3D printing in orthopaedics
Reading File
search_medical_text
3D printing additive manufacturing orthopaedics implants surgical planning
search_medical_text
3D printed bone scaffolds bioprinting fracture reconstruction
search_medical_image
3D printed orthopaedic implant custom prosthesis
GradeResponse
GradeResponse
GradeResponse
Here is a comprehensive overview of 3D printing in orthopaedics:
3D Printing in Orthopaedics
What Is Additive Manufacturing?
3D printing (additive manufacturing) builds physical structures layer-by-layer from digital models derived from CT/MRI data. In orthopaedics, it bridges preoperative planning and intraoperative execution, enabling patient-specific solutions that were not possible with conventional manufacturing.
Key Applications
1. Preoperative Planning & Surgical Models
Physical anatomical models are printed from 3D CT data to allow surgeons to:
- Visualize complex deformities before operating
- Practice osteotomies or implant placement on an exact replica
- Improve team communication and patient consent
Bailey & Love's Short Practice of Surgery (p. 576) highlights how 3D printing is increasingly used alongside computer navigation and robotics to understand deformity anatomy and plan accurate surgical corrections.
2. Patient-Specific Implants (PSI)
Custom implants are fabricated to match a patient's exact bony anatomy. The most advanced current application is in orthopaedic oncology and complex reconstruction.

Above: Custom EBM-printed pelvic prosthesis for post-tumour resection reconstruction. Porous titanium regions (~70% porosity, 600 µm pore size) at pubis-ischium and pubis-ilium interfaces promote bone ingrowth (osseointegration), while solid zones with screw holes ensure mechanical fixation.
Common uses for PSI:
| Domain | Example |
|---|---|
| Oncology | Pelvic/limb-salvage prostheses after tumour resection |
| Trauma | Craniofacial and complex acetabular reconstruction |
| Revision arthroplasty | Filling large bone defects after failed hip/knee replacements |
| Spine | Custom cages, vertebral body replacements |
| Paediatrics | Growth-sparing implants for sarcoma |
3. Manufacturing Technologies Used
| Technology | Material | Use Case |
|---|---|---|
| Selective Laser Sintering (SLS) | Titanium, CoCr | Structural implants |
| Electron Beam Melting (EBM) | Titanium (Ti-6Al-4V) | Porous-surfaced orthopaedic implants |
| Fused Deposition Modelling (FDM) | PLA, PEEK | Surgical models, guides |
| Stereolithography (SLA) | Photopolymers | Anatomical planning models |
| Bioprinting | Hydrogels + cells | Experimental scaffold tissue engineering |
4. Cutting Guides & Surgical Jigs
Patient-specific cutting guides (PSCGs) are printed to fit bone surfaces precisely, guiding saw cuts or drill holes to pre-planned positions. Used in:
- Total knee arthroplasty (TKA) alignment
- Corrective osteotomies
- Spinal pedicle screw placement
Advantages: Reduced operative time, fewer intraoperative instruments, improved angular accuracy.
5. Bone Scaffolds & Bioprinting (Experimental)
Porous 3D-printed scaffolds — made from calcium phosphate, hydroxyapatite, or titanium — serve as osteoconductive frameworks for bone ingrowth in defect repair. Bioprinting (cells + growth factors embedded in scaffolds) is still largely experimental but holds promise for cartilage and bone regeneration.
Osseointegration: Why Porosity Matters
Porous surfaces (typically 60–80% porosity, 300–900 µm pore diameter) on metal implants:
- Mimic cancellous bone architecture
- Allow vascular and bony ingrowth
- Improve long-term biological fixation, reducing aseptic loosening
This is a key design advantage of 3D-printed implants over machined ones, which cannot replicate such complex surface geometry.
