atypical pneumonia
Reading File
search_medical_text
atypical pneumonia etiology pathogens clinical presentation
search_medical_text
atypical pneumonia diagnosis treatment management antibiotics
search_medical_image
atypical pneumonia chest X-ray CT findings
GradeResponse
GradeResponse
GradeResponse
Atypical Pneumonia
Overview
"Atypical pneumonia" refers to community-acquired pneumonia (CAP) caused by a distinct group of pathogens that differ from classic ("typical") bacterial causes in their biology, clinical features, radiographic pattern, and antibiotic response. The term originates from the clinical presentation, which is often more indolent and lacks the classic lobar pattern of pneumococcal pneumonia.
Etiology
Atypical pathogens (Harrison's, p. 3800):
| Pathogen | Notes |
|---|---|
| Mycoplasma pneumoniae | Most common; especially in young adults and children; incidence increasing |
| Chlamydia pneumoniae | Increasing incidence; mild, often prolonged illness |
| Legionella spp. | Can cause severe disease (Legionnaires'); associated with water systems |
| Influenza virus | Major viral cause; seasonal |
| Adenovirus | Military recruits, children |
| Human metapneumovirus | Elderly and immunocompromised |
| RSV | Extremes of age |
| Coronaviruses (incl. SARS-CoV-2) | Can range from mild to critical illness |
In contrast, "typical" pathogens include S. pneumoniae, H. influenzae, S. aureus, and gram-negative bacilli such as Klebsiella pneumoniae.
Clinical Presentation
Atypical pneumonia classically presents with:
- Gradual onset (days to weeks vs. acute in typical pneumonia)
- Non-productive or minimally productive cough
- Low-grade fever, headache, malaise, myalgias (flu-like prodrome)
- Absence of rigors (common in pneumococcal pneumonia)
- Extra-pulmonary features: pharyngitis, earache (bullous myringitis with Mycoplasma), GI symptoms, rash, hemolytic anemia (cold agglutinins with Mycoplasma)
- Legionella can cause more severe disease with high fever, confusion, hyponatremia, and diarrhea
Physical exam findings are often disproportionately mild relative to radiographic extent.
Diagnosis
- Chest X-ray: patchy, bilateral, interstitial or reticulonodular infiltrates — often more diffuse than expected clinically
- Chest CT: bilateral ground-glass opacities (GGOs), subpleural/peripheral distribution, lower lobe predominance (see image below)
- Lab findings: Normal or mildly elevated WBC (leukocytosis less pronounced than in typical bacterial pneumonia), elevated cold agglutinins (Mycoplasma)
- Microbiological workup:
- Urinary antigen tests: Legionella (serogroup 1), S. pneumoniae
- Serology: paired titres for Mycoplasma, Chlamydia
- PCR (nasopharyngeal): respiratory viruses, Mycoplasma, Chlamydia
- Sputum culture (low yield for atypical organisms)
Radiographic example:
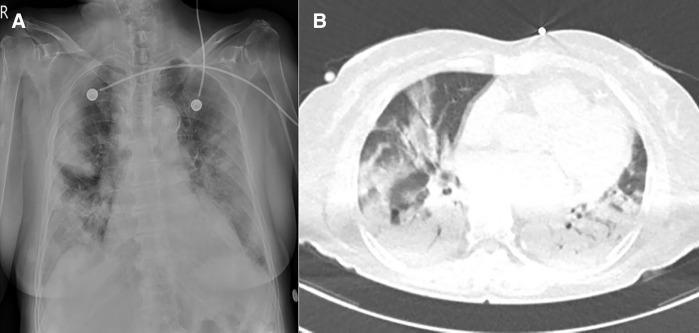
Management
Empirical Antibiotic Therapy
Per IDSA/ATS 2019 CAP Guidelines (cited in GLGCA, p. 3):
| Patient Group | Regimen |
|---|---|
| Healthy outpatient, no comorbidities | Amoxicillin or doxycycline or macrolide (azithromycin/clarithromycin) |
| Outpatient with comorbidities | β-lactam + macrolide or respiratory fluoroquinolone (levofloxacin, moxifloxacin) |
| Hospitalized (non-ICU) | β-lactam + macrolide or respiratory fluoroquinolone |
| Suspected Legionella | Fluoroquinolone or azithromycin preferred |
Key points:
- Macrolides and tetracyclines (doxycycline) cover atypical organisms; β-lactams alone do not
- IDSA/ATS recommends a minimum of 5 days of antibiotics, with clinical stability as the endpoint (resolution of vital sign abnormalities, ability to eat, normal mentation)
