Excellent. Now I have comprehensive source material. Let me deliver the corrected classification and full notes.
Classification Corrections Before Notes
Here are the errors in your classification, corrected:
Etiological Classification — Corrections
| Your Classification | Correction | Reason |
|---|
| "Coal Workers Pneumonia" | Coal Workers' Pneumoconiosis (CWP) | It is a pneumoconiosis (dust fibrosis), not a pneumonia (infection) |
| Organic dusts: "toxic chemicals" | Toxic chemicals are a separate category, not organic dusts. Organic dusts = biological/plant-origin materials | Organic dusts are grain, cotton, wood, animal proteins, fungal spores; toxic chemicals (isocyanates, fumes) belong under chemical agents |
| "Second hand tobacco use" under Other Environmental Agents | Correct placement, but the proper term is Environmental Tobacco Smoke (ETS) / Passive smoking | Minor terminology correction |
Disease-Based Classification — Corrections
| Your Classification | Correction | Reason |
|---|
| "Coal Workers Pneumonia" | Coal Workers' Pneumoconiosis (CWP) | Same as above |
| "Lung Malignancies: mesothelioma, bronchogenic carcinoma, lung cancer" | Remove "lung cancer" as a separate entry — bronchogenic carcinoma IS lung cancer; keep as: mesothelioma + bronchogenic carcinoma | Redundant listing; bronchogenic carcinoma and lung cancer are synonymous |
| Hypersensitivity Pneumonitis listed as a standalone group separate from organic dusts | Correct — HP is rightly kept as a distinct disease-based category | No change needed, this is standard |
CORRECTED CLASSIFICATIONS
✅ Etiological Classification
1. Inorganic Dusts
- Asbestosis, Silicosis, Coal Workers' Pneumoconiosis, Berylliosis
2. Organic Dusts
- Cotton dust (byssinosis), Grain dust, Other agricultural dusts (hay, mushroom compost, sugarcane bagasse)
3. Chemical Agents & Fumes
- Isocyanates (TDI/MDI), Toxic gases (chlorine, phosgene, NOx), Hydrocarbons
4. Other Environmental Agents
- Uranium, Radon, Environmental tobacco smoke (second-hand smoke), Polycyclic aromatic hydrocarbons, Biomass smoke, Diesel exhaust
✅ Disease-Based Classification
1. Pneumoconioses
- Asbestosis, Silicosis, Coal Workers' Pneumoconiosis, Berylliosis
2. Occupational Airway Diseases
- Occupational Asthma, Byssinosis, Chronic Bronchitis, COPD
3. Hypersensitivity Pneumonitis (Extrinsic Allergic Alveolitis)
- Farmer's Lung, Bird Fancier's Lung, Bagassosis, Mushroom Worker's Lung
4. Occupational Lung Infections
- Tuberculosis, Anthrax, Histoplasmosis, Aspergillosis
5. Lung Malignancies
- Mesothelioma, Bronchogenic Carcinoma (Lung Cancer)
OCCUPATIONAL LUNG DISEASES — COMPLETE NOTES
ETIOLOGICAL CLASSIFICATION & NOTES
1. INORGANIC DUSTS
These are mineral/metallic dusts that cause pneumoconioses — fibrotic lung diseases from inorganic particle deposition.
