Pityriasis rubra pilaris in 5marks format for 3rd yr dermatology post graduate theory examination
Reading File
Finding Sources
Searching the Web
pityriasis rubra pilaris dermatology 5 marks exam answer classification clinical features treatment
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I have all the information needed. Here is the comprehensive 5-marks format answer:
PITYRIASIS RUBRA PILARIS (PRP)
3rd Year Dermatology PG Theory Examination — 5 Marks
DEFINITION
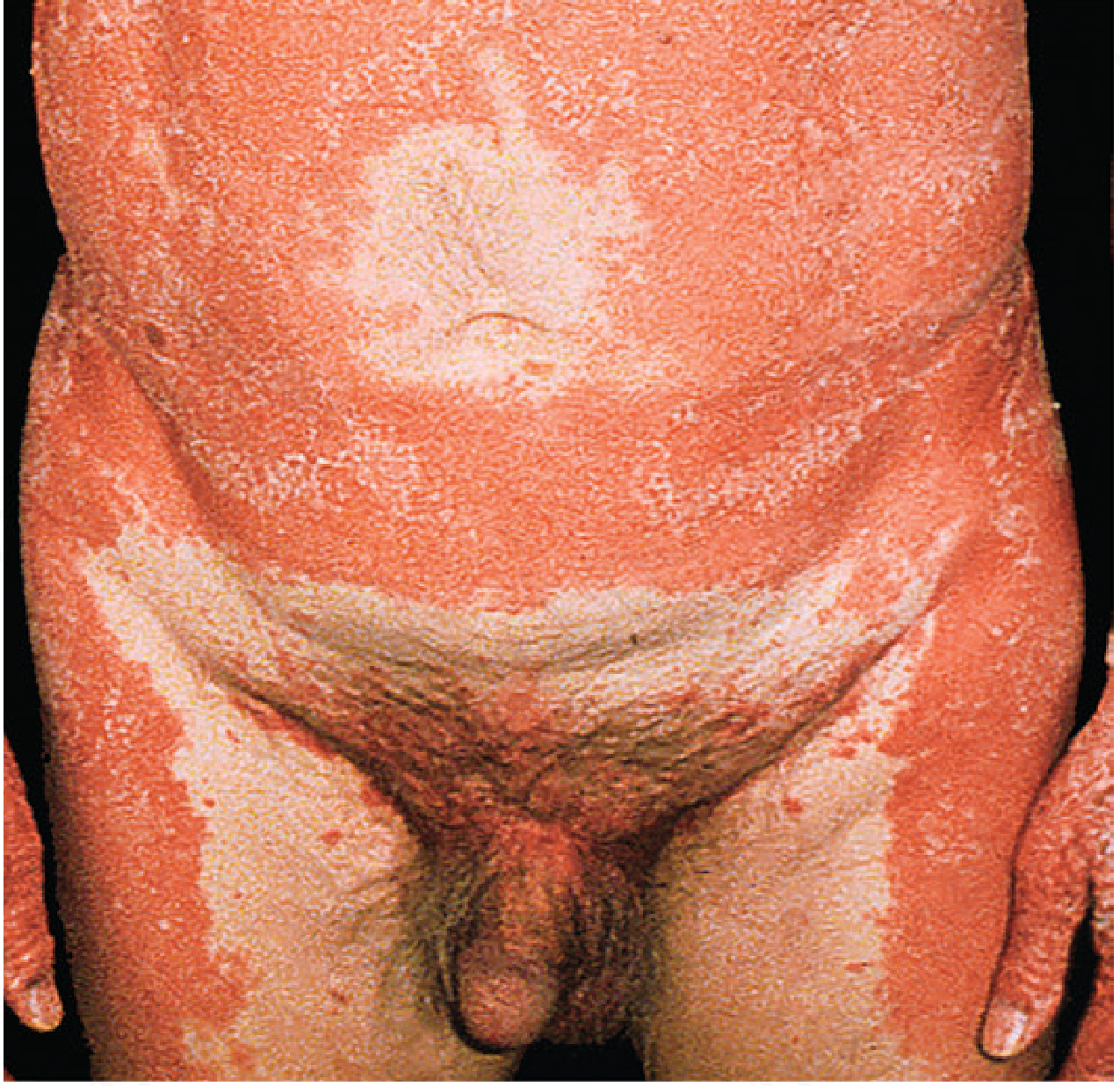
Pityriasis rubra pilaris is a rare, chronic inflammatory papulosquamous dermatosis characterised by hyperkeratotic follicular papules, salmon-coloured to red-orange scaly plaques, palmoplantar keratoderma, and well-demarcated islands of normal skin within the affected areas.
AETIOLOGY & PATHOGENESIS
| Factor | Detail |
|---|---|
| Idiopathic | Most cases (majority sporadic) |
| Genetic | ~6.5% familial; caused by CARD14 gain-of-function mutations (NF-κB pathway activation) |
| Vitamin A metabolism | Possible dysfunction leading to decreased retinol supply to epidermis |
| HIV-associated | Type VI PRP; may respond to antiretroviral therapy |
Two age peaks: first 5 years of life and fifth decade of adulthood.
