Lung Abscess and Pulmonary Gangrene
lung abscess pulmonary gangrene etiology pathophysiology clinical features diagnosis management
lung abscess pulmonary gangrene chest X-ray CT imaging
Lung Abscess & Pulmonary Gangrene
Lung Abscess
Definition
Classification
| Type | Description |
|---|---|
| Primary | Aspiration-related, occurs in otherwise healthy individuals |
| Secondary | Complicates underlying disease (bronchiectasis, malignancy, immunosuppression, septic emboli) |
| Acute | Symptoms < 6 weeks |
| Chronic | Symptoms > 6 weeks |
Etiology & Microbiology
- Anaerobes are implicated in ~90% of cases — typically 3–6 isolates per sample are identified (Harrison's, p. 5087). Common organisms include Fusobacterium nucleatum, Prevotella, Peptostreptococcus, and Bacteroides species.
- Aerobic organisms can also cause abscesses independently: Staphylococcus aureus (including MRSA), Klebsiella pneumoniae, Pseudomonas aeruginosa, Streptococcus milleri group.
- Fungal/parasitic causes (less common): Aspergillus, Entamoeba histolytica, Nocardia.
- Poor dental hygiene / dental infection (often antecedent)
- Alcohol use disorder
- Altered consciousness (seizures, general anesthesia, stroke)
- Dysphagia, esophageal disease
Pathophysiology
- Aspiration of oropharyngeal/gastric contents → colonization of dependent lung segments
- Pneumonitis → tissue necrosis due to bacterial toxins and ischemia
- Liquefaction of necrotic tissue → cavity formation
- If communication established with bronchus → air-fluid level appears
- Posterior segments of upper lobes
- Superior segments of lower lobes (when aspiration occurs supine)
Clinical Features
| Symptom | Feature |
|---|---|
| Fever & night sweats | Common |
| Malaise, weight loss | Constitutional |
| Foul-smelling/purulent sputum | Highly suggestive of anaerobic infection |
| Cough | Productive |
| Pleuritic chest pain | When pleura involved |
| Hemoptysis | Variable |
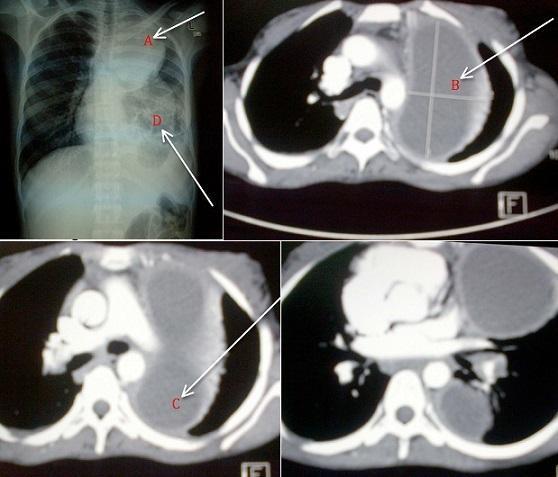
Imaging
- Thick-walled cavity, often in dependent segments
- Air-fluid level within the cavity (pathognomonic when present)
- Surrounding consolidation
- Better delineates cavity wall thickness, internal contents, satellite lesions
- Distinguishes abscess from empyema with bronchopleural fistula
Differential Diagnosis
- Necrotizing pneumonia (more diffuse, multiple small cavities)
- Empyema with bronchopleural fistula (lenticular shape, obtuse pleural angle on CT)
- Pneumatocele (thin-walled, post-infectious)
- Cavitating malignancy (irregular inner wall, no air-fluid level)
- Pulmonary tuberculosis (upper lobe, bilateral, systemic features)
- Wegener's granulomatosis / GPA (multiple nodules with cavitation)
Management
Antibiotics (Mainstay)
- First-line: Amoxicillin-clavulanate or clindamycin (anaerobic coverage)
- Alternatives: Metronidazole + penicillin, or carbapenems for resistant organisms
- Duration: Typically 4–6 weeks, continued until cavity resolves or stabilizes on imaging
- For S. aureus (MRSA): add vancomycin or linezolid
Postural Drainage
- Positioning to facilitate gravity drainage through communicating bronchus
Bronchoscopy
- Diagnostic: cultures, rule out obstructing lesion/malignancy
- Therapeutic: drainage of abscess contents in selected cases
Surgery
- Reserved for failure of medical therapy (4–6 weeks), massive hemoptysis, or suspected malignancy
- Procedures: lobectomy, pneumonectomy, or CT-guided percutaneous drainage
Complications
- Bronchopleural fistula
- Empyema thoracis
- Hemoptysis (sometimes massive)
- Septicemia
- Progression to pulmonary gangrene (see below)
- Amyloidosis (chronic cases)
Prognosis
- Overall mortality ~5–10% with appropriate therapy
- Worse prognosis: elderly, immunocompromised, large abscesses (>6 cm), S. aureus or gram-negative etiology, obstruction by malignancy
Pulmonary Gangrene
Definition
Pathophysiology
- Massive bacterial invasion → intense inflammatory response
- Thrombosis of pulmonary vasculature (arteries and veins) → ischemia
- Loss of blood supply + bacterial destruction → gangrene (wet necrosis) of entire lobes or the whole lung
- Sloughing of necrotic lung tissue → pulmonary sequestrum (free-floating necrotic tissue within a cavity — the hallmark)
Etiology
| Organism | Notes |
|---|---|
| Klebsiella pneumoniae | Classic cause, especially in diabetics/alcoholics |
| Streptococcus pneumoniae | Particularly virulent serotypes |
| Staphylococcus aureus (MRSA) | Common in post-influenza gangrene |
| Mixed anaerobes | Secondary contributor |
| Pseudomonas aeruginosa | Immunocompromised hosts |
Clinical Features
- Acute, rapidly deteriorating course (unlike subacute lung abscess)
- High fever, rigors, profound toxemia
- Copious foul, bloody, necrotic sputum
- Rapid respiratory failure
- Septic shock
Imaging Hallmarks
| Feature | Description |
|---|---|
| Pulmonary sequestrum | Sloughed necrotic lung floating within a cavity — pathognomonic |
| Massive consolidation | Involving an entire lobe or lung |
| Air-crescent sign | Necrotic fragment surrounded by air |
| Vascular cut-off on CT angiography | Thrombosis of pulmonary vessels |
| Rapid progression | Cavitation developing over hours to days |
Comparison: Lung Abscess vs. Pulmonary Gangrene
| Feature | Lung Abscess | Pulmonary Gangrene |
|---|---|---|
| Onset | Subacute (weeks) | Acute, fulminant (days) |
| Extent | Localized cavity | Lobar/whole lung destruction |
| Vascular thrombosis | Absent/minor | Prominent (central mechanism) |
| Necrotic sequestrum | Absent | Present (pathognomonic) |
| Organisms | Predominantly anaerobes | Virulent aerobes (Klebsiella, S. aureus) |
| Mortality | ~5–10% | Very high (30–50%+) |
| Surgical need | Occasional | Frequent (pneumonectomy) |
| Sputum | Foul, purulent | Bloody, necrotic, massive |
Management
- Aggressive IV antibiotics — broad-spectrum covering gram-positive, gram-negative, and anaerobes (e.g., piperacillin-tazobactam + vancomycin ± metronidazole)
- Supportive care: ICU-level, mechanical ventilation as needed
- Surgery (often required):
- Pneumonectomy or lobectomy for gangrene of a lobe/lung
- CT-guided drainage for accessible collections
- Anticoagulation: considered in selected cases due to vascular thrombosis, though evidence is limited
Prognosis
