Important terms, types of body fluid, total body water
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Reading File
Here is a comprehensive overview of body fluids, key terms, and total body water, drawn from Costanzo Physiology, Guyton & Hall, and Morgan & Mikhail's Clinical Anesthesiology.
Body Fluids: Key Terms & Compartments
Total Body Water (TBW)
Water is the largest single constituent of the body. TBW accounts for 50%–70% of body weight, with an average of 60% in a typical adult man.
The 60-40-20 Rule (a classic clinical memory aid):
| Fraction | % Body Weight | Volume (70-kg man) |
|---|---|---|
| Total Body Water | 60% | ~42 L |
| Intracellular Fluid (ICF) | 40% | ~28 L |
| Extracellular Fluid (ECF) | 20% | ~14 L |
Factors affecting TBW:
- TBW inversely correlates with body fat — fat tissue contains little water
- Thin men → highest (~70%); obese women → lowest (~50%)
- Newborns/premature infants: 70–75% of body weight
- TBW decreases progressively with age due to increased fat and loss of muscle mass
A sudden weight change (in absence of other causes) directly reflects a change in body water — e.g., a 3 kg overnight weight loss = ~3 L of water lost.
Body Fluid Compartments
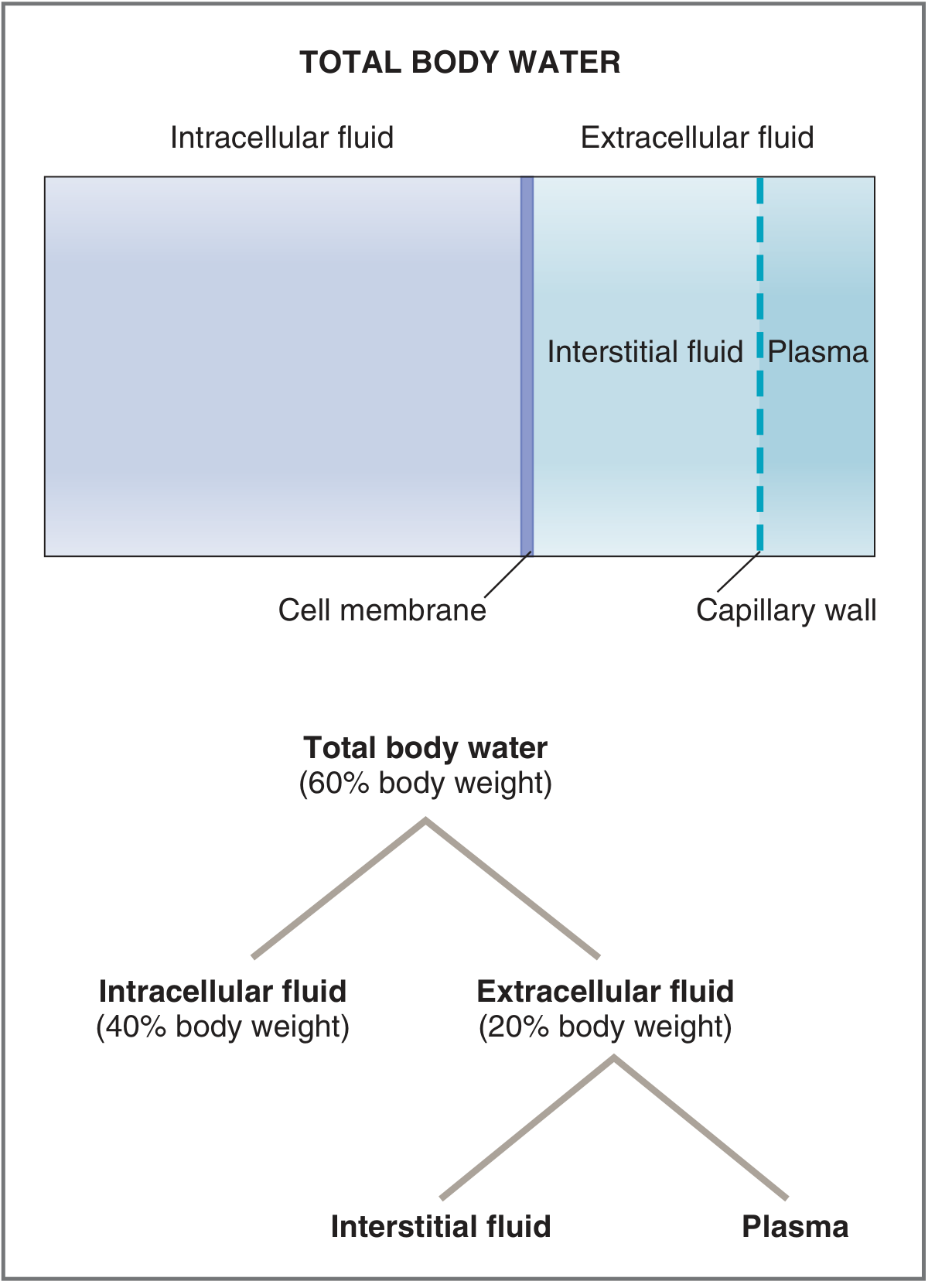
Fig. 6.4 Body fluid compartments — Costanzo Physiology 7th Ed.
TBW is divided into two major compartments separated by the cell membrane:
1. Intracellular Fluid (ICF)
- Two-thirds of TBW (≈40% of body weight, ~28 L)
- Fluid inside all cells, collectively treated as one compartment
- Major ions: K⁺ and Mg²⁺ (cations); proteins and organic phosphates (ATP, ADP, AMP) as anions
2. Extracellular Fluid (ECF)
- One-third of TBW (≈20% of body weight, ~14 L)
- All fluid outside the cells; separated from ICF by the cell membrane
- Subdivided into two main compartments separated by the capillary wall:
| Subcompartment | Fraction of ECF | Volume (~70-kg man) |
|---|---|---|
| Interstitial fluid | ~¾ of ECF (25% of TBW) | ~10.5 L |
| Plasma | ~¼ of ECF (8% of TBW) | ~3.5 L |
Plasma is the fluid circulating in blood vessels. It constitutes ~55% of blood volume; red cells make up the rest (hematocrit ~42–45%).
