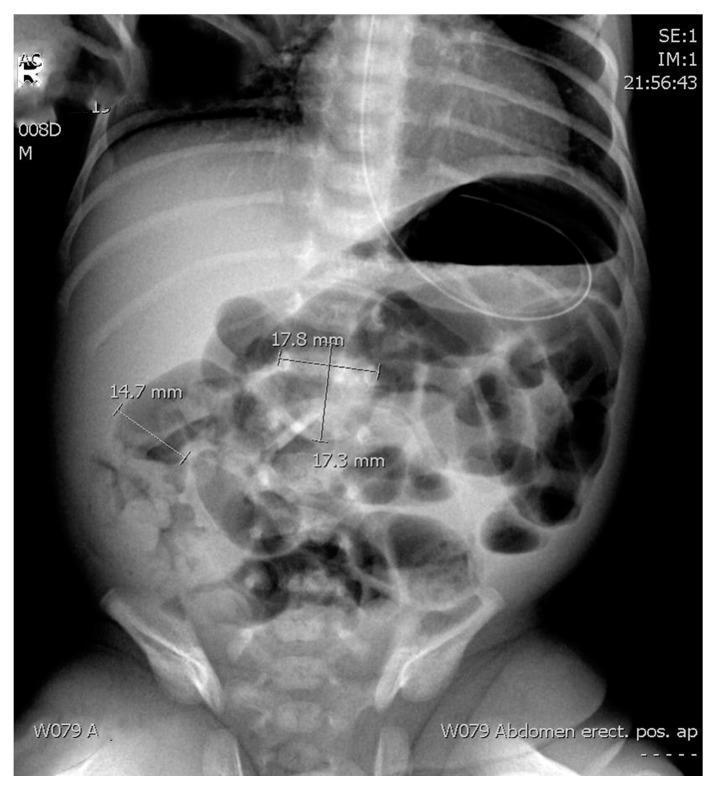
Neonatal abdominal X-ray: generalized bowel distension, dilated loops (14.7–17.8 mm), and pneumatosis intestinalis. An enteric feeding tube is in situ.
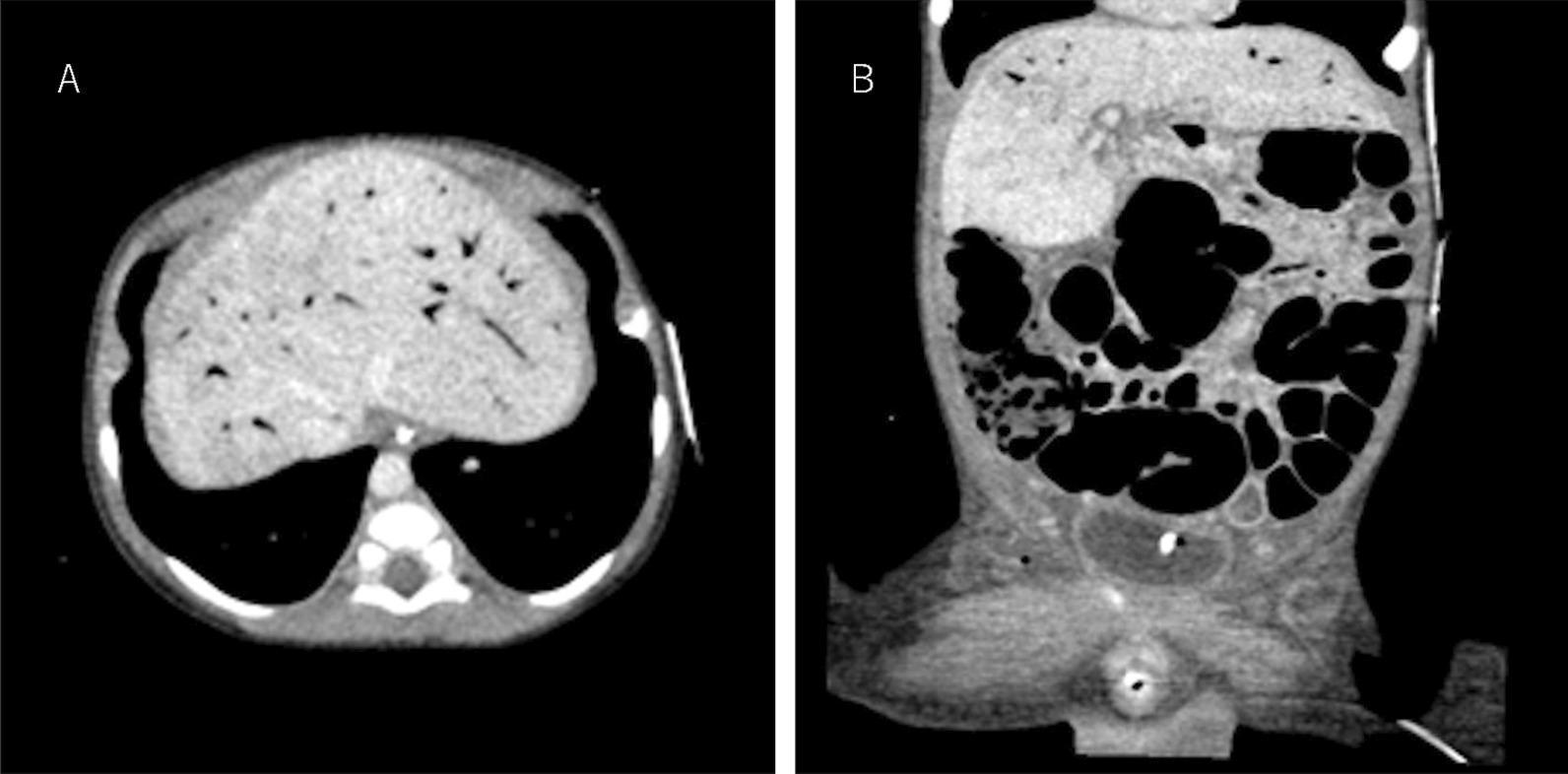
CT findings of advanced NEC: (A) portal venous gas in the liver; (B) pneumatosis intestinalis of the right colon with significant bowel dilatation.
Management
Medical (Bell Stage I–IIA, and selected IIB)
- Bowel rest + NPO
- Nasogastric tube decompression
- Broad-spectrum IV antibiotics — e.g., piperacillin/tazobactam ± metronidazole; typically 7 days from radiographic resolution of pneumatosis (individualized)
- Serial clinical assessment, labs, and abdominal X-rays (every 6–8 hours in acute phase)
- Correction of metabolic acidosis, coagulopathy, thrombocytopenia
- Vasopressor support if hemodynamically unstable
Surgical (20–40% of cases)
Absolute indication: Pneumoperitoneum (intestinal perforation)
Relative indications:
- Deteriorating clinical status despite maximal medical management
- Abdominal wall erythema/cellulitis
- Portal venous gas
- Persistent fixed loop on serial X-rays
- High clinical suspicion for bowel necrosis
Operative approach:
- Transverse superumbilical laparotomy
- Full intestinal and colonic exploration
- Resection of all frankly necrotic bowel while preserving all viable or questionable bowel ("save every centimeter")
- For extensive ischemia without frank necrosis: place a spring-loaded silo and plan second-look operation in 24–48 hours
- For well-demarcated disease: create proximal stoma + distal mucous fistula
- Ostomy reversal after ≥6 weeks once infant weighs ≥3 kg and tolerates enteral feeds; perform contrast enema ± mucous fistula study before reversal to exclude strictures
Peritoneal drain (bedside): In very-low-birth-weight neonates (<1,000 g) with suspected spontaneous intestinal perforation (SIP), a lower quadrant peritoneal drain (vessel loop or ¼-inch Penrose) is appropriate first-line therapy. — Current Surgical Therapy 14e, Schwartz's Principles of Surgery 11e