Summary
- Lung abscess = focal, subacute, predominantly anaerobic → responds well to prolonged antibiotics
- Pulmonary gangrene = lobar/diffuse, fulminant, virulent aerobic organisms, vascular thrombosis → high mortality, often requires surgery
copd
COPD chronic obstructive pulmonary disease etiology pathophysiology classification diagnosis management
COPD emphysema chest X-ray CT spirometry
Chronic Obstructive Pulmonary Disease (COPD)
Definition
| Component | Definition |
|---|---|
| Emphysema | Anatomical — destruction of alveolar walls with air space enlargement |
| Chronic Bronchitis | Clinical — productive cough for ≥3 months/year for ≥2 consecutive years |
| Small Airway Disease | Narrowing and reduction in number of small bronchioles |
Epidemiology
- One of the leading causes of morbidity and mortality worldwide (3rd leading cause of death globally)
- Affects ~10% of adults over 40
- Significantly underdiagnosed — many patients present only when disease is advanced
Etiology & Risk Factors
Environmental (Dominant)
- Cigarette smoking — accounts for ~80–90% of cases in developed countries; pack-year history is the strongest risk factor
- Biomass fuel smoke — cooking/heating fires; major cause globally, especially in women in low-income countries
- Occupational dusts/chemicals — coal dust, silica, grain dust, isocyanates
- Air pollution — outdoor (PM2.5, NO₂) and indoor
Host Factors
- Alpha-1 antitrypsin (AAT) deficiency — genetic risk; panacinar emphysema, especially in lower lobes; onset in younger non-smokers
- Abnormal lung development — low birth weight, prematurity, childhood respiratory infections
- Airway hyperresponsiveness — asthma–COPD overlap
- Genetics — multiple susceptibility loci beyond AAT
Pathophysiology
Noxious Exposure
↓
Chronic Airway Inflammation (neutrophils, macrophages, CD8+ T cells)
↓
┌────────────────────────────────────────┐
│ Airway remodeling (small airways) │ → Airflow obstruction
│ Mucus hypersecretion │ → Chronic bronchitis
│ Alveolar wall destruction (elastase > │
│ anti-elastase imbalance) │ → Emphysema
└────────────────────────────────────────┘
↓
Air trapping → Hyperinflation → ↑ Work of breathing
↓
V/Q mismatch → Hypoxemia → Hypercapnia (late)
↓
Pulmonary hypertension → Cor pulmonale
- Protease–antiprotease imbalance: excess neutrophil elastase and MMPs destroy alveolar parenchyma
- Oxidative stress: amplifies inflammation
- Loss of elastic recoil: dynamic airway collapse on expiration (air trapping)
Clinical Features
Symptoms
| Symptom | Details |
|---|---|
| Dyspnea | Progressive, initially on exertion; hallmark complaint |
| Chronic cough | Often productive; may precede dyspnea by years |
| Sputum production | Chronic; purulent during exacerbations |
| Wheeze | Especially on exertion |
| Exercise intolerance | Progressive limitation |
Signs
- Barrel chest (increased AP diameter) — emphysema
- Hyperresonance on percussion
- Diminished breath sounds, prolonged expiration
- Use of accessory muscles, pursed-lip breathing
- Cyanosis (central) — advanced disease
- Cor pulmonale: elevated JVP, peripheral edema, right ventricular heave
Classic Phenotypes
| Feature | "Pink Puffer" (Emphysema predominant) | "Blue Bloater" (Chronic Bronchitis predominant) |
|---|---|---|
| Build | Thin, cachexic | Obese |
| Cyanosis | Absent (maintains oxygenation) | Present |
| Dyspnea | Severe | Less prominent |
| Cough/sputum | Minimal | Prominent, copious |
| PaO₂ | Near normal | Low |
| PaCO₂ | Normal/low | Elevated |
| Cor pulmonale | Late | Early |
Diagnosis
Spirometry (Mandatory)
- Post-bronchodilator FEV₁/FVC < 0.70 confirms airflow obstruction
- FEV₁ % predicted classifies severity
GOLD Spirometric Grading
| GOLD Grade | FEV₁ % Predicted | Severity |
|---|---|---|
| GOLD 1 | ≥ 80% | Mild |
| GOLD 2 | 50–79% | Moderate |
| GOLD 3 | 30–49% | Severe |
| GOLD 4 | < 30% | Very Severe |
GOLD ABE Assessment (2023 Update)
| Group | Description |
|---|---|
| A | Low symptoms, low exacerbation risk |
| B | High symptoms, low exacerbation risk |
| E | ≥2 exacerbations or ≥1 hospitalization (high risk) |
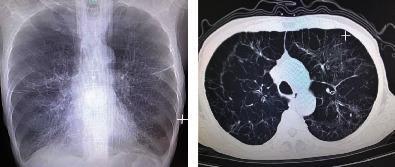
Other Investigations
- CXR: hyperinflation, flattened diaphragms, bullae, increased retrosternal airspace
- HRCT chest: better characterizes emphysema subtype, bullae, bronchiectasis
- ABG: hypoxemia, hypercapnia in advanced disease
- Alpha-1 antitrypsin level: screen all patients with COPD (especially age <45 or minimal smoking history)
- ECG/Echo: assess for cor pulmonale
- 6-minute walk test: functional assessment
- CBC: secondary polycythemia
Imaging
Management
Non-Pharmacological (Foundation)
- Smoking cessation — single most effective intervention; slows disease progression
- Pulmonary rehabilitation — improves dyspnea and exercise capacity
- Vaccination: influenza (annual), pneumococcal, COVID-19, RSV
- Nutritional support — address cachexia
- Oxygen therapy: Long-term O₂ (LTOT) if PaO₂ ≤55 mmHg or SaO₂ ≤88% at rest → improves survival
Pharmacological — Stable COPD (GOLD 2023)
Bronchodilators (Mainstay)
| Drug Class | Examples | Notes |
|---|---|---|
| SABA (Short-acting β₂ agonist) | Salbutamol, terbutaline | PRN relief |
| SAMA (Short-acting muscarinic antagonist) | Ipratropium | PRN or regular |
| LABA (Long-acting β₂ agonist) | Formoterol, salmeterol, indacaterol | Regular maintenance |
| LAMA (Long-acting muscarinic antagonist) | Tiotropium, umeclidinium, glycopyrronium | Preferred maintenance; reduces exacerbations |
| LABA + LAMA (dual bronchodilation) | Indacaterol/glycopyrronium, vilanterol/umeclidinium | Superior to mono in Group B/E |
Inhaled Corticosteroids (ICS)
- ICS + LABA: indicated when eosinophil count ≥300/µL or Group E despite dual bronchodilation
- Triple therapy (ICS + LABA + LAMA): for persistent exacerbations; reduces mortality (IMPACT trial)
- Caution: increased pneumonia risk with ICS
Other Agents
- Roflumilast (PDE-4 inhibitor): oral; for severe COPD with chronic bronchitis phenotype and frequent exacerbations (FEV₁ <50%)
- Azithromycin (long-term): reduces exacerbation frequency in select patients; monitor for hearing loss and cardiac QTc prolongation
- Mucolytics (NAC, carbocisteine): may reduce exacerbations in some patients