Interstitial fluid bathes the cells directly. It is an ultrafiltrate of plasma — formed by filtration across capillary walls. Because the capillary wall is nearly impermeable to large proteins, interstitial fluid contains little or no protein (vs. plasma, which contains ~7% protein by volume).
3. Transcellular Fluid (minor compartment)
A small, specialized subset of ECF (~1–2 L total):
- Cerebrospinal fluid (CSF)
- Synovial fluid
- Pleural fluid
- Peritoneal fluid
- Pericardial fluid
- Intraocular fluid
- Digestive secretions
Composition of the Compartments
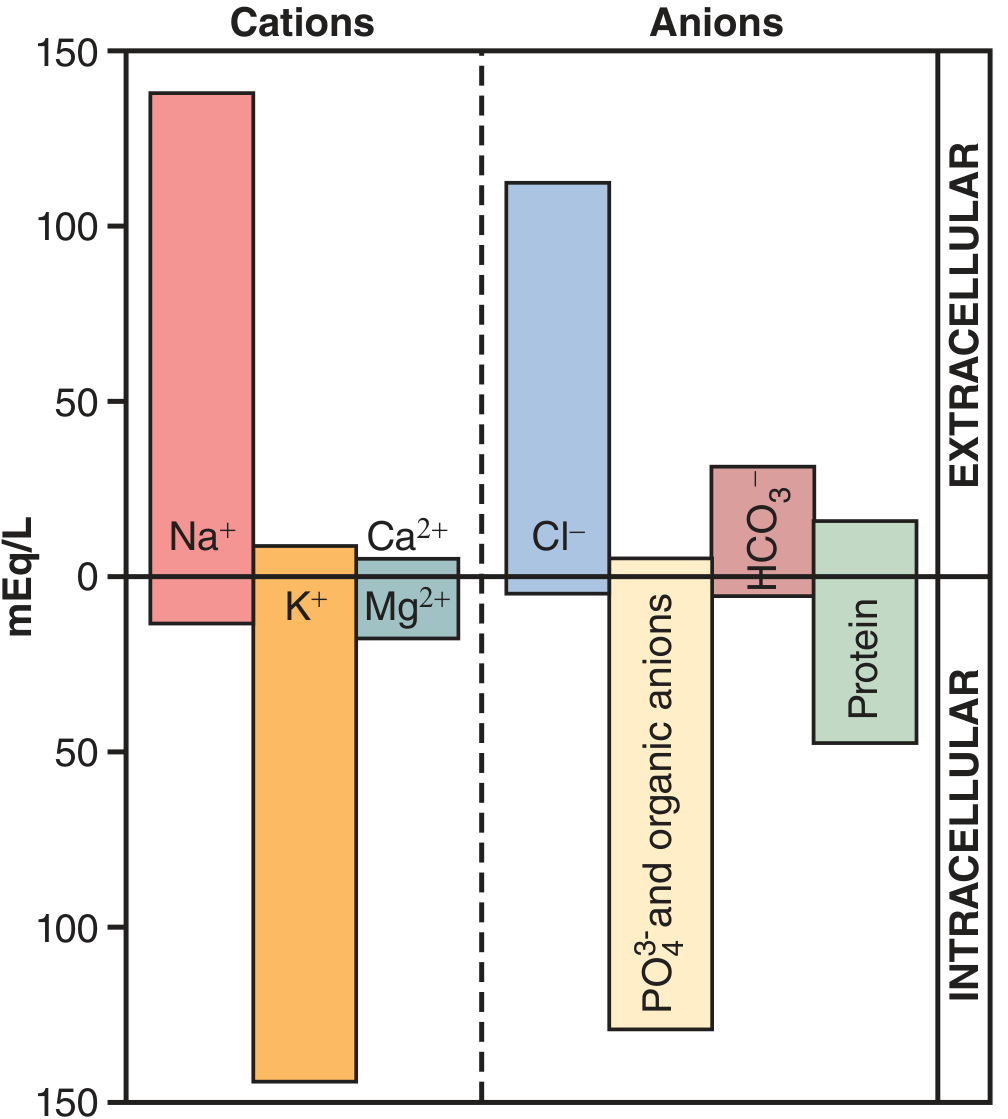
Fig. 25.2 ICF vs. ECF ionic composition — Guyton & Hall Medical Physiology
| Ion | ECF (Plasma) | ICF |
|---|---|---|
| Na⁺ | 142 mOsm/L (major cation) | 14 mOsm/L |
| K⁺ | 4.2 mOsm/L | 140 mOsm/L (major cation) |
| Cl⁻ | 106 mOsm/L (major anion) | 4 mOsm/L |
| HCO₃⁻ | 24 mOsm/L | 10 mOsm/L |
| PO₄³⁻ / HPO₄²⁻ | ~2 mOsm/L | 11 mOsm/L (major anion) |
| Proteins | High (plasma) / ~0 (interstitial) | High |
Key rule: ECF is dominated by Na⁺, Cl⁻, HCO₃⁻; ICF is dominated by K⁺, Mg²⁺, phosphates, and proteins.
