motor evoked potential in orthopaedics and neurology
Reading File
search_medical_text
motor evoked potentials MEP intraoperative neurophysiological monitoring orthopaedics spine surgery
search_medical_text
motor evoked potentials MEP neurology clinical applications transcranial magnetic stimulation
GradeResponse
GradeResponse
The retrieved results are somewhat limited, so I'll supplement with my comprehensive knowledge on MEPs. Here is a thorough overview:
Motor Evoked Potentials (MEPs): Orthopaedics & Neurology
What Are MEPs?
Motor evoked potentials are electrical responses recorded from muscles or peripheral nerves following stimulation of the motor cortex or descending corticospinal tract. They reflect the integrity of the entire motor pathway — from the motor cortex through the corticospinal tract, anterior horn cells, peripheral nerve, and neuromuscular junction to muscle.
Two main stimulation methods:
| Method | Mechanism | Use |
|---|---|---|
| Transcranial Magnetic Stimulation (TMS) | Non-invasive; electromagnetic coil induces current | Outpatient neurology diagnostics |
| Transcranial Electrical Stimulation (TES) | High-voltage electrical pulses via scalp electrodes | Intraoperative monitoring (IONM) |
MEPs in Orthopaedic Surgery
Role in Intraoperative Neurophysiological Monitoring (IONM)
MEPs are a cornerstone of IONM in spinal and orthopaedic surgery, used alongside somatosensory evoked potentials (SSEPs) and electromyography (EMG).
According to Bailey & Love's Short Practice of Surgery (28th Ed., p. 541): During surgery for scoliosis, continuous spinal cord monitoring uses SSEPs, MEPs, and free-run/stimulated EMG activity to minimise the risk of neurological damage. The risk of neurological injury is 0.4% (1 in 250).
Surgeries Requiring MEP Monitoring
| Surgery | Rationale |
|---|---|
| Scoliosis correction / spinal fusion | Distraction/instrumentation may compromise corticospinal tract |
| Cervical spine surgery (ACDF, laminectomy) | Risk of cord compression or ischaemia |
| Thoracic spine surgery | Watershed zone; vulnerable to ischaemia |
| Spinal tumour resection | Eloquent motor cortex or tract at risk |
| Aortic aneurysm repair | Spinal cord ischaemia risk (anterior cord syndrome) |
| Hip/pelvis arthroplasty | Lumbosacral plexus monitoring via EMG + MEP |
MEP vs SSEP: Complementary Roles
| Parameter | SSEP | MEP |
|---|---|---|
| Pathway monitored | Posterior column (sensory) | Corticospinal tract (motor) |
| Modality | Sensory (touch/vibration) | Motor |
| Alert to anterior cord injury? | No (critical limitation) | Yes |
| Waveform | Cortical/subcortical | Muscle CMAP |
| Continuous monitoring | Easier (averages) | More technically demanding |
Key point: SSEPs alone miss anterior cord syndrome. MEPs are essential to detect motor pathway injury independently of SSEP changes.
Warning Criteria (Intraoperative Alerts)
Standard alert thresholds triggering surgical intervention:
- Amplitude decrease ≥50% from baseline — the most widely used criterion
- Complete loss of MEP — most serious; requires immediate response
- Increased stimulation threshold (voltage needed to elicit response rises significantly)
Response to MEP Alerts
- Inform surgeon immediately
- Check anaesthetic depth (volatile agents suppress MEPs significantly)
- Raise mean arterial pressure (MAP) to ≥80 mmHg
- Reverse any recent instrumentation or distraction
- If MEPs do not recover → consider wake-up test (Stagnara test)
Anaesthetic Considerations
MEPs are highly sensitive to anaesthetic agents:
- Volatile agents (sevoflurane, isoflurane) — markedly suppress MEPs; avoid or use <0.5 MAC
- Propofol + remifentanil TIVA — preferred regimen for MEP monitoring
- Neuromuscular blockers — abolish muscle MEPs; avoid or use only partial block
- Nitrous oxide — additive suppression; generally avoided
MEPs in Neurology
Diagnostic Applications
TMS-elicited MEPs are used to assess corticospinal tract integrity in neurological diseases.
