Diabetes types, diagnosis criteria, management
diabetes mellitus types classification pathophysiology type 1 type 2 MODY gestational
diabetes diagnosis criteria fasting glucose HbA1c OGTT ADA guidelines
diabetes management treatment insulin metformin lifestyle glycemic targets
Diabetes Mellitus
Overview
Types / Classification
| Type | Pathophysiology | Key Features |
|---|---|---|
| Type 1 DM | Autoimmune destruction of pancreatic β-cells → absolute insulin deficiency | Usually childhood/young adult onset; requires insulin; positive autoantibodies (GAD, IA-2, ZnT8) |
| Type 2 DM | Combination of insulin resistance + progressive β-cell dysfunction → relative insulin deficiency | Most common (~90–95%); strongly linked to obesity, sedentary lifestyle, family history |
| Gestational DM (GDM) | Resembles T2DM; pregnancy-induced insulin resistance exceeds β-cell compensation | Affects ~5–30% of pregnancies; usually resolves post-delivery; major risk factor for future T2DM |
| MODY (Maturity-Onset Diabetes of the Young) | Monogenic; mutations in genes regulating β-cell function (e.g., GCK, HNF1A/4A) | Autosomal dominant; young, non-obese; often misdiagnosed as T1 or T2 |
| Secondary DM | Due to pancreatic disease (pancreatitis, cystic fibrosis), endocrinopathies (Cushing's, acromegaly), drugs (glucocorticoids, antipsychotics) | Treat underlying cause |
| LADA (Latent Autoimmune Diabetes in Adults) | Slow autoimmune T1DM in adults | Misclassified as T2DM; positive GAD antibodies |
Diagnostic Criteria
| Test | Diabetes | Pre-Diabetes (IFG/IGT) |
|---|---|---|
| Fasting Plasma Glucose (FPG) | ≥ 7.0 mmol/L (≥ 126 mg/dL) | 5.6–6.9 mmol/L (100–125 mg/dL) — IFG |
| 2-h Post-load Glucose (OGTT) | > 11.1 mmol/L (> 200 mg/dL) | 7.8–11.0 mmol/L (140–199 mg/dL) — IGT |
| HbA1c (added 2009, ADA/EASD/IDF) | ≥ 48 mmol/mol (≥ 6.5%) | 39–47 mmol/mol (5.7–6.4%) |
| Random Plasma Glucose | ≥ 11.1 mmol/L (≥ 200 mg/dL) + symptoms | — |
OGTT methodology: 75 g anhydrous glucose dissolved in water; 2-hour sample drawn. Endorsed by WHO, IDF, and ADA with identical cutoffs.
Special Populations
- GDM: Screened at 24–28 weeks gestation; uses either a one-step (75 g OGTT, ADA) or two-step (50 g challenge then 100 g OGTT, ACOG) approach with separate thresholds.
- Children: Same glucose criteria; T1DM more common but T2DM rising with childhood obesity.
Management
General Targets (ADA Standards of Care)
| Parameter | Target |
|---|---|
| HbA1c | < 7.0% (individualized; < 8.0% for frail/elderly) |
| Fasting glucose | 4.4–7.2 mmol/L (80–130 mg/dL) |
| Post-prandial glucose (2-h) | < 10.0 mmol/L (< 180 mg/dL) |
| Blood pressure | < 130/80 mmHg |
| LDL-cholesterol | < 1.8 mmol/L (< 70 mg/dL) in high CV risk |
Type 1 DM
- Insulin is mandatory — no alternative
- Basal-bolus regimen: Long-acting insulin (glargine, detemir, degludec) + rapid-acting insulin (aspart, lispro, glulisine) at meals
- Continuous Subcutaneous Insulin Infusion (CSII) / insulin pump: preferred in many patients
- Continuous Glucose Monitoring (CGM): standard of care; reduces HbA1c and hypoglycemia
- Closed-loop systems ("artificial pancreas"): automated insulin delivery; increasingly available
- Carbohydrate counting and flexible dosing