Important Terms
| Term | Definition |
|---|---|
| Osmole | The number of particles into which a solute dissociates in solution |
| Osmolarity | Concentration of solute particles per liter of solution (mOsm/L) |
| Osmolality | Concentration of solute particles per kilogram of solvent (mOsm/kg) |
| Tonicity | The effect of a solution on cell volume; isotonic = no change; hypotonic = cells swell; hypertonic = cells shrink |
| Mole | 6 × 10²³ molecules of a substance |
| Equivalent (Eq) | Moles × valence; used for charged (ionized) solutes |
| Hematocrit | % of blood volume occupied by red blood cells (~42–45%) |
| Osmotic pressure | Pressure needed to prevent net water movement across a semipermeable membrane toward the more concentrated side |
| Gibbs-Donnan effect | Redistribution of small ions between plasma and interstitial fluid due to the negative charge of plasma proteins — plasma has slightly higher Na⁺ and lower Cl⁻ than interstitial fluid |
| Ultrafiltrate | Fluid filtered across the capillary wall; same as plasma but lacking large proteins and cells |
Osmolarity of NaCl example: 1 mmol/L NaCl → 2 mOsm/L (NaCl dissociates into Na⁺ + Cl⁻ = 2 particles). In practice, NaCl behaves as ~75% ionized due to ionic interactions, so 1 mOsm/L difference = ~19.3 mmHg osmotic pressure.
Measuring Compartment Volumes (Dilution Principle)
| Compartment | Marker Used |
|---|---|
| Total body water | Isotopic water (D₂O, tritiated water), antipyrine |
| ECF volume | Mannitol, inulin, sulfate (cannot cross cell membranes) |
| Plasma volume | Radioactive albumin, Evans blue dye |
| ICF volume | Calculated: TBW − ECF |
| Interstitial fluid | Calculated: ECF − plasma volume |
ICF and interstitial fluid have no direct markers and must be derived by subtraction.
Sources: Costanzo Physiology 7th Ed., pp. 9, 253–255 | Guyton & Hall Medical Physiology, pp. 309–311 | Morgan & Mikhail's Clinical Anesthesiology 7th Ed., pp. 2135–2136