Regulatory & Sterilization Considerations
Onsite 3D printing at healthcare facilities carries specific risks (per GLGCA guidelines, p. 14):
- Sterilization validation is complex — gas plasma may fail to penetrate hollow structures; steam autoclaving can deform thermoplastic models
- Residual manufacturing materials (support material, solvents) must be reduced to safe levels
- Experimental devices require IRB approval and Institutional Policy Committee oversight unless they present a "non-significant risk"
- FDA (and equivalent bodies) regulate implantable 3D-printed devices as Class II/III medical devices
Advantages vs. Limitations
| Advantages | Limitations |
|---|---|
| Patient-specific fit | Higher cost vs. off-the-shelf implants |
| Complex geometry possible | Longer lead time (days–weeks) |
| Porous surfaces for osseointegration | Regulatory and sterilization challenges |
| Reduced surgical time (with cutting guides) | Fatigue properties of printed metals vary |
| Improved outcomes in complex cases | Limited long-term clinical outcome data |
Clinical Impact Summary
3D printing is most firmly established in:
- Pre-surgical planning models — now routine in complex trauma and tumour surgery
- Custom oncology implants — replacing large bone segments after sarcoma resection
- Revision arthroplasty — managing cavitary and segmental bone defects
- Spinal surgery — custom cages and interbody devices
Emerging areas include bioprinted cartilage, smart implants with embedded sensors, and robotic-assisted placement of 3D-printed constructs.
3D printing in orthopaedics
search_medical_text
3D printing additive manufacturing orthopaedics implants surgical planning
search_medical_text
3D printed bone scaffolds patient specific implants fracture reconstruction
search_medical_image
3D printed orthopaedic implant custom prosthesis surgical guide
GradeResponse
GradeResponse
GradeResponse
3D Printing in Orthopaedics
Overview
3D printing (additive manufacturing) constructs physical objects layer-by-layer from digital models derived from patient CT/MRI data. In orthopaedics, it enables patient-specific solutions — from planning models to implantable devices — that conventional subtractive manufacturing cannot replicate.
1. Preoperative Planning & Anatomical Models
Physical bone models printed from 3D CT scans allow surgeons to:
- Visualise complex deformities, fracture patterns, and tumour involvement before entering the operating theatre
- Rehearse osteotomies, reduction manoeuvres, or implant seating on an exact anatomical replica
- Improve multidisciplinary team communication and patient-informed consent
Bailey & Love's Short Practice of Surgery (28th Ed., p. 576) notes that 3D printing is increasingly used alongside computer navigation and robotics to understand deformity anatomy and plan accurate surgical corrections.
2. Patient-Specific Implants (PSI)
The most clinically advanced application — custom implants designed from a patient's own imaging data and fabricated to fit their exact anatomy.
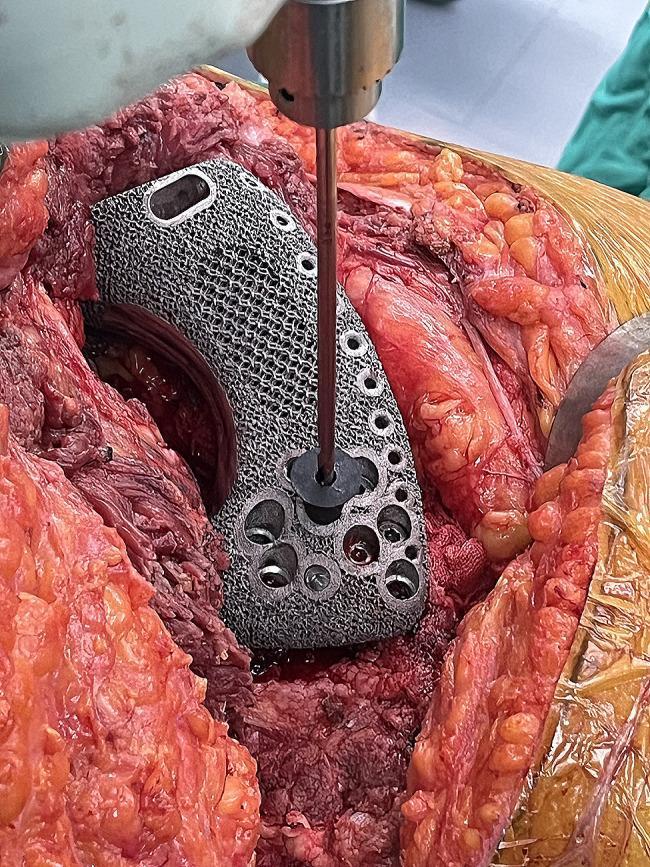
Intraoperative view of a titanium alloy (Ti-6Al-4V) hemipelvis prosthesis with porous dodecahedron lattice structure, implanted following pelvic tumour resection. A patient-specific drilling guide ensures precise screw placement. The lattice design promotes osseointegration via bone ingrowth.
