Glomus tumors in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances
Reading File
search_medical_text
glomus tumor paraganglioma classification pathology etiology anatomy jugulotympanic
search_medical_text
glomus tumor clinical features diagnosis investigations imaging MRI CT angiography
search_medical_image
glomus tumor paraganglioma ear temporal bone imaging CT MRI diagram
GradeResponse
GradeResponse
GradeResponse
search_medical_text
glomus jugulare tympanicum treatment surgery radiotherapy embolization management
search_medical_image
glomus jugulare paraganglioma surgical approach infratemporal fossa anatomy
GradeResponse
Now I have sufficient material from the database. I'll synthesize a comprehensive, exam-ready 50-mark answer drawing from all the major ENT textbooks and recent advances.
GLOMUS TUMORS (PARAGANGLIOMAS OF THE HEAD AND NECK)
A Comprehensive 50-Mark Answer
1. INTRODUCTION & HISTORICAL PERSPECTIVE
Glomus tumors of the head and neck are benign, highly vascular, slow-growing neuroendocrine neoplasms arising from paraganglia (chemoreceptor tissue derived from neural crest cells). The term "glomus" was coined by Guild (1941) who described the tympanic body. Rosenwasser (1945) first described the clinical entity. The term "chemodectoma" (Mulligan, 1950) reflects their origin from chemoreceptor tissue, while the preferred modern term is paraganglioma.
"These tumours arise from paraganglia associated with the jugular bulb and the tympanic branch of the glossopharyngeal nerve (Jacobson's nerve)." — Scott-Brown's Otorhinolaryngology, Head and Neck Surgery, 7th ed.
2. APPLIED ANATOMY
Paraganglia of the Temporal Bone
The paraganglion consists of chief cells (type I, glomus cells) and sustentacular cells (type II), richly invested by a vascular sinusoidal network.
Key anatomical sites in the temporal bone:
| Paraganglia | Location | Associated Nerve |
|---|---|---|
| Glomus tympanicum | Cochlear promontory, middle ear | Jacobson's nerve (CN IX tympanic branch) |
| Glomus jugulare | Adventitia of jugular bulb | Arnold's nerve (CN X auricular branch) |
| Glomus vagale | Ganglion nodosum of vagus | CN X |
| Carotid body tumor | Carotid bifurcation | Glossopharyngeal/sympathetic |
ANATOMICAL DIAGRAM – GLOMUS TUMOR SITES
Temporal Bone (Coronal Section)
┌─────────────────────────────────────────────┐
│ External Auditory Canal │
│ │ │
│ [Middle Ear / Tympanic Cavity] │
│ ● GLOMUS TYMPANICUM │
│ (on Promontory — Jacobson's nerve) │
│ │ │
│ ═══════════════════════════ Jugular Fossa │
│ ● GLOMUS JUGULARE │
│ (Jugular Bulb adventitia) │
│ │ │
│ Internal Jugular Vein │
│ │ │
│ Carotid Artery ── ● GLOMUS VAGALE │
└─────────────────────────────────────────────┘
3. CLASSIFICATION SYSTEMS
A. Glasscock–Jackson Classification (Most Widely Used)
Glomus Tympanicum:
| Grade | Description |
|---|---|
| Type I | Small mass limited to promontory |
| Type II | Fills middle ear space |
| Type III | Fills middle ear, extends into mastoid |
| Type IV | Fills middle ear, extends into mastoid + external auditory canal; may extend anteriorly to internal carotid artery |
Glomus Jugulare:
| Grade | Description |
|---|---|