- Sick-day rules: never stop insulin; monitor ketones
Type 2 DM
Step-wise pharmacotherapy:
- Lifestyle modification first: weight loss (≥5–10% body weight), dietary changes (low glycemic index, caloric restriction), ≥150 min/week moderate aerobic activity
- Metformin: First-line pharmacotherapy (unless contraindicated — eGFR < 30, contrast dye, hepatic failure); reduces hepatic glucose production; weight-neutral; low hypoglycemia risk
- Add-on agents (chosen by comorbidity profile):
| Drug Class | Examples | Preferred When |
|---|---|---|
| GLP-1 Receptor Agonists | Semaglutide, liraglutide, dulaglutide | ASCVD, obesity, weight loss desired |
| SGLT-2 Inhibitors | Empagliflozin, dapagliflozin, canagliflozin | Heart failure, CKD, ASCVD |
| DPP-4 Inhibitors | Sitagliptin, saxagliptin | Renal impairment (dose-adjusted), weight-neutral |
| Sulfonylureas | Glipizide, gliclazide, glibenclamide | Cost-conscious; risk of hypoglycemia |
| Thiazolidinediones | Pioglitazone | Insulin resistance; avoid in heart failure |
| Insulin | Basal then bolus as needed | T2DM not at goal despite oral agents; acute illness |
- Insulin initiation in T2DM: Start with basal insulin (10 units or 0.1–0.2 U/kg/day); titrate by 2 units every 3 days to fasting glucose target
Gestational DM
- First-line: Medical nutrition therapy (MNT) + exercise; target fasting < 5.3 mmol/L, 1-h post-meal < 7.8 mmol/L
- Pharmacotherapy if targets not met: Insulin preferred (safe, well-studied); metformin accepted but crosses placenta
- Post-delivery: retest at 6–12 weeks with 75 g OGTT; annual screening thereafter
Monitoring & Screening
- Self-monitoring of blood glucose (SMBG) or CGM: individualized frequency
- HbA1c: every 3 months until at goal, then every 6 months
- Annual: lipid panel, urine albumin-to-creatinine ratio, eGFR, dilated eye exam, foot exam
- Screening asymptomatic adults: age ≥ 35 (or younger if overweight + risk factors) every 3 years — FPG, HbA1c, or OGTT
Complications to Monitor & Prevent
| Microvascular | Macrovascular |
|---|---|
| Diabetic nephropathy | Coronary artery disease |
| Diabetic retinopathy | Peripheral artery disease |
| Diabetic neuropathy (peripheral, autonomic) | Stroke/cerebrovascular disease |
- ACE inhibitors/ARBs: first-line for diabetic nephropathy with albuminuria
- Statin therapy: indicated in most adults with DM aged > 40 or with CV risk factors
- Aspirin: for secondary prevention in established ASCVD
Define pancreatitis types causes diagnosis labs and management
pancreatitis types acute chronic classification causes etiology gallstones alcohol
pancreatitis diagnosis labs amylase lipase imaging CT severity scoring
acute pancreatitis management treatment fluid resuscitation analgesia nutrition ERCP
acute pancreatitis CT scan imaging findings peripancreatic fat stranding necrosis
Pancreatitis
Types
1. Acute Pancreatitis
| Grade | Definition |
|---|---|
| Mild | No organ failure, no local or systemic complications; resolves within a week |
| Moderately Severe | Transient organ failure (< 48 h) and/or local complications without persistent organ failure |
| Severe | Persistent organ failure (> 48 h) — single or multi-organ |
