Diabetes mellitus
Reading File
search_medical_text
diabetes mellitus classification pathophysiology type 1 type 2
search_medical_text
diabetes mellitus diagnosis criteria management treatment complications
search_medical_image
diabetes mellitus pathophysiology insulin resistance diagram
GradeResponse
GradeResponse
GradeResponse
Diabetes Mellitus
Overview
Diabetes mellitus (DM) is a group of metabolic disorders characterized by chronic hyperglycemia resulting from defects in insulin secretion, insulin action, or both. It leads to disordered carbohydrate, fat, and protein metabolism with wide-ranging systemic consequences. (Laboratory Analysis in the Diagnosis and Management of Diabetes Mellitus, p. 1)
Classification
| Type | Mechanism | Key Features |
|---|---|---|
| Type 1 DM | Autoimmune destruction of pancreatic β-cells | Absolute insulin deficiency; insulin required for survival |
| Type 2 DM | Insulin resistance + relative insulin secretory defect | Most common (~90–95% of cases); often manageable without insulin initially |
| Gestational DM (GDM) | Resembles T2DM; placental hormones cause resistance | Affects ~17% of pregnancies; major risk factor for future T2DM |
| Other specific types | Monogenic (MODY), drug-induced, pancreatic disease, endocrinopathies | Varied mechanisms |
Pathophysiology
Type 1 DM
- T-cell mediated autoimmune attack on β-cells (HLA-DR3/DR4 associations)
- Leads to near-total loss of insulin secretion
- Requires exogenous insulin for glucose uptake, prevention of ketoacidosis, and survival
Type 2 DM
The pathophysiology involves two core defects:
- Insulin resistance — peripheral tissues (muscle, fat, liver) fail to respond normally to insulin
- β-cell dysfunction — the pancreas cannot compensate with adequate insulin secretion
The diagram below illustrates the insulin signaling pathway and how disruption at multiple steps (PI3K, AKT, GLUT-4 translocation) leads to impaired glucose uptake:
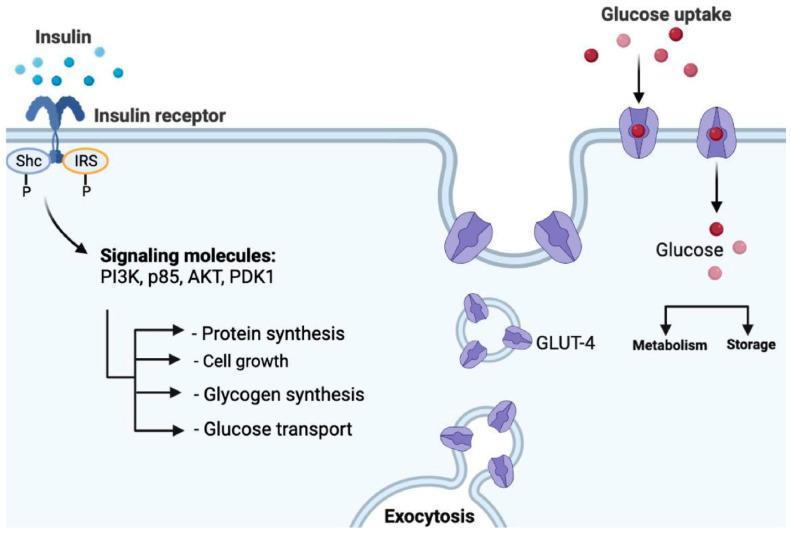
Insulin binds its receptor → phosphorylation of IRS → PI3K/AKT cascade → GLUT-4 exocytosis → glucose uptake. Defects anywhere in this chain produce insulin resistance.
