A five-year-old boy who was bitten by a dog and presented to the emergency department with multiple scratches on the upper arms and neck. His parents stated that the dog was a stray that lived in their locality (and hence could be observed). Wound toilet was performed, followed by the application of povidone-iodine. The wound was left open. The dog died two days later. Its brain was sent to the veterinary lab and tested positive for Negri bodies on histopathology. a) What are Negri bodies? b) Write in detail about the pathogenesis, laboratory diagnosis, treatment and PEP for Dog bite?
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have all the material needed for a comprehensive answer. Here is the full, structured response:
Rabies Following Dog Bite — Comprehensive Answer
a) Negri Bodies
Negri bodies are the pathognomonic histopathological finding of rabies virus infection in the nervous system. They are:
- Nature: Intracytoplasmic, round-to-oval, eosinophilic inclusion bodies found within infected neurons
- Composition: Filled with rabies virus nucleocapsids (aggregates of viral RNP — ribonucleoprotein complexes); the inclusions represent viral replication factories and storage sites
- Location: Classically found in:
- Pyramidal neurons of the hippocampus (most characteristic)
- Purkinje cells of the cerebellum
- Less frequently in cortical and brainstem neurons
- Also seen in dorsal root ganglia
- Staining: Eosinophilic (pink) on H&E; they may show internal basophilic granules (viral nucleocapsid particles) within the eosinophilic matrix
- Size: Variable; typically 2–10 µm in diameter
- Significance: Their presence is pathognomonic of rabies, but their absence does not rule out rabies — they are found in only 70–90% of infected brain tissue (not observed in at least 20% of cases, particularly rare in bats)
- Detection: Negri bodies contain rabies virus antigens detectable by immunohistochemical staining (IHC); they can also be visualized by electron microscopy, which reveals the rhabdovirus particles within
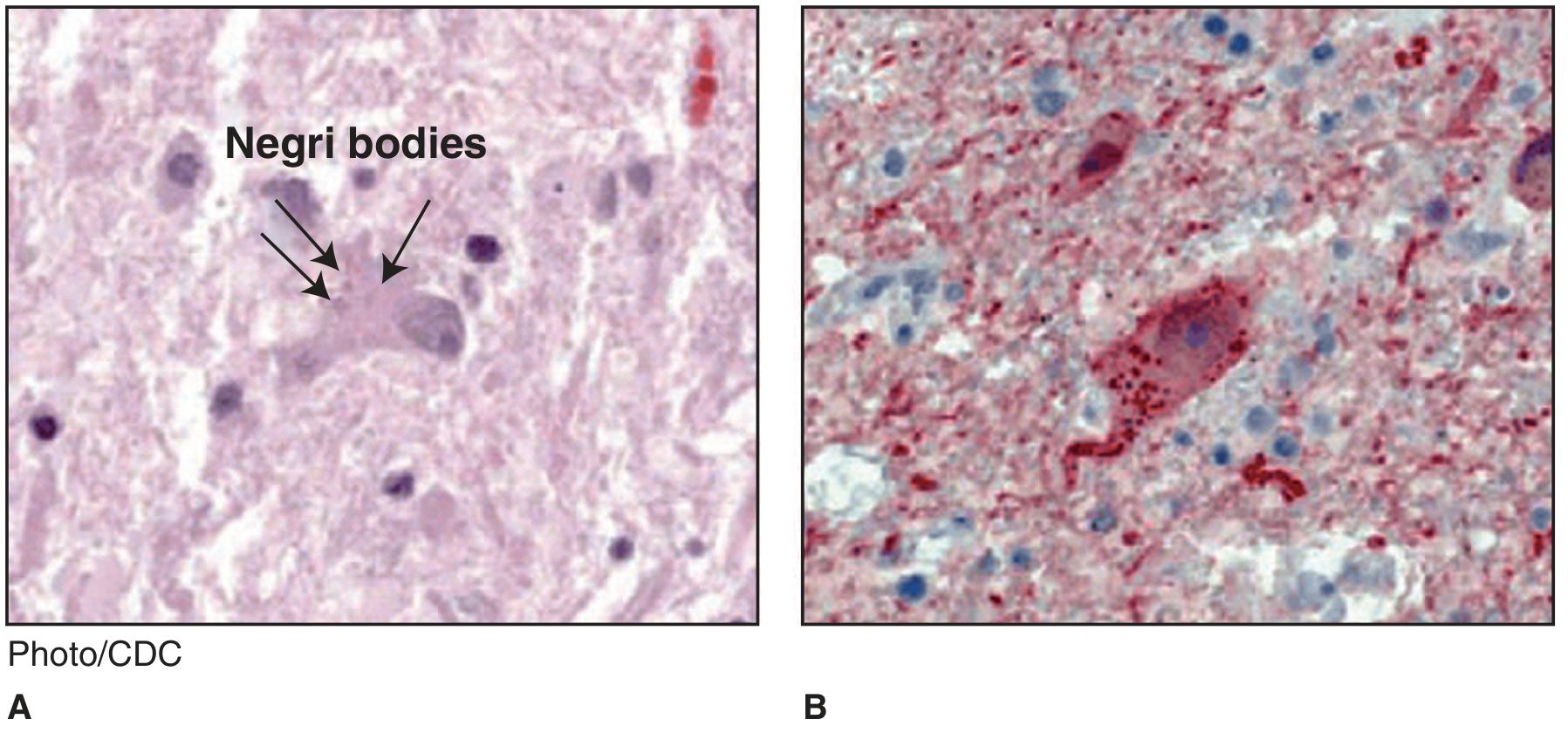
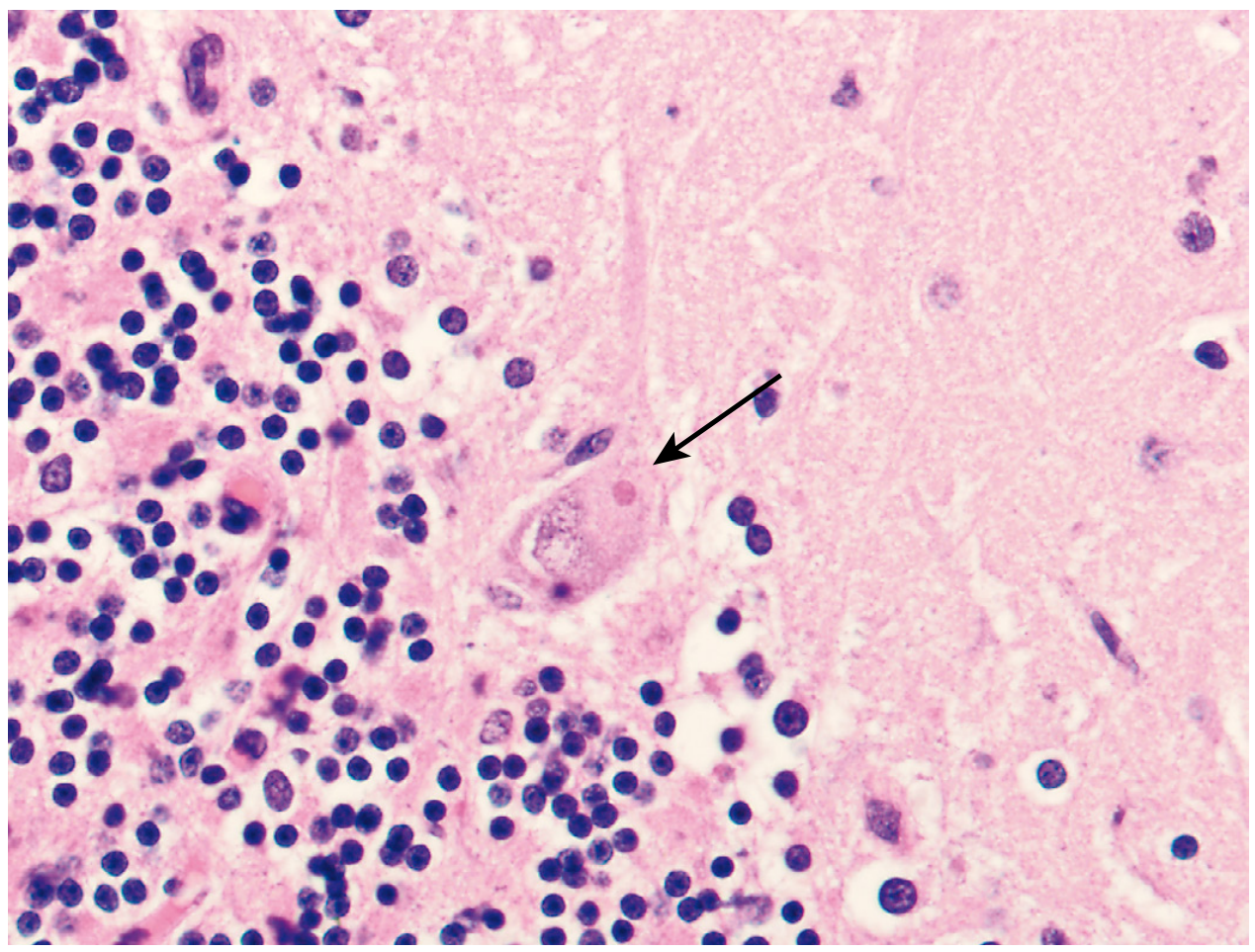
The pathologic picture also includes widespread neuronal degeneration, intense cerebral edema, vascular congestion, and a mononuclear inflammatory infiltrate most severe in the brainstem, basal ganglia, spinal cord, and dorsal root ganglia — notably, there is minimal inflammation at the hippocampus and cerebellum despite Negri body presence.
b) Pathogenesis, Laboratory Diagnosis, Treatment & PEP for Dog Bite
1. PATHOGENESIS
The Virus
Rabies virus (RABV) is a single-stranded, negative-sense RNA virus in the family Rhabdoviridae, genus Lyssavirus. It is bullet-shaped (75 × 180 nm), enveloped, with surface glycoprotein spikes. The viral glycoprotein (G protein) is the key antigen responsible for virus attachment and the induction of virus-neutralizing antibodies.
Step-by-Step Pathogenesis
| Phase | Events |
|---|---|