Key Measurements
| Parameter | Definition | Normal | Abnormal |
|---|---|---|---|
| Central Motor Conduction Time (CMCT) | MEP latency minus peripheral conduction time | <6–8 ms (upper limb) | Prolonged = demyelination or UMN lesion |
| MEP Amplitude | Height of recorded muscle response | Variable | Reduced = axonal loss or poor activation |
| Cortical Silent Period (CSP) | Post-MEP inhibition of voluntary EMG | ~100–300 ms | Shortened or absent in various conditions |
| Motor Threshold | Minimum TMS intensity to elicit MEP in 50% of trials | ~40–60% max stimulator output | Elevated = corticospinal dysfunction |
MEPs in Specific Neurological Conditions
1. Multiple Sclerosis (MS)
- Most established neurological use
- Prolonged CMCT reflects demyelination of corticospinal tracts
- MEPs can be abnormal when clinical exam and MRI are equivocal
- Used to objectively quantify disability and monitor progression
2. Amyotrophic Lateral Sclerosis (ALS)
- Upper motor neuron (UMN) signs can be subtle clinically
- Prolonged CMCT or absent MEP supports UMN involvement
- Helps distinguish ALS from pure lower motor neuron (LMN) syndromes
- Part of updated Awaji criteria evidence base
3. Stroke / Cerebrovascular Disease
- MEP presence or absence after stroke is a strong predictor of motor recovery
- Absent MEP in acute stroke → poor hand/arm recovery prognosis
- Serial MEPs track cortical reorganisation during rehabilitation
- TMS mapping identifies eloquent cortex before neurosurgery
4. Cervical Myelopathy
- CMCT prolongation occurs before severe clinical symptoms
- Helps differentiate cervical myelopathy from peripheral neuropathy
- Supports surgical decision-making in borderline cases
5. Parkinson's Disease
- Short cortical silent period; reduced MEP inhibition
- Motor cortex hyperexcitability reflected in lowered threshold
- Used in research; less routine diagnostically
6. Hereditary Spastic Paraplegia (HSP)
- Markedly prolonged CMCT or absent lower limb MEPs
- Useful in mild/subclinical cases
7. Friedreich's Ataxia
- MEPs often absent in lower limbs
- Helps differentiate from other ataxias
Therapeutic TMS (rTMS)
Repetitive TMS (rTMS) exploits MEP physiology for treatment:
- Depression: FDA-approved; 10 Hz rTMS over left dorsolateral prefrontal cortex at 120% motor threshold (Bailey & Love; rTMS guideline data, p. 14)
- Stroke rehabilitation: Low-frequency rTMS to contralesional hemisphere suppresses interhemispheric inhibition, facilitating ipsilesional motor recovery
- Chronic pain / neuropathic pain: Motor cortex stimulation protocols
- OCD: FDA-approved rTMS protocol
MEP Recording Technique
Stimulation (Intraoperative TES)
- Corkscrew scalp electrodes at C3/C4 or Cz positions
- Short train stimuli (5–7 pulses, interstimulus interval 2–4 ms)
- Avoids single-pulse cardiac/movement artefacts
Recording
- Needle electrodes in target muscles bilaterally
- Upper limb: abductor pollicis brevis (APB), abductor digiti minimi (ADM)
- Lower limb: tibialis anterior, abductor hallucis
- Recorded as compound muscle action potential (CMAP)
Summary: MEPs at a Glance
| Domain | Key Use | Gold Standard? |
|---|---|---|
| Scoliosis surgery | Real-time cord monitoring | Yes (with SSEP) |
| Cervical spine surgery | Anterior cord protection | Yes |
| MS diagnosis | CMCT prolongation | Yes |
| ALS (UMN evidence) | CMCT prolongation | Supportive |
| Stroke prognosis | MEP presence predicts recovery | Yes |
| rTMS (depression) | Motor threshold calibrates dose | Yes |
| Cervical myelopathy | Subclinical cord dysfunction | Yes |
MEPs provide information that no other single test can replicate — direct, real-time evidence of motor pathway integrity — making them indispensable in both the operating theatre and the neurology clinic.
MEPs in Orthopaedic Surgery
search_medical_text
motor evoked potentials intraoperative monitoring orthopaedic spine surgery technique alerts
search_medical_text
somatosensory evoked potentials SSEP MEP spinal cord monitoring scoliosis cervical surgery warning criteria
GradeResponse
GradeResponse
Motor Evoked Potentials (MEPs) in Orthopaedic Surgery
Why MEPs Are Used
The primary goal is real-time detection of corticospinal tract injury before it becomes irreversible. Since SSEPs only monitor the posterior (sensory) columns, they are blind to anterior cord ischaemia — the most common mechanism of paraplegia after spinal surgery. MEPs fill this critical gap.