- Interstitial Edematous Pancreatitis: most common; diffuse or focal pancreatic enlargement with enhancement on CT
- Necrotizing Pancreatitis: non-perfusion of pancreatic parenchyma ± peripancreatic necrosis on CECT; ~5–10% of cases; highest mortality
2. Chronic Pancreatitis
- End-stage: exocrine insufficiency (steatorrhea, malabsorption) + endocrine insufficiency (pancreatogenic/type 3c diabetes)
- Hallmark on imaging: pancreatic calcifications, ductal dilation, parenchymal atrophy
3. Special Subtypes
| Subtype | Key Features |
|---|---|
| Autoimmune Pancreatitis (AIP) | Type 1 (IgG4-related); Type 2 (idiopathic duct-centric); responds to steroids; can mimic pancreatic cancer |
| Hereditary/Genetic Pancreatitis | PRSS1, SPINK1, CFTR mutations; recurrent episodes from childhood |
| Groove Pancreatitis | Fibrotic mass in the groove between pancreatic head, duodenum, and CBD |
Causes / Etiology
| Category | Specific Causes |
|---|---|
| Obstructive | Gallstones (30–60% — most common overall; stones < 5 mm carry 4× higher risk), choledocholithiasis, ampullary/pancreatic tumors, pancreas divisum |
| Toxic/Metabolic | Alcohol (15–30%; 5/100,000 incidence in alcoholics — other factors like smoking and genetics modulate risk), hypertriglyceridemia (> 11.3 mmol/L / > 1000 mg/dL), hypercalcemia |
| Iatrogenic | Post-ERCP (5–10% of procedures), post-surgical, medications (thiazides, azathioprine, valproate, tetracyclines, sulfonamides, didanosine) |
| Infectious | Mumps, Coxsackie B, CMV, Ascaris lumbricoides |
| Autoimmune | AIP type 1 (IgG4), AIP type 2 |
| Genetic | PRSS1 (cationic trypsinogen), SPINK1, CFTR mutations |
| Vascular | Ischemia, vasculitis |
| Idiopathic | ~15–25% of cases |
Diagnosis
- Typical abdominal pain — epigastric, often radiating to the back, constant, severe
- Serum lipase and/or amylase ≥ 3× upper limit of normal
- Confirmatory cross-sectional imaging (CT/MRI with characteristic findings)
Laboratory Findings
| Test | Findings & Notes |
|---|---|
| Serum Lipase | More sensitive and specific than amylase; remains elevated longer (7–14 days); preferred test |
| Serum Amylase | Rises within 2–12 hours; returns to normal in 3–5 days; less specific (elevated in salivary disease, bowel perforation, renal failure) |
| WBC | Leukocytosis reflects inflammation/infection |
| Hematocrit | > 44% (hemoconcentration) — marker of severity/dehydration |
| BUN/Creatinine | BUN > 22 mg/dL on admission = marker of severity; rising creatinine = AKI |
| CRP | > 150 mg/L at 48 h strongly predicts severe pancreatitis |
| LFTs (ALT/AST, bilirubin, ALP) | ALT > 3× ULN suggests gallstone pancreatitis |
| Serum triglycerides | Rule out hypertriglyceridemic pancreatitis (> 1000 mg/dL diagnostic) |
| Serum calcium | Hypercalcemia as cause; hypocalcemia (saponification) as complication |
| Serum IgG4 | Elevated in AIP type 1 |
| Blood glucose | Hyperglycemia may reflect β-cell injury |
| ABG/Lactate | Assess for organ failure, SIRS |
Imaging
CT Abdomen with IV Contrast (CECT)
- Gold standard for staging severity and detecting necrosis
- Best performed 48–72 hours after symptom onset (necrosis may not be visible initially)