| 1. Inoculation | Virus deposited in muscle/subcutaneous tissue at bite site |
| 2. Local replication | Virus replicates in striated muscle cells near the inoculation site; binds nicotinic acetylcholine receptors at neuromuscular junctions |
| 3. Peripheral nerve entry | Virus enters peripheral nerves at neuromuscular junctions (bypassing the blood) and travels via retrograde fast axonal transport (~250 mm/day) centripetally toward the spinal cord and brainstem |
| 4. CNS dissemination | Once in the CNS, virus spreads rapidly along neuroanatomic connections via fast axonal transport; neurons are predominantly infected; astrocyte infection is unusual |
| 5. Centrifugal spread | After CNS amplification, virus spreads centrifugally along sensory and autonomic nerves to salivary glands, cornea, skin, heart, adrenal glands, kidney, and pancreas |
| 6. Salivary shedding | Virus replicates in acinar cells of the submaxillary salivary glands (highest viral titers) and is shed in saliva — enabling onward transmission |
Key determinants of incubation period:
- Distance from bite site to CNS (face/neck bites → shorter incubation)
- Severity of laceration and size of inoculum
- Bite over richly innervated areas (higher risk)
- Age and immune status of the host
Incubation period: Typically 20–90 days (range: a few days to >1 year). During this period, virus is thought to remain near the inoculation site, explaining why local wound care is effective when prompt.
