Fecal calprotectin, zinc and testosterone production
fecal calprotectin intestinal inflammation biomarker
zinc deficiency testosterone synthesis
zinc role in testosterone biosynthesis fecal calprotectin gut inflammation mechanism 2024 2025
calprotectin zinc chelation intestinal epithelium mechanism
Fecal Calprotectin, Zinc, and Testosterone Production
1. Fecal Calprotectin
What it is
Clinical uses
| Use | Detail |
|---|---|
| IBD vs. IBS differentiation | Sensitivity 83–99%, specificity 53–96% across meta-analyses; optimal cut-off ~50–60 µg/g |
| Disease activity monitoring | Falls before clinical response to treatment; predicts mucosal healing |
| Relapse prediction | Rising calprotectin in clinical remission identifies patients needing treatment escalation |
| Non-IBD causes of elevation | Colorectal carcinoma, chronic NSAID use, bacterial infections, diverticular disease |
Diagnostic algorithm
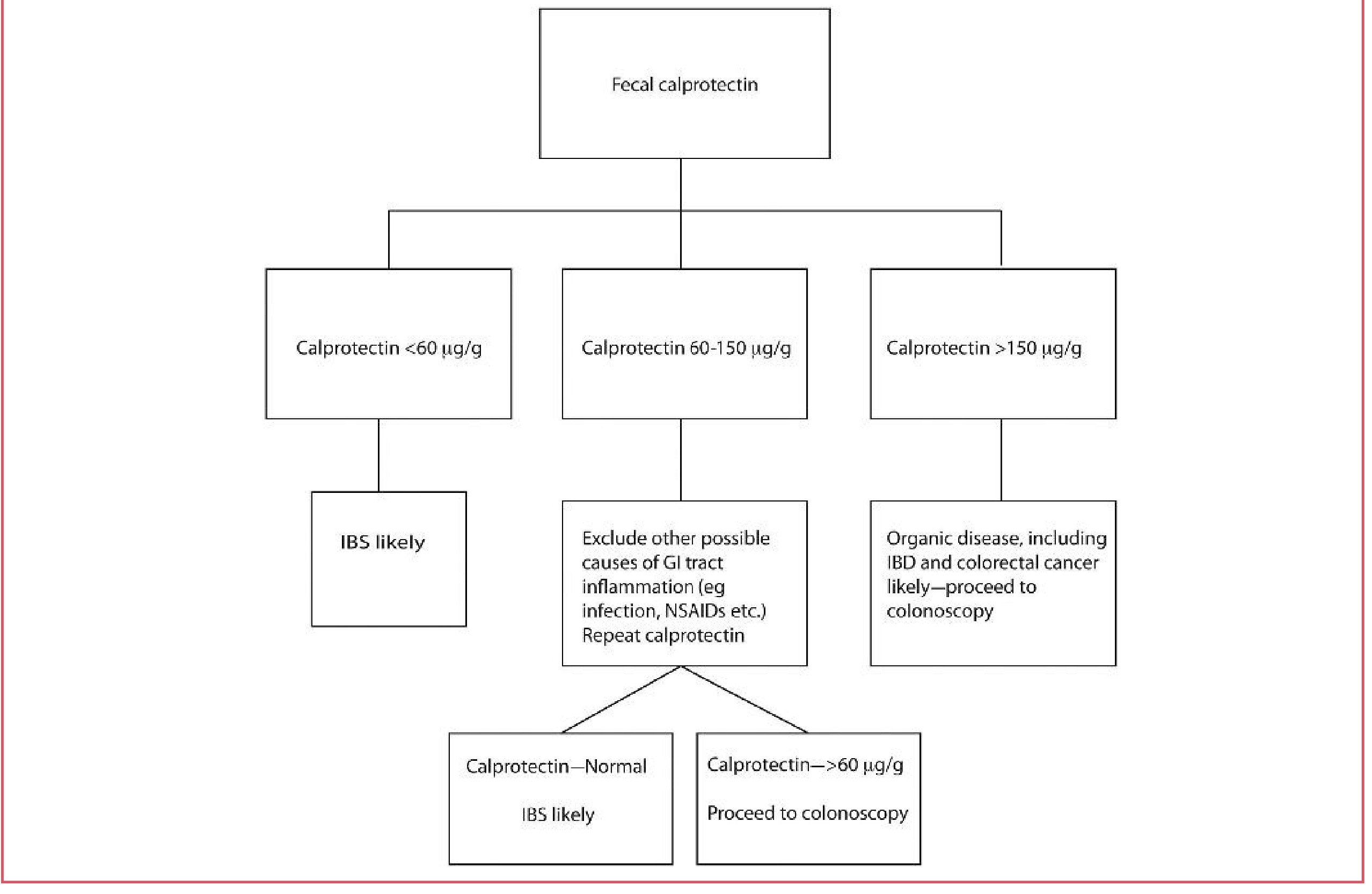
- < 60 µg/g → IBS likely
- 60–150 µg/g → Exclude infection/NSAIDs, repeat; if still > 60 µg/g → colonoscopy
- > 150 µg/g → Organic disease (IBD, colorectal cancer) likely → colonoscopy
2. Zinc and Testosterone Production
Zinc's structural and enzymatic roles in steroidogenesis
-
Steroid hormone receptor zinc fingers — All steroid receptors (androgen receptor, glucocorticoid receptor, etc.) contain a highly conserved central C domain with two zinc finger motifs responsible for DNA binding. When testosterone binds its receptor, the receptor undergoes conformational change and the activated hormone-receptor complex enters the nucleus to stimulate transcription of target genes. — Costanzo Physiology, 7th Ed., p. 3689
-