- AAT augmentation therapy: IV infusion for confirmed AAT deficiency with emphysema
Initial Treatment Algorithm (GOLD 2023)
Group A → SABA or SAMA (PRN)
Group B → LABA + LAMA (dual bronchodilation)
Group E → LABA + LAMA ± ICS (if eos ≥300)
Acute Exacerbations of COPD (AECOPD)
Definition
Causes
- Respiratory infections (bacterial: H. influenzae, S. pneumoniae, M. catarrhalis; viral: rhinovirus, influenza)
- Air pollution
- Non-compliance with therapy
- Pulmonary embolism (must exclude)
Management of AECOPD
| Intervention | Details |
|---|---|
| Short-acting bronchodilators | SABA ± SAMA (nebulized), first-line |
| Systemic corticosteroids | Prednisolone 40 mg/day × 5 days; shortens recovery |
| Antibiotics | If purulent sputum, ↑ dyspnea, or requiring ventilation — amoxicillin, doxycycline, or azithromycin |
| Controlled oxygen | Target SpO₂ 88–92% (avoid hypercapnic drive suppression) |
| NIV (BiPAP) | For hypercapnic respiratory failure (pH <7.35, PaCO₂ >6 kPa); reduces intubation and mortality |
| Invasive ventilation | When NIV fails or contraindicated |
| Thromboprophylaxis | DVT prophylaxis in hospitalized patients |
Complications
- Cor pulmonale and right heart failure
- Respiratory failure (Type I and Type II)
- Pneumothorax (especially with bullous disease)
- Pulmonary hypertension
- Secondary polycythemia
- Malnutrition and cachexia
- Anxiety and depression (significantly underdiagnosed)
- Lung cancer (shared risk factors)
Surgical/Interventional Options
| Procedure | Indication |
|---|---|
| Lung volume reduction surgery (LVRS) | Upper lobe predominant emphysema, low exercise capacity post-rehab |
| Endobronchial valve placement | Bronchoscopic LVRS equivalent; heterogeneous emphysema without collateral ventilation |
| Bullectomy | Giant bullae (>1/3 of hemithorax) |
| Lung transplantation | End-stage COPD (BODE index >7, GOLD 4) |
Prognosis & Prognostic Tools
BODE Index (predicts mortality)
| Variable | Score 0 | Score 1 | Score 2 | Score 3 |
|---|---|---|---|---|
| BMI | >21 | ≤21 | — | — |
| FEV₁ % predicted | ≥65% | 50–64% | 36–49% | ≤35% |
| mMRC dyspnea score | 0–1 | 2 | 3 | 4 |
| 6-min walk distance | ≥350 m | 250–349 m | 150–249 m | <150 m |
- Smoking cessation and LTOT are the only interventions proven to reduce mortality in stable COPD (along with triple therapy showing mortality benefit in Group E)
- 5-year mortality after first hospitalization for AECOPD: ~50%
Key Takeaways
- Spirometry is mandatory for diagnosis — clinical features alone are insufficient
- LAMA is the preferred initial maintenance bronchodilator for most patients
- ICS should not be used as monotherapy in COPD; reserve for specific indications (high eosinophils, frequent exacerbations)
- Target SpO₂ 88–92% during exacerbations — avoid suppressing hypoxic drive
- Smoking cessation at any stage alters the natural history
- Screen all COPD patients for AAT deficiency
bronchial asthma , upper airway obstruction , pulmonary edema , nutrition in copd
bronchial asthma pathophysiology diagnosis management GINA guidelines
upper airway obstruction causes diagnosis management stridor
pulmonary edema cardiogenic non-cardiogenic ARDS pathophysiology treatment
nutrition in COPD malnutrition nutritional support dietary management
bronchial asthma airway inflammation spirometry
pulmonary edema chest X-ray cardiogenic
1. Bronchial Asthma
Definition
Pathophysiology
Allergen / Trigger exposure
↓
Th2-mediated immune response
→ IL-4, IL-5, IL-13 release
→ IgE production (sensitization)
↓
Mast cell & eosinophil activation
↓
┌─────────────────────────────────────────┐
│ Bronchoconstriction (acute, reversible) │
│ Airway mucosal edema │
│ Mucus hypersecretion │
│ Airway remodeling (chronic) │
└─────────────────────────────────────────┘
↓
Airflow obstruction → Wheeze, dyspnea, chest tightness
- Subepithelial fibrosis
- Smooth muscle hypertrophy
- Goblet cell hyperplasia
- Angiogenesis
Classification of Severity (GINA / NAEPP)
| Severity | Daytime Symptoms | Nocturnal | FEV₁ % Predicted | FEV₁/FVC |
|---|---|---|---|---|
| Intermittent | ≤2 days/week | ≤2×/month | ≥80% | Normal |
| Mild Persistent | >2 days/week | 3–4×/month | ≥80% | Normal |
| Moderate Persistent | Daily | >1×/week | 60–79% | Reduced |
| Severe Persistent | Continuous | Frequent | <60% | Reduced |
Triggers
| Category | Examples |
|---|---|
| Allergens | Dust mites, pollen, pet dander, mold |
| Infections | Rhinovirus, RSV, influenza |
| Occupational | Isocyanates, flour, latex |
| Drugs | NSAIDs (aspirin-exacerbated asthma), beta-blockers |
| Environmental | Cold air, exercise, air pollution, smoke |
| Emotional | Stress, laughter |
| GERD | Microaspiration/vagal reflex |
Clinical Features
- Episodic wheeze, dyspnea, chest tightness, cough (often worse at night/early morning)
- Symptoms vary and are reversible
- Between attacks: patient may be completely asymptomatic
- Diurnal variation: PEF lowest in early morning ("morning dipping")
- Tachypnea, tachycardia
- Prolonged expiration, expiratory wheeze
- Use of accessory muscles
- Pulsus paradoxus (>10 mmHg drop in SBP on inspiration — indicates severe attack)
- Silent chest = very severe (no air movement)
Diagnosis
| Test | Finding |
|---|---|
| Spirometry | FEV₁/FVC <0.70; ≥12% and ≥200 mL improvement in FEV₁ post-bronchodilator |
| Peak expiratory flow (PEF) | >10% diurnal variability |
| Methacholine challenge | Positive (PC₂₀ <8 mg/mL) if spirometry normal but asthma suspected |
| FeNO (exhaled NO) | ≥25 ppb suggests eosinophilic airway inflammation |
| Skin prick / RAST | Identifies allergen sensitization |
| Eosinophil count / IgE | Elevated in atopic asthma |
Management (GINA 2023 Stepwise Approach)
Controller + Reliever Therapy
| GINA Step | Preferred Controller | Reliever |
|---|---|---|
| Step 1 | As-needed low-dose ICS-formoterol | ICS-formoterol PRN |
| Step 2 | Low-dose ICS daily | SABA or ICS-formoterol PRN |
| Step 3 | Low-dose ICS + LABA | ICS-formoterol PRN |
| Step 4 | Medium/high-dose ICS + LABA | ICS-formoterol PRN |
| Step 5 | Add-on: tiotropium, anti-IL-5, anti-IL-4Rα, anti-IgE | ICS-formoterol PRN |
Key GINA 2023 update: SABA-alone reliever is no longer recommended at any step. ICS-containing reliever (ICS-formoterol) is preferred to reduce exacerbation risk.