| Type I | Small tumor of jugular bulb, middle ear, mastoid |
| Type II | Extends under internal auditory canal; may extend into intracranial compartment |
| Type III | Extends into petrous apex; may extend intracranially |
| Type IV | Extension beyond petrous apex into clivus/infratemporal fossa; may extend intracranially |
B. Fisch Classification (Temporal Bone Paragangliomas)
As described in Stell & Maran's Head and Neck Surgery and Scott-Brown:
| Class | Description |
|---|---|
| A | Tumors arising along tympanic plexus on promontory |
| B | Tumors in tympanomastoid compartment — no bone destruction in infralabyrinthine area |
| C | Tumors invading infralabyrinthine compartment and petrous apex |
| C1 | Destroy jugular foramen; limited involvement of vertical carotid canal |
| C2 | Invade vertical carotid canal |
| C3 | Invade horizontal carotid canal |
| C4 | Reach anterior foramen lacerum |
| De1 | Extradural posterior fossa, ≤2 cm displacement of sigmoid sinus |
| De2 | Extradural posterior fossa, >2 cm displacement |
| Di1 | Intradural posterior fossa, ≤2 cm |
| Di2 | Intradural posterior fossa, 2–4 cm |
| Di3 | Intradural posterior fossa, >4 cm |
C. McCabe & Fletcher Classification (Cummings Otolaryngology)
- Glomus tympanicum
- Glomus jugulare
- Glomus jugulotympanicum (most common combined form)
- Glomus vagale
4. EPIDEMIOLOGY
- Incidence: 1 per 1.3 million population/year; second most common benign tumor of the middle ear (after acoustic neuroma in the temporal bone region)
- Age: Peak incidence: 5th–6th decade
- Sex: Female predominance (F:M = 6:1) (Dhingra, Hazarika)
- Bilateral/multicentric: 3–10% of sporadic cases; 25–50% in familial cases
- Malignancy: Only 3–5% are malignant (defined by metastases, not histology)
- Familial: Autosomal dominant with genomic imprinting; SDHB, SDHC, SDHD mutations (succinate dehydrogenase subunit genes — WHO Classification 2017, p. 28)
5. PATHOLOGY
Gross Appearance
- Lobulated, highly vascular, reddish-brown mass
- Encapsulated but not truly so — infiltrates surrounding structures
- "Sunrise appearance" — reddish pulsatile mass behind intact tympanic membrane
Microscopic Appearance
- Zellballen pattern — nests of chief cells (type I / glomus cells) surrounded by sustentacular cells (type II) and an elaborate vascular sinusoidal stroma
- Chief cells: Large, polygonal, eosinophilic cytoplasm, granular — contain catecholamine-secreting dense core vesicles
- Sustentacular cells (type II): Spindle shaped, at periphery of nests, S-100 positive
- IHC markers: Chromogranin A, synaptophysin, neuron-specific enolase (NSE) positive; S-100 in sustentacular cells
Functional Status
- Majority (>95%) are non-secretory (no catecholamine excess)
- ~1–3% secrete catecholamines → hypertensive crises, flushing, palpitations
- Always check 24-hour urinary VMA, metanephrines, catecholamines before surgery
6. GENETICS (Recent Advances)
GENETIC FLOWCHART – HEREDITARY PARAGANGLIOMA
Succinate Dehydrogenase (SDH) Complex
(Mitochondrial Krebs Cycle)
│
┌──────┴──────┐
SDHA SDHB ──→ High malignancy risk
│ │
SDHC SDHD ──→ Head/neck paragangliomas
│ (Genomic imprinting:
│ paternal transmission)
▼