Clinical Stages:
| Stage | Duration | Features |
|---|---|---|
| Incubation | 20–90 days | Asymptomatic |
| Prodrome | 2–10 days | Fever, malaise, headache, anorexia; paresthesias/pain/pruritus at wound site (hallmark) |
| Acute neurologic (encephalitic/furious, 80%) | 2–7 days | Anxiety, agitation, hyperexcitability, hallucinations, autonomic dysfunction, hydrophobia, aerophobia, hypersalivation ("foaming at mouth"), priapism |
| Acute neurologic (paralytic/dumb, 20%) | 2–10 days | Flaccid ascending paralysis, quadriparesis, facial paralysis — may mimic Guillain-Barré |
| Coma → Death | 0–14 days | Cardiorespiratory failure; multiple organ failure |
Hydrophobia and aerophobia result from dysfunction of infected brainstem neurons that normally inhibit inspiratory neurons near the nucleus ambiguus, producing exaggerated defense reflexes. Mortality is nearly 100% once symptoms develop.
2. LABORATORY DIAGNOSIS
Multiple specimens and techniques are required, as no single test is 100% sensitive antemortem. Negative tests do not exclude rabies.
Antemortem (In Living Patients)
| Test | Specimen | Notes |
|---|---|---|
| Direct Fluorescent Antibody (DFA) test | Skin biopsy (nape of neck), corneal impression smear | DFA detects rabies virus antigen in cutaneous nerves at hair follicle bases; skin biopsy preferred; corneal smears have low yield |
| RT-PCR | Saliva, CSF, skin biopsy, brain tissue | Highly sensitive and specific; detects viral RNA; can identify specific viral variant to trace source |
| Virus-neutralizing antibodies | Serum, CSF | In unvaccinated patients, serum antibodies = diagnostic; CSF antibodies = diagnostic regardless of vaccination status; may not appear until late in disease |
| Virus isolation | Saliva, brain tissue | Cell culture (mouse neuroblastoma cells) or mouse inoculation |
Postmortem (In Dead Animals/Humans)
| Test | Specimen | Notes |
|---|---|---|
| DFA (gold standard) | Brain tissue (hippocampus, cerebellum, brainstem) | Standard postmortem test; highly sensitive and specific; rapid result |
| Histopathology (H&E) | Brain tissue | Demonstrates Negri bodies in 70–90% of cases; presence is pathognomonic; absence does not exclude rabies |
| Immunohistochemistry (IHC) | Brain tissue | More sensitive than H&E alone; detects antigen even in Negri body–negative specimens |
| RT-PCR | Brain tissue | Detects viral RNA; useful when DFA is equivocal; allows variant identification |
| Mouse inoculation test | Brain suspension | Intracerebral inoculation; develops disease in 6–12 days; confirmatory but slow |
In this case: The dog's brain tested positive for Negri bodies on H&E, confirming rabies. The child is fully exposed and requires immediate PEP.
3. TREATMENT
There is no established curative treatment for clinical rabies once symptoms develop. Management is essentially supportive.
- Palliative/Supportive care: Sedation, analgesia, mechanical ventilation, management of autonomic instability
- "Milwaukee Protocol" (therapeutic coma): A combination of ketamine-induced coma, ribavirin, and amantadine was used in one famously recovered unvaccinated patient in 2004. However, subsequent application of this protocol in >60 cases has failed to replicate the result, and it is no longer recommended as standard practice
- Expert consultation is mandatory before embarking on experimental therapy
- Prevention of secondary transmission: Barrier precautions for healthcare workers; rabies is not transmitted person-to-person through casual contact
- Prognosis: Almost uniformly fatal; of ~33 documented survivors worldwide, all but 2 had received at least one dose of rabies vaccine before illness onset
4. POST-EXPOSURE PROPHYLAXIS (PEP) FOR DOG BITE
PEP must be initiated immediately after a confirmed or suspected rabies exposure; it is highly effective if started promptly during the incubation period, before virus enters the CNS.