CLASSIFICATION — GRIFFITHS (1980), Modified
| Type | Name | % Cases | Key Features | Course |
|---|---|---|---|---|
| I | Classic adult | >50% | Erythroderma, follicular papules, islands of sparing, waxy palmoplantar keratoderma; cephalocaudal spread | Resolves in ~3 years |
| II | Atypical adult | 5% | Ichthyosiform scaling, follicular hyperkeratosis, sparse scalp hair; no cephalocaudal progression | >20 years |
| III | Classic juvenile | 10% | Similar to Type I but onset age 5–10 years | Resolves in 1–2 years |
| IV | Circumscribed juvenile | 25% | Prepubertal; sharply demarcated erythematous plaques on elbows/knees ± palmoplantar keratoderma; resembles psoriasis | Uncertain |
| V | Atypical juvenile | 5% | Early onset, chronic; most familial cases; follicular hyperkeratosis + scleroderma-like changes of hands/feet; linked to CARD14 | Chronic, relapsing |
| VI | HIV-associated | — | Follicular papules, prominent follicular plugging; acne conglobata, hidradenitis suppurativa, lichen spinulosus | Responds to ART |
CLINICAL FEATURES
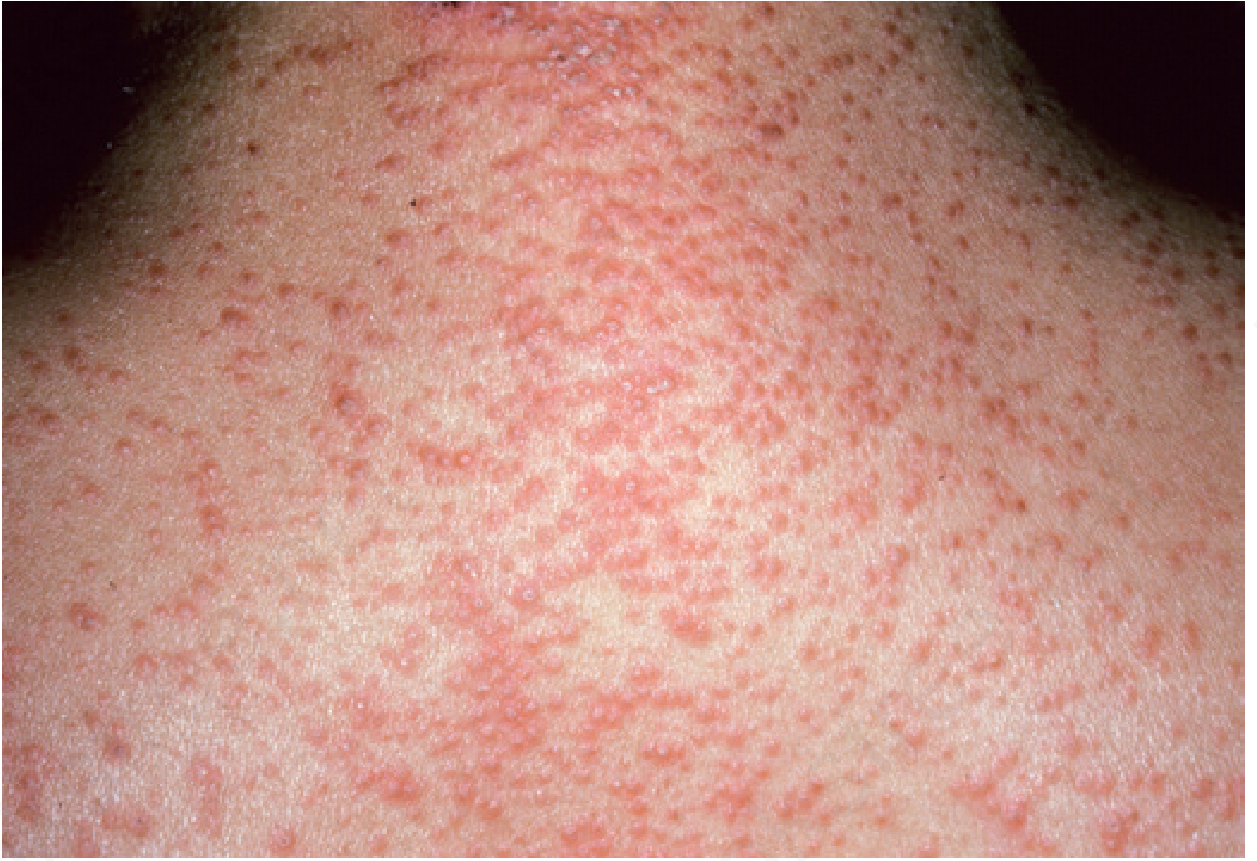
Primary lesion: Pinhead-sized, acuminate (pointed), salmon-coloured to reddish-brown follicular papules topped by a central horny plug (with embedded hair fragment). The surface feels like a "nutmeg grater."
Evolution (Type I, cephalocaudal):
- Scaliness and erythema of scalp → spreads to face, neck