Biologic Therapies (Step 5, Severe Asthma)
| Biologic | Target | Indication |
|---|---|---|
| Omalizumab | Anti-IgE | Allergic asthma, high IgE |
| Mepolizumab / Reslizumab | Anti-IL-5 | Eosinophilic asthma (eos ≥300) |
| Benralizumab | Anti-IL-5Rα | Eosinophilic asthma |
| Dupilumab | Anti-IL-4Rα | Type-2 asthma ± atopic dermatitis |
| Tezepelumab | Anti-TSLP | Broad severe asthma (any phenotype) |
Acute Severe Asthma (Status Asthmaticus)
Severity Assessment
| Parameter | Moderate | Severe | Life-Threatening |
|---|---|---|---|
| SpO₂ | >92% | <92% | <90% |
| PEF | 50–75% predicted | 33–50% | <33% |
| Speech | Sentences | Words | Unable |
| Consciousness | Normal | Agitated | Drowsy/confused |
| PaCO₂ | <45 | 45+ | Rising (impending fatigue) |
Treatment
- Oxygen: Target SpO₂ 94–98%
- Nebulized SABA: Salbutamol 2.5–5 mg, repeat every 20 min × 3
- Ipratropium bromide: add to SABA in severe attack
- Systemic corticosteroids: Prednisolone 40–50 mg PO or IV hydrocortisone 100 mg QID
- IV magnesium sulfate: 1.2–2 g IV over 20 min for severe/life-threatening attack
- IV aminophylline: second-line
- Heliox: reduces turbulent airflow in critical obstruction
- NIV/Intubation: last resort — intubation in asthma is high risk (dynamic hyperinflation)
2. Upper Airway Obstruction (UAO)
Definition
Classification
| By Onset | By Location | By Nature |
|---|---|---|
| Acute | Supraglottic | Fixed (equal on inspiration + expiration) |
| Chronic | Glottic/subglottic | Variable intrathoracic |
| Tracheal | Variable extrathoracic |
Causes
Acute UAO (Emergencies)
| Cause | Features |
|---|---|
| Foreign body aspiration | Sudden onset, children or elderly; café coronary sign |
| Anaphylaxis / Angioedema | Rapid angioedema of glottis/tongue; urticaria, hypotension |
| Epiglottitis | H. influenzae type b or adults (S. pyogenes); tripod position, drooling, "hot potato" voice |
| Croup (Laryngotracheobronchitis) | Parainfluenza virus; children; barking cough, steeple sign on X-ray |
| Ludwig's angina | Floor-of-mouth cellulitis; rapidly progressive |
| Retropharyngeal abscess | Posterior pharyngeal wall abscess |
| Trauma / Burns | Inhalation injury; thermal/chemical airway burns |
Chronic UAO
| Cause | Features |
|---|---|
| Obstructive sleep apnea | Recurrent nocturnal obstruction, snoring, daytime somnolence |
| Laryngeal carcinoma | Progressive hoarseness → stridor |
| Tracheal stenosis | Post-intubation/tracheostomy; fixed obstruction |
| Goiter / Thyroid mass | Compression of trachea; tracheal deviation |
| Vocal cord paralysis | Unilateral/bilateral; post-thyroidectomy, recurrent laryngeal nerve injury |
| Tracheomalacia | Weakness of tracheal cartilage; expiratory collapse |
| Subglottic stenosis | Congenital or acquired (Wegener's/GPA) |
Pathophysiology of Airflow Dynamics
| Type | Inspiration | Expiration | Cause |
|---|---|---|---|
| Fixed obstruction | ↓ | ↓ (equal) | Tracheal stenosis, goiter |
| Variable extrathoracic | ↓ (worsens) | Normal | Vocal cord paralysis, epiglottitis |
| Variable intrathoracic | Normal | ↓ (worsens) | Tracheomalacia |
- Flow-volume loop is diagnostic: flattening of the inspiratory limb (extrathoracic), expiratory limb (intrathoracic), or both (fixed)
Clinical Features
- Stridor (inspiratory = supraglottic/glottic; biphasic = subglottic/tracheal)
- Dyspnea, use of accessory muscles
- Hoarseness, dysphonia, dysphagia
- Cyanosis and altered consciousness in severe obstruction
- Paradoxical breathing (chest sucks in on inspiration) in complete obstruction
Diagnosis
| Investigation | Purpose |
|---|---|
| Flow-volume loop (spirometry) | Pattern of obstruction (fixed vs. variable) |
| Lateral neck X-ray | Epiglottitis (thumbprint sign), croup (steeple sign) |
| CT neck/chest | Mass, abscess, tracheal narrowing |
| Direct/flexible laryngoscopy | Visualize larynx, vocal cords |
| Bronchoscopy | Subglottic/tracheal assessment |
Management
Emergency
| Situation | Intervention |
|---|---|
| Foreign body | Heimlich maneuver → bronchoscopic retrieval |
| Anaphylaxis | IM adrenaline 0.5 mg (1:1000) → airway secured |
| Epiglottitis | Secure airway (intubation/tracheostomy) + IV ceftriaxone ± dexamethasone |
| Croup (moderate-severe) | Nebulized adrenaline + oral/IM dexamethasone |
| Angioedema | Adrenaline, IV antihistamines, corticosteroids; FFP or C1-esterase inhibitor for hereditary angioedema |
| Trauma/burns | Early intubation before edema progresses |
Definitive / Chronic
- Tracheostomy: bypasses obstruction; emergency or elective
- Tracheal dilation/stenting: post-intubation stenosis
- Surgery: laryngeal carcinoma (laryngectomy), goiter, tumor resection
- CPAP: obstructive sleep apnea
- Voice therapy / reinnervation: vocal cord paralysis
3. Pulmonary Edema
Definition
Classification: Cardiogenic vs. Non-Cardiogenic
| Feature | Cardiogenic | Non-Cardiogenic (ARDS) |
|---|---|---|
| Mechanism | ↑ hydrostatic pressure (↑ PCWP >18 mmHg) | ↑ capillary permeability (normal PCWP) |
| Cause | LV failure, mitral stenosis, fluid overload | Sepsis, pneumonia, aspiration, trauma, pancreatitis |
| Fluid protein | Low (transudate) | High (exudate) |
| BNP | Markedly elevated | Normal/mildly elevated |
| Echo | LV dysfunction | Normal LV function |
| CXR | Cardiomegaly, cephalization, Kerley B lines | Bilateral infiltrates, normal heart size |
| Response to diuretics | Good | Poor |
Pathophysiology
Cardiogenic
LV dysfunction / ↑ filling pressures
↓
↑ Pulmonary capillary wedge pressure (>18 mmHg)
↓
Fluid transudation: interstitium → alveoli
↓
Interstitial edema → Alveolar flooding
↓
V/Q mismatch → Hypoxemia → Dyspnea
Non-Cardiogenic (ARDS)
Systemic/pulmonary insult (sepsis, aspiration)
↓
Endothelial injury + inflammatory mediators
↓
↑ Capillary permeability
↓
Protein-rich exudate floods alveoli
↓
Surfactant destruction → Alveolar collapse
↓
Refractory hypoxemia (P/F ratio <300)
Clinical Features