General Pathogenesis (All Pneumoconioses)
- Dust particles <10 µm reach alveoli
- Phagocytosed by alveolar macrophages
- Macrophage activation → release of cytokines (IL-1β, TNF-α, TGF-β)
- Progressive fibrosis, impaired mucociliary clearance
- Fibrosis severity depends on: particle size, solubility, inherent fibrogenicity, dose, and duration
A. SILICOSIS
| Feature | Detail |
|---|
| Agent | Crystalline silica (SiO₂ — quartz, cristobalite, tridymite) |
| Occupations | Sandblasting, tunneling, granite quarrying, stone cutting, foundry work, ceramics, glass manufacturing |
| Fibrogenicity | Very high — silica is the most fibrogenic inorganic dust |
Types:
| Type | Exposure Intensity | Latency | Key Features |
|---|
| Chronic (Classic) | Low–moderate | >10 years | Upper-lobe rounded nodules; often asymptomatic early |
| Accelerated | Moderate–heavy | 5–10 years | Faster progression; similar radiological pattern |
| Acute (Silicoproteinosis) | Very heavy (sandblasting in enclosed spaces) | <5 years (as little as 10 months) | Resembles pulmonary alveolar proteinosis; "crazy paving" on HRCT; rapidly progressive |
Pathology:
- Silicotic nodule — concentric whorls of hyalinized collagen with birefringent silica crystals at center
- Nodules coalesce → Progressive Massive Fibrosis (PMF)
Clinical Features:
- Dyspnea on exertion (progressive)
- Dry cough
- Late: respiratory failure, cor pulmonale
- Acute form: fever, weight loss, rapid deterioration
Radiology:
- Simple: small rounded opacities, upper lobe predominant
- Complicated/PMF: masses ≥1 cm, upper lobe conglomerate nodules
- Eggshell calcification of hilar lymph nodes — pathognomonic
- Acute: miliary infiltrates, "crazy paving" pattern on HRCT
Complications:
- Silicotuberculosis — silica impairs macrophage mycobacterial killing; TB risk ↑ 2–3×
- PMF — severe restrictive deficits, premature death
- Lung cancer — IARC Group 1 carcinogen (Harrison's p. 7989)
- Caplan syndrome — silicotic nodules + seropositive rheumatoid arthritis
- Erasmus syndrome — silicosis + scleroderma
- Chronic renal disease (rare)
Diagnosis:
- Occupational history + CXR/HRCT
- PFTs: restrictive (mixed in PMF)
- IGRA/Mantoux — screen for TB
- Biopsy rarely needed
Management:
- Remove from exposure
- Whole-lung lavage (acute form)
- Treat TB promptly and aggressively
- Supportive: O₂, bronchodilators, vaccinations
- Lung transplantation (end-stage)
B. COAL WORKERS' PNEUMOCONIOSIS (CWP) — "Black Lung"
| Feature | Detail |
|---|
| Agent | Coal dust (mixture of organic + inorganic particles; anthracite has higher silica content) |
| Occupations | Underground coal mining, surface mining, coal processing |
| Key point | Effects of coal dust + cigarette smoke are additive (Harrison's p. 7983) |
Pathology:
- Coal macule — focal accumulation of dust-laden macrophages around respiratory bronchioles (earliest lesion)
- Coal nodule — collagen deposition around coal macule
- PMF — masses >1 cm in upper lobes
Types:
| Type | Radiological Finding | Clinical Impact |
|---|
| Simple CWP | Nodules <1 cm, upper/mid zones | Usually no pulmonary impairment |
| Complicated CWP / PMF | Nodules ≥1 cm, coalescing upper lobe masses | Severe functional deficits, premature mortality |
Complications:
- PMF — progressive respiratory failure
- Caplan Syndrome — first described in coal miners; CWP nodules + seropositive RA (Harrison's p. 7983)
- COPD — coal dust causes chronic bronchitis and airflow obstruction independently
- Cor pulmonale
Diagnosis:
- CXR: upper-lobe rounded opacities; ILO classification