- Predilection for sides of neck, extensor surfaces (especially dorsum of proximal phalanges — "knuckle pads")
- Papules coalesce into red-orange scaling plaques
- Progresses to erythroderma over 2–3 months
Pathognomonic features:
- Islands of sparing (nappes claires) — sharply demarcated areas of normal skin within erythroderma (hallmark sign)
- Waxy, diffuse, yellowish palmoplantar keratoderma — extends up the sides of the soles ("sandal distribution")
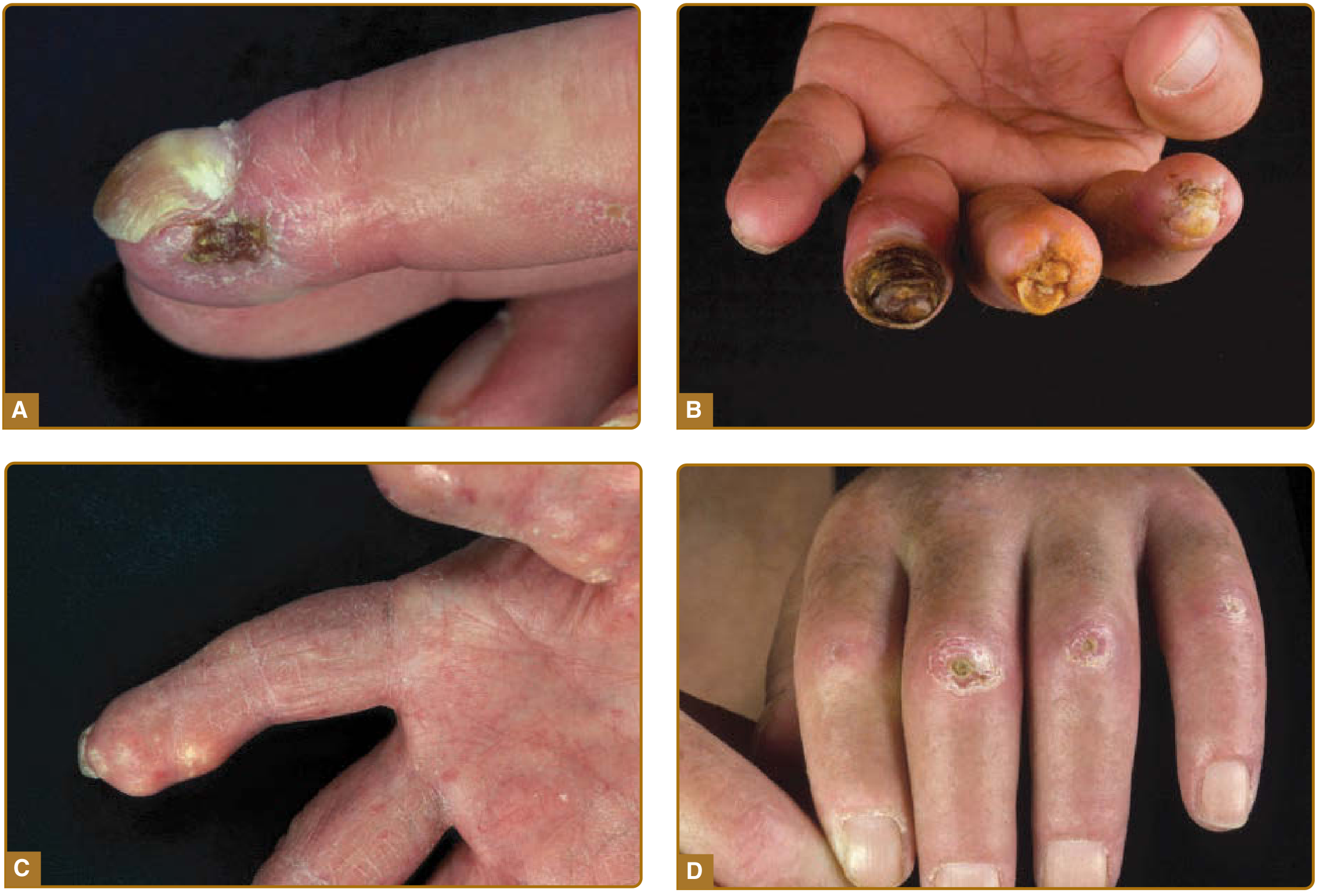
Nail changes: Thickening, splinter haemorrhages, subungual hyperkeratosis; nails are never pitted (important distinction from psoriasis)
Other features: Ectropion (with facial involvement), Koebner phenomenon, pruritus (variable), arthritis (rare), protein-losing enteropathy (rare in extensive disease)
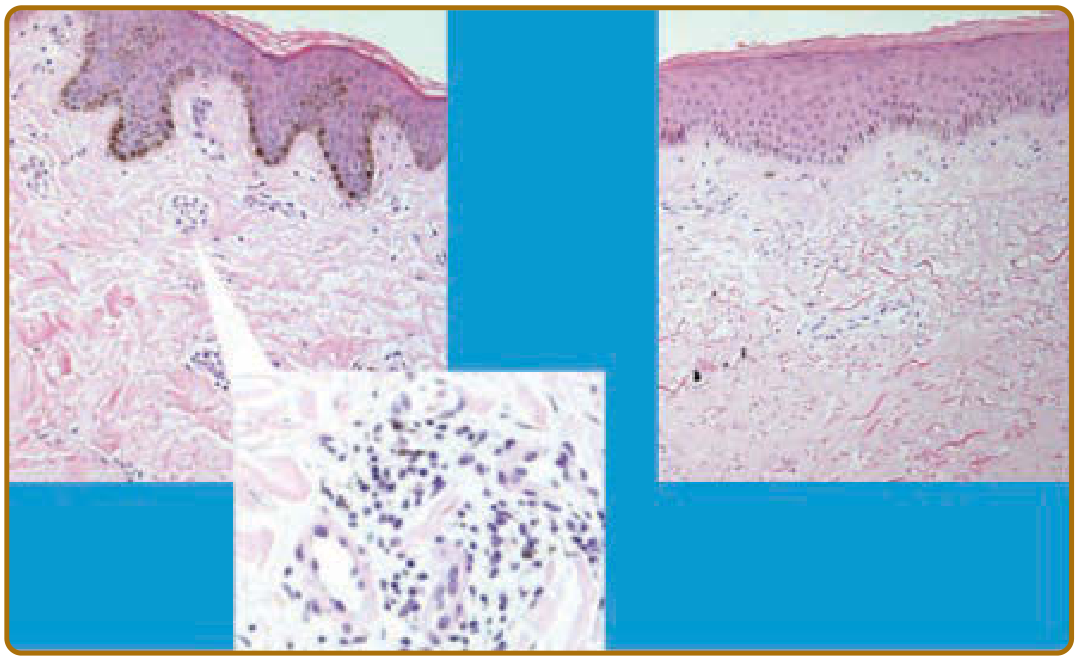
HISTOPATHOLOGY
| Feature | Significance |
|---|---|
| Alternating orthokeratosis and parakeratosis in a checkerboard (horizontal and vertical) pattern | Pathognomonic |
| Hyperkeratosis with follicular plugging | Diagnostic |
| Acanthosis with broad rete ridges | Seen in established disease |
| Focal acantholytic dyskeratosis | Characteristic |
| Perivascular lymphocytic infiltrate (sparse) | Non-specific |
| Absent suprapapillary plate thinning and Munro microabscesses | Distinguishes from psoriasis |
DIAGNOSIS
Clinical diagnosis is primary. Histopathology confirms in doubtful cases.