| Symptom/Sign | Details |
|---|---|
| Acute dyspnea | Sudden onset, worse lying flat |
| Orthopnea / PND | Cardiogenic hallmarks |
| Pink frothy sputum | Alveolar flooding |
| Crepitations (crackles) | Bilateral, basal (ascending in severity) |
| Wheeze | "Cardiac asthma" — bronchospasm from peribronchial edema |
| Tachycardia, hypertension | Sympathetic activation |
| Cold, clammy extremities | Cardiogenic shock |
| SpO₂ ↓, cyanosis | Hypoxemia |
Imaging
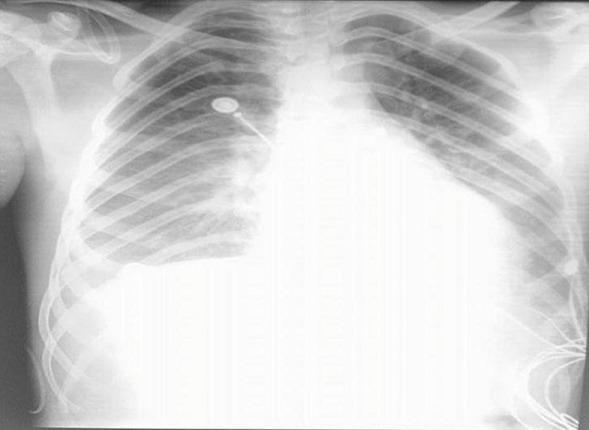
Radiological Stages of Pulmonary Edema (Cardiogenic)
| Stage | CXR Finding | PCWP |
|---|---|---|
| I — Vascular redistribution | Upper lobe vascular engorgement (cephalization) | 12–18 mmHg |
| II — Interstitial edema | Kerley B lines, peribronchial cuffing, haziness | 18–25 mmHg |
| III — Alveolar edema | Bilateral confluent "bat-wing" opacities | >25 mmHg |
Diagnosis
| Test | Finding |
|---|---|
| CXR | As above |
| ABG | Hypoxemia, respiratory alkalosis early; respiratory acidosis late |
| BNP / NT-proBNP | ↑↑ in cardiogenic (BNP >400 pg/mL strongly suggests HF) |
| Echo | LV systolic/diastolic function, LVEF, valvular disease |
| PCWP (Swan-Ganz) | >18 mmHg = cardiogenic |
| Troponin, ECG | Exclude ACS as precipitant |
| CBC, CRP, cultures | If non-cardiogenic (sepsis screen) |
Management
Cardiogenic Pulmonary Edema (Acute)
| Intervention | Details |
|---|---|
| Upright positioning | Sit patient up; reduces venous return |
| Oxygen | Target SpO₂ ≥94% |
| NIV (CPAP/BiPAP) | First-line for respiratory failure; reduces intubation rate and mortality |
| IV furosemide | 40–80 mg IV bolus; venodilation within minutes, then diuresis |
| IV nitrates | GTN infusion; reduce preload and afterload; avoid if SBP <90 |
| Morphine | IV 2–4 mg; reduces anxiety and preload (use with caution — may worsen outcomes) |
| Treat precipitant | ACS → revascularization; AF → rate control; hypertensive crisis → IV nitrates |
| Inotropes | Dobutamine if low-output cardiogenic shock |
| Mechanical support | IABP, Impella in refractory cardiogenic shock |
Non-Cardiogenic (ARDS) Management
| Intervention | Details |
|---|---|
| Lung-protective ventilation | TV 6 mL/kg IBW, plateau pressure <30 cmH₂O, PEEP titration |
| Prone positioning | ≥16 hours/day for moderate-severe ARDS (P/F <150); reduces mortality |
| Conservative fluid strategy | Once hemodynamically stable; negative fluid balance reduces ventilator days |
| Corticosteroids | Methylprednisolone for ARDS >7–14 days (fibroproliferative phase); also in COVID-ARDS |
| Treat underlying cause | Antibiotics for sepsis/pneumonia, etc. |
| Neuromuscular blockade | Cisatracurium for 48 h in severe ARDS (P/F <150) |
| ECMO | Refractory ARDS unresponsive to optimization |
4. Nutrition in COPD
Why Nutrition Matters in COPD
Mechanisms of Malnutrition in COPD
| Mechanism | Explanation |
|---|---|
| ↑ Energy expenditure | Increased work of breathing (hyperinflation); respiratory muscles work harder |
| ↓ Oral intake | Dyspnea during eating; early satiety (diaphragm flattening compresses stomach) |
| Systemic inflammation | IL-6, TNF-α cause anorexia and muscle catabolism |
| Hypoxia | Reduces appetite and GI motility |
| Drug side effects | Theophylline → nausea; corticosteroids → metabolic derangements |
| Depression/anxiety | Reduces appetite and motivation to eat |
Consequences of Malnutrition in COPD
- Muscle wasting (sarcopenia) → reduced respiratory muscle strength → ventilatory failure
- Impaired immune function → increased susceptibility to infections/exacerbations
- Reduced exercise capacity and quality of life
- Osteoporosis (compounded by corticosteroid use)
- Increased hospital admissions and mortality
- BMI <21 kg/m² is an independent predictor of mortality (component of BODE index)
Nutritional Assessment
| Tool | Details |
|---|---|
| BMI | <21 kg/m² = poor prognosis in COPD |
| FFMI (Fat-Free Mass Index) | Better measure of muscle wasting than BMI |
| Mid-arm circumference | Proxy for muscle mass |
| Hand-grip strength | Sarcopenia assessment |
| MNA / SGA / NRS-2002 | Validated malnutrition screening tools |
| Serum albumin / prealbumin | Protein status (low = worse prognosis) |
Nutritional Requirements in COPD
| Nutrient | Recommendation |
|---|---|
| Calories | 27–35 kcal/kg/day; up to 45–50 kcal/kg in severe malnutrition |
| Protein | 1.2–1.7 g/kg/day (higher end if on corticosteroids or during exacerbations) |
| Carbohydrates | Moderate restriction — CHO metabolism produces more CO₂ per O₂ consumed (↑RQ); high-CHO diets worsen hypercapnia |
| Fats | Higher fat content preferred — lower respiratory quotient (RQ=0.7 vs CHO RQ=1.0); less CO₂ produced |
| Omega-3 fatty acids | Anti-inflammatory; may reduce exacerbation frequency |
| Vitamin D | Commonly deficient; supplement to maintain 25-OH-VitD >50 nmol/L; improves respiratory muscle function |
| Antioxidants | Vitamins C, E; reduce oxidative stress |
| Magnesium, Phosphate | Critical for respiratory muscle function; correct deficiencies |
Dietary & Practical Recommendations
- Small, frequent meals (4–6 meals/day) — reduces post-meal dyspnea and bloating
- Eat main meals when least breathless (often earlier in the day)
- Avoid gas-forming foods (carbonated drinks, beans, cabbage) — worsen diaphragmatic compression
- Bronchodilators before meals — reduce dyspnea during eating
- Supplemental oxygen during meals if SpO₂ drops on eating
- High-calorie, high-protein oral supplements (e.g., Ensure, Fortisip) if inadequate oral intake
- Dental care — poor dentition is common and limits food intake
Nutritional Support Algorithm
Screen all COPD patients (BMI, weight trend, muscle mass)
↓
BMI <21 or unintentional weight loss >10% in 6 months?
↓
YES → Dietary counseling + high-calorie, high-protein diet
↓
Inadequate with diet alone?
↓
YES → Oral nutritional supplements (ONS)
↓
Unable to meet needs orally (severe, mechanically ventilated)?
