Hep c
Asking for Preferences
Reading File
Finding Sources
Searching PubMed
"hepatitis C virus" AND pathophysiology
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive data from multiple authoritative textbooks. Here is a thorough overview of Hepatitis C — its virology, pathogenesis, epidemiology, and clinical course.
Hepatitis C Virus (HCV) — Overview & Pathophysiology
1. The Virus
HCV is a small (~55 nm), enveloped, positive-sense single-stranded RNA virus, the sole member of the genus Hepacivirus within the Flaviviridae family (which also includes dengue, Zika, West Nile, and yellow fever viruses).
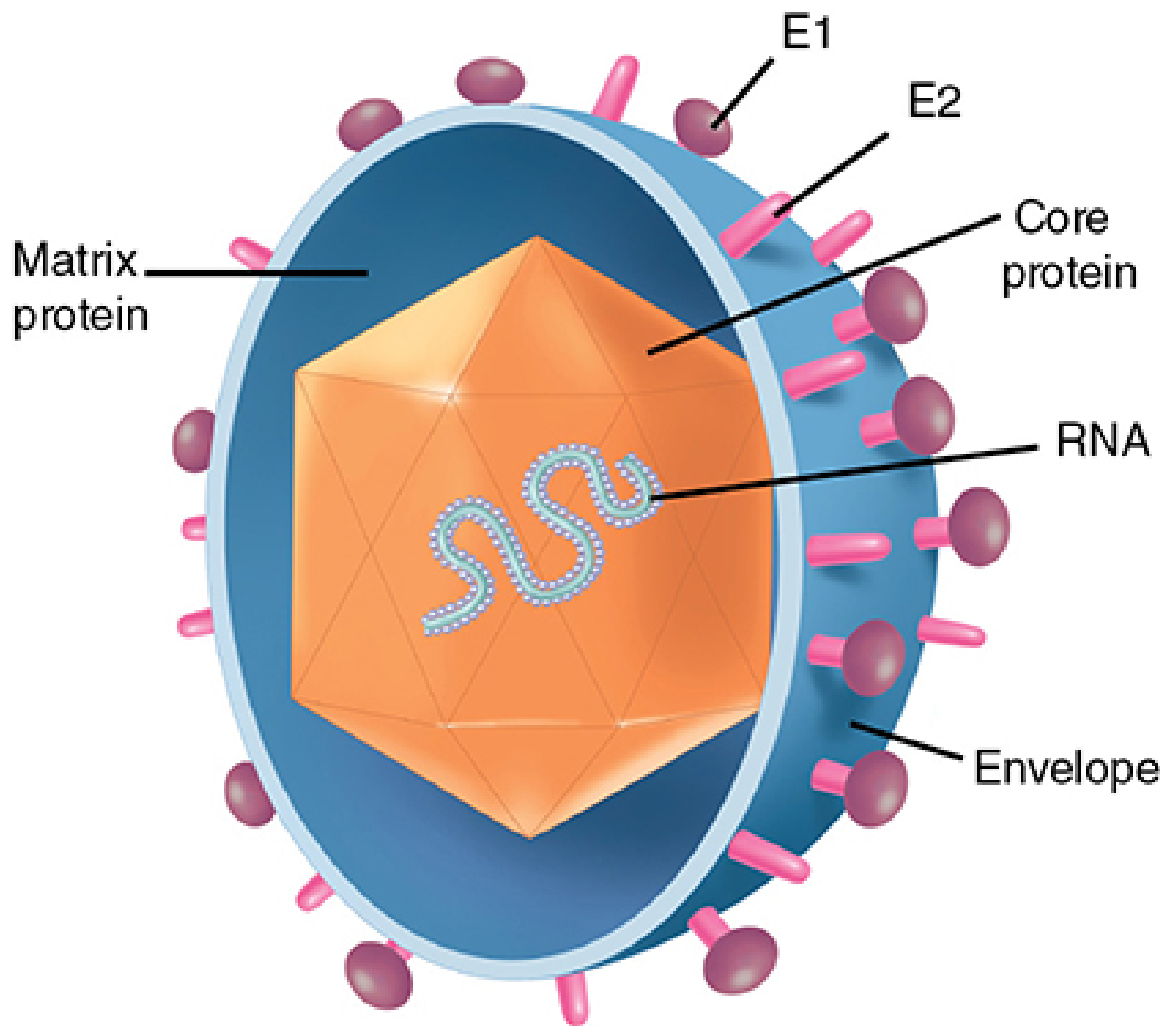
Genome (~9.6 kb) encodes a single large polyprotein that is cleaved by both host and viral proteases into 10 proteins:
| Category | Proteins | Function |
|---|---|---|
| Structural | Core, E1, E2 | Capsid, envelope glycoproteins (cell entry via CD81, SR-B1, claudin-1, occludin) |
| Ion channel | p7 | Membrane ion channel |
| Non-structural | NS2, NS3/4A, NS4B, NS5A, NS5B | Polyprotein cleavage, RNA helicase/protease, replication complex, RNA-dependent RNA polymerase (RdRp) |
The NS5B RNA-dependent RNA polymerase lacks proofreading ability, generating enormous genetic diversity — including quasispecies (viral variants within a single host) and distinct genotypes (≥6) globally. This diversity is the primary obstacle to vaccine development and underlies immune evasion.
2. Genotypes
| Genotype | Geographic predominance | Notes |
|---|---|---|
| 1 (1a, 1b) | North America, Western Europe, Japan | Most common globally; worst response to IFN-based therapy |
| 2 | Worldwide | Best response to IFN therapy |
| 3 | South Asia | Highest rate of spontaneous clearance |
| 4 | Central Africa, Middle East | Highest frequency of chronicity after acute infection |
| 5 | Southern Africa | |