- CT Severity Index (Balthazar Score): grades A–E based on pancreatic inflammation + necrosis percentage
CT Findings — Necrotizing Pancreatitis
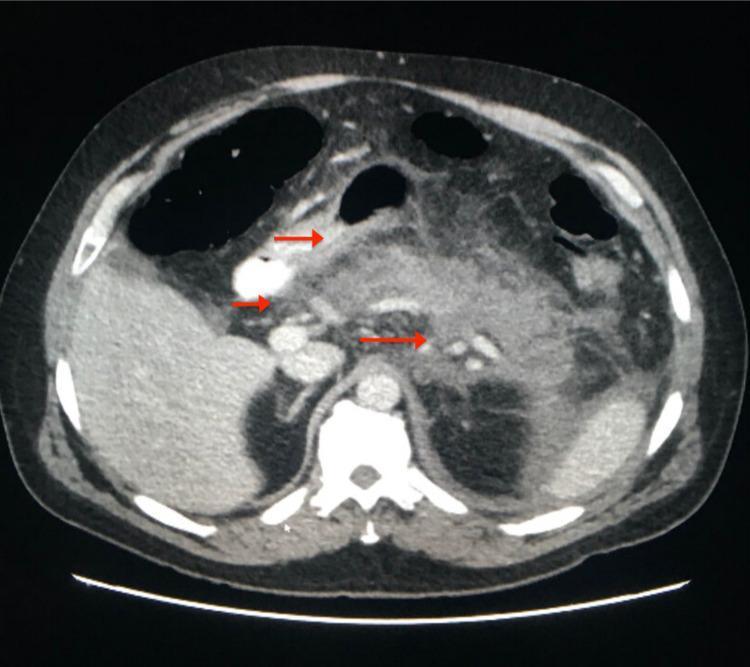
| Finding | Significance |
|---|---|
| Pancreatic enlargement, edema | Interstitial pancreatitis |
| Peripancreatic fat stranding | Inflammation extending beyond pancreas |
| Non-enhancing parenchyma | Pancreatic necrosis |
| Peripancreatic fluid collections (APFC) | Early (< 4 weeks); no wall |
| Walled-Off Necrosis (WON) | Encapsulated, mature (> 4 weeks); may require drainage |
| Pseudocyst | Fluid collection with wall, no solid component |
Other Imaging Modalities
| Modality | Use |
|---|---|
| Ultrasound (RUQ) | First-line to detect gallstones; limited pancreatic visualization due to bowel gas |
| MRI/MRCP | Preferred for biliary anatomy, ductal evaluation, avoiding radiation; detects choledocholithiasis |
| ERCP | Therapeutic (not diagnostic) — stone extraction, sphincterotomy |
| EUS | Detects small stones, evaluates pancreatic duct; guides drainage procedures |
Severity Scoring Systems
| Score | Components | Notes |
|---|---|---|
| Ranson's Criteria | 11 parameters (5 at admission, 6 at 48 h); ≥ 3 = severe | Older; cannot be completed until 48 h |
| APACHE II | 12 physiological variables | Can be used serially; > 8 = severe |
| BISAP Score | BUN > 25, impaired mental status, SIRS, age > 60, pleural effusion; ≥ 3 = severe | Simple; performed at admission |
| CT Severity Index | Balthazar grade + % necrosis; max 10 points; ≥ 6 = severe | Requires CT |
Management
Acute Pancreatitis
1. Initial Resuscitation (First 12–24 Hours)
- IV fluid resuscitation: Lactated Ringer's preferred over normal saline (reduces SIRS, acidosis); 250–500 mL/hr initially; titrate to urine output > 0.5 mL/kg/hr, HR < 100, MAP 65–85 mmHg
- Monitor closely: BUN, creatinine, hematocrit, urine output
2. Pain Management
- IV opioids (morphine, hydromorphone, fentanyl) — effective and safe; old concerns about morphine causing sphincter of Oddi spasm are not clinically significant
- NSAIDs (ketorolac) as adjunct
3. Nutrition
- Mild pancreatitis: Oral feeding tolerated as soon as patient can eat (low-fat, soft diet); no need for bowel rest
- Severe/moderately severe: Enteral nutrition (nasojejunal or nasogastric) preferred over parenteral — maintains gut barrier, reduces infectious complications; start within 24–72 hours
- TPN only if enteral route not feasible
4. Antibiotics
- Not routinely indicated for sterile pancreatitis
- Reserve for proven/suspected infected necrosis — guided by FNA culture or clinical deterioration with fever + rising CRP; use carbapenems (imipenem, meropenem) or fluoroquinolones + metronidazole (penetrate pancreatic tissue)
5. Cause-Specific Interventions
| Cause | Intervention |
|---|---|
| Gallstone pancreatitis | Early ERCP (within 24 h) if concurrent cholangitis or CBD obstruction; cholecystectomy during same admission (mild) or after recovery (severe) to prevent recurrence |
| Hypertriglyceridemia | Insulin infusion (lowers TGs rapidly); plasmapheresis in extreme cases; fibrates/omega-3 long-term |
| AIP | Oral prednisolone 0.6–1 mg/kg/day; excellent response |
6. Management of Complications