- HRCT: confirms nodules, PMF masses
- PFTs: mixed obstructive + restrictive pattern
Management:
- Remove from exposure
- Supportive: bronchodilators, O₂
- No disease-modifying therapy available
- Lung transplantation (end-stage)
C. ASBESTOSIS
| Feature | Detail |
|---|
| Agent | Asbestos fibers — amphiboles (crocidolite, amosite) most fibrogenic; chrysotile (serpentine) also causes disease |
| Occupations | Construction/insulation workers, shipbuilders, asbestos miners, brake lining workers, demolition |
| Latency | 15–40 years from first exposure |
| Distribution | Lower lobe fibrosis (opposite of silicosis/CWP) |
Pathology:
- Long fibers deposited in alveoli → incomplete macrophage clearance → persistent inflammation → interstitial fibrosis
- Asbestos bodies (ferruginous bodies) — asbestos fibers coated with iron-protein complex; found in BAL/biopsy
- Fibrosis is lower lobe, subpleural predominant
Clinical Features:
- Progressive exertional dyspnea
- Dry cough
- Bibasilar fine Velcro crackles (hallmark)
- Digital clubbing (advanced)
- Cor pulmonale (late)
- Pleural disease: plaques, benign effusion, diffuse thickening, rounded atelectasis
Radiology:
- CXR: bilateral lower-lobe reticulation, honeycombing; calcified pleural plaques ("holly leaf" pattern)
- HRCT: subpleural reticulation, traction bronchiectasis, honeycombing; lower-lobe predominant
PFTs: Restrictive — ↓FVC, ↓TLC, ↓DLCO; FEV₁/FVC preserved or elevated
Asbestos-Related Malignancies:
| Malignancy | Key Points |
|---|
| Lung Cancer (Bronchogenic carcinoma) | Most common asbestos cancer; all histological types; latency ≥15–19 years; synergistic with smoking (multiplicative risk — 50–90× in smokers vs non-exposed non-smokers) (Harrison's p. 7978) |
| Mesothelioma (pleural/peritoneal) | NOT associated with smoking; even brief exposure sufficient; latency 30–40 years; very poor prognosis |
Management:
- No specific therapy — supportive care same as any diffuse interstitial fibrosis (Harrison's p. 7978)
- Smoking cessation (critical)
- Annual low-dose CT surveillance for lung cancer
- O₂ therapy, pulmonary rehabilitation
- Mesothelioma: cisplatin + pemetrexed; nivolumab + ipilimumab (unresectable)
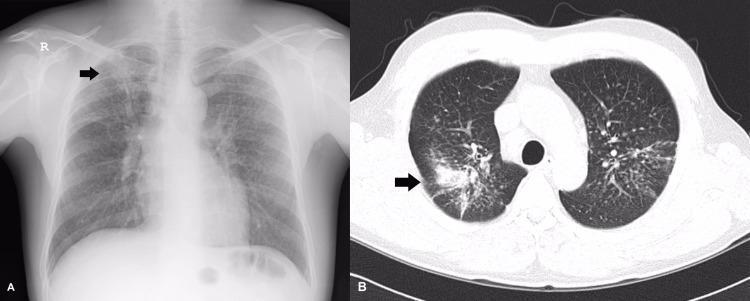
D. BERYLLIOSIS (Chronic Beryllium Disease — CBD)
| Feature | Detail |
|---|
| Agent | Beryllium metal/oxide/salts |
| Occupations | Aerospace, nuclear weapons/reactors, electronics, ceramics, dental alloy manufacturing |
| Mechanism | Type IV hypersensitivity (T-cell mediated); NOT a direct toxic/fibrotic effect |
| Genetic susceptibility | HLA-DPB1 Glu69 allele — increases risk; ~2–6% of exposed workers develop CBD |
Pathology: Non-caseating granulomas — histologically identical to sarcoidosis
Clinical Features:
- Mimics sarcoidosis clinically and radiologically
- Dyspnea, cough, fatigue, chest pain
- Bilateral hilar lymphadenopathy
- Skin, liver, lymph node involvement possible
Key Diagnostic Test:
- Beryllium Lymphocyte Proliferation Test (BeLPT) — blood or BAL
- Positive BeLPT = beryllium sensitization → differentiates from sarcoidosis
Radiology: Bilateral micronodules, hilar lymphadenopathy, upper-lobe fibrosis (mimics sarcoidosis)
Management:
- Remove from beryllium exposure
- Corticosteroids (same as sarcoidosis approach)
- Long-term follow-up (risk of progression to fibrosis)
2. ORGANIC DUSTS
Organic dusts are biologically derived (plant, animal, fungal origin). They primarily cause hypersensitivity reactions and airway diseases rather than fibrosis from direct toxicity.