| 6 | East/Southeast Asia |
— Sherris & Ryan's Medical Microbiology, 8th Ed.; Jawetz, Melnick & Adelberg's Medical Microbiology, 28th Ed.
3. Epidemiology & Transmission
- ~1% of the world population has been infected; >70 million chronic carriers worldwide (>3 million in the US)
- Sub-Saharan Africa, South America, and Asia have the highest prevalence (up to 10% in some populations)
- Egypt has ~20% HCV prevalence, linked to mass parenteral treatment campaigns for schistosomiasis (1950s–1980s) using reused needles
Transmission routes (decreasing prevalence):
- Injecting drug use (~80%)
- Clotting factor recipients (pre-1987 era)
- Blood transfusion recipients (pre-screening era)
- Chronic hemodialysis (~10%)
- High-risk sexual practices
- Healthcare workers (~1%)
- Mother-to-child vertical transmission (3–10%); higher with HIV coinfection or high viral load
- No risk with breastfeeding
Incubation period: average 6–7 weeks; seroconversion ~8–9 weeks post-exposure; ~90% anti-HCV positive by 5 months.
— Jawetz, Melnick & Adelberg's Medical Microbiology, 28th Ed.
4. Cell Entry & Replication
- HCV circulates in blood associated with lipoproteins forming lipoviroparticles (LVPs)
- LVPs bind heparan sulfate proteoglycans on hepatocyte surfaces
- Productive entry requires sequential interaction of E2 glycoprotein with SCARB1 → CD81 → claudin-1 → occludin
- Internalization via clathrin-mediated, EGFR-dependent endocytosis
- pH-dependent membrane fusion at the endosome releases the genome into the cytoplasm
- The positive-sense RNA is translated via an IRES (internal ribosome entry site) directly into the polyprotein
- Replication occurs via a negative-sense RNA intermediate template, catalyzed by NS5B RdRp
- New virions are assembled and secreted
— Yamada's Textbook of Gastroenterology, 7th Ed.; Sherris & Ryan's Medical Microbiology, 8th Ed.