According to Bailey & Love's Short Practice of Surgery (28th Ed., p. 541): Continuous spinal cord monitoring using SSEPs, MEPs, and free-run/stimulated EMG is standard practice during corrective spinal surgery. The risk of neurological injury is 0.4% (1 in 250) — a risk MEP monitoring aims to prevent or detect early.
Anatomical Basis
MEPs travel via the corticospinal tract (CST), which runs in the anterior and lateral columns of the spinal cord — the region most vulnerable to:
- Mechanical compression by instrumentation
- Anterior spinal artery ischaemia
- Distraction forces during deformity correction
This is exactly why SSEP alone is insufficient: it monitors the posterior columns only.
Surgeries Where MEP Monitoring Is Used
| Surgery | Primary Risk Monitored |
|---|---|
| Scoliosis correction / spinal fusion | Cord distraction, pedicle screw malposition |
| Cervical spine surgery (ACDF, posterior decompression) | Cord compression, ischaemia |
| Thoracic spine surgery (tumour, deformity) | Watershed ischaemia of anterior cord |
| Lumbar decompression / fusion | Nerve root and conus injury |
| Spinal tumour resection | Surgical resection near eloquent tracts |
| Aortic surgery (thoracoabdominal) | Anterior spinal artery compromise |
| Pelvic / acetabular fracture fixation | Lumbosacral plexus, sciatic nerve |
MEP Technique in the Operating Theatre
Stimulation
- Transcranial electrical stimulation (TES) via corkscrew scalp electrodes
- Electrode positions: C1/C2 or C3/C4 (international 10-20 system) — contralateral placement targets opposite limbs
- Short train stimuli: 4–7 pulses, interstimulus interval 2–4 ms, high voltage (up to 200–400 V)
- Short trains allow the stimulus to summate and traverse injured/partially blocked cord segments
Recording
- Surface or intramuscular needle electrodes in target muscles
- Recorded as compound muscle action potential (CMAP)
Typical muscle targets:
| Level Monitored | Upper Limb Muscles | Lower Limb Muscles |
|---|---|---|
| Cervical | Abductor pollicis brevis (APB), deltoid, biceps | — |
| Thoracic | — | Tibialis anterior, abductor hallucis |
| Lumbar/Sacral | — | Gastrocnemius, EHL, anal sphincter |
MEP vs SSEP: Critical Comparison
| Feature | SSEP | MEP |
|---|---|---|
| Pathway | Posterior column (sensory) | Corticospinal tract (motor) |
| Column of cord | Dorsal | Anterior / lateral |
| Detects anterior cord syndrome? | ❌ No | ✅ Yes |
| Continuous monitoring | Easier (averaged responses) | Intermittent (movement artefact) |
| Anaesthetic sensitivity | Moderate | High (especially volatile agents) |
| Waveform | Cortical/subcortical peaks | Muscle CMAP |
| Clinical significance of loss | Sensory deficit risk | Motor deficit / paralysis risk |
Combined MEP + SSEP monitoring is the standard of care — they are complementary, not interchangeable.
Alert Criteria (Warning Signs)
When these thresholds are crossed, the surgical team must be notified immediately:
| Alert | Threshold | Significance |
|---|---|---|
| Amplitude decrease | ≥50% reduction from baseline | Most widely used criterion |
| Complete MEP loss | All responses absent | Most serious — high risk of permanent deficit |
| Threshold increase | Stimulus voltage needed rises significantly | Early sign of deteriorating cord conduction |
| Latency increase | ≥10% prolongation | Suggests demyelination or ischaemia |
The 50% amplitude drop rule is the universally accepted alert criterion. Complete loss mandates immediate surgical action.
Response Protocol to MEP Alerts
A structured stepwise response is followed when an alert is triggered:
- Notify the surgeon immediately — pause instrumentation
- Check anaesthetic depth — volatile agents, bolus propofol, or recent neuromuscular blockade can cause false positives
- Optimise haemodynamics — raise MAP to ≥80 mmHg (or >10 mmHg above baseline) to improve cord perfusion
- Correct mechanical cause — remove distraction, back off implant, decompress if applicable
- Irrigate wound with warm saline (cord temperature matters)
- Reassess MEPs after each corrective step
- If MEPs do not recover → perform wake-up test (Stagnara test) to clinically confirm motor function
Anaesthetic Management for MEP Monitoring
Anaesthetic choice is critical — MEPs are the most anaesthetic-sensitive of all evoked potentials.