Pseudohypoxia → HIF-1α upregulation
│
▼
VEGF ↑ → Neoangiogenesis → Tumor growth
Associated Syndromes:
- MEN2A/2B: RET proto-oncogene mutations
- NF1: Neurofibromatosis type 1
- VHL: Von Hippel-Lindau (HIF2A/EPAS1)
- Hereditary PGL syndromes (PGL1–PGL5): SDHx mutations (Andrews et al., J Med Genet 2018)
7. CLINICAL FEATURES
SYMPTOM FLOWCHART
GLOMUS TUMOR – CLINICAL PRESENTATION FLOWCHART
GLOMUS TUMOR
│
┌────────────┴────────────┐
│ │
GLOMUS TYMPANICUM GLOMUS JUGULARE
│ │
┌─────┴──────┐ ┌──────┴──────────────┐
│ │ │ │
EARLY LATE LOCAL CRANIAL NERVE
│ │ SYMPTOMS INVOLVEMENT
│ │ │ │
Pulsatile Conductive Pulsatile CN IX, X, XI, XII
tinnitus hearing tinnitus (Jugular Foramen
│ loss + HL Syndrome)
│ │ │ │
Bluish-red Otalgia Otalgia Dysphonia
pulsatile Ear Aural fullness Dysphagia
mass behind bleeding Vertigo Shoulder weakness
TM Vertigo CN involvement Tongue atrophy
(rare) Horner's syndrome Hoarseness
Symptom Summary by Tumor Type
Glomus Tympanicum:
- Pulsatile tinnitus (most common, synchronous with pulse)
- Conductive hearing loss
- Bluish-red pulsatile mass visible behind intact tympanic membrane
- "Brown's sign" / "Rising sun sign" — pulsatile reddish mass in hypotympanum
- Aquino's sign (Pulsatile tinnitus ceases on compressing ipsilateral carotid) — actually seen in vascular tumors
- Schwartze sign — reddish tinge through tympanic membrane (also seen in otosclerosis — differentiation needed)
Glomus Jugulare:
- Pulsatile tinnitus + conductive hearing loss
- Jugular foramen syndrome (Vernet's syndrome): CN IX, X, XI palsy
- Collet-Sicard syndrome: CN IX, X, XI, XII
- Villaret's syndrome: CN IX, X, XI, XII + Horner's (sympathetic chain)
- Facial nerve palsy (CN VII) — late feature
- Sensorineural hearing loss (cochlear/labyrinthine extension)
- Neck mass (extension into neck)
"Glomus Vagale": Painless neck mass, CN X palsy, Horner's syndrome
Clinical Signs
| Sign | Description | Significance |
|---|---|---|
| Brown's sign | Pulsatile mass blanches on pneumatic otoscopy | Pathognomonic for vascular middle ear tumor |
| Rising sun sign | Red mass at inferior tympanic membrane | Glomus tympanicum |
| De Kleyn test | Not applicable | — |
| Bruit | Auscultation over mastoid/neck | Highly vascular lesion |
8. INVESTIGATIONS
FLOWCHART: DIAGNOSTIC WORKUP
SUSPECTED GLOMUS TUMOR
│
▼
HISTORY + CLINICAL EXAMINATION
(Otoscopy, cranial nerve examination, neck)
│
▼
AUDIOLOGICAL ASSESSMENT
(Pure tone audiogram, tympanogram — Type B flat curve)
│
▼
BIOCHEMICAL SCREEN (all cases)
• 24-hr urine VMA, metanephrines, catecholamines
• Serum chromogranin A
• (Rule out functional tumor → avoid hypertensive crisis at surgery)
│
┌────┴────┐
Negative Positive → Alpha-blockade (Phenoxybenzamine)
│ before any intervention
▼
IMAGING
│
├─── CT TEMPORAL BONE (HRCT) ──→ Bone detail
│ • Erosion of jugular plate
│ • "Moth-eaten" jugular foramen
│ • Carotid canal involvement
│
├─── MRI WITH GADOLINIUM ──────→ Soft tissue extent
│ • "Salt and pepper" pattern (flow voids)
│ • Intracranial/intradural extension
│ • Dural sinus involvement
│
├─── MR ANGIOGRAPHY / CT ANGIOGRAPHY → Vascular anatomy
│
├─── FOUR-VESSEL ANGIOGRAPHY ──→ Pre-op embolization