Key Organic Dust Sources:
| Source | Disease | Notes |
|---|
| Cotton dust | Byssinosis | Endotoxin-mediated; "Monday fever" |
| Grain dust | Grain fever, OA, chronic bronchitis | Grain workers, farmers |
| Moldy hay | Farmer's Lung (HP) | Thermophilic actinomycetes |
| Bird feathers/droppings | Bird Fancier's Lung (HP) | Avian proteins |
| Sugarcane bagasse | Bagassosis (HP) | Thermoactinomyces sacchari |
| Mushroom compost | Mushroom Worker's Lung (HP) | Thermophilic actinomycetes |
| Wood dust | OA, sinonasal cancer | Hardwoods (oak, beech) |
(Details of each disease covered under Disease-Based Classification below)
3. CHEMICAL AGENTS & FUMES
| Agent | Occupation | Disease |
|---|
| Isocyanates (TDI, MDI) | Polyurethane foam, spray painting | Occupational asthma (most common chemical cause of OA) |
| Chlorine gas | Water treatment, chemical industry | Acute chemical pneumonitis, pulmonary edema |
| Phosgene | Plastics, dye industry | Delayed (up to 24h) pulmonary edema |
| Nitrogen oxides (NOₓ) | Welding, silo gas ("silo filler's disease") | Bronchiolitis obliterans, pulmonary edema |
| Ammonia | Refrigerants, fertilizers | Upper airway injury, tracheobronchitis |
| Zinc oxide fumes | Welding, galvanizing | Metal fume fever — flu-like, self-limiting, 4–8h post-exposure |
| PTFE (Teflon) fumes | Manufacturing | Polymer fume fever — similar to metal fume fever |
| Hydrogen sulfide | Sewers, mining | Systemic toxicity (cytochrome oxidase inhibitor) |
4. OTHER ENVIRONMENTAL AGENTS
According to Harrison's (p. 7989), occupational exposures contribute to approximately 10% of all lung cancer cases.
A. Uranium & Radon
- Uranium — direct radiation-induced carcinogen; lung cancer risk in miners and processing workers
- Radon (radon daughters) — emitted from rock formations in underground mines; alpha particle emission → DNA damage → lung cancer; risk not limited to uranium mines — any underground mining where radon is present (Harrison's p. 7989)
- Combined radon + smoking: synergistic lung cancer risk
B. Environmental Tobacco Smoke (Secondhand Smoke)
- Classified as an occupational/environmental carcinogen (Harrison's p. 7989)
- Causes lung cancer (adenocarcinoma most common in non-smokers)
- Also causes COPD, asthma exacerbations in exposed workers (hospitality, service industry workers historically)
C. Polycyclic Aromatic Hydrocarbons (PAHs)
- Sources: coke oven emissions, diesel exhaust, coal tar, asphalt
- PAHs → DNA adducts → lung cancer (squamous cell carcinoma predominant)
- Also: bladder cancer, skin cancer
D. Biomass Smoke
- Exposure in agricultural workers, developing-world indoor cooking
- Causes: COPD, chronic bronchitis, pulmonary hypertension, lung cancer (long-term exposure)
- WHO estimates biomass smoke causes ~4 million deaths/year globally
E. Diesel Exhaust
- IARC Group 1 carcinogen (since 2012)
- Contains NOₓ, PAHs, fine particulate matter
- Lung cancer risk (especially adenocarcinoma) in truck drivers, miners, dock workers
- Also contributes to COPD and asthma
F. Other Proven/Suspected Respiratory Carcinogens (Harrison's p. 7989)
| Agent | Primary Cancer |
|---|
| Arsenic compounds | Lung, skin |
| Chromium (hexavalent) | Lung |
| Nickel carbonyl (nickel smelting) | Lung, nasal |
| Bis(chloromethyl) ether | Lung (small cell) |
| Vinyl chloride | Sarcomas |
| Beryllium | Lung cancer |
| Silica | Lung cancer |
| Mustard gas | Lung, larynx |
| Formaldehyde | Nasal/nasopharyngeal |
DISEASE-BASED CLASSIFICATION & NOTES
1. PNEUMOCONIOSES
(Covered in full detail under Etiological Classification — Inorganic Dusts above)
Summary Comparison Table:
| Feature | Silicosis | CWP | Asbestosis | Berylliosis |
|---|
| Agent | Crystalline SiO₂ | Coal dust | Asbestos fibers | Beryllium |
| Fibrosis location | Upper lobes | Upper lobes | Lower lobes | Upper lobes |
| Nodule type | Silicotic nodules (whorled collagen) | Coal macules/nodules | Interstitial fibrosis | Non-caseating granulomas |
| Pathognomonic sign | Eggshell calcification (hilar) | PMF upper-lobe masses | Calcified pleural plaques | Positive BeLPT |
| TB risk | ↑↑↑ | ↑ | Normal | Normal |
| Malignancy | Lung cancer | Lung cancer (smoking) | Lung cancer + Mesothelioma | — |
| Caplan syndrome | Yes | Yes (first described) | No | No |
| Treatment | Supportive ± WLL (acute) | Supportive | Supportive | Corticosteroids |
2. OCCUPATIONAL AIRWAY DISEASES
A. OCCUPATIONAL ASTHMA (OA)
Definition: Asthma caused or exacerbated by workplace exposures — either through immunological sensitization or direct irritant mechanisms.