| Agent | Effect on MEPs | Recommendation |
|---|---|---|
| Volatile agents (sevoflurane, desflurane, isoflurane) | Markedly suppress | Avoid or use <0.5 MAC |
| Nitrous oxide (N₂O) | Additive suppression | Avoid |
| Propofol (infusion) | Mild suppression | Preferred |
| Remifentanil / fentanyl | Minimal effect | Safe; use freely |
| Ketamine | May enhance MEPs | Useful adjunct |
| Neuromuscular blockers (NMBs) | Abolish muscle MEPs | Avoid or use only partial block |
| Dexmedetomidine | Mild suppression | Generally acceptable |
Preferred anaesthetic technique: TIVA (Total Intravenous Anaesthesia) — propofol + remifentanil infusion, no volatile agent, no NMB.
The Wake-Up Test (Stagnara Test)
Used as a backup or confirmatory test when MEPs are lost or unreliable:
- Anaesthesia is lightened until the patient can follow commands
- Patient asked to move hands, then feet
- Positive response (movement preserved) = reassuring
- Limitations: patient cooperation required, one-time snapshot, awareness risk, cannot be repeated frequently
MEPs have largely replaced the wake-up test as the primary monitoring tool, but it remains a valid adjunct when MEP signals are uninterpretable.
Specific Orthopaedic Contexts
Scoliosis Surgery
- Highest-risk spinal operation for neurological injury
- Distraction forces across multiple levels can compromise anterior cord perfusion
- MEPs + SSEPs + EMG used together
- Alert during rod rotation or final compression/distraction manoeuvres
Cervical Spine Surgery
- Risk of cord injury from retraction, instability, or direct compression
- Upper limb MEPs (APB, deltoid) are particularly important
- Myelopathic patients may have reduced baseline MEP amplitudes — establishing a good baseline before incision is critical
Pedicle Screw Placement
- Stimulated EMG (not MEP per se) is used to detect medial wall breach
- A screw stimulation threshold <6–8 mA suggests medial breach into the canal
- Free-run EMG detects nerve root irritation in real time
Thoracic Surgery / Aortic Cross-Clamping
- Anterior spinal artery watershed zone (T4–T9) is most vulnerable
- MEP loss during aortic cross-clamping → reposition clamp, insert intercostal reimplantation, raise MAP
Limitations of MEP Monitoring
| Limitation | Clinical Impact |
|---|---|
| Cannot be used continuously (movement artefact) | Intermittent snapshots only |
| High false-positive rate with anaesthetic changes | Requires skilled neurophysiologist to interpret |
| Technically demanding setup | Not available at all centres |
| May be absent at baseline in severe myelopathy | Limits utility in advanced cord compression |
| Does not monitor sensory pathways | Must use with SSEP |
Summary
MEPs in orthopaedic surgery serve one primary function: detecting corticospinal tract injury in real time before permanent neurological damage occurs. They are indispensable for any surgery near the spinal cord or major nerve roots, and when combined with SSEPs and EMG, provide comprehensive coverage of the entire neuraxis. The anaesthetic team, surgeon, and neurophysiologist must work in close coordination for IONM to be effective.
navigation in Orthopaedics
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Searching Images
computer assisted navigation orthopaedic surgery intraoperative tracking system
I now have excellent textbook content and clinical images. Here is the comprehensive answer:
Navigation in Orthopaedic Surgery
Definition
Surgical navigation — also called Computer-Assisted Surgery (CAS) or Image-Guided Surgery (IGS) — is a real-time intraoperative system that tracks the position of surgical instruments and implants in three dimensions relative to the patient's anatomy, displayed on a monitor much like a GPS system.
Core Components of a Navigation System
| Component | Function |
|---|---|
| Imaging source | Provides anatomical map (preoperative CT/MRI, intraoperative fluoroscopy, or 3D O-arm CT) |
| Localiser / camera unit | Optical infrared or electromagnetic detector that tracks tracker positions in space |
| Reference trackers | Rigid arrays with reflective spheres fixed to patient bones and instruments |
| Navigation workstation | Software that registers patient anatomy and displays real-time instrument position |
| Navigated instruments | Probes, drills, reamers, saws fitted with tracking arrays |
How It Works: The Workflow
1. Image Acquisition
- Preoperative CT/MRI → imported into workstation; virtual 3D model constructed
- Intraoperative fluoroscopy (most common) → C-arm images taken after patient positioning, automatically calibrated
- Intraoperative 3D CT (O-arm, Vario 3D) → highest accuracy; spin CT acquired on table, registered automatically
According to Rockwood & Green's Fractures in Adults (10th Ed., p. 232): "Computer-assisted surgical navigation utilizing fluoroscopy is an emerging technology applied to percutaneous pelvic and acetabular screw placement. Fluoroscopy is commonly used for surgical navigation because of its flexibility, convenience, lower radiation exposure, and lower cost."