5α-Reductase activity — Zinc influences the conversion of testosterone to the more potent 5α-dihydrotestosterone (DHT) in peripheral tissues. The biotransformation of these steroids is complex and zinc-dependent. — Fitzpatrick's Dermatology, p. 3212
-
Direct effect on the hypothalamic-pituitary-gonadal axis — Zinc deficiency in humans suppresses circulating testosterone. The landmark study by Prasad et al. (1996) — cited in Tietz Textbook of Laboratory Medicine — demonstrated a direct relationship between zinc status and serum testosterone levels in healthy adults. Both zinc restriction in young men and zinc supplementation in marginally zinc-deficient elderly men produced significant, corresponding changes in serum testosterone.
-
Prostate and seminal zinc — The prostate accumulates the highest concentrations of zinc in the body; prostatic secretions are rich in zinc, which plays a role in spermatozoa motility and function. — Campbell-Walsh-Wein Urology
Zinc deficiency → hypogonadism pathway
↓ Dietary zinc / ↓ intestinal absorption
↓
↓ Leydig cell testosterone synthesis
↓ LH pulsatility (zinc modulates GnRH/LH axis)
↓
Hypogonadotropic hypogonadism phenotype
(low testosterone, oligospermia, delayed puberty in children)
3. The Calprotectin–Zinc–Testosterone Connection
Calprotectin chelates zinc in the gut lumen
Intestinal inflammation reduces zinc bioavailability
- Calprotectin upregulates the zinc-absorptive transporter Slc39a4 (ZIP4) in intestinal epithelial cells, suggesting a compensatory but potentially insufficient adaptation.
- The metal-chelating function of calprotectin is required for its effects on epithelial zinc metabolism and cell proliferation — a metal-binding-deficient calprotectin mutant lost these effects.