NEC vs. Spontaneous Intestinal Perforation (SIP)
| Feature | NEC | SIP |
|---|---|---|
| Pathology | Widespread patchy ischemia + necrosis | Focal terminal ileal perforation |
| Shock at presentation | Almost universal | Often absent initially |
| Associated factors | Prematurity, enteral feeds | Indomethacin, early postnatal steroids |
| Pneumoperitoneum | Later stage | Presenting feature |
| Surgical approach | Laparotomy with resection | Peritoneal drain first-line in <1,000 g |
Complications
- Short bowel syndrome (SBS) — most devastating long-term complication after massive resection; requires intestinal rehabilitation ± transplantation
- Post-NEC strictures — fibrous healing can cause obstruction; colonic strictures most common; investigate with contrast enema before ostomy reversal
- Recurrent NEC
- Cholestatic liver disease — secondary to prolonged TPN
- Neurodevelopmental impairment — NEC is an independent risk factor for adverse neurodevelopmental outcomes in VLBW infants
- Growth failure
Prevention
| Strategy | Evidence |
|---|---|
| Human breast milk (preferably mother's own milk) | Meta-analysis: ~4-fold reduction in NEC (RR 0.25) vs. formula |
| Standardized feeding protocols | Shown to reduce incidence in VLBW infants |
| Probiotics | Multiple RCTs and meta-analyses support risk reduction; implementation varies by institution |
| Antenatal corticosteroids | Protective (accelerates gut maturation) |
| Infection control | Aborts epidemic clusters |
| Avoidance of unnecessary antibiotics | Reduces dysbiosis |
| Donor human milk (if mother's own milk unavailable) | Preferred over formula for high-risk premature infants |
Mothers of infants <32 weeks' gestation should be actively encouraged and supported in providing breast milk; early antenatal counseling increases lactation initiation without increasing maternal stress. — Creasy & Resnik's Maternal-Fetal Medicine
Prognosis
- Overall mortality: 10–50% (varies by center and disease severity)
- Surgical NEC mortality: approaches 50%