2. Registration
- The process of matching the virtual image model to the actual patient anatomy in real space
- Methods:
- Point-to-point registration: surgeon touches anatomical landmarks with a tracked probe
- Surface matching: probe swept over bone surface; software matches contour to CT model
- Image-based (automatic): O-arm/C-arm auto-registers after intraoperative scan — no manual step required
3. Tracking
- A reference frame (star-shaped array with retroreflective spheres) is rigidly fixed to the patient's bone (e.g., spinous process pin, iliac crest pin, or tibial pin)
- Instruments fitted with tracking arrays
- The camera continuously tracks both patient and instrument positions
- Real-time position displayed on workstation in 2D/3D reconstructions
4. Guided Surgery
- Surgeon views instrument trajectory on screen relative to anatomy in axial, sagittal, coronal, and 3D views
- Allows precise planning and execution of cuts, screw trajectories, implant placement
Navigation Technologies: Types
| Technology | Image Source | Accuracy | Use Case |
|---|---|---|---|
| Fluoroscopy-based navigation | Intraoperative C-arm | ±2–3 mm | Fracture fixation, pelvic screws |
| CT-based navigation | Preoperative CT | ±1–2 mm | Spine (pedicle screws), pelvis |
| Intraoperative 3D CT (O-arm) | Intraop CT spin | <1 mm | Spine, complex deformity |
| Imageless navigation | Kinematic data (no imaging) | ±2–3 mm | TKA alignment, THA |
| Robotic-assisted navigation | CT + robotic arm | <1 mm | TKA, THA, spine screws |
| Electromagnetic navigation | EM field tracking | ±2–3 mm | Minimally invasive, no line-of-sight |
Clinical Applications
1. Total Knee Arthroplasty (TKA)
The most widely studied application.
Purpose: Achieve neutral mechanical axis alignment (target: 0° ± 3° of the mechanical axis)
Methods:
- Imageless CAS: sensors capture knee kinematics (range of motion, femoral/tibial axes) without imaging; software guides cutting blocks
- CT-based / robotic: preoperative plan mapped onto patient in theatre
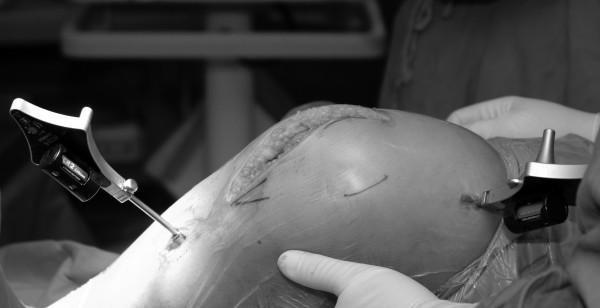
Evidence:
Schwartz's Principles of Surgery (11th Ed., p. 1937): "Computer navigation in total joint arthroplasty has been shown to minimize outliers in alignment, but there has been no proven benefit in survival or function secondary to computer-navigated or robotic-assisted joint replacement."
Miller's Review of Orthopaedics (9th Ed.): "Evidence supports not using intraoperative navigation because there is no difference in outcomes or complications" (conventional TKA instrumentation).
Interpretation: Navigation reduces malalignment outliers (components placed outside ±3° of neutral) but has not yet demonstrated superior long-term clinical outcomes over conventional instrumentation.