| Complication | Management |
|---|---|
| Infected necrosis | Step-up approach: percutaneous drainage → endoscopic/surgical necrosectomy if no improvement |
| Walled-off necrosis (WON) | Endoscopic transmural drainage (EUS-guided cystogastrostomy) preferred over surgery |
| Pseudocyst | Drainage if symptomatic (endoscopic, percutaneous, or surgical) |
| Organ failure (ARDS, AKI) | ICU care, mechanical ventilation, renal replacement therapy as needed |
| Abdominal compartment syndrome | Decompressive measures; surgery if refractory |
Chronic Pancreatitis
| Problem | Management |
|---|---|
| Pain | Analgesics (step-wise: paracetamol → NSAIDs → opioids); pancreatic enzyme supplementation may reduce pain; endoscopic therapy (ductal stenting, stone extraction); celiac plexus block; surgery (Frey/Puestow procedure) for refractory pain with dilated duct |
| Exocrine insufficiency | Pancreatic enzyme replacement therapy (PERT): lipase ≥ 40,000–50,000 IU per meal; taken with meals; fat-soluble vitamin supplementation |
| Endocrine insufficiency (Type 3c DM) | Insulin therapy; highly brittle — risk of hypoglycemia; glucagon secretion also impaired |
| Nutritional deficiencies | Fat-soluble vitamins (A, D, E, K), B12; nutritional support |
| Abstinence | Alcohol and smoking cessation — slows progression |
Asthma defination diagnosis and management
asthma definition pathophysiology airway inflammation hyperresponsiveness bronchoconstriction
asthma diagnosis spirometry reversibility criteria GINA guidelines classification severity
asthma stepwise treatment management SABA ICS LABA biologic therapy GINA steps
asthma pathophysiology airway inflammation diagram bronchoconstriction
Asthma
Definition
Pathophysiology
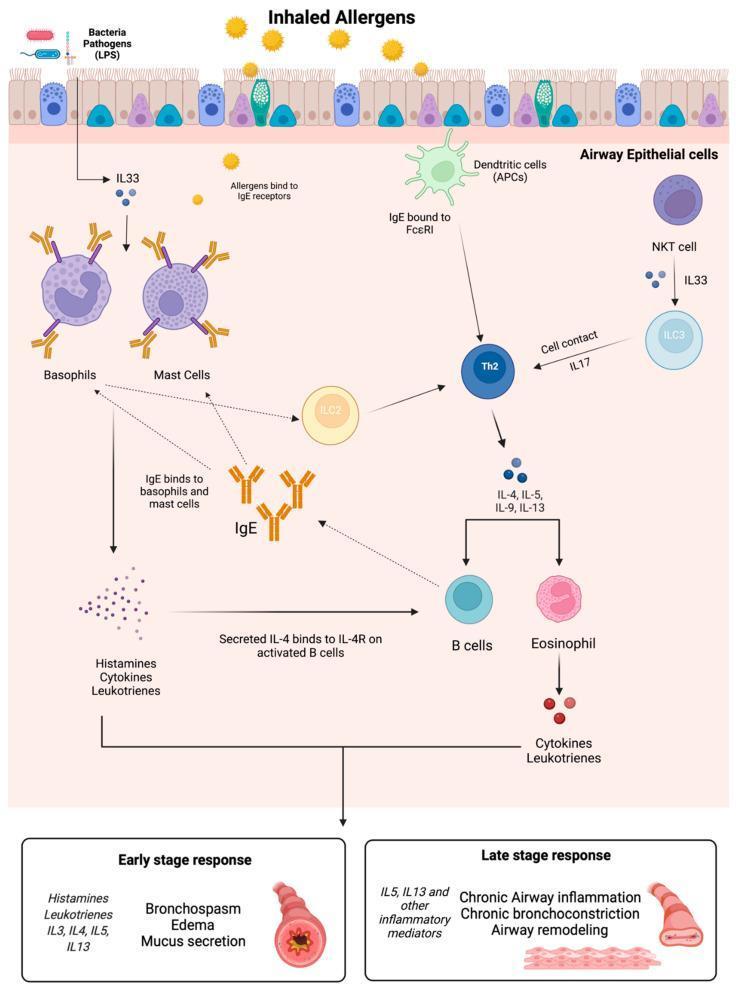
| Phase | Mechanism | Clinical Result |
|---|---|---|
| Early (acute) | IgE-mediated mast cell degranulation → histamine, leukotrienes, prostaglandins | Bronchospasm, edema, mucus hypersecretion within minutes |
| Late (chronic) | Th2/ILC2-driven eosinophil and cytokine (IL-4, IL-5, IL-13) recruitment | Chronic airway inflammation, hyperresponsiveness, structural remodeling |
Triggers / Risk Factors
| Category | Examples |
|---|---|
| Allergens | House dust mite, pollen, cockroach, pet dander, mold |
| Respiratory infections | Rhinovirus, RSV, influenza |
| Exercise | Exercise-induced bronchoconstriction (EIB) |
| Occupational | Isocyanates, flour dust, latex, animals (baker's asthma, etc.) |
| Drugs | NSAIDs/aspirin (Samter's triad), beta-blockers, ACE inhibitors (cough) |
| Environmental | Cold air, tobacco smoke, air pollution, strong odors |
| Comorbidities | Allergic rhinitis, GERD, obesity, obstructive sleep apnea |
| Genetic predisposition | Atopy (personal/family history of eczema, rhinitis, food allergy) |
Clinical Features
- Wheeze (expiratory > inspiratory)
- Dyspnea / chest tightness
- Cough (especially nocturnal; may be the only symptom — cough-variant asthma)
- Expiratory wheeze on auscultation
- Prolonged expiratory phase
- Use of accessory muscles (severe)
- Silent chest = very severe obstruction (no airflow to generate wheeze — ominous sign)
- Pulsus paradoxus > 10 mmHg (severe)
Diagnosis