- Survivors are at significant risk for SBS, strictures, and neurodevelopmental delay
- Quality improvement initiatives in recent years have successfully reduced NEC incidence even in the highest-risk populations
Recent literature (2024–2025): Reviews by Roberts et al. (Paediatr Drugs, 2024, PMID 38564081) and Zhang et al. (Gut Microbes, 2025, PMID 39826099) highlight evolving understanding of the microbiome's role in NEC pathogenesis and emerging therapeutic targets. The microbiome perspective — specifically Bifidobacterium-dominant colonization via human milk — is increasingly recognized as central to both prevention and future treatment strategies.
Sources: Schwartz's Principles of Surgery 11e · Current Surgical Therapy 14e · Creasy & Resnik's Maternal-Fetal Medicine · Robbins & Cotran Pathologic Basis of Disease · Robbins & Kumar Basic Pathology
Why gain weight
Asking for Preferences
Reading File
Searching the Web
causes of weight gain physiology mechanisms
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Here is a comprehensive answer covering why humans gain weight — from basic physiology to medical causes:
Why Do Humans Gain Weight?
Weight gain occurs when energy intake chronically exceeds energy expenditure, causing the body to store the surplus primarily as fat. But this simple equation sits on top of a highly complex biological system.
1. The Energy Balance Equation
Body weight = Energy In − Energy Out
- Energy In: Calories from food and drink
- Energy Out: Made up of four components:
- Resting metabolic rate (RMR) — ~60–70% of total expenditure; energy used at rest for organ function, circulation, breathing
- Thermic effect of food (TEF) — ~10%; energy used to digest and metabolize food
- Voluntary exercise — variable
- NEAT (Non-Exercise Activity Thermogenesis) — energy from spontaneous movement like fidgeting, posture, daily activities; highly variable between individuals and a major determinant of weight gain susceptibility
When energy intake persistently exceeds all four of these, the surplus is converted to triglycerides and stored in adipose tissue.
