Hey guys, I'm studying about the mantles and lymphomas through the cell cycle regulators. So I came to know that in a lymph node, in the B area, like where the follicles are. So the region immediate next to the follicle is called mantle and above the mantle is called marginal margin. So there can lymphomas that arise from the follicle called follicular lymphoma, can arise from the mantle called mantle cell lymphoma, and can arise from margin called marginal cell lymphoma. So in this mantle cell lymphoma, there is a translocation of 11 and 14 chromosomes. The chromosome 14 usually have a gene for immunoglobulin heavy chain, which gets translocated to the locus, where on the chromosome 11 we have CD or cyclin B1. Hence it's get translocated instead of Ig immunoglobulin heavy chain, CD1 overproduced more. Hence CD1 is overproduced more. Then from the cell cycle as you know, G1, from G1 entering into the S phase is a stoply and highly regulated step in the cell cycle. And for that to undergo for the cells to undergo from G1 phase to S phase, we need the cyclin, the cyclin dependent kinases, cyclins and cyclin dependent kinases which will push the molecule to enter into the S phase of the cell cycle. So this is highly related because in USMLE classical beginners, they say that lymph node biopsy is given and the region above the follicle has shown so and so, hence it could be, the answer would be mantle cell lymphoma.
mantle cell lymphoma t(11;14) cyclin D1 CCND1 pathogenesis
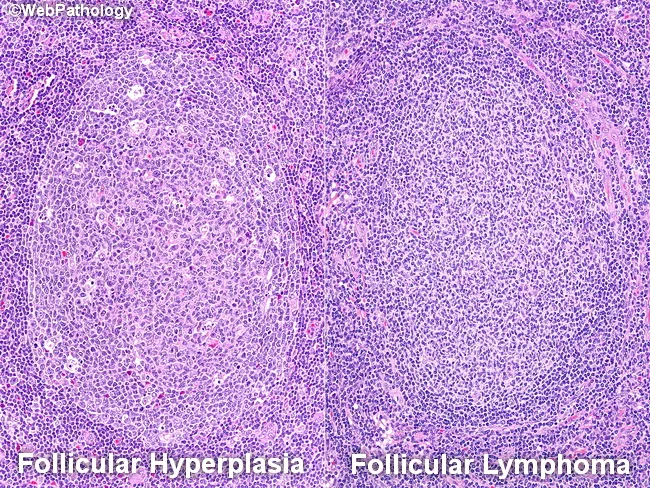
lymph node B cell zones follicle mantle zone marginal zone lymphoma classification
lymph node follicle mantle marginal zone diagram
Mantle Cell Lymphoma & Cell Cycle Regulation — A Complete Breakdown
Lymph Node B-Cell Architecture
| Zone | Location | Cell of Origin | Lymphoma |
|---|---|---|---|
| Germinal Center | Core of follicle | Germinal center B cells | Follicular Lymphoma |
| Mantle Zone | Immediately surrounds germinal center | Naive mantle B cells | Mantle Cell Lymphoma |
| Marginal Zone | Outside the mantle | Memory-like marginal B cells | Marginal Zone Lymphoma |
Your description is correct — mantle is immediately surrounding the follicle, and marginal zone is beyond that. The USMLE-style biopsy clue would describe a lymphoma arising from the rim around the follicle (mantle) vs. the outer rim/sinuses (marginal).
The Translocation: t(11;14) — One Critical Correction
- Chromosome 14q32 carries the immunoglobulin heavy chain (IgH) gene, which has an extremely active promoter (B cells are constantly producing Ig)
- Chromosome 11q13 carries CCND1, the gene encoding Cyclin D1
- In t(11;14), the CCND1 gene on chromosome 11 is translocated next to the IgH promoter on chromosome 14
Result: The powerful IgH promoter now drives constitutive overexpression of Cyclin D1 — far beyond normal levels.
Why Cyclin D1? — The Cell Cycle Connection
G0/G1 ──────────────────► S phase ──► G2 ──► M
↑
Restriction point (R)
Most tightly regulated step
- Cyclin D1 (and D2, D3) binds CDK4/CDK6
- The Cyclin D–CDK4/6 complex phosphorylates Rb (retinoblastoma protein)
- Phosphorylated Rb releases E2F transcription factors
- E2F drives transcription of genes needed for S phase entry (DNA synthesis machinery)
- Cyclin E–CDK2 then completes the Rb phosphorylation to lock in S-phase entry
- Cyclin D1 is massively overexpressed due to the IgH promoter driving CCND1 continuously
- The Cyclin D1–CDK4/6 complex is constitutively active
- Rb is perpetually phosphorylated
- Cells are locked into cycling — they cannot pause at G1 or respect normal anti-proliferative signals
- Result: uncontrolled B-cell proliferation → lymphoma
Immunophenotype — USMLE Key Points
- CD19⁺, CD20⁺ (B-cell markers)
- CD5⁺ (shared with CLL — a common USMLE trap)
- CD10⁻, CD23⁻ (distinguishes from CLL which is CD23⁺, and follicular lymphoma which is CD10⁺)
- Cyclin D1⁺ (the definitive marker — staining for cyclin D1 on biopsy is diagnostic)
The USMLE question stem will often give you CD5⁺ and ask you to distinguish MCL from CLL. The differentiator: CD23⁻ + Cyclin D1⁺ + t(11;14) = MCL.