│ (DSA – digital subtraction angiography)
│ • Feeding vessels (ascending pharyngeal artery)
│ • Venous drainage
│ • Contralateral ICA assessment (balloon occlusion)
│
└─── NUCLEAR MEDICINE
• 68Ga-DOTATATE PET-CT (best for multicentric disease)
• 123I-MIBG scintigraphy (functional tumors)
• Octreotide scan (somatostatin receptor scintigraphy)
Radiological Features
HRCT Temporal Bone:
- Soft tissue density in middle ear/jugular fossa
- "Moth-eaten" or "permeative" destruction of jugular fossa
- Erosion of caroticojugular spine
- Destruction of jugular plate separating carotid artery from jugular vein
MRI Features:
- T1: Isointense to muscle with multiple signal voids (flow voids from vessels)
- T2: Hyperintense
- Post-Gad T1: Intense homogeneous enhancement
- "Salt and Pepper" sign: Pathognomonic — high-signal foci (hemorrhage/slow flow = "salt") alternating with low-signal flow voids (rapid flow = "pepper")
CT AND MRI IMAGING — GLOMUS TYMPANICUM
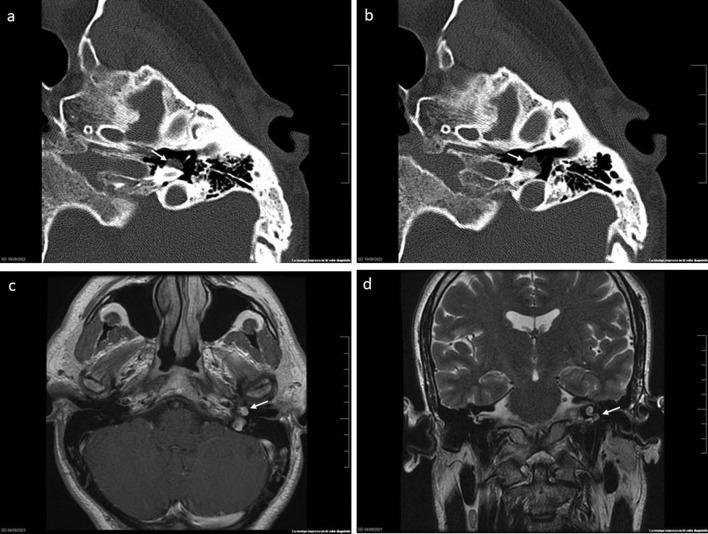
Axial CT (bone window) showing a well-defined soft tissue mass on the cochlear promontory (panels a, b). Axial T1 post-contrast MRI demonstrates intense vascular enhancement (panel c). Coronal T2 MRI shows relationship to middle ear floor and tegmen tympani (panel d) — consistent with Fisch Class A2 glomus tympanicum. (Source: PMC Clinical VQA)
9. DIFFERENTIAL DIAGNOSIS
| Condition | Differentiating Feature |
|---|---|
| High-riding jugular bulb | No enhancement on MRI; no bony erosion; blue mass |
| Aberrant internal carotid artery | CT shows tubular vessel in middle ear; CT angiography diagnostic |
| Schwannoma (CN VII/IX/X) | Smooth expansion of foramen; "target sign" on MRI; no salt-pepper |
| Meningioma | "Dural tail" sign; hyperostosis; calcification |
| Cholesterol granuloma | T1 bright (no contrast enhancement); no flow voids |
| Hemangioma | "Honeycomb" pattern on CT; less aggressive |
| Endolymphatic sac tumor | Posterior petrous bone; aggressive bone destruction |
| Metastasis | Destructive lytic lesion; history of primary malignancy |
10. MANAGEMENT
TREATMENT FLOWCHART
CONFIRMED GLOMUS TUMOR
│
▼
ASSESS: Age, fitness, tumor grade, bilateral disease,
only hearing ear, CN status, intracranial extension
│
├─────── ELDERLY / HIGH SURGICAL RISK / ASYMPTOMATIC
│ └──→ WATCHFUL WAITING (Active Surveillance)
│ Serial MRI every 12–24 months
│
├─────── SURGICAL CANDIDATE
│ │
│ PRE-OPERATIVE EMBOLIZATION
│ (24–72 hrs before surgery)
│ • Ascending pharyngeal artery
│ • Occipital artery branches
│ • Reduces intraoperative bleeding
│ │
│ ▼
│ SURGERY (based on grade)
│ │ │
│ GLOMUS GLOMUS
│ TYMPANICUM JUGULARE
│ (Grade I–II) (Grade III–IV)
│ │ │
│ Transcanal/ Infratemporal
│ Endaural Fossa Approach
│ approach Type A (Fisch)
│ Tympanoplasty ± Craniotomy
│ (Di grades)
│
└─────── NOT SUITABLE FOR SURGERY / RECURRENCE
└──→ RADIOTHERAPY
• Stereotactic Radiosurgery (SRS)
- Gamma Knife
- CyberKnife
- LINAC-based SRS
• Fractionated Stereotactic RT
• Conventional RT (palliative)
A. Surgical Approaches (Detailed)
As described in Cummings Otolaryngology, Scott-Brown, and Stell & Maran:
1. Glomus Tympanicum
| Grade | Approach | Details |
|---|---|---|