Differential diagnosis:
- Psoriasis (most important) — PRP has no nail pitting, no Auspitz sign; islands of sparing; checkerboard parakeratosis on histology
- Ichthyosis, seborrhoeic dermatitis, erythroderma of other causes
TREATMENT
Topical
- Emollients (mainstay of symptomatic relief)
- Keratolytics: salicylic acid, urea
- Topical corticosteroids, calcineurin inhibitors, vitamin D analogues
Systemic (first-line)
| Drug | Dose | Notes |
|---|---|---|
| Acitretin (oral retinoid) | 0.5–1 mg/kg/day | First-line; most effective; monitor LFTs, lipids |
| Isotretinoin | 0.5–1 mg/kg/day | Alternative retinoid |
| Methotrexate | 15–25 mg/week | Second-line; combined with retinoids in extensive disease; monitor liver toxicity |
Second-line / Refractory
- Biologics: TNF-α inhibitors (infliximab, etanercept, adalimumab); ustekinumab (anti-IL-12/23); anti-IL-17 agents
- Cyclosporine (5 mg/kg/day)
- Azathioprine (100–150 mg/day)
- Apremilast (PDE4 inhibitor)
- Fumaric acid esters
Phototherapy
- PUVA (psoralen + UVA) / Narrowband UVB — adjunctive role; used with caution as some PRP can worsen with UVB
HIV-associated (Type VI)
- Antiretroviral therapy (ART) — may lead to clearance
PROGNOSIS
- Type I (classic adult): spontaneous remission in ~80% within 3 years
- Type III (classic juvenile): remission within 1–2 years
- Type II and V: chronic, lasting >20 years; may require long-term retinoids
- Type IV: unpredictable; may resolve in late teens
Key exam points to remember:
- Follicular papules + islands of sparing + waxy PPK = classic triad
- Checkerboard pattern of parakeratosis on histology is pathognomonic
- Nails never pitted → key distinction from psoriasis
- CARD14 mutations → familial/Type V PRP
- Acitretin is first-line systemic treatment
Sources: Fitzpatrick's Dermatology 9e (Ch. 29), Andrews' Diseases of the Skin, Dermatology 2-Vol Set 5e
What is scleroderma?discuss pathogenesis of scleroderma in 10 marks format for 3rd yr dermatology post graduate theory examination
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Searching the Web
scleroderma systemic sclerosis pathogenesis vascular fibrosis autoimmunity 2024 review
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Loading Image
Loading Image
I now have all the content needed for a comprehensive 10-marks answer. The Fitzpatrick's data was extremely rich, covering the full pathogenesis triad (vasculopathy, immune dysregulation, fibrosis), classification, clinical features, histopathology, and treatment. Let me compile the complete answer:
SCLERODERMA (SYSTEMIC SCLEROSIS)
3rd Year Dermatology PG Theory Examination — 10 Marks
DEFINITION
Scleroderma (Systemic Sclerosis, SSc) is a multisystemic autoimmune connective tissue disease characterised by the triad of:
- Vasculopathy (obliterative microangiopathy)
- Immune dysregulation (autoimmunity, inflammation)
- Progressive fibrosis of the skin and internal organs
The term scleroderma (Greek: skleros = hard, derma = skin) encompasses both localised (morphoea) and systemic forms.
EPIDEMIOLOGY
- Female : Male ratio = 3:1 to 14:1
- Age of onset: 30–50 years
- SSc has the highest case-specific mortality of any autoimmune rheumatic disease
- Incidence: 0.6–16 per million; Prevalence: 2–233 per million
- Blacks develop disease younger and with greater severity than whites
CLASSIFICATION
A. Based on Extent of Skin Involvement (LeRoy, 1988)
| Feature | Diffuse Cutaneous SSc (dcSSc) | Limited Cutaneous SSc (lcSSc) |
|---|---|---|
| Skin involvement | Proximal + distal extremities, trunk, face | Distal to knees/elbows + face only |
| RP onset | Within 1 year of skin changes | Precedes skin changes by years |
| Autoantibody | Anti-Scl-70 (anti-topoisomerase I), anti-RNA Pol III | Anticentromere antibody (50–70%) |
| Organ involvement | Lung fibrosis, renal crisis, cardiac | Isolated PAH, CREST features |
| Acronym | — | CREST syndrome |
CREST = Calcinosis + Raynaud's + Esophageal dysmotility + Sclerodactyly + Telangiectasia
B. Other Subsets
- SSc sine scleroderma — vascular + immunologic features without skin sclerosis (1.5%)
- Early/undifferentiated SSc — Raynaud's + autoantibodies, not yet fulfilling ACR criteria
- Overlap syndromes — mixed connective tissue disease (MCTD), SSc–myositis, SSc–lupus
C. Localised Scleroderma (Morphoea) — skin only, no systemic involvement
- Plaque, linear, generalised, pansclerotic subtypes
PATHOGENESIS
The pathogenesis of SSc involves three interacting and overlapping processes:
GENETIC PREDISPOSITION + ENVIRONMENTAL TRIGGERS
↓
VASCULAR INJURY
↓
IMMUNE DYSREGULATION
↓
FIBROBLAST ACTIVATION
↓
PROGRESSIVE FIBROSIS
1. GENETIC SUSCEPTIBILITY
- HLA associations: HLA-DRB1, DQB1, DPB1 alleles linked to SSc and specific autoantibody profiles
- Non-HLA genes: STAT4, IRF5, CD247 (T cell signalling), BANK1, BLK (B cell activation)
- CARD14 mutations in familial cases (also seen in PRP)
- ~6.5% of cases are familial
- Concordance in identical twins is only ~5%, emphasising the role of environmental triggers
Environmental triggers: Silica dust (190× increased risk in silicotic miners), polyvinyl chloride, bleomycin, organic solvents, silicone implants (controversial)
2. VASCULOPATHY — The Initiating Event
Vascular injury is believed to be the earliest pathogenic event, preceding fibrosis by years.