One More Correction: "CD1" vs. "Cyclin D1"
Visual Reference
Summary for USMLE
| Feature | Mantle Cell Lymphoma |
|---|---|
| Cell of origin | Mantle zone B cell |
| Translocation | t(11;14) |
| Mechanism | IgH promoter → CCND1 overexpression |
| Effect | Rb permanently phosphorylated → G1→S blockade bypassed |
| Key markers | CD5⁺, CD20⁺, CD23⁻, Cyclin D1⁺ |
| Prognosis | Aggressive; median survival ~3–5 years |
Make a note without tables because as bullet points because I can't copy any tables and no more pictures also. Thank you. Make a high yield, ultra-high yield USMLE notes.
Mantle Cell Lymphoma — Ultra-High Yield USMLE Notes
Lymph Node B-Cell Zone Architecture (Inside → Outside)
- Innermost = Germinal Center → gives rise to Follicular Lymphoma
- Middle ring = Mantle Zone → gives rise to Mantle Cell Lymphoma
- Outer ring = Marginal Zone → gives rise to Marginal Zone Lymphoma
- USMLE stem clue: "lymph node biopsy shows lymphoma arising from the region immediately surrounding the follicle" = Mantle Cell Lymphoma
The Translocation — t(11;14)
- Chromosome 14q32 carries the Immunoglobulin Heavy Chain (IgH) gene
- The IgH locus has an extremely powerful, constitutively active promoter (B cells are constantly making antibodies)
- Chromosome 11q13 carries CCND1, the gene that encodes Cyclin D1
- In t(11;14), the CCND1 gene jumps next to the IgH promoter
- Result: the hyperactive IgH promoter now drives massive, uncontrolled overexpression of Cyclin D1
- Present in >90% of MCL cases (Harrison's, p. 3254)
- Remaining ~10% overexpress Cyclin D2, D3, or E via other mechanisms
Why Cyclin D1 Overexpression Causes Lymphoma — Cell Cycle Mechanism
- The cell cycle checkpoint between G1 and S phase is the most tightly regulated step
- This is called the Restriction Point (R point)
- Normal sequence to cross the R point:
- Cyclin D1 binds CDK4/CDK6 → forms an active complex
- Cyclin D1–CDK4/6 complex phosphorylates Rb (Retinoblastoma protein)
- Phosphorylated Rb releases E2F transcription factors
- E2F drives expression of genes needed for S phase (DNA synthesis)
- Cyclin E–CDK2 then completes the process and locks the cell into S phase
- In MCL, Cyclin D1 is overexpressed → Cyclin D1–CDK4/6 is constitutively active
- Rb is permanently phosphorylated → E2F is always free
- Cells bypass G1 completely and enter S phase without restraint
- Result: uncontrolled B-cell proliferation → lymphoma
Immunophenotype — The Most Tested Part
- CD19 positive — B-cell marker
- CD20 positive — B-cell marker
- CD5 positive — this is the classic USMLE trap (shared with CLL)
- CD23 NEGATIVE — key differentiator from CLL (CLL is CD23 positive)
- CD10 NEGATIVE — differentiates from Follicular Lymphoma (FL is CD10 positive)
- Cyclin D1 POSITIVE — the definitive, pathognomonic marker of MCL
- t(11;14) on FISH or cytogenetics = confirmatory
MCL vs CLL vs Follicular Lymphoma — The Classic USMLE Trio
- MCL: CD5⁺, CD23⁻, Cyclin D1⁺, t(11;14) → aggressive
- CLL/SLL: CD5⁺, CD23⁺, Cyclin D1⁻, no t(11;14) → indolent
- Follicular Lymphoma: CD5⁻, CD10⁺, BCL-2⁺, t(14;18) → indolent
Important Naming Correction
- The overexpressed protein is Cyclin D1 — encoded by gene CCND1
- This is NOT "CD1" — CD1 is a completely different surface antigen found on dendritic cells and thymocytes
- On USMLE, the answer choices will say Cyclin D1 or CCND1, not CD1
Clinical Pearls
- MCL is considered an aggressive lymphoma — median survival 3–5 years
- It is the lymphoma most associated with t(11;14)
- The classic demographic: older males
- Often presents with diffuse lymphadenopathy, splenomegaly, and frequent GI involvement (lymphomatous polyposis of the colon — a classic USMLE association)
- Bone marrow and peripheral blood involvement is common at diagnosis
- Mantle cell lymphoma is not curable with standard chemotherapy; autologous stem cell transplant used in eligible patients
One-Line Memory Hooks
- "11 goes to 14, Cyclin D1 goes wild, G1 checkpoint gone, B cells divide forever"
- "CD5⁺ but CD23⁻ = think Mantle, not CLL"