Diagnostic Criteria (GINA 2023)
- Characteristic symptom pattern (variable wheeze, dyspnea, cough, chest tightness)
- Objective evidence of variable expiratory airflow limitation (confirmatory pulmonary function testing)
Spirometry — Key Tests
| Test | Criterion | Significance |
|---|---|---|
| FEV₁/FVC ratio | < 0.70 (< LLN) | Confirms obstructive pattern |
| Bronchodilator reversibility | FEV₁ increase ≥ 12% AND ≥ 200 mL post SABA | Confirms reversibility — hallmark of asthma |
| FEV₁% predicted | < 60% = high risk for exacerbations | Guides severity assessment |
| Peak Expiratory Flow (PEF) | Variability > 10% diurnal variation | Useful for monitoring, especially at home |
Note: Normal spirometry does not exclude asthma — testing during a symptomatic period or bronchoprovocation challenge (methacholine, mannitol) may be needed.
Bronchial Provocation Testing
- Used when spirometry is normal but asthma is suspected
- Methacholine challenge: PC₂₀ < 8 mg/mL = airway hyperresponsiveness (positive)
- Useful to rule out asthma (high sensitivity — negative test makes asthma unlikely)
Additional Investigations
| Investigation | Purpose |
|---|---|
| FeNO (Fractional Exhaled NO) | ≥ 40 ppb supports eosinophilic inflammation; guides ICS therapy |
| Blood eosinophil count | ≥ 300 cells/μL = type 2 inflammation; guides biologic selection |
| Total IgE / Specific IgE (RAST) | Atopic status; guides omalizumab eligibility |
| Skin prick testing | Identifies specific allergic triggers |
| CXR | Typically normal; useful to exclude infection, pneumothorax, foreign body |
| ABG | Severe exacerbation: hypoxia + initially respiratory alkalosis; then CO₂ retention = respiratory failure |
Classification
By Symptom Control (GINA)
| Control Level | Daytime Symptoms | Night Waking | Reliever Use | Activity Limitation |
|---|---|---|---|---|
| Well Controlled | ≤ 2 days/week | None | ≤ 2 days/week | None |
| Partly Controlled | > 2 days/week | Any | > 2 days/week | Any |
| Uncontrolled | 3+ features of partly controlled | — | — | — |
By Severity (Based on Treatment Required to Achieve Control)
| Severity | Description |
|---|---|
| Mild | Well-controlled on Steps 1–2 (as-needed SABA or low-dose ICS) |
| Moderate | Controlled on Step 3 (low-dose ICS/LABA) |
| Severe | Requires Steps 4–5 or uncontrolled despite high-dose treatment |
Management
GINA Stepwise Approach (Adults & Adolescents ≥ 12 years)
| Step | Preferred Controller | Preferred Reliever | Notes |
|---|---|---|---|
| Step 1 | None OR low-dose ICS (when SABA used) | As-needed SABA | Very mild; prefer ICS-formoterol over SABA alone |
| Step 2 | Low-dose ICS daily | As-needed SABA | ICS is the cornerstone of asthma treatment |
| Step 3 | Low-dose ICS + LABA | As-needed SABA or ICS-formoterol | MART (Maintenance and Reliever Therapy) preferred with budesonide-formoterol |
| Step 4 | Medium/high-dose ICS + LABA | As-needed ICS-formoterol | Add LAMA (tiotropium) if uncontrolled |
| Step 5 | High-dose ICS + LABA + add-on biologic | As-needed ICS-formoterol | Refer to specialist |
GINA 2019+ Update: ICS-containing reliever (budesonide-formoterol) preferred over SABA alone at all steps — reduces exacerbation risk even in mild asthma.