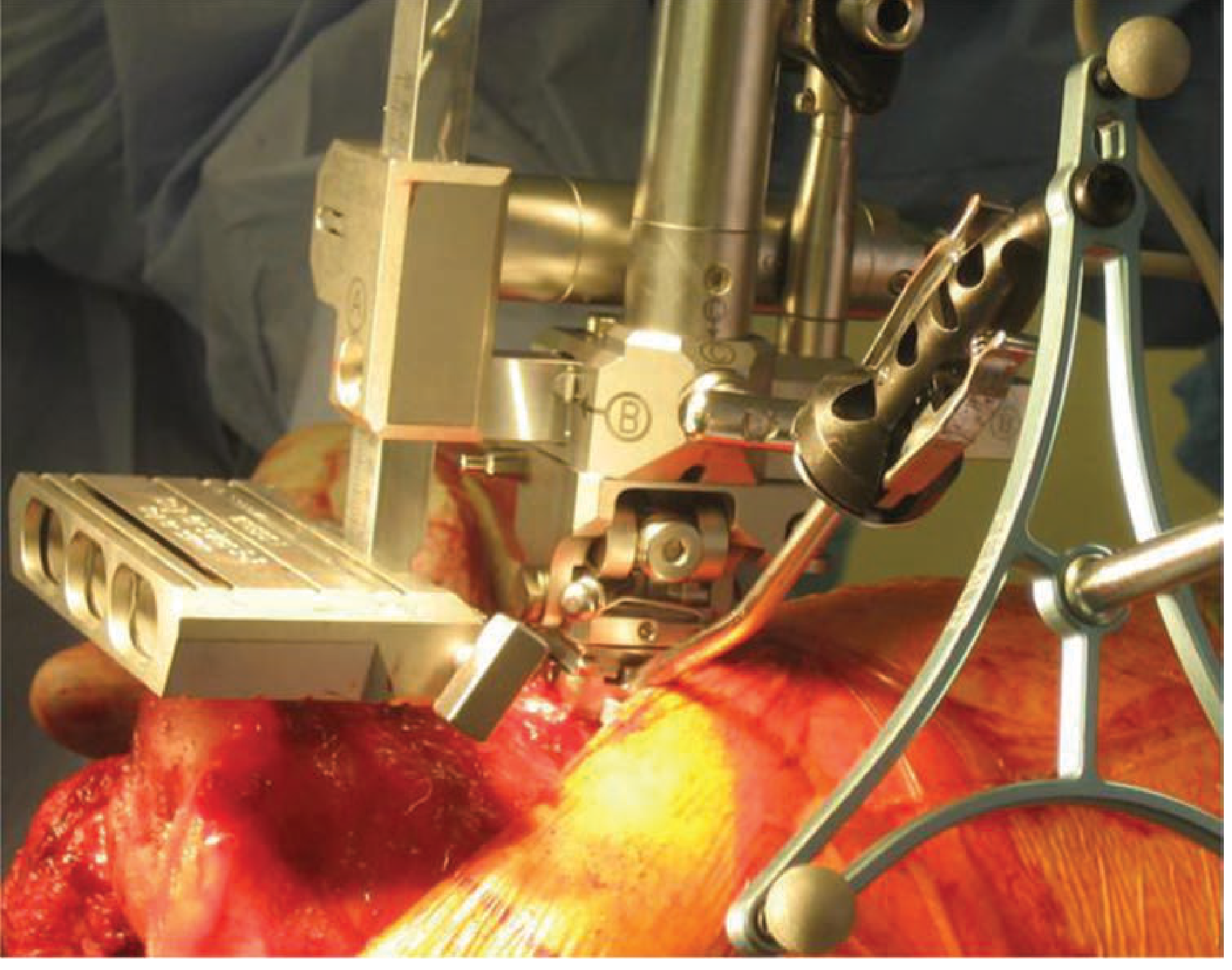
2. Total Hip Arthroplasty (THA)
Purpose: Accurate cup orientation (target: 40° inclination, 15° anteversion — the "safe zone")
- Cup malposition is the leading cause of dislocation and impingement
- Imageless CAS or robot-assisted (e.g., MAKO, Stryker) guides acetabular reaming and cup insertion
- Leg length and offset can be measured intraoperatively with navigation
3. Pedicle Screw Placement (Spine)
The most impactful application in terms of safety.