Types:
| Type | Mechanism | Latency | Examples |
|---|
| Sensitizer-induced OA (immunological) | IgE or T-cell mediated | Weeks–years | Isocyanates, latex, flour, animal dander |
| Irritant-induced OA / RADS | Non-immunological, direct airway injury | Acute (minutes–hours after single massive exposure) | Chlorine gas, ammonia, strong acids |
Key Sensitizing Agents:
| Agent Type | Agents | Occupation |
|---|
| High molecular weight (HMW) | Latex, flour, animal dander, enzymes (subtilisin), grain | Healthcare, bakers, lab workers, farmers |
| Low molecular weight (LMW) | Isocyanates (TDI/MDI), colophony (rosin), anhydrides, metals (platinum, chromium), wood dust | Spray painters, electronics, woodworkers |
| Irritants (RADS) | Chlorine, ammonia, strong acids/alkalis | Chemical industry, firefighters |
Clinical Features:
- Work-related pattern — symptoms worse at work, improve on weekends and holidays (key history clue)
- Wheeze, chest tightness, dyspnea, rhinitis/conjunctivitis
- RADS: acute onset after single massive exposure; symptoms persist >3 months
Diagnosis:
- Detailed occupational history
- Serial peak expiratory flow (PEF) monitoring — at work vs away from work (standard diagnostic tool)
- Methacholine challenge — documents non-specific bronchial hyperresponsiveness
- Specific inhalation challenge (SIC) — gold standard (performed in specialist centers)
- Skin prick test / specific IgE for HMW allergens
Management:
- Early removal from exposure — prognosis directly related to duration of continued exposure after diagnosis
- Inhaled corticosteroids + bronchodilators
- If removal not possible: strict exposure reduction + PPE
- Compensation and medicolegal assessment
B. BYSSINOSIS ("Monday Fever" / "Brown Lung")
| Feature | Detail |
|---|
| Agent | Cotton dust (also flax, hemp) |
| Active component | Endotoxin from gram-negative bacteria contaminating cotton; cotton bracts |
| Mechanism | Non-immunological; endotoxin triggers bronchoconstriction and neutrophilic airway inflammation |
| Occupations | Cotton textile workers (card room workers highest risk), rope makers |
Classic Presentation — "Monday Fever":
- Chest tightness, cough, dyspnea on return to work after weekend
- Symptoms improve as the working week progresses (tachyphylaxis)
- Chronic byssinosis: persistent symptoms, COPD-like picture
Schilling Grading:
| Grade | Symptoms |
|---|
| 0 | No symptoms |
| ½ | Occasional chest tightness on Mondays only |
| 1 | Chest tightness every Monday |
| 2 | Chest tightness on Monday and other days |
| 3 | Permanent disability with chronic symptoms |
Diagnosis:
- Occupational history + Monday symptom pattern
- ≥10% fall in FEV₁ across a Monday work shift — diagnostic criterion
- CXR normal until late stages
Management:
- Dust control measures (engineering controls)
- Bronchodilators
- Smoking cessation
- Advanced disease: COPD management principles
C. OCCUPATIONAL CHRONIC BRONCHITIS
- Chronic productive cough for >3 months/year for ≥2 consecutive years, attributable to occupational dust/fume exposure
- Agents: coal dust, silica, grain dust, cadmium fumes, wood dust
- Distinguishable from smoking-related bronchitis only by careful exposure history
- Management: same as non-occupational chronic bronchitis (inhaled therapies, smoking cessation, exposure reduction)
D. OCCUPATIONAL COPD
- Occupational exposures account for 10–20% of COPD cases (ATS statement); up to 31% in never-smokers (GOLD 2025, p. 22)
- Agents: coal dust, silica, cadmium, grain dust, isocyanates, vapors/gases/dusts/fumes (VGDF)
- Risk is higher in less regulated work environments globally
- COPD attributable to occupational exposure is underdiagnosed as the clinical picture is identical to smoking-induced COPD
- Pesticide inhalation → increased COPD incidence, even in non-smokers (GOLD 2025, p. 22)
- Management: standard COPD guidelines (GOLD strategy) + mandatory removal from exposure
3. HYPERSENSITIVITY PNEUMONITIS (HP) / Extrinsic Allergic Alveolitis (EAA)
Definition: Diffuse parenchymal lung disease caused by immune-mediated reaction (Type III + IV hypersensitivity) to inhaled organic antigens in sensitized individuals.