Chronic hyperzincemia paradoxically raises calprotectin
Clinical implication: IBD → zinc deficiency → low testosterone
| Step | Mechanism |
|---|---|
| Active IBD (↑ fecal calprotectin) | Neutrophil-derived calprotectin sequesters luminal Zn²⁺ |
| Malabsorption + chelation | Zinc absorption is impaired |
| Zinc deficiency | ↓ LH pulsatility, ↓ Leydig cell function, ↓ testosterone |
| Clinical picture | Fatigue, reduced libido, hypogonadism symptoms in IBD patients |
Key References
- Tietz Textbook of Laboratory Medicine, 7th Ed. — Calprotectin, fecal markers
- Costanzo Physiology, 7th Ed. — Steroid hormone receptor zinc finger mechanism
- Prasad AS et al. (1996). "Zinc status and serum testosterone levels of healthy adults." Nutrition 12:344–8 [cited in Tietz, ref. 923]
- PMC11913417 (2025) — Calprotectin regulates intestinal zinc metabolism via Slc39a4 upregulation in organoids
- PMID 40943053 (2025, Systematic Review/Meta-Analysis) — Non-invasive markers in ulcerative colitis including fecal calprotectin
Search for recent research on zinc bioavailability in different IBD phenotypes
zinc deficiency AND "inflammatory bowel disease"
zinc bioavailability AND "Crohn's disease" AND absorption
zinc bioavailability IBD Crohn ulcerative colitis phenotype 2024 2025
zinc bioavailability Crohn disease ulcerative colitis phenotype small bowel location absorption mechanism
zinc transporter ZIP4 ZnT IBD intestinal epithelium
zinc supplementation Crohn disease clinical trial outcomes
zinc deficiency prevalence IBD CD UC phenotype location clinical outcomes predictors
Zinc Bioavailability in Different IBD Phenotypes: Recent Research
Overview
1. Prevalence by IBD Phenotype
Meta-analyses
| IBD Subtype | Population | Zinc Deficiency Prevalence |
|---|---|---|
| Crohn's disease | n = 1,677 | 54% (95% CI: 51–56%) |
| Ulcerative colitis | n = 806 | 41% (95% CI: 38–45%) |
| Overall | Combined | 50% (95% CI: 48–52%) |
One in two IBD patients suffers from zinc deficiency. Heterogeneity was high (I² = 96%), reflecting differences in cut-off values and populations.
- Pooled prevalence: 35% (95% CI: 19–52%; I² = 98.5%) across the broader IBD population
- Subgroup: CD = 40% (95% CI: 21–59%) vs. UC = 33% (95% CI: 18–51%)
The numerical differences between the two meta-analyses likely reflect different inclusion criteria and cut-off definitions, but the directional finding is consistent: CD > UC.
2. Why CD Has Greater Zinc Deficiency: Phenotype-Specific Mechanisms
Anatomy of absorption
"Zn deficiency has been most well-established in Crohn's disease... primarily caused by reduced absorption of dietary Zn, even when the tissue appears normal or even in remission." — Frontiers in Nutrition, 2025
CD disease location and phenotype (Montreal Classification)
| CD Location | % of cohort |
|---|---|
| L1 (ileal) | 15.8% |
| L2 (colonic) | 9.6% |
| L3 (ileocolonic) | 73.7% |
| L4 (upper GI) | 2.3% |
| CD Behaviour | % of cohort |
|---|---|
| B1 (non-stricturing/non-penetrating) | 29.7% |
| B2 (stricturing) | 18.2% |
| B3 (penetrating) | 43.5% |
| Perianal disease | 38.3% |
- Zinc deficiency was significantly more prevalent in CD vs. UC (OR favoring UC: 0.60, 95% CI 0.37–0.98)
- Younger age and shorter disease duration independently associated with zinc deficiency — suggesting nutritional deterioration happens early and doesn't necessarily self-correct
- Males were less likely to be zinc-deficient (OR 1.48 for normal zinc in males)
- Prior bowel resection was not significantly associated with zinc deficiency in multivariate analysis, though biologically expected
Local vs. systemic deficiency in UC
"Measurements of systemic Zn (i.e., circulating Zn levels) may not accurately reflect tissue Zn status in patients with ulcerative colitis." — Frontiers in Nutrition, 2025
3. Mechanisms Reducing Zinc Bioavailability in IBD
A. Reduced absorptive surface (CD)
- Transmural inflammation → villous atrophy and crypt distortion
- Fistulae and strictures → rapid transit, bypassing absorptive segments
- Prior ileal resection → loss of absorptive mucosa
B. Increased luminal chelation (both phenotypes)
- Elevated fecal calprotectin (S100A8/A9) sequesters luminal Zn²⁺ via nutritional immunity — previously discussed
- Inflamed mucosa increases secretion of zinc-chelating metalloproteins