Step 1 — Wound Care (Most Critical First Step)
- Thorough wound washing with soap and water for at least 15 minutes
- Application of povidone-iodine or 70% alcohol (viricidal)
- Leave wound open (do not primarily suture — promotes viral retention)
- Tetanus prophylaxis and antibiotics as indicated
This step alone can prevent rabies — local wound care is the single most important immediate intervention.
Step 2 — Assess Need for PEP
| Animal | Situation | Action |
|---|---|---|
| Dog/cat/ferret (healthy, available) | Bite occurred | Observe for 10 days — if animal remains healthy, PEP not needed; if animal dies or develops signs of rabies → euthanize, test brain, initiate PEP immediately |
| Dog/cat/ferret (died, brain positive for rabies) | As in this case | Initiate PEP immediately |
| Wild animal (bat, skunk, raccoon, fox) | Any bite | PEP mandatory regardless |
| Rodents, rabbits | Bite | Rarely require PEP (consult public health) |
In this case: The dog died and brain confirmed Negri bodies (rabies positive) → Full PEP must be initiated immediately.
Step 3 — Rabies Immune Globulin (RIG) — Passive Immunization
- Purpose: Provides immediate, passive antibody protection while the vaccine stimulates active immunity
- Human Rabies Immune Globulin (HRIG): 20 IU/kg body weight — preferred
- Equine RIG (ERIG): 40 IU/kg — used when HRIG unavailable; lower adverse effect rate with modern purified products
- Administration: As much as anatomically feasible should be infiltrated directly into and around the wound; remainder given IM at a site distant from vaccine
- Timing: Must be given on Day 0 (same day as first vaccine dose), ideally; can be given up to Day 7 of vaccine series but not after
- Key rule: RIG is not given to previously vaccinated persons (would suppress their active immune response)
Step 4 — Rabies Vaccine — Active Immunization
Available vaccines (all inactivated):
- Human Diploid Cell Vaccine (HDCV) — grown in MRC-5 cell line, inactivated with β-propiolactone
- Purified Chick Embryo Cell Vaccine (PCEC) — grown in chicken fibroblasts, inactivated with β-propiolactone
Schedule for Previously Unvaccinated Persons:
| Day | Vaccine Dose |
|---|---|
| Day 0 | 1st dose (+ RIG given simultaneously at different site) |
| Day 3 | 2nd dose |
| Day 7 | 3rd dose |
| Day 14 | 4th dose |
(In immunocompromised patients, a 5th dose on Day 28 is sometimes added)
Schedule for Previously Vaccinated Persons:
- RIG is NOT given
- Only 2 booster doses of vaccine: Day 0 and Day 3
Pre-exposure Prophylaxis (PrEP) — For High-Risk Individuals
- Indicated for: veterinarians, laboratory workers, spelunkers, wildlife workers, travelers to rabies-endemic areas
- Schedule: 2 doses on Days 0 and 7
- Periodic serum neutralizing antibody titers monitored; boosters when titers fall below protective levels
Summary Algorithm for This Case
Dog bite (5-year-old) → Wound toilet + povidone-iodine + open wound ✓
↓
Dog observed (available) — dog died Day 2
↓
Brain: Negri bodies positive = CONFIRMED RABIES
↓
IMMEDIATE PEP:
• HRIG 20 IU/kg — infiltrate into wound + IM remainder (Day 0)
• Rabies vaccine (HDCV or PCEC) — Days 0, 3, 7, 14
• Wound care (already done) ✓
• Tetanus prophylaxis
Sources: Jawetz, Melnick & Adelberg's Medical Microbiology 28e | Harrison's Principles of Internal Medicine 22e (2025) | Robbins, Cotran & Kumar Pathologic Basis of Disease | Bradley & Daroff's Neurology in Clinical Practice
This is a shared conversation. Sign in to Orris to start your own chat.