Mechanism:
- Type III (immune complex deposition) — acute phase
- Type IV (T-lymphocyte-mediated, delayed) — chronic granulomatous inflammation
- Result: lymphocytic alveolitis → non-caseating granulomas → fibrosis (if chronic/untreated)
Key Diseases:
| Disease | Causative Antigen | Source |
|---|
| Farmer's Lung | Saccharopolyspora rectivirgula (thermophilic actinomycetes) | Moldy hay/grain |
| Bird Fancier's Lung | Avian serum proteins, feathers, excreta | Pigeons, parrots, budgerigars |
| Bagassosis | Thermoactinomyces sacchari | Moldy sugarcane bagasse |
| Mushroom Worker's Lung | Thermophilic actinomycetes | Mushroom compost |
| Malt Worker's Lung | Aspergillus clavatus | Moldy malt |
| Humidifier Lung | Bacteria, amoeba (Naegleria), fungi | Contaminated HVAC/humidifiers |
| Cheese Worker's Lung | Penicillium casei | Moldy cheese |
Clinical Forms:
| Form | Onset | Exposure Pattern | Features |
|---|
| Acute | 4–8 hours post-exposure | Intermittent heavy exposure | Flu-like: fever, chills, myalgia, cough, dyspnea; resolves 24–72h |
| Subacute | Days–weeks | Repeated moderate exposure | Progressive cough, dyspnea, weight loss, fatigue |
| Chronic | Months–years | Continuous low-level exposure | Progressive irreversible fibrosis; indistinguishable from IPF |
Clinical Features (Acute):
- Fever, chills, myalgia — "flu-like" 4–8 hours after antigen exposure
- Cough, dyspnea, crackles on auscultation
- Resolves spontaneously on antigen removal
Diagnosis:
- Detailed antigen exposure history + temporal relationship
- HRCT: Ground-glass opacities, centrilobular nodules, mosaic attenuation (air trapping); fibrosis in chronic form
- BAL: Lymphocytosis; CD4:CD8 ratio <1 (vs. sarcoidosis where ratio >3.5) — key differentiator
- Precipitating IgG antibodies (precipitins) against causative antigen — confirms exposure, not disease activity
- Lung biopsy: non-caseating granulomas, lymphocytic alveolitis, bronchiolocentric distribution
- Inhalation challenge — gold standard (rarely performed)
PFTs: Restrictive pattern + ↓DLCO
Differential Diagnosis: Sarcoidosis (BAL CD4:CD8 >3.5, no exposure history), IPF (no granulomas, older age)
Management:
- Antigen avoidance — cornerstone; most important intervention
- Acute/subacute: Corticosteroids (prednisolone 40–60 mg/day, tapered over months) — accelerate resolution
- Chronic fibrotic HP: Nintedanib (antifibrotic) — approved for progressive fibrotic ILD including HP
- Supplemental O₂, pulmonary rehabilitation
- Lung transplantation for end-stage disease
4. OCCUPATIONAL LUNG INFECTIONS
Certain occupations increase exposure to specific pathogens, causing characteristic lung infections.