↓
Enteral nutrition (nasogastric/PEG)
↓
Use high-fat, low-carbohydrate enteral formula (e.g., Pulmocare)
to minimize CO₂ production
Pulmonary-Specific Enteral Formulas
| Formula | Fat % | CHO % | RQ | Use |
|---|---|---|---|---|
| Standard (e.g., Ensure) | 30% | 55% | ~0.90 | General use |
| Pulmocare / Respifor | 55% | 28% | ~0.77 | COPD/respiratory failure; reduces CO₂ load |
Exercise + Nutrition: Synergistic Effect
- Pulmonary rehabilitation + nutritional supplementation together are more effective than either alone for improving muscle mass, 6-minute walk distance, and quality of life
- Anabolic agents (testosterone, megestrol acetate) explored in severe sarcopenia but evidence remains limited
Summary Table
| Topic | Key Points |
|---|---|
| Bronchial Asthma | Reversible airflow obstruction; Th2/eosinophilic inflammation; stepwise ICS-based therapy; biologics for severe asthma; ICS-formoterol preferred reliever |
| Upper Airway Obstruction | Stridor = hallmark; extrathoracic vs. intrathoracic vs. fixed patterns; emergency airway control for acute causes |
| Pulmonary Edema | Cardiogenic (↑PCWP, BNP↑, responds to diuretics/NIV) vs. non-cardiogenic ARDS (↑permeability, lung-protective ventilation, prone positioning) |
| Nutrition in COPD | Malnutrition = independent mortality predictor; high-fat, low-CHO diet; small frequent meals; correct Vit D/Mg/phosphate; pulmonary enteral formulas reduce CO₂ production |
pleural effusion,dry pleuritis, pneumothorax
pleural effusion causes transudates exudates diagnosis management thoracentesis
dry pleuritis pleurisy causes clinical features diagnosis treatment
pneumothorax spontaneous tension classification management chest drain
pleural effusion pneumothorax chest X-ray imaging
pneumothorax tension pneumothorax chest X-ray collapsed lung
pleural effusion,dry pleuritis, pneumothorax
1. Pleural Effusion
Definition
Anatomy & Physiology of the Pleural Space
- Fluid normally enters via the parietal pleura (systemic capillaries) and drains via lymphatics
- Effusion occurs when:
- Fluid production exceeds drainage (↑ hydrostatic pressure or ↑ capillary permeability)
- Lymphatic drainage is impaired
- Fluid enters from adjacent compartments (abdomen, chest wall)
Classification: Transudate vs. Exudate
Light's Criteria (1972 — Gold Standard)
| Criterion | Exudate |
|---|---|
| Pleural fluid protein / Serum protein | > 0.5 |
| Pleural fluid LDH / Serum LDH | > 0.6 |
| Pleural fluid LDH | > 2/3 upper limit of normal serum LDH |
Causes
| Transudate | Exudate |
|---|---|
| Congestive heart failure (most common) | Parapneumonic / empyema |
| Liver cirrhosis (hepatic hydrothorax) | Malignancy (lung, breast, lymphoma, mesothelioma) |
| Nephrotic syndrome | Tuberculosis |
| Hypoalbuminemia | Pulmonary embolism |
| Hypothyroidism | Autoimmune (SLE, RA, Sjögren's) |
| Peritoneal dialysis | Pancreatitis |
| Constrictive pericarditis | Viral pleuritis |
| Meigs' syndrome | Chylothorax (lymphatic obstruction) |
| Hemothorax (trauma, malignancy) | |
| Drugs (amiodarone, nitrofurantoin, methotrexate) |
Clinical Features
| Symptom / Sign | Details |
|---|---|
| Dyspnea | Progressive; proportional to size of effusion |
| Pleuritic chest pain | Exudative effusions (especially parapneumonic, PE, TB) |
| Dry cough | Compression of adjacent lung |
| Decreased chest expansion | Ipsilateral |
| Stony dull percussion | Hallmark; dull over fluid |
| Absent/reduced breath sounds | Over the effusion |
| Bronchial breathing | At upper border of effusion (compressed lung) |
| Tracheal/mediastinal shift | Away from large effusion; toward small effusion (if collapse) |
| Aegophony | "E → A" change at fluid level |
Investigations
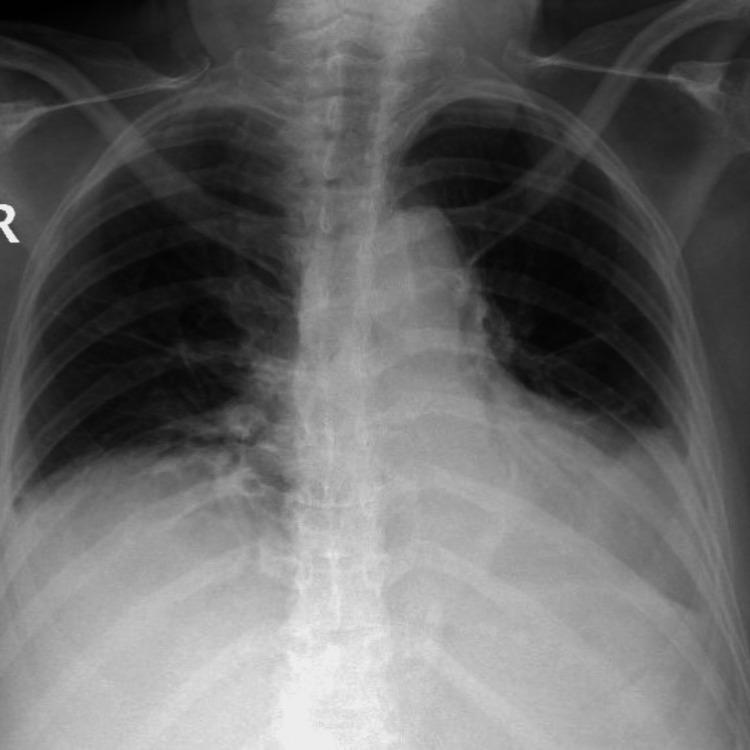
Imaging
- Small effusion (>200 mL): blunting of costophrenic angle
- Moderate: homogeneous basal opacity with meniscus sign
- Large: complete hemithorax opacification with contralateral mediastinal shift
- Subpulmonic effusion: apparent elevated hemidiaphragm
- Detects as little as 20 mL
- Guides thoracentesis (current standard of care — Harrison's, p. 7875)
- Distinguishes free fluid from loculated/solid
- Identifies underlying cause (malignancy, lymphadenopathy, loculations)
- Characterizes effusion (simple, complex, empyema)
Pleural Fluid Analysis
| Test | Significance |
|---|---|
| Appearance | Clear (transudate), turbid (exudate/empyema), bloody (malignancy/hemothorax/PE), milky (chylothorax) |
| Protein + LDH | Light's criteria (transudate vs. exudate) |
| Glucose | Low (<60 mg/dL) → empyema, RA, TB, malignancy |
| pH | <7.2 → empyema (drain required); <7.0 → frank pus |
| Cell count + differential | Neutrophils → parapneumonic/PE; Lymphocytes → TB/malignancy |
| Cytology | Malignant cells (positive in ~60% of malignant effusions) |
| Gram stain + culture | Empyema diagnosis |
| ADA (adenosine deaminase) | >40 U/L strongly suggests TB |
| Triglycerides | >110 mg/dL → chylothorax |
| Hematocrit (fluid/blood) | >0.5 → hemothorax |
| Mesothelin | Elevated in mesothelioma |
| Amylase | Elevated in pancreatitis-related effusion or esophageal rupture |
Management
General Approach
Pleural effusion detected
↓
Clinical context clear? (e.g., bilateral effusions + CHF)
↓
YES → Treat underlying cause first
↓
No response or unilateral/exudative features?