- "Follicle core = Follicular, Follicle rim = Mantle, Beyond rim = Marginal"
This one small ultra short, maybe like a flow chart, whatever I speak. So basically, BCL2 is an important regulator of apoptosis, right? So, in case of follicular lymphoma, why it's happening only in the follicles, because in general, the apoptosis is not very much needed, but where the apoptosis is super needed is in the follicles of the lymph node, because this is the spot where the B cells undergo the somatic hypermutation and the apoptosis should undergo. But if it's not happening due to a mutation, that is translocation of 14, 18. 14 is where the immunoglobulin heavy chain is present and 18 is where BCL2 is present. If there is translocation, so that BCL2, which is on the chromosome 18, to Ig heavy chain locus, which is on chromosome 14, which will result in increased BCL2. When there is increased BCL2, then the mitochondrial membrane is further stabilized and it will prohibit or inhibit apoptosis. So B cells that would normally undergo apoptosis during hyperzomatic mutation in the lymph node germinal center will accumulate and lead into lymphoma. This in a very conceptual way, I want an ultra short note or maybe a flow chart. This is why BCL2 is overexpressed in follicles and hence it happens more in the follicles because this is where the apoptotic activity is much needed in the body.
Follicular Lymphoma — BCL2 & Apoptosis Failure
Why the Germinal Center Specifically?
- Germinal center is where B cells undergo somatic hypermutation
- Most B cells after hypermutation are useless or autoreactive
- These bad B cells must be eliminated via apoptosis
- Germinal center is therefore one of the highest apoptotic demand zones in the body
- If apoptosis fails here → bad B cells accumulate → lymphoma
The Translocation — t(14;18)
- Chromosome 14q32 → IgH gene (powerful, always-on promoter)
- Chromosome 18q21 → BCL2 gene
- Translocation moves BCL2 next to the IgH promoter
- Result → BCL2 massively overexpressed
Flow: How BCL2 Blocks Apoptosis
Somatic hypermutation in germinal center
↓
B cell should receive apoptotic signal
↓
Normally: BAX/BAK punch holes in mitochondrial membrane
↓
Cytochrome C leaks out → Caspase cascade → Cell dies
↓↓ BUT in t(14;18) ↓↓
BCL2 overexpressed → stabilizes mitochondrial membrane
↓
BAX/BAK blocked → no cytochrome C release
↓
Apoptosis inhibited
↓
Useless/mutated B cells SURVIVE and accumulate
↓
Follicular Lymphoma
One-Line Summary
- t(14;18) → BCL2 overexpression → mitochondrial membrane locked → apoptosis blocked → germinal center B cells that should die, survive → Follicular Lymphoma
USMLE Hook
- BCL2 = anti-apoptotic (think: BCL2 = Blocks Cell death Locus)
- Follicular lymphoma = indolent because cells accumulate slowly (they aren't proliferating fast, they just aren't dying)
- Contrast with MCL: cells are proliferating too fast (Cyclin D1) vs FL: cells are dying too slowly (BCL2)
one quick line, you know, which is conceptual and logical. So to easily remember, you know, that choriocarcinoma usually happens through a hematogenous, but it makes sense because what is a choriocarcinoma? Choriocarcinoma is nothing but a malignancy of the placental villi or trophoblast, right? The trophoblast in general in the body, it is actually designed to look out for a blood vessel and invade it. So hence, if there is a tumor in the trophoblast, so the placental villi, then it will automatically search for the blood vessel and it will spread through hematogenous spread, right? That I want as a classic logic one step, one line step answer.
Am I right, you can correct the sentence according to the knowledge you have.
Okay, or else, can you give me a little idea about the immunochemical stains and immunohistochemical stains and immunohistochemistry where the antibody is taken and the brown pigment is added and then this is going, I'm going to add on the poorly differentiated cell biopsy type. If the antibody takes the color or something, then how, what's the principle, how it's happening? A short, ultra short flowchart.