Drug Classes
| Drug Class | Examples | Mechanism | Role |
|---|---|---|---|
| ICS (Inhaled Corticosteroids) | Beclomethasone, budesonide, fluticasone, ciclesonide | Suppress airway inflammation | Cornerstone controller |
| SABA (Short-acting β₂-agonist) | Salbutamol (albuterol), terbutaline | Bronchodilation (rapid, 15 min) | Reliever / rescue |
| LABA (Long-acting β₂-agonist) | Formoterol, salmeterol | Sustained bronchodilation (≥12 h) | Always combined with ICS; never monotherapy |
| LAMA (Long-acting muscarinic antagonist) | Tiotropium | Reduce cholinergic bronchoconstriction | Add-on at Steps 4–5 |
| LTRA (Leukotriene receptor antagonists) | Montelukast | Block LTD4 receptors | Alternative or add-on; useful in aspirin-exacerbated asthma, allergic rhinitis comorbidity |
| Methylxanthines | Theophylline | PDE inhibitor; mild bronchodilation + anti-inflammatory | Add-on; narrow therapeutic window; largely replaced by biologics |
| Oral corticosteroids | Prednisolone | Broad anti-inflammatory | Short courses for exacerbations; chronic use only in severe refractory asthma |
| Biologics | See below | Target specific inflammatory pathways | Step 5; type 2/eosinophilic or allergic asthma |
Biologic Therapies (Step 5 — Severe Asthma)
| Drug | Target | Indication |
|---|---|---|
| Omalizumab | Anti-IgE | Allergic asthma; total IgE 30–1500 IU/mL; sensitization confirmed |
| Mepolizumab / Reslizumab | Anti-IL-5 | Severe eosinophilic asthma; blood eos ≥ 150–300/μL |
| Benralizumab | Anti-IL-5Rα | Severe eosinophilic asthma; rapid eosinophil depletion |
| Dupilumab | Anti-IL-4Rα (blocks IL-4 + IL-13) | Type 2 asthma ± comorbid atopic dermatitis, nasal polyps |
| Tezepelumab | Anti-TSLP | Broad severe asthma (including non-eosinophilic); reduces all exacerbations |
Acute Exacerbation Management
Severity Assessment
| Feature | Moderate | Severe | Life-Threatening |
|---|---|---|---|
| SpO₂ | ≥ 92% | < 92% | < 92% + silent chest |
| Speech | Sentences | Words | Cannot speak |
| RR | Increased | > 25/min | > 30/min |
| HR | < 110 | ≥ 110 | Bradycardia |
| PEF | 50–70% | < 50% predicted | < 33% |
| PaCO₂ | Normal | Normal/rising | > 45 mmHg (respiratory failure) |
Treatment Protocol
- Oxygen: Titrate to SpO₂ 93–95% (avoid hyperoxia)
- SABA: Salbutamol 2.5–5 mg nebulized every 20 minutes × 3 (or MDI 4–8 puffs), then reassess
- Ipratropium bromide: Add 0.5 mg nebulized in moderate–severe attacks (reduces hospitalizations)
- Systemic corticosteroids: Prednisolone 40–50 mg oral (or IV methylprednisolone 1 mg/kg) — start early; course 5–7 days
- Magnesium sulfate IV: 2 g IV over 20 minutes for severe/life-threatening attacks not responding to initial treatment
- Heliox: Consider in severe refractory obstruction (reduces turbulent airflow)
- Intubation & mechanical ventilation: Last resort; use permissive hypercapnia strategy
Non-Pharmacological Management
- Allergen avoidance: HDMA covers, pet removal, cockroach control
- Smoking cessation: Reduces ICS effectiveness; accelerates decline
- Allergen immunotherapy (AIT): Subcutaneous or sublingual; reduces sensitization and exacerbations in allergic asthma
- Breathing exercises: Buteyko, physiotherapy — reduce reliever use, improve quality of life
- Weight loss: Significant benefit in obese patients (reduces airway inflammation, improves control)
- Asthma action plan: Written self-management plan for every patient — recognizing worsening, when to increase treatment, when to seek emergency care
- Vaccination: Annual influenza; pneumococcal for severe/oral steroid-dependent asthma
Monitoring
| Parameter | Frequency |
|---|---|
| Symptom control assessment (GINA/ACQ/ACT score) | Every visit |
| Spirometry | At diagnosis, 3–6 months after starting ICS, then annually |
| FeNO / blood eosinophils | At diagnosis; before starting/stepping up biologics |
| Inhaler technique review | Every visit |
| Adherence check | Every visit |