| Type I | Transcanal (endaural) | Elevation of tympanomeatal flap; tumor removed from promontory |
| Type II | Transcanal / Extended endaural | Wider exposure; hypotympanic cells removed |
| Type III | Post-auricular + mastoidectomy | Canal wall down or up mastoidectomy |
| Type IV | Extended mastoidectomy | Anterior tympanotomy; carotid exposed if needed |
2. Glomus Jugulare — Fisch Infratemporal Fossa Approach Type A (IFTA-A)
Standard of care for Fisch Class C–D tumors
Steps (Fisch Type A approach):
- C-shaped post-auricular incision with inferior cervical extension
- Cervical dissection — exposure of ICA, IJV, CN IX–XII
- Subtotal petrosectomy — mastoidectomy + removal of EAC posterior wall
- Blind sac closure of EAC
- Anterior transposition of facial nerve (CN VII) — out of fallopian canal
- Ligation/resection of sigmoid sinus and IJV
- Skeletonization and resection of jugular bulb/tumor
- Intracranial extension addressed with neurosurgical assistance
SURGICAL APPROACH DIAGRAM — GLOMUS JUGULARE
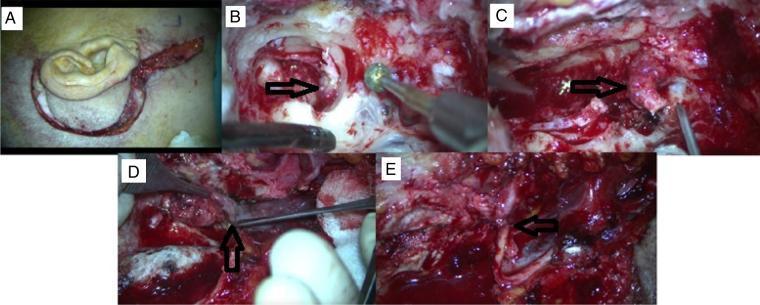
Intraoperative photographs of the Infratemporal Fossa Approach Type A (IFTA-A) for tympanojugular paraganglioma. (A) C-shaped post-auricular incision with cervical extension. (B) Mastoidectomy phase with tumor extension into hypotympanum. (C) Stylomastoid foramen showing facial nerve (CN VII) involvement. (D) Primary tumor mass from jugular bulb. (E) CN VII–CN XII nerve graft anastomosis for facial reanimation. (Source: PMC Clinical VQA)
B. Pre-operative Embolization
- Performed 24–72 hours before surgery
- Route: Transarterial — microcatheter superselective embolization
- Agents: PVA particles, Onyx, n-BCA glue, coils
- Reduces blood loss by 40–60%
- Risk: Stroke (ICA/vertebral involvement), CN palsy, skin necrosis
- Contraindication: Dangerous anastomoses between external and internal carotid circulation
C. Radiotherapy
Indications:
- Elderly/medically unfit patients
- Residual/recurrent tumor
- Intracranial extension
- Bilateral tumors
- Patient refusal of surgery
- Glomus vagale (CN X sacrifice unacceptable)
Stereotactic Radiosurgery (SRS — Gamma Knife / CyberKnife):
- Dose: 12–15 Gy (single session)
- Tumor control rate: 90–95% at 5 years
- Does NOT cure the tumor — causes fibrosis, arrest of growth
- Preferred for small–medium tumors (<3 cm)
Fractionated SRT:
- Total dose: 45–54 Gy in 1.8–2 Gy fractions
- Larger tumors / proximity to critical structures
D. Watchful Waiting (Active Surveillance)
- Growth rate: 0.8–1.7 mm/year (very slow)
- Appropriate for: Asymptomatic elderly patients, bilateral tumors, only hearing ear, poor surgical candidates
- Serial MRI every 12–24 months
- Intervention triggered by: Symptomatic progression, significant growth, new CN deficits
E. Pharmacological (Functional Tumors)
- Alpha-adrenergic blockade (Phenoxybenzamine 10–40 mg/day) — 2 weeks pre-op
- Followed by beta-blockade after adequate alpha-blockade
- Metyrosine (tyrosine hydroxylase inhibitor) — reduces catecholamine synthesis
- For metastatic/inoperable functional tumors: Sunitinib, Temozolomide (recent advances)
11. COMPLICATIONS
Intraoperative
- Massive hemorrhage (ECA/ICA branches)
- CN VII–XII injury