C. Dietary phytate load
- Many IBD patients self-restrict to low-residue diets rich in grains and cereals, increasing the phytate/zinc molar ratio — a validated predictor of poor zinc bioavailability. High phytate intake reduces net zinc absorption by competitive chelation in the gut lumen. — Frontiers in Nutrition, 2025
D. ZIP/ZnT transporter dysregulation
- The SLC39 (ZIP) family facilitates zinc uptake into cells; SLC30 (ZnT) family exports zinc from cells
- Inflammation downregulates ZIP4 expression in enterocytes (the primary absorptive transporter)
- Calprotectin compensatorily upregulates Slc39a4 (ZIP4) in organoid models — but whether this is sufficient in vivo during severe IBD flares remains unknown (PMC11913417, 2025)
- ZnT2 deletion in animal models paradoxically improved outcomes in infectious colitis, possibly via reduced TLR4/NF-κB signaling — highlighting the complexity of targeting these pathways therapeutically
E. Increased fecal losses
- Exudative enteropathy from inflamed mucosa → protein and zinc loss into lumen
- Diarrhea increases stool zinc excretion
F. Medication effects
- Corticosteroids alter zinc metabolism
- Some immunosuppressants and biologics may modestly affect trace element homeostasis
4. Zinc Supplementation Evidence in IBD
- Zinc supplementation relieves IBD severity, particularly in zinc-deficient patients
- Mechanisms: immunomodulation (Th1/Th2/Th17 rebalancing), intestinal epithelial barrier repair, gut microbiota normalization, antioxidant activity (inhibiting IL-1β and IL-18)
- Animal models: dietary zinc at 160 ppm was optimal; higher doses (400–1000 ppm) conferred no additional benefit and may be counterproductive
- Clinical severity scores (CDAI, endoscopic indices) improved with zinc repletion
- Children with IBD face amplified risk because zinc is critical for growth and immune development
- Routine zinc screening is recommended; deficiency in pediatric CD can impair growth velocity independently of caloric intake
- ESPEN 2023 guidelines endorse zinc monitoring as part of IBD nutritional assessment
5. Research Gaps and Emerging Questions
| Gap | Status |
|---|---|
| Colonic zinc homeostasis in UC | Nearly entirely unexplored |
| Whether tissue zinc deficiency in UC predicts outcomes | No prospective data |
| Optimal zinc form and dose for IBD supplementation | No RCTs with standardized endpoints |
| Effect of biologic therapy (anti-TNF, anti-integrin) on zinc restoration | Limited data |
| Genetic polymorphisms in ZIP/ZnT transporters and IBD susceptibility | Early data from Dragasevic et al. 2022 [PMID: 36295058] |
| Whether zinc repletion reduces calprotectin levels and impacts IBD course | Correlation data available; causal RCT data lacking |
Summary Table
| Feature | Crohn's Disease | Ulcerative Colitis |
|---|---|---|
| Zinc deficiency prevalence | ~40–54% | ~33–41% |
| Primary mechanism | Malabsorption (small bowel disease) | Reduced intake; local mucosal deficiency |
| Key risk factors | Ileocolonic location, penetrating phenotype, young age | Active flare, restricted diet |
| Serum zinc as surrogate | Reasonably reflects absorptive status | May underestimate local tissue deficiency |
| Supplementation evidence | Moderate; benefits in deficient patients | Less studied |
| Current guideline recommendation | Routine screening; supplement if deficient (ESPEN 2023) | Screen; supplement if deficient |
Key References
| Evidence | Citation |
|---|---|
| [Systematic Review · 2022] | Zupo R et al. "Prevalence of Zinc Deficiency in IBD." Nutrients 14:4052. [PMID: 36235709] |
| [Review · 2025] | Peng X et al. "Zinc and IBD: From Clinical Study to Animal Experiment." Biol Trace Elem Res 2025. [PMID: 38805169] |
| [Review · 2025] | Galeazzi T et al. "Micronutrient Deficiencies in Pediatric IBD." Nutrients 17:1425. [PMID: 40362741] |
| [Review · 2022] | Dragasevic S et al. "Genetic Aspects of Micronutrients Important for IBD." Life (Basel). [PMID: 36295058] |
| [Meta-Analysis · 2025] | "Zinc Deficiency Among Patients With IBD." PMC12951249 |
| [Cohort Study · 2025] | "Prevalence and Impact of Zinc Deficiency on Clinical Outcomes in IBD." PMC12610508 |
| [Review · 2025] | "Roles of zinc and zinc transporters in IBD." Front Nutr 2025 (doi: 10.3389/fnut.2025.1649658) |