A. TUBERCULOSIS (TB)
| Aspect | Detail |
|---|
| At-risk occupations | Healthcare workers, miners (silicosis ↑ risk 2–3×), prisoners, immunosuppressed workers |
| Key occupational association | Silicotuberculosis — silica impairs macrophage mycobacterial killing; miners are at significantly elevated TB risk |
| Clinical features | Cough >2 weeks, hemoptysis, fever, night sweats, weight loss, apical infiltrates/cavitation on CXR |
| Diagnosis | Sputum AFB smear/culture; GeneXpert (NAAT); IGRA or Mantoux |
| Management | Standard RIPE therapy (Rifampicin, Isoniazid, Pyrazinamide, Ethambutol); DOT; screen contacts |
B. ANTHRAX (Pulmonary / Inhalation Anthrax)
| Aspect | Detail |
|---|
| Agent | Bacillus anthracis — spores |
| At-risk occupations | Wool sorters ("woolsorter's disease"), hide/leather workers, abattoir workers, laboratory workers |
| Pathogenesis | Inhaled spores → germination in mediastinal lymph nodes → hemorrhagic mediastinitis → septicemia |
| Clinical features | Biphasic: initial flu-like illness (2–5 days) → sudden deterioration with hemorrhagic mediastinitis, pleural effusions, septic shock |
| CXR/CT | Widened mediastinum, hemorrhagic pleural effusions (hallmark) |
| Diagnosis | Blood culture, serology, PCR |
| Management | IV ciprofloxacin or doxycycline; anthrax antitoxin; pleural drainage |
| Prognosis | High mortality (>80%) if untreated; vaccination available for at-risk workers |
C. HISTOPLASMOSIS
| Aspect | Detail |
|---|
| Agent | Histoplasma capsulatum (dimorphic fungus) |
| At-risk occupations | Cave explorers (spelunkers), construction/demolition workers (disturbing soil), farmers, archaeologists; endemic in Mississippi/Ohio River valleys |
| Pathogenesis | Inhaled microconidia → phagocytosis by macrophages → dissemination in immunocompromised |
| Clinical forms | Acute pulmonary (mild flu-like in most), chronic cavitary histoplasmosis (smokers/emphysema — upper-lobe cavitation resembling TB) (Harrison's p. 6196), disseminated (immunocompromised) |
| CXR | Hilar lymphadenopathy, calcified granulomas ("buckshot" calcifications), upper-lobe cavities (chronic) |
| Diagnosis | Urine/serum Histoplasma antigen (most sensitive), serology, culture |
| Management | Mild acute: observation; Moderate–severe/chronic: Itraconazole 6–12 months; Severe/disseminated: Liposomal amphotericin B → step down to itraconazole |
D. ASPERGILLOSIS
| Aspect | Detail |
|---|
| Agent | Aspergillus fumigatus (most common), A. flavus, A. niger |
| At-risk occupations | Construction workers, demolition, farmers, compost workers, healthcare workers (immunosuppressed patients) |
| Forms | |
| Allergic Bronchopulmonary Aspergillosis (ABPA) | Hypersensitivity reaction in asthmatics/CF; central bronchiectasis, mucus plugging, eosinophilia |
| Aspergilloma | Fungal ball in pre-existing cavity (TB, sarcoid); hemoptysis; "crescent sign" on CXR |
| Chronic Pulmonary Aspergillosis (CPA) | Subacute/chronic; upper-lobe cavities, weight loss |
| Invasive Pulmonary Aspergillosis (IPA) | Immunocompromised; angioinvasion → halo sign on CT, wedge-shaped infarcts; high mortality |
| Diagnosis | Serum galactomannan, β-D-glucan, BAL culture/PCR, CT chest (halo sign in IPA) |
| Management | ABPA: corticosteroids + itraconazole; Aspergilloma: monitoring ± surgical resection; IPA: Voriconazole (first-line); isavuconazole (alternative) |
5. LUNG MALIGNANCIES
Occupational exposures account for approximately 10% of all lung cancers (Harrison's p. 7989).