2. Hormonal & Neurological Regulation
The brain — specifically the hypothalamus — acts as the master controller of appetite and body weight:
- Leptin — secreted by adipose tissue in proportion to fat mass; acts on hypothalamic receptors to suppress appetite and increase energy expenditure. In obesity, leptin resistance develops (similar to insulin resistance), so high leptin levels fail to suppress hunger — a key driver of progressive weight gain. — Fitzpatrick's Dermatology
- Ghrelin — the "hunger hormone," secreted by the stomach; rises before meals and promotes food intake
- GLP-1, PYY, CCK — gut hormones released after eating that signal satiety to the brain
- Insulin — promotes fat storage; chronically elevated insulin (as in insulin resistance) favors fat accumulation, especially visceral fat
- Melanocortin pathway (POMC/MC4R) — mutations in MC4R account for up to 6% of severe early-onset obesity; this pathway is downstream of leptin signaling
- AgRP — a hypothalamic antagonist that drives feeding behavior when activated
When satiety signals are impaired or hunger signals are chronically elevated, the set point for body weight shifts upward. — Fitzpatrick's Dermatology, Fuster & Hurst's The Heart 15e
3. Common Causes of Weight Gain
A. Behavioral / Lifestyle
- Excess caloric intake (especially ultra-processed, energy-dense foods)
- Physical inactivity / sedentary lifestyle
- Poor sleep (raises ghrelin, lowers leptin, increases hunger)
- Chronic stress (elevates cortisol → increases appetite for energy-dense foods)
- Emotional/stress eating
B. Physiological / Life Stages
- Aging — resting metabolic rate declines ~1–2% per decade after age 30; muscle mass decreases (sarcopenia), reducing calorie burn
- Pregnancy — intentional weight gain for fetal development
- Menopause — estrogen decline shifts fat distribution toward visceral/abdominal fat
- Puberty — normal developmental weight and fat gain
C. Endocrine / Medical Causes
| Condition | Mechanism |
|---|---|
| Hypothyroidism | Low thyroid hormone → reduced metabolic rate, fluid retention |
| Cushing's syndrome | Excess cortisol → central adiposity, muscle wasting |
| PCOS | Insulin resistance → hyperinsulinemia → fat storage |
| Hypothalamic disorders | Damage to satiety centers (tumors, trauma) → uncontrolled hyperphagia |
| Type 2 diabetes / insulin resistance | Hyperinsulinemia promotes fat storage; impaired fat mobilization |
| Growth hormone deficiency | Reduced lipolysis, increased fat mass |
| Depression | Reduced activity, appetite dysregulation |
D. Medications (Iatrogenic Weight Gain)
- Corticosteroids — increase appetite, promote central fat redistribution
- Antipsychotics (olanzapine, clozapine) — block dopamine/serotonin receptors, increase appetite
- Antidepressants (TCAs, mirtazapine, paroxetine)
- Insulin and sulfonylureas — fat-anabolic effects
- Beta-blockers — reduce metabolic rate, cause fatigue limiting exercise
- Antihistamines, anticonvulsants (valproate, gabapentin)
- Hormonal contraceptives — some formulations
E. Genetic Factors
Obesity is largely polygenic — familial studies show strong heritability. Rare monogenic causes include leptin deficiency, leptin receptor mutations, POMC deficiency, and MC4R mutations. Most common obesity, however, involves the interplay of many genes with environmental and behavioral factors.
4. The Vicious Cycle of Weight Gain
Once weight gain begins, comorbid factors can perpetuate further weight gain:
Excess weight → inflammation + insulin resistance → depression + joint pain + sleep apnea → reduced activity + emotional eating + fatigue → more weight gain
Visceral fat (intra-abdominal) is particularly harmful: it secretes inflammatory adipokines, reduces adiponectin (a protective hormone), worsens insulin resistance, and accelerates cardiovascular and metabolic disease. — Fuster & Hurst's The Heart 15e
Summary
Weight gain is ultimately driven by a positive energy balance, but the reasons that energy balance tilts positive are multifactorial — behavioral, hormonal, genetic, pharmaceutical, and medical. No single cause applies to everyone, which is why effective weight management requires identifying the individual's specific drivers.
Why weight gain here ti this patient before surgery
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Searching the Web
NEC necrotizing enterocolitis weight gain before ostomy reversal surgery requirement
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.