- Shorter courses supported by meta-analyses and RCTs
Supportive Care
- Adequate hydration, antipyretics, rest
- Supplemental oxygen if hypoxic
- ICU admission criteria: severe CAP (CURB-65 or PSI scoring)
Key Distinguishing Features vs. Typical Pneumonia
| Feature | Atypical | Typical |
|---|---|---|
| Onset | Gradual | Acute |
| Cough | Dry/non-productive | Productive, purulent |
| Fever | Low-grade | High, often with rigors |
| X-ray | Bilateral interstitial/GGO | Lobar consolidation |
| WBC | Normal/mildly elevated | Markedly elevated |
| β-lactam response | Poor | Good |
| Extra-pulmonary features | Common | Rare |
Complications
- Respiratory failure (especially with Legionella, viral pneumonia)
- Pleural effusion
- Hemolytic anemia (Mycoplasma — cold agglutinins)
- Neurological complications (Mycoplasma — encephalitis, Guillain-Barré)
- Myocarditis, pericarditis (rare, Mycoplasma)
Special Populations
- Young adults/college students: Mycoplasma predominates ("walking pneumonia")
- Elderly/immunocompromised: RSV, metapneumovirus, and Legionella more severe
- Epidemic/cluster setting: Legionella (water source exposure), adenovirus (military)
- Immunocompromised: Broader differential including PCP (Pneumocystis jirovecii), CMV, fungal
lung abscess and pulmonary gangrene definition,etiology, pathophysiology, clinical presentation, diagnosis, differential diagnosis, management, complications
search_medical_text
lung abscess definition etiology pathophysiology
search_medical_text
lung abscess clinical presentation diagnosis chest X-ray CT
search_medical_text
lung abscess management treatment antibiotics drainage surgery
search_medical_text
pulmonary gangrene definition pathophysiology management
search_medical_image
lung abscess chest X-ray CT scan air fluid level
GradeResponse
GradeResponse
GradeResponse
Lung Abscess & Pulmonary Gangrene
LUNG ABSCESS
Definition
A lung abscess is a localized area of necrosis and cavitation of the lung parenchyma resulting from microbial infection, forming a dominant cavity >2 cm in diameter, often with surrounding pneumonitis. It can be single or multiple (Harrison's, p. 3840).
Classification:
| Type | Criteria |
|---|---|
| Primary | Aspiration in a previously healthy host |
| Secondary | Complicates an underlying condition (malignancy, immunosuppression, bronchial obstruction) |
| Acute | Duration <4–6 weeks |
| Chronic | Duration >6 weeks |
Etiology
Aspiration is the dominant mechanism and underpins most primary lung abscesses. Key predisposing conditions and organisms:
Predisposing Conditions:
- Altered consciousness (alcoholism, seizures, general anesthesia, drug overdose)
- Dysphagia (neurological disease, esophageal disorders)
- Poor dental hygiene / periodontal disease
- Oropharyngeal/GI malignancy
- Bronchial obstruction (endobronchial tumor, foreign body)
- Immunocompromised states (HIV, transplant, corticosteroid use)
- Septic emboli (IV drug use, right-sided endocarditis)
- Hematogenous spread from distant infection
Causative Organisms:
| Category | Organisms |
|---|---|
| Anaerobes (most common) | Prevotella, Fusobacterium, Bacteroides, Peptostreptococcus |
| Aerobic/Facultative | S. aureus (incl. MRSA), K. pneumoniae, S. pyogenes, P. aeruginosa |
| Special settings | Nocardia, Actinomyces, Aspergillus, Entamoeba histolytica, Mycobacterium |
Pathophysiology
- Aspiration of oropharyngeal contents (anaerobe-rich) → organisms seed dependent lung segments