A. BRONCHOGENIC CARCINOMA (Lung Cancer)
Occupational Carcinogens Causing Lung Cancer (Harrison's p. 7989):
| Carcinogen | Source/Occupation | IARC Classification |
|---|
| Asbestos | Construction, shipbuilding | Group 1 |
| Silica | Mining, sandblasting | Group 1 |
| Radon/Uranium | Underground mining | Group 1 |
| Diesel exhaust | Transport, mining | Group 1 |
| Secondhand tobacco smoke | Hospitality, offices | Group 1 |
| Polycyclic aromatic hydrocarbons | Coke ovens, chimney sweeping | Group 1 |
| Chromium (hexavalent) | Electroplating, welding | Group 1 |
| Arsenic | Pesticide production, smelting | Group 1 |
| Nickel carbonyl | Nickel smelting | Group 1 |
| Bis(chloromethyl) ether | Chemical industry | Group 1 — causes small cell carcinoma |
| Beryllium | Aerospace, nuclear | Group 1 |
| Vinyl chloride | PVC manufacturing | Group 1 — angiosarcomas/sarcomas |
Important Interactions:
- Asbestos + Smoking → multiplicative (not merely additive) lung cancer risk; up to 50–90× baseline risk in asbestos workers who smoke (Harrison's p. 7978)
- Radon + Smoking → synergistic elevated lung cancer risk
Histological Types by Exposure:
- Asbestos, silica, PAHs: all types (squamous, adenocarcinoma, SCLC)
- Bis(chloromethyl) ether: specifically small cell lung cancer
- Secondhand smoke in non-smokers: predominantly adenocarcinoma
Management: Standard lung cancer management (surgery, chemotherapy, immunotherapy, targeted therapy based on stage and molecular markers)
B. MESOTHELIOMA
| Feature | Detail |
|---|
| Agent | Asbestos (ALL fiber types; amphiboles > chrysotile) |
| Types | Pleural (most common), peritoneal, pericardial |
| Smoking | NOT associated with smoking — key distinguishing feature from lung cancer (Harrison's p. 7978) |
| Latency | 30–40 years (very long) |
| Exposure required | Even brief/low-level asbestos exposure is sufficient |
| Clinical features | Progressive dyspnea, pleuritic chest pain, recurrent pleural effusions, weight loss |
| CXR/CT | Circumferential pleural thickening, pleural effusion, encasing the lung |
| Histology | Epithelioid (best prognosis), sarcomatoid, biphasic |
| Diagnosis | CT chest, thoracoscopy + biopsy; calretinin, WT-1, mesothelin immunohistochemistry |
| Biomarker | Serum mesothelin (SMRP) — surveillance in exposed workers |
| Staging | TNM staging (IASLC/IMIG system) |
| Management | Unresectable: Nivolumab + Ipilimumab (first-line immunotherapy); Cisplatin + Pemetrexed (first-line chemotherapy); surgery (pleurectomy/decortication) for selected early-stage cases |
| Prognosis | Median survival 12–18 months from diagnosis; epithelioid type has best prognosis |
GENERAL PRINCIPLES IN OCCUPATIONAL LUNG DISEASE
Exposure History — Must Cover:
- All jobs held (chronological list with durations)
- Specific agents encountered and intensity of exposure
- Temporal relationship of symptoms to work schedule
- Use of personal protective equipment (PPE)
- Co-exposures — especially cigarette smoking (major effect modifier)
- Latency period — time from first exposure to first symptom
- Hobbies and home exposures (birds, humidifiers, molds)
PFT Pattern Summary:
| Disease | Pattern |
|---|
| Asbestosis, Silicosis (simple), CWP (simple) | Restrictive |
| PMF (silicosis/CWP complicated) | Mixed restrictive + obstructive |
| Occupational Asthma, Byssinosis | Obstructive (reversible) |
| Occupational COPD, Chronic Bronchitis | Obstructive (partially reversible) |
| Hypersensitivity Pneumonitis (acute/subacute) | Restrictive + ↓DLCO |
| Hypersensitivity Pneumonitis (chronic/fibrotic) | Restrictive + ↓DLCO |
Prevention Framework:
Hierarchy of Controls (most → least effective):
1. Elimination — remove the hazardous agent entirely
2. Substitution — replace with a less hazardous material
3. Engineering controls — local exhaust ventilation, wet drilling, enclosure
4. Administrative controls — job rotation, reduced shift duration
5. Personal Protective Equipment (PPE) — respirators, masks (last resort)
Sources: Harrison's Principles of Internal Medicine, 21st Edition (pp. 7970, 7978–7989); GOLD 2025 Report (p. 22); supplemented from standard pulmonology curriculum.