Mechanism:
- Endothelial cell (EC) injury → triggered by auto-antibodies, reactive oxygen species (ROS), granzyme B from cytotoxic T cells, and viral antigens (CMV molecular mimicry)
- Injured EC → loss of normal vasoregulatory function → imbalance between:
- Vasoconstrictors (↑ endothelin-1, ↑ thromboxane A₂)
- Vasodilators (↓ nitric oxide, ↓ prostacyclin)
- Platelet activation → release of TGF-β, PDGF, serotonin → smooth muscle proliferation
- Intimal proliferation and adventitial fibrosis → luminal narrowing → ischaemia
Vascular consequences:
| Feature | Mechanism |
|---|---|
| Raynaud's phenomenon | Episodic vasospasm of digital arteries |
| Digital ulcers / gangrene | Obliterative endarteritis + ischaemia |
| Pulmonary arterial hypertension (PAH) | Intimal hyperplasia, medial hypertrophy of pulmonary arterioles |
| Scleroderma renal crisis | Hyperplastic arteriolosclerosis → RAAS activation |
| Nailfold capillaroscopy changes | Giant loops, avascular areas, haemorrhages |
Impaired angiogenesis:
- Despite ischaemia, paradoxically defective neovascularisation occurs — VEGF is elevated but its receptor (VEGFR-2) is dysfunctional
- Circulating endothelial progenitor cells (EPCs) are reduced and dysfunctional
3. IMMUNE DYSREGULATION — The Amplifying Event
Both innate and adaptive immunity are dysregulated.
Innate Immunity:
- Type I interferons (IFN-α, IFN-β) are elevated — "interferon signature" seen in SSc
- Toll-like receptors (TLR3, TLR7, TLR8, TLR9) activated by endogenous nucleic acids released from damaged cells → NF-κB activation → pro-inflammatory cytokines (IL-1, IL-6, TNF-α)
- Mast cells — increased in SSc skin; release histamine, TGF-β, chymase → fibroblast activation
- Macrophages — M2 polarisation (alternatively activated) → IL-4, IL-13, TGF-β → pro-fibrotic
Adaptive Immunity:
T cells:
- CD4⁺ T helper cells (Th2 skewing) predominate in lesional skin
- Th2 cytokines (IL-4, IL-13) → directly stimulate fibroblast collagen synthesis
- Th17 cells → IL-17 → synergises with TGF-β in fibrosis
- Regulatory T cells (Tregs) → reduced/dysfunctional → loss of immune tolerance
- CD8⁺ T cells (cytotoxic) → injure endothelial cells via perforin/granzyme B
B cells:
- Autoantibodies — hallmark of SSc (almost mutually exclusive):
| Antibody | SSc Subset | Clinical Association |
|---|---|---|
| Anti-Scl-70 (anti-topoisomerase I) | dcSSc | Interstitial lung fibrosis |
| Anticentromere (ACA) | lcSSc / CREST | Isolated PAH, calcinosis |
| Anti-RNA Polymerase III | dcSSc | Scleroderma renal crisis, cancer association |
| Anti-U1-RNP | Overlap/MCTD | Mixed features |
| Anti-U3-RNP (anti-fibrillarin) | dcSSc | Severe systemic involvement |
| Anti-PM-Scl | SSc-myositis overlap | Muscle involvement |
- Autoantibodies against PDGFR (platelet-derived growth factor receptor) → directly stimulate fibroblast ROS production and collagen synthesis
- B cells also act as antigen-presenting cells perpetuating T cell activation
4. FIBROBLAST ACTIVATION AND FIBROSIS — The End-Effector
The culmination of vasculopathy and immune dysregulation is persistent, autonomous fibroblast activation — the hallmark of SSc.
Key pro-fibrotic mediators:
| Mediator | Source | Effect |
|---|---|---|
| TGF-β (most important) | Platelets, macrophages, T cells, EC | ↑ Collagen I, III synthesis; ↑ TIMP; ↓ MMP → net ECM accumulation |
| PDGF | Platelets, macrophages | Fibroblast proliferation, migration |
| IL-4, IL-13 | Th2 cells | Direct fibroblast stimulation |
| Endothelin-1 | Injured EC | Fibroblast activation + vasoconstriction |
| CTGF (connective tissue growth factor) | Fibroblasts (TGF-β induced) | Amplifies TGF-β fibrotic signalling |
| Lysophosphatidic acid (LPA) | Platelets | Fibroblast activation via LPA receptors |
TGF-β Signalling (Central Pathway):
TGF-β binds TGF-βRI/II receptors
↓
SMAD2/3 phosphorylation → SMAD4 complex
↓
Nuclear translocation → Transcription of COL1A1, COL3A1, ACTA2, CTGF
↓
↑ Collagen I & III, fibronectin, TIMP-1
↓
↓ Matrix metalloproteinases (MMP-1, MMP-3)
↓
NET FIBROSIS (dermis, lungs, kidneys, GI, heart)
Myofibroblast transformation:
- Normal fibroblasts → activated myofibroblasts (α-smooth muscle actin positive, α-SMA⁺)
- Myofibroblasts are autonomous — continue producing ECM even without external TGF-β stimulation
- Epigenetic modifications (DNA methylation, histone modification) lock fibroblasts in a pro-fibrotic state
Epithelial-Mesenchymal Transition (EMT):
- Epithelial cells (e.g., alveolar epithelium) transition to mesenchymal/myofibroblast phenotype under TGF-β → contributes to lung fibrosis
INTEGRATED PATHOGENESIS — SUMMARY DIAGRAM
GENETIC SUSCEPTIBILITY (HLA, STAT4, IRF5)
+
ENVIRONMENTAL TRIGGER (silica, virus, oxidants)
↓
┌───────────────────┐
│ VASCULAR INJURY │ ← EC apoptosis, ROS, anti-EC antibodies
│ (earliest event) │
└────────┬──────────┘
↓
Platelet activation → TGF-β, PDGF release
Raynaud's, digital ulcers, PAH, renal crisis
↓
┌──────────────────────┐
│ IMMUNE ACTIVATION │ ← T cells (Th2/Th17), B cells, macrophages (M2)
│ Autoantibodies │ IFN signature, IL-4, IL-13, IL-17