- Initial pneumonitis develops in the dependent zones (posterior segments of upper lobes, superior segments of lower lobes — favored in recumbent position)
- Tissue necrosis from bacterial enzymes, toxins, and host inflammatory response
- Liquefaction of necrotic tissue → abscess cavity forms
- Communication with a bronchus → air enters the cavity, creating the classic air-fluid level on imaging
- Surrounding wall of granulation tissue and fibrosis forms over time
- If drainage is inadequate → chronic abscess with thick fibrotic wall
Clinical Presentation
Subacute onset (days to weeks):
- Cough — initially dry, then productive of foul-smelling, purulent sputum (pathognomonic of anaerobic infection)
- Hemoptysis (can be massive)
- Fever, chills, night sweats
- Pleuritic chest pain
- Anorexia, weight loss (especially chronic abscess)
- Fatigue, malaise
Physical Examination:
- Decreased breath sounds over the affected area
- Amphoric/cavernous breath sounds (hollow, blowing quality over large cavity)
- Dullness to percussion (surrounding consolidation/fluid)
- Clubbing — may be present in chronic abscess
- Foul breath (halitosis) in anaerobic infections
- Fever and tachycardia
Diagnosis
Laboratory:
- Leukocytosis with left shift (neutrophilia)
- Elevated CRP, ESR
- Anemia (chronic disease in prolonged cases)
- Blood cultures (positive in hematogenous spread)
Microbiological:
- Sputum Gram stain and culture (limited by oropharyngeal contamination)
- Bronchoscopic samples (BAL, protected specimen brush) — preferred for anaerobes
- Transthoracic needle aspiration (if peripheral)
- Pleural fluid culture (if effusion present)
Imaging (Harrison's, p. 3845):
- Chest X-ray: thick-walled cavitary lesion with air-fluid level — the classic finding
- CT chest (preferred): better delineates cavity wall thickness, satellite lesions, underlying endobronchial obstruction or malignancy; distinguishes lung abscess from empyema
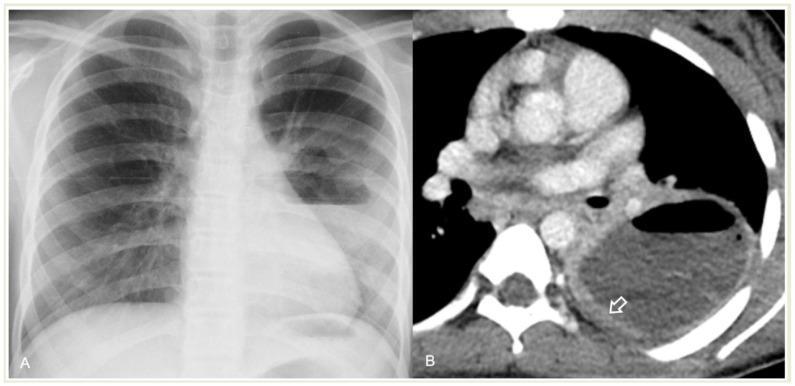
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Empyema with bronchopleural fistula | Air-fluid level spans the full width of hemithorax; D-shaped on CT; no enhancing lung wall |
| Cavitating carcinoma | Irregular, eccentric wall; no surrounding pneumonitis; mass in older smoker |
| Tuberculosis | Upper lobe predominance, satellite nodules, calcification, positive AFB |
| Pulmonary gangrene | Sloughed necrotic lung tissue floating in cavity ("floating island" sign on CT) |
| Necrotizing pneumonia | Multiple small cavities (<2 cm), more extensive parenchymal involvement |
| Fungal cavity (Aspergilloma) | Fungus ball with "air crescent sign" on CT; immunocompromised host |
| Wegener's/GPA | Bilateral, irregular cavities; ANCA positive; multi-organ involvement |
| Hydatid cyst | "Water lily sign"; exposure history; serology positive |
| Septic emboli | Multiple peripheral nodules/cavities; IV drug use; positive blood cultures |
Management
1. Antibiotic Therapy (Cornerstone of Treatment)
Empirical regimen must cover anaerobes and relevant aerobes:
| Setting | Regimen |
|---|---|
| Community-acquired (aspiration) | Amoxicillin-clavulanate OR clindamycin + β-lactam |
| Preferred first-line | Amoxicillin-clavulanate (covers anaerobes + aerobes) |
| Penicillin allergy | Clindamycin OR metronidazole + a β-lactam |
| Hospital-acquired / MRSA risk | Add vancomycin or linezolid |
| Pseudomonas risk | Piperacillin-tazobactam or carbapenem |
| Duration | 4–6 weeks (until radiographic resolution or a small stable residual scar) |
Note: Metronidazole alone is insufficient (poor activity against microaerophilic streptococci).