5. Pathogenesis of Liver Injury
HCV is not directly cytopathic — liver injury is principally immune-mediated.
Innate immune evasion
- Host pattern recognition receptors detect HCV RNA motifs → trigger IFN and cytokine production
- HCV NS3/4A protease cleaves key innate immune signaling adaptors (MAVS/IPS-1, TRIF), blocking type-I IFN responses
- HCV core, NS4B, and NS5B suppress the NF-κB pathway, reducing antiapoptotic proteins and increasing susceptibility to TNF-α–mediated cell death
Adaptive immune response
- HLA class I-restricted intrahepatic cytotoxic T lymphocytes (CTLs/CD8+ T cells) target core, envelope, and NS protein antigens
- CTLs eliminate infected hepatocytes via apoptosis and IFN-γ–mediated inhibition of viral replication
- CD4+ T cells secrete proinflammatory cytokines contributing to hepatocyte death
- The immune response is characteristically slow and weak, owing to HCV's evolved immune evasion repertoire
TH1/TH2 imbalance in chronic disease
- TH1 cytokines (IL-2, TNF-α) → aggressive hepatic disease
- TH2 cytokines (IL-10) → milder presentation
- Chronic overexpression of TNF-α triggers a "cytokine storm," causing ongoing liver damage
- TNF-α and IL-6 drive insulin resistance in hepatocytes → promotes fibrosis and hepatocarcinogenesis
Genetic host factors
- IL28B CC haplotype (codes for interferon-λ3) → associated with spontaneous HCV clearance; non-CC alleles impair clearance
- A frameshift variant creating IFN-λ4 (ΔG polymorphism) is linked to persistent infection
- MHC class II DR5 allele → lower incidence of cirrhosis
- NK cells: in spontaneous clearance, NK cells show enhanced IFN-γ and activating receptor expression; in persistent infection, NK cytotoxicity is dysfunctional
Viral quasispecies and immune escape
- The lack of NS5B proofreading generates hypervariable regions (HVR1, HVR2) in the E2 envelope glycoprotein
- Continuous mutation allows newly generated HCV variants to escape preexisting humoral and cellular immune responses → a key mechanism driving chronicity
— Harrison's Principles of Internal Medicine, 22nd Ed.; Sherris & Ryan's Medical Microbiology, 8th Ed.; Jawetz, Melnick & Adelberg's, 28th Ed.
6. Clinical Course
| Phase | Features |
|---|---|
| Acute | 80% asymptomatic or mild; 20–30% jaundice; 10–20% nonspecific symptoms (anorexia, malaise, abdominal pain) |
| Chronicity rate | 70–80% of exposed individuals develop chronic infection |
| Chronic | Slowly progressive, often asymptomatic for decades |
| End-stage | Cirrhosis, liver failure, hepatocellular carcinoma (HCC) |
Chronic HCV is a leading cause of cirrhosis and HCC globally. US mortality from HCV exceeds that of HIV.
7. Extrahepatic Manifestations
Immune complex deposition drives extrahepatic disease:
- Mixed cryoglobulinemia (vasculitis)
- Glomerulonephritis
- Arthritis
- Lichen planus, porphyria cutanea tarda (skin)
- Lymphoma (B-cell)
Recent Evidence
A 2024 systematic review (PMID: 38385567) found that HCV eradication by direct-acting antivirals (DAAs) significantly reduces liver steatosis and carotid atherosclerosis, underscoring that HCV's metabolic and vascular effects extend well beyond the liver.
Sources: Harrison's Principles of Internal Medicine 22nd Ed. | Sherris & Ryan's Medical Microbiology 8th Ed. | Jawetz, Melnick & Adelberg's Medical Microbiology 28th Ed. | Yamada's Textbook of Gastroenterology 7th Ed.
This is a shared conversation. Sign in to Orris to start your own chat.