gene therapy in fracture healing and orthopaedics
Reading File
search_medical_text
gene therapy fracture healing bone repair mechanisms
search_medical_text
gene therapy orthopaedics delivery vectors viral non-viral
search_medical_text
BMP growth factors gene transfer bone regeneration
search_medical_image
gene therapy bone fracture healing diagram
GradeResponse
GradeResponse
GradeResponse
GradeResponse
search_medical_text
gene therapy clinical applications musculoskeletal bone nonunion
search_medical_text
viral vectors adenovirus AAV lentivirus gene delivery orthopaedic
GradeResponse
GradeResponse
Here is a comprehensive overview of gene therapy in fracture healing and orthopaedics:
Gene Therapy in Fracture Healing & Orthopaedics
What Is Gene Therapy?
Gene therapy uses a nucleic acid sequence (DNA or RNA) as the active therapeutic agent rather than a conventional drug or protein. The goal is to deliver a gene of interest into target cells, directing them to produce a specific growth factor or signaling molecule locally and continuously — an advantage over repeated protein injections which have short half-lives and systemic side effects (Harrison's Principles, p. 13361).
Why Gene Therapy for Bone?
Fracture healing and bone regeneration depend on a tightly orchestrated cascade involving growth factors, cytokines, and transcription factors. Conventional delivery of recombinant proteins (e.g., BMP-2 injections) suffers from:
- Short biological half-life at the injury site
- Supraphysiologic doses required (increasing cost and ectopic bone formation risk)
- Systemic dissemination causing off-target effects
Gene therapy overcomes these by enabling sustained, localized production of the therapeutic molecule directly at the repair site.
Key Target Genes in Orthopaedic Gene Therapy
| Gene / Factor | Role in Bone Healing | Application |
|---|---|---|
| BMP-2, BMP-4, BMP-7 | Master osteoinductive signals; recruit MSCs and drive osteoblast differentiation | Fracture non-union, spinal fusion, critical-size defects |
| BMP-6 | Chondrogenic and osteogenic differentiation | Cartilage and bone repair |
| GDF-5 (BMP-14) | Upregulated in osteogenesis; tendon/ligament repair | Rotator cuff, ACL, fracture healing |
| VEGF | Angiogenesis; vascular invasion of callus | Avascular necrosis, large defects |
| LMP-1 (LIM Mineralization Protein) | Intracellular osteogenic enhancer | Spinal fusion |
| Runx2 / Osterix | Osteoblast transcription factors | Osteoporotic fracture, non-union |
| PTHrP / PTH (1-34) | Anabolic bone signaling | Osteoporosis-related fractures |
| Sox9 | Chondrogenesis | Articular cartilage repair, OA |
| IGF-1 | Mitogen for osteoblasts; promotes callus maturation | Delayed union |
| FGF-2 | Proliferation of periosteal cells; early callus formation | Fracture repair |
| Anti-RANKL / OPG | Inhibit osteoclast-mediated resorption | Osteoporosis, peri-implant osteolysis |
| sclerostin siRNA (SOST) | Inhibit Wnt pathway suppressor → enhance bone formation | Osteoporosis, non-union |
Gene Delivery Vectors
Viral Vectors
| Vector | Key Features | Orthopaedic Relevance |
|---|---|---|
| Adeno-Associated Virus (AAV) | Non-integrating; episomal; long-term expression in non-dividing cells; minimal immunogenicity; tropism for muscle, CNS, hepatocytes | Preferred for cartilage and tendon (non-dividing cells); systemic osteoporosis therapy (Harrison's, p. 13371) |
| Adenovirus (Ad) | High transduction efficiency; transient expression (weeks); some immune response | Acute fracture repair; ex vivo cell modification |
| Lentivirus | Stable genomic integration; long-term expression; transduces dividing cells | Stem cell-based therapies; ex vivo approaches |
| Retrovirus | Integrating; requires dividing cells | Limited use due to insertional mutagenesis risk |
Non-Viral Vectors
| Vector | Features | Status |
|---|---|---|
| Lipid nanoparticles (LNP) | mRNA or DNA delivery; low immunogenicity; scalable | Emerging; used in mRNA-BMP delivery |
| Calcium phosphate / hydroxyapatite | Biomimetic; combined with scaffolds | Pre-clinical; scaffold-integrated delivery |
| Electroporation / gene gun | Physical methods; direct DNA transfer | Research settings |
| Polymer nanoparticles (PLGA) | Sustained release; biodegradable | Scaffold-based applications |
AAV vectors are especially attractive for bone because skeletal muscle — a common depot — is a long-lived cell population where episomal DNA persists without integration risk (Harrison's, p. 13371).
Delivery Strategies
1. In Vivo Gene Therapy
The vector is injected directly into the fracture site, joint, or periosteal tissue. Simple and clinically translatable but requires precise delivery and carries immune exposure risk.
2. Ex Vivo Gene Therapy
Cells (MSCs, periosteal cells, fibroblasts) are harvested from the patient, transduced in culture, then reimplanted at the defect site. Advantages:
- Quality control of transduced cells
- Reduced systemic vector exposure
- Cells serve as both a delivery vehicle and a regenerative scaffold
3. In Situ Gene Therapy
Vector is incorporated into a scaffold or matrix (e.g., collagen sponge, demineralized bone matrix, hydrogel). As the scaffold degrades, vector is released locally — spatiotemporally controlled delivery matching the healing timeline.
Applications in Orthopaedics
Fracture Non-Union and Delayed Union
The most studied indication. BMP-2 and BMP-7 gene transfer via adenoviral vectors or AAV has demonstrated accelerated callus formation and bridging in animal models of critical-size defects. Ex vivo approaches using MSCs transduced with BMP-2 are particularly promising, avoiding high-dose recombinant protein use.