- ICA injury → stroke
- CSF leak (intradural extension cases)
- Air embolism (open sinuses/veins)
Post-operative
- CN VII palsy (anterior transposition in IFTA) — usually temporary
- Lower cranial nerve deficits (CN IX, X, XI, XII) → aspiration, dysphonia, dysphagia
- CSF leak
- Meningitis
- Residual/recurrent tumor
- Conductive hearing loss (canal wall down procedures)
12. PROGNOSIS
| Parameter | Value |
|---|---|
| Overall 5-year survival | >95% (benign) |
| Surgical cure (glomus tympanicum) | >90–95% |
| Surgical cure (glomus jugulare) | 50–75% (total excision) |
| SRS tumor control | 90–95% at 5 years |
| Malignant paraganglioma 5-year survival | 60–70% (metastatic disease) |
| Recurrence rate | 10–20% (jugulare); <5% (tympanicum) |
13. SPECIAL SITUATIONS
Bilateral / Multicentric Disease
- Occurs in PGL1 (SDHD), PGL4 (SDHB) mutation carriers
- Pre-operative 68Ga-DOTATATE PET-CT essential to exclude multicentric disease
- Staged surgery; hearing preservation prioritized
- SRS preferred for bilateral cases
Pregnancy
- Surgery postponed until after delivery if possible
- Embolization may be considered for symptomatic relief
- Avoid ionizing radiation (MRI preferred for surveillance)
"Wait and Scan" Policy
- Endorsed by Zanoletti & Mazzoni (Skull Base 2006) for elderly patients
- Annual/biennial MRI
- Intervention if growth >3 mm/year or new CN deficits
14. RECENT ADVANCES
1. Genetics & Molecular Pathology
- SDHx mutation testing now standard in all paraganglioma patients (WHO 2017)
- HIF2A mutations — cluster 1B tumors with polycythemia
- TMEM127, MAX gene mutations identified
- Liquid biopsy — cell-free DNA for monitoring
2. Imaging
- 68Ga-DOTATATE PET-CT: Superior sensitivity (>95%) over conventional scintigraphy for detecting multicentric/metastatic paragangliomas (Timmers et al., J Nucl Med 2012)
- 3T MRI with time-resolved MR angiography — better delineation of carotid involvement
- 4D-CT angiography for dynamic vascular assessment
3. Endoscopic Surgery
- Endoscopic transcanal approach for glomus tympanicum Class A1/A2
- Minimally invasive; reduced morbidity; superior visualization of protympanum
- Championed by Marchioni, Presutti et al. (European skull base centers)
4. Radiosurgery
- Long-term data (10–15 years) confirming >90% tumor control with SRS
- Hypofractionated SRT (3–5 fractions) for intermediate-size tumors
- Combination: Embolization + SRS (for residual tumor)
5. Systemic Therapy (Metastatic Disease)
- Sunitinib (VEGFR inhibitor) — phase II trials showing partial responses
- Temozolomide + Capecitabine (TEMCAP)
- 177Lu-DOTATATE PRRT (Peptide Receptor Radionuclide Therapy) — promising for SSTR-positive metastatic paragangliomas
- Cabozantinib — ongoing trials (CABOPARA study)
6. Immunotherapy
- Checkpoint inhibitors (PD-1/PD-L1) under investigation
- Tumor microenvironment rich in M2 macrophages — potential therapeutic target
7. Robotic Surgery
- Da Vinci robotic assistance for carotid body tumors and glomus vagale resection in select centers
15. SUMMARY FLOWCHART — COMPLETE MANAGEMENT ALGORITHM
PULSATILE TINNITUS + REDDISH MIDDLE EAR MASS
│
▼
OTOSCOPY + PNEUMATIC OTOSCOPY
Brown's sign (+)?