Diagnosis
ADA Diagnostic Criteria (any one of the following, confirmed on repeat testing unless unequivocal hyperglycemia):
| Test | Diagnostic Threshold |
|---|---|
| Fasting plasma glucose (FPG) | ≥ 126 mg/dL (7.0 mmol/L) |
| 2-hour plasma glucose (75g OGTT) | ≥ 200 mg/dL (11.1 mmol/L) |
| HbA1c | ≥ 6.5% (48 mmol/mol) |
| Random glucose + symptoms | ≥ 200 mg/dL (11.1 mmol/L) |
Prediabetes (increased risk):
- FPG: 100–125 mg/dL (impaired fasting glucose)
- 2h OGTT: 140–199 mg/dL (impaired glucose tolerance)
- HbA1c: 5.7–6.4%
Clinical Presentation
Classic symptoms ("3 Ps"):
- Polyuria — osmotic diuresis from glucosuria
- Polydipsia — secondary to fluid loss
- Polyphagia — cellular starvation despite hyperglycemia
Additional findings: weight loss, blurred vision, fatigue, recurrent infections, slow wound healing
T2DM is often asymptomatic at diagnosis; T1DM may present acutely with diabetic ketoacidosis (DKA).
Management
Individualized Glycemic Targets
- HbA1c < 7% is the general target for most non-pregnant adults
- Less stringent (< 8%) for elderly, limited life expectancy, or frequent hypoglycemia
- More stringent (< 6.5%) in younger, newly diagnosed with no hypoglycemia risk
Type 1 DM
- Insulin therapy is mandatory (basal-bolus regimens or continuous subcutaneous insulin infusion)
- Carbohydrate counting, continuous glucose monitoring (CGM)
Type 2 DM — Stepwise Approach
- Lifestyle modification — Medical nutrition therapy, physical activity, weight loss (first-line)
- Metformin — First-line pharmacotherapy (reduces hepatic glucose output, low cost, weight-neutral)
- Add-on agents based on comorbidities:
| Drug Class | Benefit Beyond Glucose | Notes |
|---|---|---|
| GLP-1 agonists (e.g., semaglutide) | CV protection, weight loss | Injectable (oral semaglutide available) |
| SGLT-2 inhibitors (e.g., empagliflozin) | CV & renal protection, HF benefit | Preferred in CKD/HF |
| DPP-4 inhibitors | Weight-neutral | Well tolerated |
| Sulfonylureas | Low cost | Hypoglycemia risk |
| Insulin | Universal efficacy | Required when other agents fail |
- Insulin therapy when HbA1c targets not met with oral/injectable non-insulin agents (Eye Care of the Patient with Diabetes Mellitus, p. 23)
Cardiovascular & Other Risk Factor Management
- Blood pressure control (target < 130/80 mmHg in most)
- Statin therapy (high-intensity in those with CVD or high risk)
- Antiplatelet therapy (aspirin) in established CVD
Complications
Microvascular
| Complication | Mechanism | Manifestation |
|---|---|---|
| Diabetic nephropathy | Mesangial expansion, hyperfiltration | Proteinuria → CKD → ESRD |
| Diabetic retinopathy | Pericyte loss, neovascularization | Leading cause of adult blindness |
| Diabetic neuropathy | Axonal degeneration, demyelination | Distal symmetric polyneuropathy, autonomic neuropathy |
Macrovascular
- Accelerated atherosclerosis → coronary artery disease, stroke, peripheral artery disease
- Leading cause of mortality in T2DM patients
Acute Metabolic
| Emergency | Type | Key Features |
|---|---|---|
| DKA | Primarily T1DM | Hyperglycemia, anion-gap acidosis, ketonemia; pH < 7.3 |
| HHS | T2DM | Extreme hyperglycemia (> 600 mg/dL), hyperosmolarity, no significant ketosis |
| Hypoglycemia | Any treated DM | BG < 70 mg/dL; symptoms: tremor, diaphoresis, confusion |
Monitoring
- HbA1c every 3 months until stable, then every 6 months
- Self-monitoring of blood glucose (SMBG) or CGM
- Annual: urine albumin-to-creatinine ratio, eGFR, lipid panel, ophthalmologic exam, foot exam
- Immunizations: influenza, pneumococcal, hepatitis B (for adults with DM)
Special Populations
- Gestational DM: Screen at 24–28 weeks; managed with diet ± insulin (metformin/glyburide as alternatives); risk of macrosomia, neonatal hypoglycemia
- Elderly: Hypoglycemia is particularly dangerous; de-intensify therapy as needed
- CKD: Adjust drug doses; prefer SGLT-2 inhibitors and GLP-1 agonists with proven renal benefit
This is a shared conversation. Sign in to Orris to start your own chat.