General principles for fluid balance,
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive overview of the general principles of fluid balance, compiled from Schwartz's Surgery, Mulholland & Greenfield's Surgery, and Costanzo Physiology.
General Principles of Fluid Balance
1. Principle of Osmotic Equilibrium
All fluid compartments are at osmotic equilibrium with each other. Because cell membranes are freely permeable to water (but not most solutes), any change in osmolality in one compartment triggers water redistribution until equilibrium is restored.
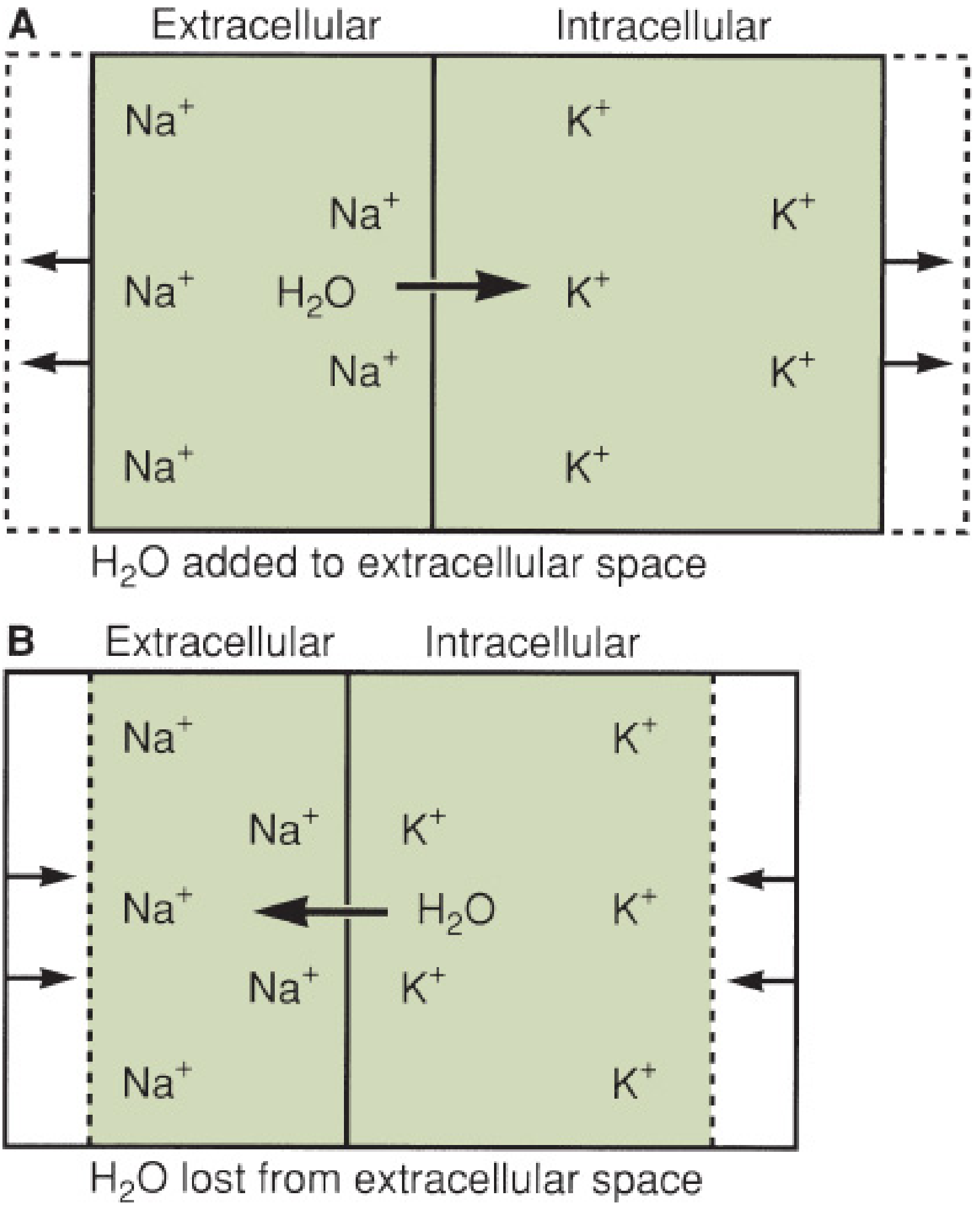
Fig. 11.1 — Mulholland & Greenfield's Surgery 7e: (A) Free water added to ECF → osmolality falls → water shifts INTO cells. (B) Free water lost from ECF → osmolality rises → water shifts OUT of cells.
Normal plasma osmolality = 290–310 mOsm/L in all compartments simultaneously.
Serum osmolality formula:
P_osm = 2 × [Na⁺] + Glucose/18 + BUN/2.8
Since Na⁺ is the dominant ECF solute, serum [Na⁺] is the primary indicator of water balance, not volume status.
2. Normal Daily Fluid Exchange (Intake vs. Output)
| Input | Volume |
|---|---|
| Oral liquids | ~1500 mL |
| Water from solid food | ~500 mL |
| Total | ~2000 mL/day |
| Output | Volume |
|---|---|
| Urine | 800–1200 mL |
| Stool | ~250 mL |
| Insensible losses (skin 75%, lungs 25%) | ~600 mL |
| Total | ~2000 mL/day |
Insensible losses increase with fever, hyperventilation, and hypermetabolism. The kidneys must produce a minimum of 500–800 mL/day to clear metabolic waste, regardless of intake.
Dietary sodium intake is typically 3–5 g/day. The kidney can adjust urinary sodium between 1 mEq/day (maximum conservation) and 5000 mEq/day (maximum excretion).
3. Principle of Electroneutrality
The milliequivalents of cations must always equal anions in every compartment. This principle governs ionic composition and is the basis for understanding acid-base disturbances and anion gap calculations.