↓
Thoracentesis (diagnostic ± therapeutic)
↓
Analyze fluid → Light's criteria + full panel
↓
Guide specific management
Specific Management
| Type | Management |
|---|---|
| Transudative (CHF) | Diuretics; treat heart failure |
| Transudative (cirrhosis/hepatic hydrothorax) | Salt restriction, diuretics; TIPS; liver transplant |
| Parapneumonic (uncomplicated) | Antibiotics alone |
| Complicated parapneumonic / Empyema | Chest drain + antibiotics; fibrinolytics (tPA/DNase) if loculated; surgery if persistent |
| Malignant | Therapeutic thoracentesis for symptom relief; pleurodesis (talc) for recurrent; indwelling pleural catheter (IPC) |
| TB effusion | Anti-TB therapy (HRZE × 2 months → HR × 4 months); corticosteroids may reduce fibrous thickening |
| Hemothorax | Chest drain; surgical intervention if >1.5 L initial or ongoing bleeding |
| Chylothorax | Low-fat diet / TPN; octreotide; surgical ligation of thoracic duct |
Pleurodesis
- Aims to obliterate pleural space by inducing inflammation/fibrosis
- Agents: Talc (most effective), bleomycin, doxycycline
- Indication: recurrent malignant or benign symptomatic effusions
2. Dry Pleuritis (Fibrinous Pleuritis)
Definition
Pathophysiology
Pleural injury / inflammation
↓
Increased vascular permeability → fibrin-rich exudate
↓
Fibrin deposits on visceral + parietal pleura
↓
Roughened pleural surfaces rub together on breathing
↓
PLEURITIC CHEST PAIN (friction rub)
↓
If inflammation progresses → fluid accumulation → wet pleuritis (effusion)
Causes
| Category | Examples |
|---|---|
| Infections | Viral (Coxsackie B, influenza, EBV — most common), bacterial pneumonia, TB, fungal |
| Autoimmune | SLE (serositis), Rheumatoid arthritis, Sjögren's, MCTD |
| Pulmonary | Pulmonary embolism/infarction, pneumonia (parapneumonic) |
| Cardiac | Pericarditis (pleuropericarditis) — Dressler's syndrome post-MI |
| Neoplastic | Pleural metastases, mesothelioma |
| Drugs | Hydralazine, procainamide, isoniazid (drug-induced lupus) |
| Metabolic | Uraemia |
| Trauma | Rib fracture, thoracic surgery |
| Idiopathic | No cause identified (~25%) |
Clinical Features
Symptoms
| Feature | Description |
|---|---|
| Pleuritic chest pain | Sharp, stabbing, well-localized; worsens with inspiration, coughing, sneezing; relieved by breath-holding or lying on affected side |
| Dyspnea | Due to splinting (voluntary shallow breathing to avoid pain) |
| Fever | Depending on underlying cause |
| Cough | Dry, painful |
| Referred pain | Diaphragmatic pleuritis → shoulder tip pain (phrenic nerve C3,4,5) |
Signs
| Sign | Details |
|---|---|
| Pleural friction rub | Pathognomonic — leathery, creaking sound heard in both inspiration and expiration; localized; does not clear with coughing |
| Reduced chest expansion | Ipsilateral (splinting) |
| Tachypnea | Rapid shallow breathing |
| Normal percussion and breath sounds | (No fluid) |
Key distinction: Pleural friction rub vs. pericardial friction rub — pleural rub disappears when patient holds breath; pericardial rub persists.
Investigations
| Investigation | Purpose |
|---|---|
| CXR | Usually normal; may show underlying pneumonia, small effusion, or rib fracture |
| USS chest | Confirms no significant effusion |
| ECG | Exclude pericarditis (saddle-shaped ST elevation in pleuropericarditis) |
| CBC, CRP, ESR | Inflammatory markers |
| Viral serology | If viral cause suspected |
| ANA, anti-dsDNA, RF, ANCA | If autoimmune cause suspected |
| D-dimer / CTPA | If PE suspected |
| Blood cultures | If bacterial cause suspected |
Management
Symptomatic (Core Treatment)
| Treatment | Details |
|---|---|
| NSAIDs | First-line analgesic and anti-inflammatory — ibuprofen 400–600 mg TDS, indomethacin 25–50 mg TDS |
| Colchicine | 0.5 mg BD × 3 months; particularly effective in recurrent pleuritis and pleuropericarditis; reduces recurrence |
| Corticosteroids | Reserved for NSAID-refractory cases or autoimmune-related pleuritis; prednisolone 0.2–0.5 mg/kg/day |
| Opioids | Short-term for severe pain unresponsive to NSAIDs |
Treat Underlying Cause
- Viral → supportive
- Bacterial pneumonia → antibiotics
- TB → anti-TB therapy
- PE → anticoagulation
- SLE/RA → disease-modifying agents, corticosteroids
- Uraemic pleuritis → dialysis
Patient Advice
- Rest, avoid exertion during acute phase
- Deep breathing exercises once pain controlled (prevent atelectasis)
- Splinting with pillow against chest wall when coughing
3. Pneumothorax
Definition
Classification
Pneumothorax
├── Spontaneous
│ ├── Primary (PSP) — no underlying lung disease
│ └── Secondary (SSP) — complicates existing lung disease
├── Traumatic — penetrating/blunt chest trauma, iatrogenic
└── Tension pneumothorax — air enters but cannot escape (one-way valve) → EMERGENCY
Primary Spontaneous Pneumothorax (PSP)
- Young, tall, thin males (20–30 years)
- Due to rupture of apical subpleural blebs/bullae
- No underlying lung disease
- Smoking increases risk 20-fold
- Recurrence rate: ~30% first episode, ~60% after second
Secondary Spontaneous Pneumothorax (SSP)
- Complicates underlying lung pathology
- More serious — less reserve lung function
| Cause | Examples |
|---|---|
| Obstructive | COPD (most common), asthma, CF |
| Infectious | Pneumocystis jirovecii pneumonia, TB, necrotizing pneumonia |
| Interstitial | LAM, Langerhans cell histiocytosis, IPF |
| Connective tissue | Marfan syndrome, Ehlers-Danlos syndrome |
| Malignancy | Cavitating lung cancer |
Iatrogenic Pneumothorax
- Central line insertion (subclavian/internal jugular)
- Thoracentesis, pleural biopsy
- Mechanical ventilation (barotrauma)
- CT-guided lung biopsy
Tension Pneumothorax — EMERGENCY
One-way valve mechanism:
Air enters pleural space on inspiration → cannot exit on expiration
↓
Progressive ↑ intrapleural pressure
↓
Complete ipsilateral lung collapse
↓
Mediastinal shift → contralateral lung compression
↓
↓ Venous return → ↓ Cardiac output → Obstructive SHOCK
↓
DEATH if not immediately decompressed
- Severe respiratory distress + tachycardia
- Tracheal deviation away from affected side
- Absent breath sounds ipsilateral
- Hypotension + raised JVP (Beck's triad variant)
- Cyanosis
Do NOT wait for CXR in suspected tension pneumothorax — treat immediately.