Purpose: Accurate screw placement within the pedicle, avoiding breach into the canal, foramen, or vascular structures
Without navigation: fluoroscopy-based freehand — breach rate 15–40% (literature range)
With CT navigation: breach rate drops to <5%
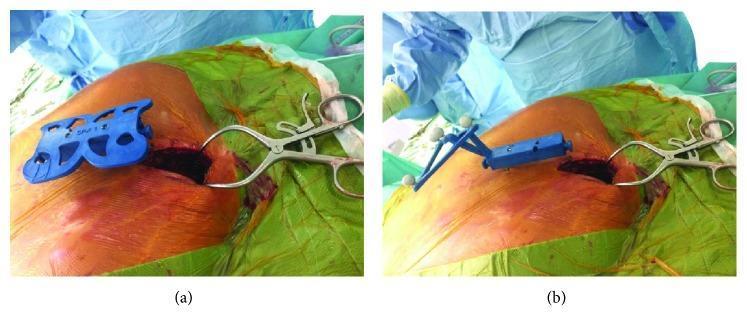
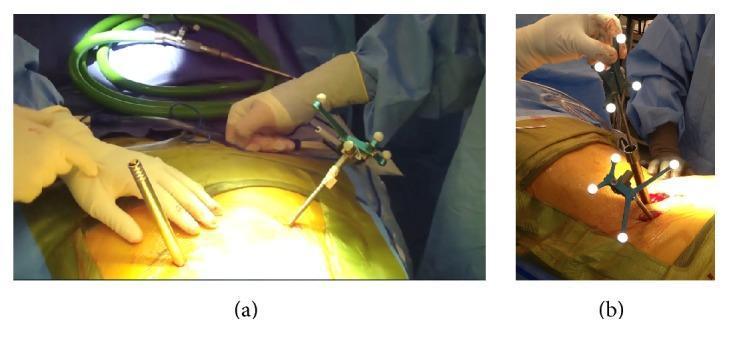
Rockwood & Green's (10th Ed., p. 895): "Intraoperative CT systems such as the O-arm (Medtronic), Vario 3D by Ziehm, or Surgivisio (eCential) can be coupled with navigation systems and may improve reduction and fixation in trauma surgery. Intraoperative CT coupled to robotics is currently used in spine and arthroplasty surgery."
Systems used: Medtronic StealthStation, Brainlab Spine, Surgivisio, ROSA Spine (Zimmer Biomet)
4. Fracture Fixation & Trauma Surgery
Applications:
- Percutaneous pelvic and acetabular screw placement (iliosacral, supra-acetabular, column screws)
- Femoral nail antegrade/retrograde — guidewire placement
- Tibial plateau and pilon fractures
- Distal radius fixation
Advantage: Reduces radiation exposure to surgeon by allowing navigation-guided drilling without continuous fluoroscopy
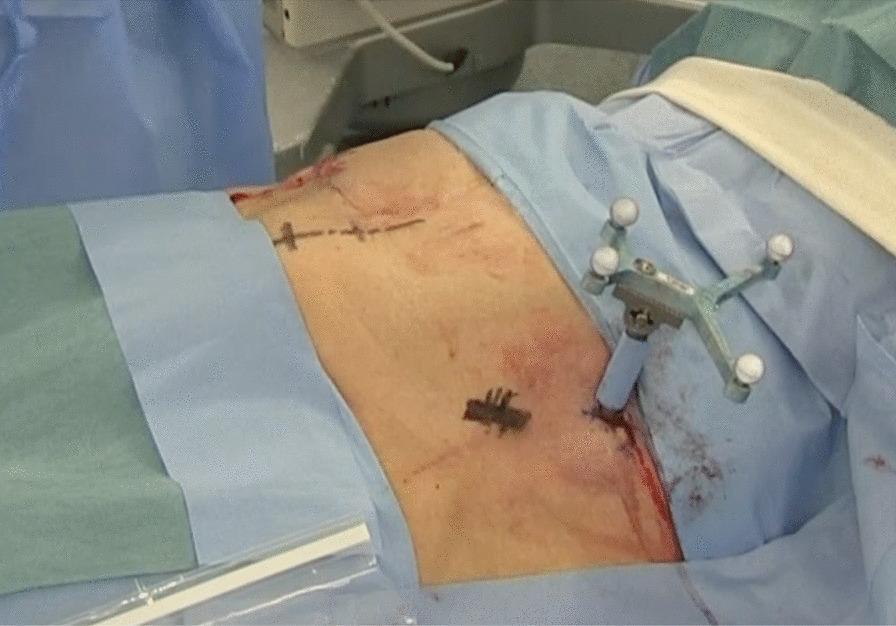
Rockwood & Green's (10th Ed.): Computer-assisted fluoroscopy navigation has "been applied to percutaneous pelvic and acetabular screw placement with expected continued progress."