│ (ACA, anti-Scl-70) │
└──────────┬───────────┘
↓
┌──────────────────────────┐
│ FIBROBLAST ACTIVATION │ ← TGF-β (SMAD pathway), PDGF, CTGF, ET-1
│ Myofibroblast │
│ transformation (α-SMA⁺) │
└──────────┬───────────────┘
↓
┌──────────────────────────┐
│ PROGRESSIVE FIBROSIS │
│ Skin, lungs, GI, │
│ kidneys, heart │
└──────────────────────────┘
HISTOPATHOLOGY
| Stage | Features |
|---|---|
| Early (oedematous) | Perivascular mononuclear (lymphocytic) infiltrate; mucin deposition; capillary dilation |
| Late (fibrotic) | Dense homogenous collagen bundles in reticular dermis (Types I & III) running parallel to skin surface; absent adnexa (hair follicles, sweat glands); effaced rete ridges; avascular dermis |
CLINICAL FEATURES (Brief Overview)
| System | Features |
|---|---|
| Skin | Raynaud's → puffy fingers → sclerodactyly → tight facies (beaked nose, microstomia, perioral furrowing) |
| Lungs | Interstitial lung disease (ILD), PAH — most common cause of death |
| GI | Dysphagia, GERD, Barrett's, malabsorption, pseudo-obstruction |
| Kidney | Scleroderma renal crisis — acute hypertension + renal failure (5–10%) |
| Heart | Pericardial effusion, arrhythmias, diastolic dysfunction |
| MSK | Tendon friction rubs (pathognomonic of dcSSc), arthralgia, myositis |
DIAGNOSIS
ACR/EULAR 2013 Classification Criteria (score-based):
- Skin thickening of fingers extending proximal to MCP joints (score 9 — sufficient alone)
- Sclerodactyly, fingertip pitting scars/digital ulcers
- Abnormal nailfold capillaroscopy
- PAH and/or ILD
- Raynaud's phenomenon
- SSc-specific autoantibodies (ACA, anti-Scl-70, anti-RNA Pol III)
- Total score ≥9 = definite SSc
TREATMENT PRINCIPLES
| Target | Drug |
|---|---|
| Raynaud's / vasculopathy | CCBs (nifedipine), PDE-5 inhibitors (sildenafil), endothelin antagonists (bosentan), IV prostacyclins (iloprost) |
| PAH | Bosentan, sildenafil, riociguat, epoprostenol |
| ILD | Mycophenolate mofetil (first-line), cyclophosphamide, nintedanib |
| Skin fibrosis | Methotrexate (early dcSSc), autologous HSCT (severe progressive) |
| Renal crisis | ACE inhibitors (captopril) — medical emergency |
| GI | PPIs, prokinetics |
| Immunosuppression | Methotrexate, mycophenolate, hydroxychloroquine (overlap) |
| Biologics | Rituximab (B cell depletion), tocilizumab (anti-IL-6R) |
PROGNOSIS
- dcSSc: 10-year survival ~60–70%; main causes of death = PAH, lung fibrosis, renal crisis
- lcSSc: better prognosis; main late complication = isolated PAH
- Scleroderma renal crisis: >90% mortality before ACE inhibitors; now manageable if treated early
Key exam points to remember:
- SSc pathogenesis = Vasculopathy → Immune dysregulation → Fibrosis (in sequence)
- TGF-β / SMAD pathway is the central driver of fibrosis
- Earliest event = endothelial injury → Raynaud's
- Anti-Scl-70 → dcSSc + ILD; Anticentromere → lcSSc/CREST + PAH; Anti-RNA Pol III → renal crisis
- ACE inhibitors are life-saving in scleroderma renal crisis
- SSc has highest mortality of all autoimmune rheumatic diseases
Sources: Fitzpatrick's Dermatology 9e (Chapter 63 — Systemic Sclerosis, Moinzadeh, Denton, Black & Krieg), Andrews' Diseases of the Skin
What is scleroderma?discuss pathogenesis of scleroderma in 10 marks format for 3rd yr dermatology post graduate theory examination
This is the same question you just asked. Here is the complete answer from the previous response — formatted for your 10-marks theory exam:
SCLERODERMA (SYSTEMIC SCLEROSIS)
3rd Year Dermatology PG Theory Examination — 10 Marks
DEFINITION
Scleroderma (Systemic Sclerosis, SSc) is a multisystemic autoimmune connective tissue disease characterised by the triad of:
- Vasculopathy (obliterative microangiopathy)
- Immune dysregulation (autoimmunity + inflammation)
- Progressive fibrosis of the skin and internal organs
The term derives from Greek: skleros = hard, derma = skin.
EPIDEMIOLOGY
- F : M ratio = 3:1 to 14:1
- Age of onset: 30–50 years
- Blacks develop disease younger and with greater severity
- SSc carries the highest case-specific mortality of any autoimmune rheumatic disease
CLASSIFICATION
A. Based on Extent of Skin Involvement (LeRoy, 1988)
| Feature | Diffuse Cutaneous SSc (dcSSc) | Limited Cutaneous SSc (lcSSc) |
|---|---|---|
| Skin involvement | Proximal + distal limbs, trunk, face | Distal to knees/elbows + face only |
| Raynaud's onset | Within 1 year of skin changes | Precedes skin changes by years |
| Key autoantibody | Anti-Scl-70, anti-RNA Pol III | Anticentromere (50–70%) |
| Organ involvement | ILD, renal crisis, cardiac | Isolated PAH, CREST features |
CREST = Calcinosis + Raynaud's + Esophageal dysmotility + Sclerodactyly + Telangiectasia
B. Other Subsets
- SSc sine scleroderma — vascular + immunological features, no skin sclerosis (~1.5%)
- Early/undifferentiated SSc — Raynaud's + autoantibodies, not yet fulfilling ACR criteria
- Overlap syndromes — SSc + myositis, SSc + SLE, MCTD
C. Localised Scleroderma (Morphoea)
Skin-only involvement; no systemic disease. Subtypes: plaque, linear, generalised, pansclerotic.