4. How Fluids Distribute After Administration
The distribution of an administered fluid depends entirely on its solute content:
| Fluid Type | Distribution | Effect |
|---|---|---|
| Free water | Distributes across ALL compartments equally | Expands all three compartments proportionally |
| Isotonic saline (0.9% NaCl) | Stays in ECF only (Na⁺ is confined to ECF) | Expands interstitial space ~3× more than plasma |
| Hypertonic saline | Draws water from ICF into ECF | Shrinks cells, expands ECF |
| Colloids (albumin) | Stay in intravascular space | Expand plasma volume preferentially |
"Although the administration of sodium-containing fluids expands intravascular volume, it also expands the interstitial space by approximately three times as much as the plasma." — Schwartz's Surgery 11e
5. Classification of Fluid Balance Disorders
Three categories, which may coexist but each requires individual correction:
| Category | Key Feature | Example |
|---|---|---|
| Volume disturbance | Isotonic gain or loss of salt solution; mainly affects ECF | Hemorrhage, diarrhea |
| Concentration disturbance | Change in free water → alters serum [Na⁺] and osmolality | Hyponatremia, hypernatremia |
| Composition disturbance | Change in specific ion (not Na⁺) without major osmolarity change | Hypokalemia, hypercalcemia |
Extracellular volume deficit is the most common fluid disorder in surgical patients. Acute deficits cause cardiovascular and CNS signs (tachycardia, hypotension); chronic deficits also cause tissue signs (reduced skin turgor, sunken eyes). Lab clues: elevated BUN, urine osmolality > serum osmolality, urine sodium < 20 mEq/L.
6. Regulatory Mechanisms — How the Body Maintains Fluid Balance
A. Osmoreceptors & ADH (Vasopressin)
- Specialized cells in the anterolateral hypothalamus detect changes in ECF tonicity
- Respond to changes as small as 1–2% in plasma osmolality
- A rise in osmolality → ADH released from posterior pituitary → kidneys reabsorb free water → osmolality falls back to normal
- ADH also stimulated non-osmotically by a 10–20% drop in blood volume — baroreceptors override osmoreceptors in severe hypovolemia