2. Postural Drainage & Physiotherapy
- Position patient to facilitate drainage through the relevant bronchus
- Chest physiotherapy to aid expectoration
- Avoid in patients with large cavities at risk of aspiration of purulent contents
3. Bronchoscopy
- Indications: exclude endobronchial obstruction (foreign body, tumor), obtain microbiological samples, facilitate drainage
- NOT used for therapeutic drainage of abscess itself (risk of soiling other lung segments)
4. Percutaneous Drainage (CT-guided)
- Indicated for: large abscess (>6 cm), failure of antibiotic therapy after 4–6 weeks, organisms resistant to antibiotics
- Increasingly preferred over surgery in poor surgical candidates
5. Surgery
- Reserved for failure of medical therapy (10–15% of cases)
- Indications:
- No response to antibiotics after 6–8 weeks
- Massive hemoptysis
- Suspected underlying malignancy
- Bronchopleural fistula
- Abscess >6 cm unresponsive to drainage
- Procedure: lobectomy or segmentectomy (pneumonectomy reserved for pulmonary gangrene)
Complications
- Bronchopleural fistula — communication between abscess and pleural space
- Empyema — most common complication; requires drainage
- Massive hemoptysis — erosion of pulmonary vessels; life-threatening
- Pyopneumothorax — abscess ruptures into pleural space
- Septicemia / bacteremia — hematogenous dissemination
- Brain abscess — haematogenous spread (especially anaerobes)
- Pulmonary gangrene — extension of necrosis to a large segment/lobe
- Chronic lung abscess — fibrotic thick-walled cavity, persistent infection
- Amyloidosis — in very chronic cases (rare)
PULMONARY GANGRENE
Definition
Pulmonary gangrene is a severe, life-threatening form of necrotizing pulmonary infection characterized by massive necrosis of one or more lobes (or an entire lung) with sloughing of necrotic lung tissue that remains within the pleural cavity or cavity. It represents the most extreme end of the spectrum:
Pneumonia → Necrotizing pneumonia → Lung abscess → Pulmonary gangrene
Unlike a simple lung abscess (localized, walled-off), pulmonary gangrene involves extensive, non-circumscribed necrosis of entire lobes with vascular thrombosis.
Etiology
Pulmonary gangrene is usually caused by highly virulent organisms or occurs in immunocompromised hosts:
| Pathogen | Notes |
|---|---|
| Klebsiella pneumoniae | Most common cause in adults; classic in alcoholics/diabetics |
| Staphylococcus aureus (MRSA) | Produces Panton-Valentine leukocidin (PVL); post-influenza |
| Pseudomonas aeruginosa | Immunocompromised, ICU patients |
| Streptococcus pneumoniae | Severe necrotizing pneumonia |
| Aspergillus spp. | Immunocompromised; angioinvasive |
| Anaerobes | Aspiration in debilitated patients |
| Acinetobacter baumannii | Hospital-acquired, ventilator-associated |
Risk factors: Diabetes, alcoholism, immunosuppression (HIV, organ transplant), malnutrition, prior structural lung disease, septic embolism.