PATHOGENESIS
The pathogenesis of SSc involves three interacting, overlapping processes set against a background of genetic susceptibility and environmental triggers:
GENETIC PREDISPOSITION + ENVIRONMENTAL TRIGGERS
↓
VASCULAR INJURY (initiating event)
↓
IMMUNE DYSREGULATION (amplifying event)
↓
FIBROBLAST ACTIVATION (end-effector)
↓
PROGRESSIVE FIBROSIS (skin, lungs, GI, kidneys, heart)
1. GENETIC SUSCEPTIBILITY
- HLA associations: HLA-DRB1, DQB1, DPB1 alleles — linked to specific autoantibody profiles
- Non-HLA genes: STAT4, IRF5 (interferon pathway), CD247 (T cell signalling), BANK1, BLK (B cell activation)
- ~6.5% of cases are familial
- Twin concordance is only ~5% → environmental triggers are essential
Environmental triggers:
- Silica dust — underground coal/gold miners have 190× increased risk of SSc
- Polyvinyl chloride, organic solvents, bleomycin (dose-dependent, reversible)
- Viral triggers — CMV molecular mimicry (anti-UL83 antibodies cross-react with anti-topoisomerase I)
2. VASCULOPATHY — The Initiating Event
Vascular injury is the earliest pathogenic event, preceding fibrosis by years.
Mechanism of endothelial injury:
- Anti-endothelial cell antibodies (AECA) → complement activation → EC apoptosis
- Reactive oxygen species (ROS) from activated neutrophils and ischaemia-reperfusion
- CD8⁺ cytotoxic T cells → perforin/granzyme B → EC lysis
- CMV-derived antigens → molecular mimicry → autoimmune EC attack
Consequences of EC injury:
| Mechanism | Result |
|---|---|
| ↑ Endothelin-1, ↑ Thromboxane A₂ | Vasoconstriction |
| ↓ Nitric oxide (NO), ↓ Prostacyclin | Loss of vasodilation |
| Platelet activation → TGF-β, PDGF, serotonin | Smooth muscle proliferation, intimal fibrosis |
| Intimal hyperplasia + adventitial fibrosis | Luminal narrowing → ischaemia |
Clinical vascular manifestations:
- Raynaud's phenomenon — episodic vasospasm of digital arteries (earliest sign)
- Digital ulcers / gangrene — obliterative endarteritis
- PAH — intimal hyperplasia + medial hypertrophy of pulmonary arterioles
- Scleroderma renal crisis (SRC) — hyperplastic arteriolosclerosis → RAAS activation → malignant hypertension
- Nailfold capillaroscopy — giant loops, avascular areas, haemorrhages (diagnostic tool)
Impaired angiogenesis (paradox):
- Despite ischaemia, VEGF is elevated but its receptor VEGFR-2 is dysfunctional
- Circulating endothelial progenitor cells (EPCs) are reduced and functionally impaired
- → Net result: inadequate vessel repair → progressive vascular obliteration

3. IMMUNE DYSREGULATION — The Amplifying Event
Both innate and adaptive immunity are dysregulated and amplify vascular injury and fibrosis.
Innate Immunity:
- Type I interferons (IFN-α, IFN-β) — elevated; "interferon signature" present in SSc blood
- TLR3, TLR7, TLR8, TLR9 activated by endogenous nucleic acids from damaged cells → NF-κB → IL-1, IL-6, TNF-α
- Mast cells — increased in SSc lesional skin; release TGF-β, chymase, histamine → fibroblast activation
- Macrophages — M2 (alternatively activated) polarisation → IL-4, IL-13, TGF-β → pro-fibrotic milieu
Adaptive Immunity — T cells:
- Th2 skewing in lesional skin — IL-4, IL-13 → directly stimulate collagen synthesis by fibroblasts
- Th17 cells — IL-17 → synergises with TGF-β to drive fibrosis
- Tregs (regulatory T cells) — reduced and dysfunctional → loss of peripheral tolerance
- CD8⁺ cytotoxic T cells — injure endothelium via perforin/granzyme B
Adaptive Immunity — B cells and Autoantibodies:
| Autoantibody | SSc Subset | Key Clinical Association |
|---|---|---|
| Anti-Scl-70 (anti-topoisomerase I) | dcSSc | Interstitial lung disease (ILD) |
| Anticentromere (ACA) | lcSSc / CREST | Isolated PAH, calcinosis, better prognosis |
| Anti-RNA Polymerase III | dcSSc | Scleroderma renal crisis; cancer-associated SSc |
| Anti-U1-RNP | Overlap / MCTD | Mixed connective tissue features |
| Anti-U3-RNP (anti-fibrillarin) | dcSSc | Severe systemic, PAH, skeletal myopathy |
| Anti-PM-Scl | SSc–myositis overlap | Muscle inflammation |
| Anti-Th/To | lcSSc | PAH, ILD |
These autoantibodies are almost mutually exclusive in any one patient and serve as prognostic markers.
- Anti-PDGFR antibodies → directly stimulate fibroblast ROS production and collagen synthesis
- B cells also function as antigen-presenting cells → perpetuate T cell autoimmunity
4. FIBROBLAST ACTIVATION AND FIBROSIS — The End-Effector
The culmination of vasculopathy and immune dysregulation is persistent, autonomous fibroblast activation — the defining feature of SSc.