B. Baroreceptors & Volume Control
- Stretch receptors located in: intrathoracic capacitance vessels, atria, aortic arch, carotid arteries, intrarenal afferent arterioles, hepatic and CSF receptors
- Detect effective circulating volume (the fraction of ECF actually perfusing organs)
- Signal through neural and hormonal pathways to adjust renal Na⁺ and water handling
C. Renin-Angiotensin-Aldosterone System (RAAS)
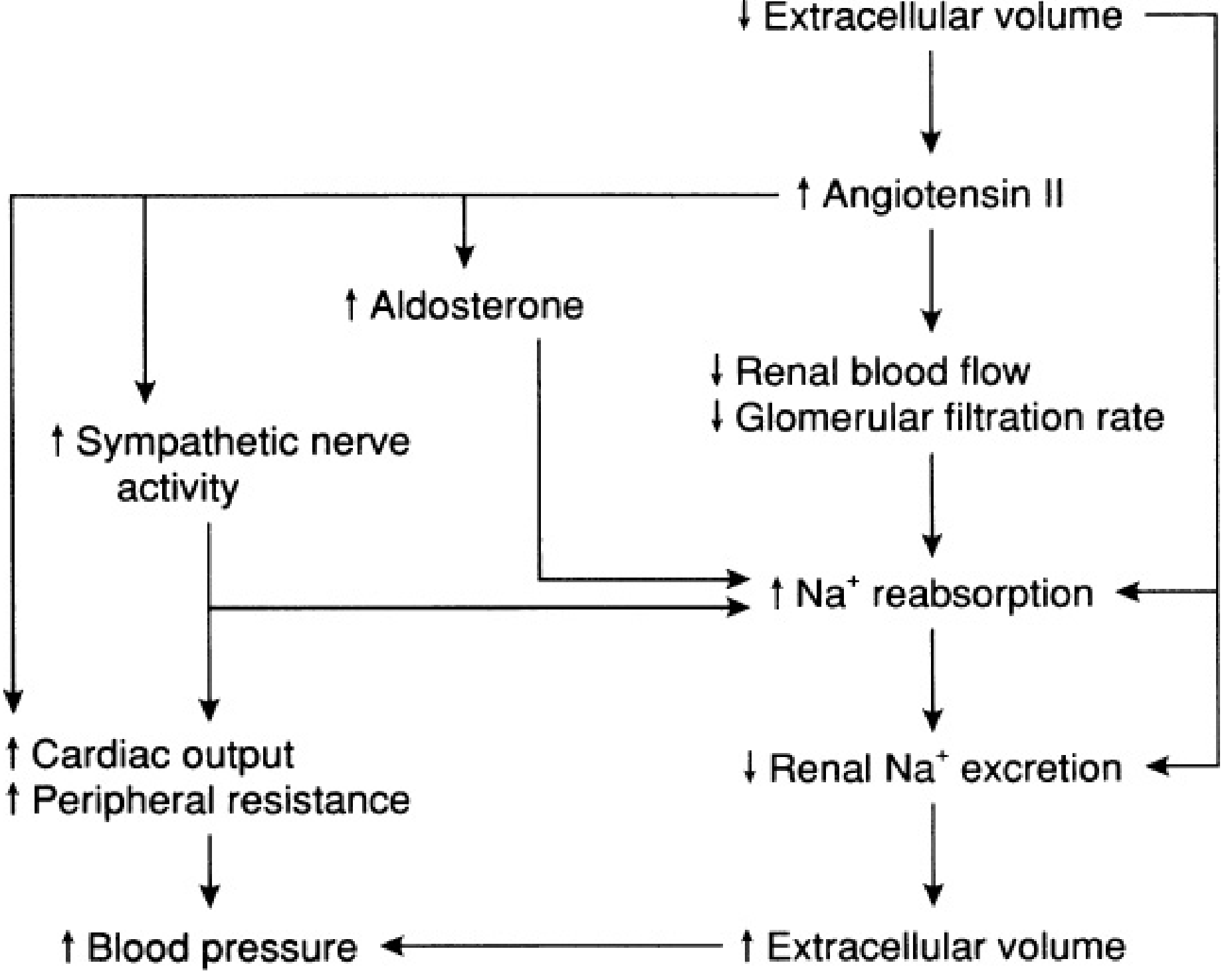
Fig. 11.5 — Mulholland & Greenfield's Surgery 7e: Effects of ↑ Angiotensin II in response to ↓ ECF volume.
Stimulus → Renin release from juxtaglomerular cells when:
- Arterial pressure falls
- NaCl delivery to macula densa decreases
- β-adrenergic activity increases
Cascade:
- Renin cleaves angiotensinogen (liver) → Angiotensin I
- ACE (pulmonary endothelium) → Angiotensin II
- Angiotensin II effects: vasoconstriction, ↑ aldosterone, ↓ GFR, ↑ Na⁺ reabsorption, ↑ ADH, thirst
- Aldosterone (adrenal zona glomerulosa): opens epithelial Na⁺ channels in distal tubule → Na⁺ reabsorption + obligate K⁺ loss via ROMK channel
D. Atrial Natriuretic Peptide (ANP) — Counter-regulatory
- Released by atrial myocytes in response to atrial wall distension (volume overload)
- Directly inhibits renal Na⁺ reabsorption → natriuresis and diuresis
- Counter-balances RAAS when volume is excessive
7. The Serum Sodium–TBW Relationship
Since Na⁺ is confined to ECF, serum [Na⁺] reflects TBW content, not just sodium:
Serum [Na⁺] = (Na⁺_e + K⁺_e) / TBW
Because total body solute (Na⁺ + K⁺) remains relatively stable, changes in TBW are inversely proportional to serum [Na⁺]:
- Hyponatremia → excess TBW (water overload)
- Hypernatremia → deficit of TBW (free water deficit)
Summary Flowchart
↓ Volume / ↑ Osmolality
↓
Osmoreceptors + Baroreceptors activated
↓
ADH release → ↑ free water reabsorption (kidney)
RAAS activation → Na⁺ + water retention
Thirst → ↑ water intake
↓
ECF volume and osmolality restored
Sources: Schwartz's Principles of Surgery 11e, pp. 111–113 | Mulholland & Greenfield's Surgery 7e, pp. 658–669 | Costanzo Physiology 7th Ed., pp. 253–254
This is a shared conversation. Sign in to Orris to start your own chat.