Pathophysiology
- Virulent organism infects lung parenchyma → intense inflammatory response
- Vascular invasion and thrombosis of pulmonary vasculature (arteries and veins) → ischemia
- Loss of blood supply → massive coagulative necrosis of entire segments or lobes
- Liquefaction of necrotic tissue → large irregular cavities with sloughed, devitalized lung tissue
- The necrotic lung fragment detaches and floats in the fluid-filled cavity — the hallmark "pulmonary sequestrum" or "floating island" sign on CT
- Secondary infection by mixed flora accelerates destruction
- May extend to pleural space → pyopneumothorax, bronchopleural fistula
Clinical Presentation
- Acute, severely ill patient — unlike the subacute course of simple lung abscess
- High-grade fever, rigors, sepsis
- Productive cough with foul-smelling, often bloody-purulent sputum
- Massive hemoptysis (common; can be life-threatening)
- Severe pleuritic chest pain
- Dyspnea, tachypnea, cyanosis
- Rapid deterioration → respiratory failure, septic shock
- Examination: signs of consolidation/pleural effusion; toxic appearance
Diagnosis
Imaging:
- Chest X-ray: large area of consolidation progressing to a large, irregular cavitary lesion; may see a dense shadow within the cavity (sloughed lung)
- CT chest (diagnostic of choice):
- Large, irregular cavitary lesion with thick walls
- "Floating island" / pulmonary sequestrum sign: sloughed necrotic lung fragment floating within the fluid-filled cavity
- Extensive surrounding consolidation, pleural effusion
- Vascular cut-off or thrombosis in adjacent vessels (CT angiography)
- Pyopneumothorax if bronchopleural fistula develops
Laboratory:
- Severe leukocytosis or leukopenia (ominous)
- Elevated procalcitonin, CRP
- Blood cultures — frequently positive
- Sputum culture, BAL
- Consider bronchoscopy for sampling
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Lung abscess | Smaller (<lobe), walled-off, responds to antibiotics; no sequestrum |
| Necrotizing pneumonia | Multiple small cavities, no sequestrum, less severe |
| Empyema necessitatis | Pleural-based; D-shaped; no intrapulmonary cavity |
| Cavitating malignancy | Gradual course; irregular wall; no systemic toxicity |
| Pulmonary infarction | Hampton's hump; peripheral; vascular occlusion on CTPA |
| Invasive aspergillosis | Air crescent sign; immunocompromised; galactomannan positive |
Management
Pulmonary gangrene carries high mortality (30–60%) and requires aggressive multi-modal treatment:
1. ICU Admission & Supportive Care
- High-flow oxygen / mechanical ventilation if respiratory failure
- Vasopressors for septic shock
- Nutritional support (often severely malnourished)
2. Aggressive Antibiotic Therapy
- Broad-spectrum from the outset; narrowed based on cultures
- Cover gram-negatives, S. aureus (MRSA), and anaerobes:
- Piperacillin-tazobactam or carbapenem (meropenem/imipenem) PLUS
- Vancomycin or linezolid (MRSA cover) PLUS
- Metronidazole (additional anaerobic cover if not already included)
- Duration: prolonged (6–8 weeks minimum), guided by clinical and radiological response
3. Drainage Procedures
- Chest tube (tube thoracostomy): mandatory if empyema or pyopneumothorax
- CT-guided percutaneous drainage: if accessible collection
- Bronchoscopy: to clear secretions and obtain cultures
4. Surgery (Definitive for Non-Responders)
- Often unavoidable in pulmonary gangrene
- Pneumonectomy (removal of entire lung) is frequently required due to extent of destruction
- Lobectomy if gangrene is confined to a single lobe
- High surgical mortality in the acute phase — timing is critical (ideally after stabilization)
- Decortication if organized empyema present
5. Adjunctive Measures
- Antifungal therapy if fungal co-infection suspected
- Immunoglobulin (IVIG) in severe S. aureus PVL-toxin-mediated disease (off-label)
- Hyperbaric oxygen — limited evidence, used in some centers
Complications
- Massive, life-threatening hemoptysis
- Bronchopleural fistula — very common; prolongs hospital stay
- Pyopneumothorax — rupture into pleural space
- ARDS (acute respiratory distress syndrome)
- Septic shock and multi-organ failure
- Mediastinitis — if infection spreads
- Post-pneumonectomy complications: empyema, respiratory failure
- Death — mortality 30–60% even with aggressive management
Comparison: Lung Abscess vs. Pulmonary Gangrene
| Feature | Lung Abscess | Pulmonary Gangrene |
|---|---|---|
| Extent | Localized cavity >2 cm | Lobar/whole-lung necrosis |
| Onset | Subacute (weeks) | Acute, fulminant |
| Key organisms | Anaerobes, S. aureus | Klebsiella, MRSA, Pseudomonas |
| CT hallmark | Thick-walled cavity, air-fluid level | "Floating island" sequestrum |
| Vascular thrombosis | Absent | Present (central to pathogenesis) |
| Response to antibiotics | Usually good (85–90%) | Poor; often needs surgery |
| Surgical need | 10–15% | Majority |
| Mortality | <5% with treatment | 30–60% |
| Typical surgery | Lobectomy (rare) | Pneumonectomy |
This is a shared conversation. Sign in to Orris to start your own chat.