Key pro-fibrotic mediators:
| Mediator | Source | Effect on Fibroblasts |
|---|---|---|
| TGF-β (most important) | Platelets, macrophages, T cells, EC | ↑ Collagen I & III; ↑ TIMP; ↓ MMP → net ECM accumulation |
| PDGF | Platelets, macrophages | Proliferation, migration, ROS production |
| IL-4, IL-13 | Th2 cells | Direct collagen synthesis stimulation |
| Endothelin-1 | Injured EC | Fibroblast activation + vasoconstriction |
| CTGF (connective tissue growth factor) | Fibroblasts (TGF-β–induced) | Amplifies TGF-β signalling (autocrine loop) |
| Lysophosphatidic acid (LPA) | Platelets | Fibroblast activation via LPA receptors |
TGF-β / SMAD Signalling Pathway (Central):
TGF-β → TGF-βR I/II (serine-threonine kinase)
↓
SMAD2/3 phosphorylation
↓
SMAD2/3 + SMAD4 complex (nuclear translocation)
↓
Transcription of:
• COL1A1, COL3A1 (Collagen I & III)
• ACTA2 (α-smooth muscle actin)
• CTGF, fibronectin, TIMP-1
↓
↓ MMP-1, MMP-3 (collagen-degrading enzymes)
↓
NET DERMAL AND VISCERAL FIBROSIS
Non-SMAD pathways also activated: PI3K/Akt, MAPK/ERK, Wnt/β-catenin, JAK/STAT
Myofibroblast Transformation:
- Normal dermal fibroblasts → activated myofibroblasts (α-SMA⁺, contractile)
- Myofibroblasts produce vast amounts of collagen, fibronectin, and CTGF
- They become autonomous — continue ECM synthesis even without external TGF-β (due to autocrine loops)
- Epigenetic locking: DNA methylation of anti-fibrotic genes (FLI1, PPAR-γ); histone modifications → constitutive pro-fibrotic gene expression
Epithelial-Mesenchymal Transition (EMT):
- Under TGF-β, alveolar/tubular epithelial cells transition to myofibroblast phenotype
- Contributes to pulmonary fibrosis and renal interstitial fibrosis
INTEGRATED PATHOGENESIS SUMMARY
GENETIC SUSCEPTIBILITY (HLA, STAT4, IRF5)
+
ENVIRONMENTAL TRIGGER (silica, CMV, oxidants)
↓
┌─────────────────────────┐
│ VASCULAR INJURY │ ← AECA, ROS, CD8⁺ T cells
│ (EC apoptosis/damage) │
└──────────┬──────────────┘
│
Platelet activation: TGF-β, PDGF, serotonin
Imbalance: ↑ET-1, ↑TXA₂ vs ↓NO, ↓Prostacyclin
→ Raynaud's, digital ulcers, PAH, SRC
↓
┌─────────────────────────────────┐
│ IMMUNE DYSREGULATION │
│ Th2/Th17, M2 macrophages, │
│ Mast cells, Type I IFN │
│ Autoantibodies (ACA, Scl-70, │
│ RNA Pol III) │
└──────────────┬──────────────────┘
│ TGF-β, IL-4, IL-13, PDGF, CTGF, ET-1
↓
┌─────────────────────────────────┐
│ FIBROBLAST ACTIVATION │
│ TGF-β/SMAD pathway │
│ Myofibroblast transformation │
│ Epigenetic locking │
└──────────────┬──────────────────┘
↓
┌─────────────────────────────────┐
│ PROGRESSIVE FIBROSIS │
│ Skin → Lungs → GI → Kidney │
│ → Heart │
└─────────────────────────────────┘
HISTOPATHOLOGY

| Stage | Histological Features |
|---|---|
| Early (oedematous) | Perivascular lymphocytic infiltrate; mucin deposition; capillary dilation; interstitial oedema |
| Late (fibrotic) | Dense homogenous collagen bundles (Types I & III) in reticular dermis, running parallel to skin surface; loss of hair follicles, sweat glands; effaced rete ridges; avascular/hypovascular dermis |
CLINICAL FEATURES — OVERVIEW
| System | Manifestations |
|---|---|
| Skin | Raynaud's → puffy fingers (early) → sclerodactyly → tight facies (beaked nose, microstomia, perioral radial furrowing), calcinosis, telangiectasias |
| Lungs | ILD (dcSSc), PAH (lcSSc) — most common cause of death |
| GI | Dysphagia, GERD, Barrett's oesophagus, malabsorption, pseudo-obstruction |
| Kidney | Scleroderma renal crisis — sudden malignant hypertension + acute renal failure (5–10%) |
| Heart | Pericardial effusion, arrhythmias, diastolic dysfunction, cor pulmonale |
| MSK | Tendon friction rubs (pathognomonic of dcSSc), arthralgia, inflammatory myopathy |
DIAGNOSIS
ACR/EULAR 2013 Criteria (score-based; ≥9 = definite SSc):
- Skin thickening proximal to MCPJs — score 9 (sufficient alone)
- Sclerodactyly, fingertip pitting scars/digital ulcers
- Abnormal nailfold capillaroscopy
- PAH / ILD
- Raynaud's phenomenon
- SSc-specific autoantibodies (ACA, anti-Scl-70, anti-RNA Pol III)
TREATMENT PRINCIPLES
| Target | Drug(s) |
|---|---|
| Raynaud's / digital ulcers | CCBs (nifedipine), PDE-5 inhibitors (sildenafil), prostacyclins (iloprost IV) |
| PAH | Bosentan (endothelin antagonist), sildenafil, riociguat, epoprostenol |
| ILD | Mycophenolate mofetil (first-line), cyclophosphamide, nintedanib |
| Skin fibrosis | Methotrexate (early dcSSc); autologous HSCT (severe progressive) |
| Renal crisis | ACE inhibitors (captopril) — medical emergency; life-saving |
| GI | PPIs, prokinetics (metoclopramide) |
| Biologics | Rituximab (anti-CD20), tocilizumab (anti-IL-6R) |
Key exam points — must-know:
- Pathogenesis triad: Vasculopathy → Immune dysregulation → Fibrosis (sequential and interdependent)
- TGF-β / SMAD pathway is the central driver of fibrosis — know it step-by-step
- Earliest clinical event = Raynaud's phenomenon (endothelial injury)
- Anti-Scl-70 → dcSSc + ILD | Anticentromere → lcSSc/CREST + PAH | Anti-RNA Pol III → Renal crisis
- ACE inhibitors in renal crisis reduced mortality from >90% to manageable levels
- SSc has highest mortality of all autoimmune rheumatic diseases
- Myofibroblast transformation + epigenetic locking = why fibrosis is irreversible once established
Sources: Fitzpatrick's Dermatology 9e — Chapter 63 (Moinzadeh, Denton, Black & Krieg); Andrews' Diseases of the Skin
What is recalcitrant dermatophytosis ?How do you manage the different types of tines causing dermatophytosis in 10marks format for 3rd yr dermatology post graduate theory examination
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.