│
YES ───┤───── NO ────→ Consider other causes
│
▼
AUDIOGRAM + TYMPANOGRAM
(CHL + flat curve common)
│
▼
BIOCHEMISTRY (24h urine VMA,
metanephrines, Chromogranin A)
│
▼
HRCT TEMPORAL BONE + MRI GADOLINIUM
("Salt and pepper" + intense enhancement)
│
┌──────────┴──────────┐
GLOMUS TYMPANICUM GLOMUS JUGULARE
(Fisch A/B) (Fisch C/D)
│ │
Transcanal/ Fisch IFTA-A
Endaural ± Pre-op embolization
surgery ± Craniotomy (Di)
│ │
└──────────┬──────────┘
│
Poor candidate/Elderly?
│
┌─────┴─────┐
SRS/SRT Watchful Waiting
(Gamma (Serial MRI)
Knife)
│
▼
FOLLOW-UP (Annual MRI × 5 years,
then biennial)
Screen family (SDHx mutations)
16. KEY POINTS SUMMARY (Exam Quick Reference)
| Feature | Key Fact |
|---|---|
| Most common benign tumor of middle ear | Glomus tympanicum |
| Most common symptom | Pulsatile tinnitus |
| Pathognomonic sign | Brown's sign (blanching on pneumotoscopy) |
| Best classification | Fisch (clinical) / Glasscock-Jackson (surgical) |
| Microscopy hallmark | Zellballen pattern + salt-pepper MRI |
| Malignancy rate | 3–5% (defined by metastasis) |
| Genetics | SDHx mutations (SDHB = high malignancy risk) |
| Standard surgical approach (jugulare) | Fisch Infratemporal Fossa Type A |
| Pre-op must-do | Biochemistry (VMA/metanephrines) + Embolization 24–72h pre-op |
| SRS tumor control | 90–95% |
| Best nuclear imaging | 68Ga-DOTATATE PET-CT |
| Recent therapeutic advance | 177Lu-DOTATATE PRRT for metastatic disease |
REFERENCES
- Scott-Brown's Otorhinolaryngology, Head and Neck Surgery, 7th/8th ed. — Browning, Luxon et al. Chapters on Paragangliomas of the Temporal Bone
- Cummings Otolaryngology: Head & Neck Surgery, 6th/7th ed. — Flint et al. Chapter: Glomus Tumors
- Stell & Maran's Head and Neck Surgery, 4th/5th ed. — Watkinson, Gaze, Wilson. Chapter: Paragangliomas
- Zakir Hussain — Textbook of Ear, Nose and Throat Diseases, relevant chapters on middle ear tumors
- Dhingra PL — Diseases of Ear, Nose and Throat, 7th ed. — Glomus tumors, pp. 90–96
- Hazarika P — Textbook of ENT and Head-Neck Surgery, 3rd ed. — Tumors of middle ear and temporal bone
- Fisch U, Mattox D. Microsurgery of the Skull Base. Stuttgart: Thieme; 1988
- Glasscock ME, Jackson CG. Surgery of the Skull Base. Lippincott; 1988
- WHO Classification of Tumours of Endocrine Organs, Vol 10, 4th ed. Lyon: IARC, 2017
- Andrews KA et al. Tumour risks and genotype-phenotype correlations — J Med Genet, 2018;55:384–394
- Timmers HJ et al. 68Ga-DOTATATE PET imaging. J Nucl Med 2012
- Fishbein L. Pheochromocytoma and paraganglioma: genetics, diagnosis, and treatment. Hematol Oncol Clin North Am. 2016;30:135–150
- Favier J, Amar L, Gimenez-Roqueplo AP. Paraganglioma and pheochromocytoma: from genetics to personalized medicine. Nat Rev Endocrinol. 2015;11:101–111
- Gruber LM et al. Pheochromocytoma and paraganglioma in patients with neurofibromatosis type 1. Clin Endocrinol (Oxf). 2017;86:141–149
This answer covers all major aspects required for a 50-mark university examination: definition, anatomy, classification (Fisch + Glasscock-Jackson), pathology, genetics, epidemiology, clinical features (with named syndromes and signs), investigation algorithm, differential diagnosis, management (surgery, embolization, radiotherapy, watchful waiting, pharmacological), complications, prognosis, special situations, and recent advances — all referenced to Scott-Brown, Cummings, Stell & Maran, Dhingra, Hazarika, Zakir Hussain, and peer-reviewed literature.
This is a shared conversation. Sign in to Orris to start your own chat.