Clinical Features
| Feature | PSP | SSP | Tension |
|---|---|---|---|
| Onset | Sudden, at rest | Sudden | Rapid deterioration |
| Pain | Ipsilateral pleuritic | Variable | Severe |
| Dyspnea | Mild–moderate | Severe (less reserve) | Profound |
| Tracheal shift | Absent | Variable | Away from side |
| Haemodynamics | Stable | May be compromised | Shock |
| Percussion | Hyperresonant | Hyperresonant | Hyperresonant |
| Breath sounds | ↓ ipsilateral | ↓ ipsilateral | Absent |
Investigations
| Investigation | Finding |
|---|---|
| CXR (PA erect) | Visible pleural line with absent lung markings; estimate size |
| CT chest | Gold standard for size, underlying blebs/bullae; confirms diagnosis when CXR equivocal |
| Arterial Blood Gas | Hypoxemia ± hypercapnia (SSP) |
| ECG | Tachycardia; axis shift (large pneumothorax) |
| Ultrasound | Loss of lung sliding; absent B-lines; M-mode: barcode sign (replaces seashore sign) |
Imaging
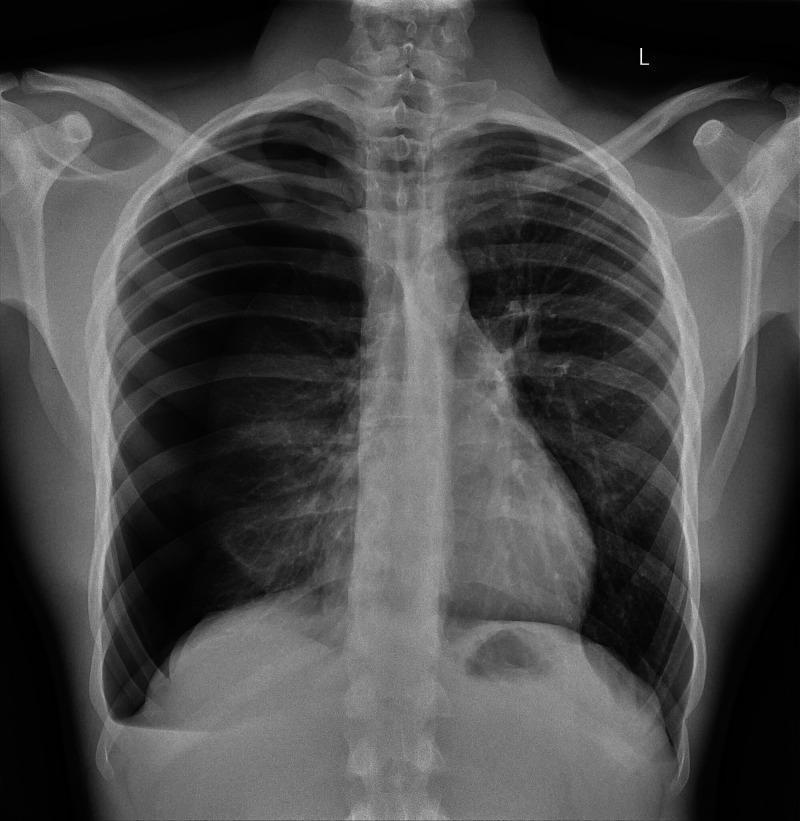
Size Estimation
BTS Classification
| Size | CXR measurement (apex to cupola) |
|---|---|
| Small | <2 cm rim |
| Large | ≥2 cm rim |
ACCP Classification
| Size | Estimation |
|---|---|
| Small | <3 cm apex-to-cupola distance |
| Large | ≥3 cm |
Management
Tension Pneumothorax (Immediate)
- Needle thoracostomy (emergency decompression):
- 2nd intercostal space, midclavicular line — large-bore cannula (14G)
- Definitive: chest tube insertion (5th ICS, anterior axillary line) immediately after
- High-flow oxygen
- Do NOT delay for imaging
Spontaneous Pneumothorax — BTS Guidelines
Primary Spontaneous Pneumothorax (PSP)
PSP confirmed on CXR
↓
Breathless AND large (≥2 cm)?
↓
NO → Conservative management (discharge, review in 2–4 weeks)
↓
YES → Aspiration (16–18G cannula, 2nd ICS MCL)
↓
Successful (<2 cm residual, improved symptoms)?
↓
YES → Discharge + review NO → Chest drain insertion
Secondary Spontaneous Pneumothorax (SSP)
SSP confirmed
↓
Any size SSP = hospital admission
↓
Small (<1 cm) + minimally symptomatic → Observation + O₂
↓
1–2 cm → Aspiration
↓
>2 cm OR failed aspiration → Chest drain
Chest Drain Management
| Aspect | Detail |
|---|---|
| Site | Triangle of safety: 4th–5th ICS, anterior axillary line |
| Size | 8–14 Fr (small-bore Seldinger) for pneumothorax |
| Underwater seal | Single-bottle system; fluid level swings with respiration (tidaling) |
| Bubbling | Continuous bubbling = air leak; cessation = lung re-expanded |
| Suction | Low-pressure (-10 to -20 cmH₂O) if lung fails to re-expand |
| Removal | Once bubbling stopped and CXR confirms re-expansion |
Persistent Air Leak (>5–7 days)
| Option | Details |
|---|---|
| Chemical pleurodesis | Talc slurry or tetracycline via chest drain |
| Video-assisted thoracoscopic surgery (VATS) | Bullectomy + mechanical pleurodesis; preferred for recurrent PSP or persistent air leak |
| Open thoracotomy | Reserved for failed VATS |
Indications for Surgery (VATS/Thoracotomy)
- Second ipsilateral pneumothorax
- First contralateral pneumothorax
- Bilateral simultaneous pneumothorax
- Persistent air leak >5–7 days despite chest drain
- Failure of lung to re-expand
- High-risk occupation (pilot, diver) — even after first episode
- Haemopneumothorax
Oxygen Therapy in Pneumothorax
- High-flow 100% oxygen (reservoir mask) accelerates pleural air absorption 3–4 times faster than breathing room air (replaces nitrogen in pleural space with rapidly absorbed oxygen)
- Use with caution in SSP with COPD (target SpO₂ 88–92%)
Complications
| Complication | Notes |
|---|---|
| Tension pneumothorax | Life-threatening; can occur spontaneously or during ventilation |
| Haemopneumothorax | Blood + air in pleural space; large vessel or adhesion tear |
| Recurrence | PSP 30% first recurrence; SSP even higher |
| Re-expansion pulmonary edema | Rapid re-expansion of collapsed lung after drainage; unilateral edema |
| Empyema | Infection of pleural space (post-chest drain complication) |
| Chronic pneumothorax | Air persists > 3 months |
| Respiratory failure | Particularly in SSP with underlying disease |
Summary Comparison Table
| Feature | Pleural Effusion | Dry Pleuritis | Pneumothorax |
|---|---|---|---|
| Pathology | Fluid in pleural space | Fibrinous pleural inflammation; no fluid | Air in pleural space |
| Key symptom | Dyspnea | Pleuritic chest pain | Sudden dyspnea + pain |
| Percussion | Stony dull | Normal | Hyperresonant |
| Breath sounds | Absent/reduced | Normal | Absent/reduced |
| Pathognomonic sign | Meniscus on CXR | Pleural friction rub | Visible pleural line on CXR |
| Key investigation | Thoracentesis + Light's criteria | Clinical diagnosis; CXR normal | CXR ± CT |
| Core treatment | Treat cause ± drain ± pleurodesis | NSAIDs + colchicine | Needle decompression / chest drain / VATS |
| Emergency variant | Tension hydrothorax (rare) | None | Tension pneumothorax |