Clinical Domains for PSI
| Domain | Application |
|---|---|
| Orthopaedic oncology | Pelvic, femoral, humeral reconstruction after sarcoma resection |
| Revision arthroplasty | Filling large cavitary/segmental defects after failed hip or knee replacements |
| Trauma | Comminuted periarticular fractures, acetabular defects |
| Spine | Custom vertebral body cages, interbody devices |
| Paediatrics | Growth-sparing implants in skeletally immature patients |
3. Patient-Specific Cutting Guides & Jigs
Printed guides contour precisely to the bone surface, directing saw cuts and drill holes to pre-planned positions. No intraoperative fluoroscopy or mechanical alignment rods needed.
Uses:
- Total knee arthroplasty (TKA) — femoral and tibial resections
- Corrective osteotomies (tibial, femoral, forearm)
- Pedicle screw placement in spinal deformity surgery
Benefits: Reduced operative time, fewer instrument trays, improved angular accuracy, less soft-tissue disruption.
4. Printing Technologies & Materials
| Technology | Material | Primary Use |
|---|---|---|
| Electron Beam Melting (EBM) | Ti-6Al-4V | Porous structural implants |
| Selective Laser Sintering/Melting (SLS/SLM) | Titanium, CoCr | Load-bearing implants |
| Fused Deposition Modelling (FDM) | PLA, PEEK | Surgical models, guides, orthoses |
| Stereolithography (SLA) | Photopolymers | High-resolution planning models |
| Bioprinting | Hydrogels + cells | Experimental scaffold/tissue engineering |
5. Why Porosity Matters: Osseointegration
A major advantage of 3D-printed metal implants over machined ones is the ability to create precisely controlled porous surfaces that mimic cancellous bone:
- Porosity: 60–80%
- Pore size: 300–900 µm
- Architecture: Interconnected lattices (dodecahedron, gyroid, etc.)
This promotes vascular and bony ingrowth, improving biological fixation and reducing long-term aseptic loosening — the leading cause of revision arthroplasty.
6. Bone Scaffolds & Bioprinting (Experimental)
Porous scaffolds made from hydroxyapatite, β-tricalcium phosphate, or titanium act as osteoconductive frameworks for bone defect repair. True bioprinting — embedding osteoblasts, chondrocytes, or mesenchymal stem cells within hydrogel carriers — aims to regenerate bone and cartilage de novo. Currently confined to research settings but advancing rapidly.
7. Regulatory & Sterilization Considerations
Per GLGCA guidelines (p. 14), onsite manufacture of 3D-printed devices carries specific risks:
- Gas plasma sterilization fails to penetrate hollow internal geometries
- Steam autoclaving can deform thermoplastic models
- Residual manufacturing materials (support structures, solvents) must be reduced to safe levels
- IRB and Institutional Policy Committee oversight is required for experimental onsite-printed devices
- Implantable devices are regulated as Class II/III medical devices by the FDA (and equivalent bodies globally)
8. Advantages vs. Limitations
| Advantages | Limitations |
|---|---|
| Anatomically precise fit | Higher unit cost than off-the-shelf implants |
| Complex porous geometry achievable | Longer lead time (days to weeks) |
| Improved osseointegration | Sterilization validation complexity |
| Reduced operative time (with cutting guides) | Variable fatigue/mechanical properties |
| Enables limb salvage in oncology | Limited long-term RCT outcome data |
| Improved surgical training models | Regulatory pathway can be lengthy |
Summary: Where 3D Printing Is Established vs. Emerging
| Status | Application |
|---|---|
| Established | Surgical planning models, oncology PSI, revision arthroplasty augments, cutting guides |
| Growing | Spinal cages, trauma reconstruction, orthotic/prosthetic devices |
| Experimental | Bioprinted cartilage, drug-eluting scaffolds, smart implants with sensors |
3D printing is most transformative where anatomy is uniquely distorted — tumour resection, complex revision, and rare congenital deformities — and is progressively moving into routine arthroplasty and trauma care.
This is a shared conversation. Sign in to Orris to start your own chat.