Spinal Fusion
LMP-1 gene transfer and BMP-2 gene therapy have been investigated for augmenting posterolateral spinal fusion. Rodent and rabbit models show superior fusion rates compared to autograft controls.
Osteoporosis
Systemic AAV delivery of sclerostin antibody genes or OPG (osteoprotegerin) to suppress RANKL-mediated osteoclastogenesis represents a potential "one-shot" alternative to repeated anti-resorptive injections.
Avascular Necrosis (AVN) of the Femoral Head
VEGF gene therapy to restore vascularity, combined with BMP therapy for osteogenesis, is under investigation in animal models of steroid-induced AVN.
Articular Cartilage & Osteoarthritis
- IL-1Ra (interleukin-1 receptor antagonist) gene transfer to synoviocytes reduces cartilage-degrading cytokine signaling — the rationale behind the Invossa (TissueGene-C) construct (Phase III trials in knee OA)
- Sox9 and TGF-β1 gene therapy promotes chondrogenesis for focal cartilage defects
- FGF-18 gene transfer for chondroprotection
Tendon and Ligament Repair
GDF-5, PDGF-B, and bFGF gene transfer has shown improved collagen organization and mechanical strength in rotator cuff and ACL repair models.
Peri-Implant Osteolysis
Gene therapy targeting RANKL/OPG balance to reduce aseptic loosening of orthopaedic implants is a preclinical area of active research.
Fracture Healing Interventions — Overview
The following infographic illustrates the spectrum of strategies used to accelerate bone fracture healing, placing gene/pharmacologic approaches in context:
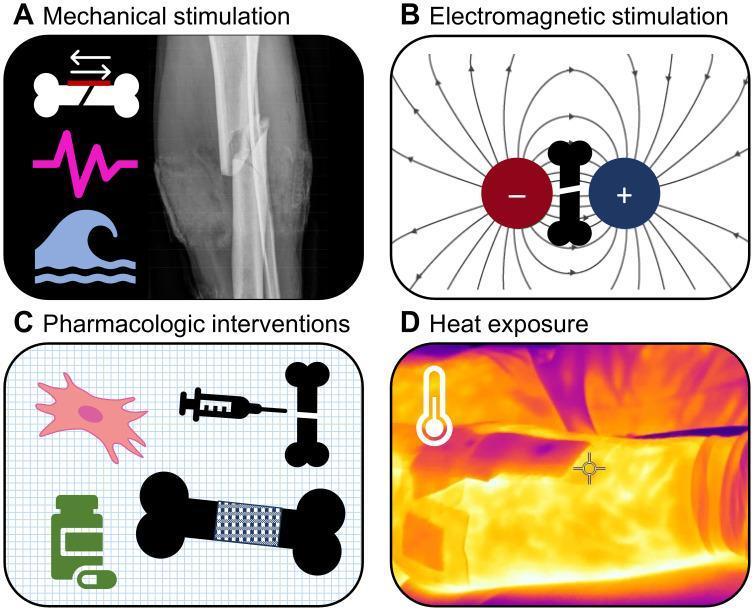
Pharmacologic and cellular (gene therapy) approaches are among the key categories alongside mechanical and electromagnetic stimulation.
Challenges and Limitations
| Challenge | Detail |
|---|---|
| Immunogenicity | Viral vectors, especially adenovirus, trigger immune responses that can eliminate transduced cells |
| Insertional mutagenesis | Integrating vectors (retrovirus, lentivirus) risk oncogene activation |
| Ectopic bone formation | Supraphysiologic BMP expression can cause heterotopic ossification |
| Transient vs. sustained expression | Acute healing requires temporary expression; persistent expression can be harmful |
| Regulatory and manufacturing hurdles | GMP production of viral vectors is expensive and complex |
| Immune pre-existing antibodies | Many patients have neutralizing antibodies to AAV serotypes |
| Translation gap | Most data from rodent models; large animal and human trials remain limited |
Clinical Translation Status
- Invossa (TissueGene-C): Retrovirally transduced chondrocytes expressing TGF-β1 for knee OA — reached Phase III in the US (FDA placed on clinical hold in 2019 for cell identity concerns; ongoing revision)
- AAV-OPG for periprosthetic osteolysis: Phase I trials
- BMP-2 gene-activated matrices: Pre-clinical/early Phase I
- Most orthopaedic gene therapy remains in pre-clinical or early Phase I/II stages
Future Directions
- CRISPR/Cas9 in vivo bone editing: Correcting OI (osteogenesis imperfecta) mutations or activating endogenous osteogenic genes
- mRNA-based BMP delivery (avoiding DNA integration entirely; similar to COVID-19 vaccine technology) — transient, non-integrating, immunologically safer
- Scaffold-integrated gene-activated matrices (GAMs): 3D-printed scaffolds carrying viral or non-viral vectors for critical-size defect reconstruction
- Cell-free exosome delivery: MSC-derived exosomes loaded with miRNAs or mRNA to stimulate osteogenesis without cellular immunogenicity
- Combinatorial approaches: Gene therapy + biologic scaffolds + physical stimulation (ultrasound, electromagnetic fields) for synergistic healing
Summary
Gene therapy in orthopaedics represents a shift from single-dose protein delivery to sustained, cell-directed biosynthesis of osteogenic, chondrogenic, and angiogenic signals. The most advanced applications target fracture non-union and osteoarthritis. AAV is the preferred vector for long-term gene expression in non-dividing musculoskeletal tissues, while ex vivo MSC-based strategies offer the safest route to clinical translation. The field is moving toward mRNA and CRISPR-based platforms that reduce integration risk and improve regulatory acceptability.
This is a shared conversation. Sign in to Orris to start your own chat.