5. Complex Spinal Deformity
- Navigated osteotomies (e.g., posterior vertebral column resection) guided in real time
- Multiplanar CT reconstructions allow precise bone removal and implant trajectory planning
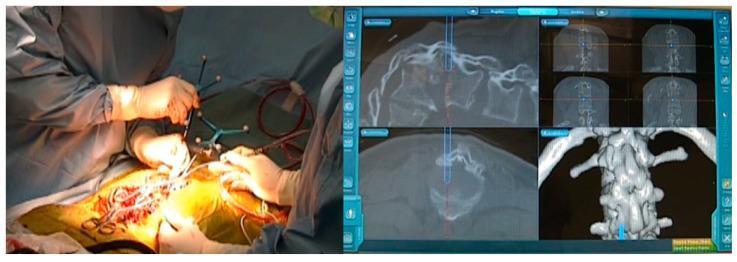
Robotic-Assisted Navigation
Robotics adds a physical constraint or active assistance on top of navigation:
| System | Type | Application |
|---|---|---|
| MAKO (Stryker) | Semi-active (haptic boundary) | TKA, UKA, THA |
| ROSA Knee (Zimmer Biomet) | Semi-active | TKA |
| Mazor X (Medtronic) | Active arm guidance | Pedicle screws |
| ROSA Spine | Active arm guidance | Spine screws |
| Navio (Smith & Nephew) | Imageless robotic | UKA, TKA |
Robotic types:
- Passive: navigation display only, surgeon controls instrument freely
- Semi-active (haptic): robotic arm creates a virtual boundary; surgeon cannot cut outside planned zone
- Active: robot moves autonomously to planned position, surgeon supervises
Miller's Anesthesia (10th Ed.): "Many robotic spine surgery platforms have been introduced, but their acceptance has [been variable] — robots provide intraoperative image-guided navigation combined with robotic assistance for 3D reconstruction for screw placement."
Advantages of Navigation
| Advantage | Detail |
|---|---|
| Improved accuracy | Reduces outliers in implant positioning and screw placement |
| Reduced radiation | Less fluoroscopy needed intraoperatively |
| Minimally invasive compatibility | Enables MIS approaches without direct visualisation |
| Real-time feedback | Surgeon sees instrument position before committing |
| Documentation | Digital record of intraoperative alignment and implant position |
| Revision planning | Intraoperative data guides revision decisions |
Limitations and Disadvantages
| Limitation | Detail |
|---|---|
| Increased setup time | Registration and tracker fixation adds 15–30 minutes |
| Cost | Navigation systems expensive; disposables costly per case |
| Learning curve | Requires training; accuracy depends on correct registration |
| Registration error | Poorly performed registration renders the whole system inaccurate |
| Line-of-sight issues | Optical systems require clear view between camera and trackers |
| Reference frame movement | If tracker shifts after registration, all subsequent data is invalid |
| No proven outcome benefit (TKA) | Alignment improved but clinical scores and survival unchanged in RCTs |
| Soft tissue kinematics | Navigation does not assess ligament balance in real time (though kinematic sensors now address this) |
Patient-Specific Instrumentation (PSI) — A Related Concept
PSI uses preoperative CT/MRI to manufacture custom cutting jigs that fit uniquely to the patient's bone surfaces, pre-planned in a virtual environment. It shares navigation's goal of accuracy without an intraoperative system.
Miller's Review of Orthopaedics: "Evidence supports not using patient-specific instrumentation compared to conventional instrumentation for TKA because there is no difference in pain or functional outcomes."
Summary Table: Navigation by Application
| Procedure | Navigation Type | Key Benefit | Evidence Strength |
|---|---|---|---|
| TKA | Imageless / robotic (MAKO) | Reduces malalignment outliers | Strong (no outcome benefit proven) |
| THA | Imageless / robotic | Cup orientation accuracy | Moderate |
| Pedicle screw | CT-based / O-arm robotic | Reduces breach rate | Strong |
| Pelvic screws | Fluoroscopy-based | Corridor accuracy, less radiation | Moderate |
| Spinal deformity | CT navigation | Complex osteotomy precision | Moderate |
| Fracture fixation | Fluoroscopy navigation | MIS guidance, radiation reduction | Emerging |
Navigation represents a significant advance in precision orthopaedic surgery. Its greatest proven benefit is in spinal pedicle screw placement, while in joint replacement its role continues to evolve alongside robotic platforms.
— Schwartz's Principles of Surgery 11th Ed., p. 1937; Rockwood & Green's Fractures in Adults 10th Ed., pp. 232, 895; Miller's Review of Orthopaedics 9th Ed.
This is a shared conversation. Sign in to Orris to start your own chat.