Immunohistochemistry (IHC) — How It Works
Core Principle
- Poorly differentiated tumor cells lose their shape and structure
- You cannot tell origin by morphology alone
- But cells still express proteins specific to their origin
- IHC detects those proteins using antibodies
Flow
Tumor biopsy (poorly differentiated)
↓
Apply PRIMARY ANTIBODY
(targets specific protein on tumor cell)
↓
Primary antibody binds to its antigen on the cell
↓
Apply SECONDARY ANTIBODY
(targets the primary antibody)
↓
Secondary antibody is tagged with an ENZYME (HRP)
↓
Add substrate (DAB - diaminobenzidine)
↓
Enzyme converts substrate → BROWN PRECIPITATE
↓
Brown color = protein is PRESENT = antigen positive
No color = protein ABSENT = antigen negative
One-Line Principle
USMLE Hook
- Brown color = positive stain = antigen present
- This tells you the cell of origin of a poorly differentiated tumor
- e.g., brown with Cytokeratin → carcinoma, brown with Vimentin → sarcoma, brown with CD45 → lymphoma, brown with S-100/HMB-45 → melanoma
I didn't understand properly the flowchart. First, we have a biopsy. For that tissue, first I am going to add some antibody. I don't know some primary antibody. After that, I will give some time for the biopsy to get absorbed and then I will add the second antibody which has an enzyme and also a substrate to it. So the enzyme which will convert the substrate into brown precipitate if the antibody reacts with the protein.
IHC — Corrected Flowchart
Tumor biopsy on glass slide
↓
Add PRIMARY ANTIBODY
(specific to protein you're testing for)
↓
Wait — primary antibody binds to its target
protein on the cell (if present)
↓
Add SECONDARY ANTIBODY
(this one carries the enzyme — HRP)
(secondary binds to the primary antibody)
↓
Add SUBSTRATE (DAB)
↓
If primary found its target:
enzyme is present → converts DAB → BROWN COLOR
↓
Brown = protein present = antigen positive
No brown = protein absent = antigen negative
One correction to your description
- The substrate is not attached to the secondary antibody
- The enzyme is attached to the secondary antibody
- The substrate is added separately afterward
- Enzyme + Substrate react together → brown precipitate
Simple Analogy
- Primary antibody = detective that finds the suspect (protein)
- Secondary antibody = reporter following the detective, carrying a camera (enzyme)
- Substrate = film in the camera — only develops (turns brown) if the reporter was there
Now I understood. And the simple analogy, you can make it a little more simple, like primary antibody, it's like a detective. It will find if there is a protein present and go attached to it. Secondary antibody goes with the enzyme and it will go and attach to the primary antibody. It's like a reporter. And we'll add the substrate a little time later. So, if the protein is present in every, all three goes to that particular cell. So, and the substrate will, the substrate will be degraded by the enzyme and it will produce a brown pigment. And hence, if the brown pigment is present, then we have to say that the antigen is positive. And if not, the protein is negative. now i want whole concept super neat
Immunohistochemistry (IHC) — Complete Concept
The Flow
Tumor biopsy on glass slide
↓
Add PRIMARY ANTIBODY
(the detective — searches for its specific protein)
↓
If protein is present → primary antibody locks onto it
↓
Add SECONDARY ANTIBODY carrying an ENZYME
(the reporter — follows and attaches to the primary antibody)
↓
Add SUBSTRATE (DAB)
↓
Enzyme breaks down the substrate → BROWN PIGMENT forms
↓
BROWN = all three came together = protein is PRESENT
NO BROWN = primary found nothing = protein is ABSENT
The Analogy
- Primary antibody = Detective — finds and locks onto the suspect (protein)
- Secondary antibody + enzyme = Reporter — follows the detective and sits on top of him
- Substrate = Reporter's camera film — only develops into a brown picture if the reporter was there
The Readout
- Brown color = Antigen POSITIVE → protein is expressed in that cell
- No color = Antigen NEGATIVE → protein is absent in that cell
Why It Matters in USMLE
- Poorly differentiated tumors all look the same under the microscope
- IHC reveals their true identity by detecting origin-specific proteins
- Cytokeratin positive → Carcinoma (epithelial origin)
- Vimentin positive → Sarcoma (mesenchymal origin)
- CD45 positive → Lymphoma
- S-100 / HMB-45 positive → Melanoma
- PSA positive → Prostate carcinoma
- AFP positive → Hepatocellular carcinoma or Yolk sac tumor