Define IVUS and working principle - Study catheter- based transducers - Imaging mechanism inside
IVUS intravascular ultrasound definition working principle catheter transducer
IVUS intravascular ultrasound catheter imaging mechanism
IVUS — Intravascular Ultrasound
1. Definition
According to Harrison's Principles of Internal Medicine, 21st Edition (p. 6916): "IVUS provides a more accurate anatomic assessment of the coronary artery and the degree of coronary atherosclerosis... used to image atherosclerotic plaque precisely, determine luminal cross-sectional area, and measure vessel size."
2. Working Principle
| Step | Description |
|---|---|
| 1. Pulse emission | The transducer emits short bursts of high-frequency ultrasound (typically 20–60 MHz) into surrounding tissue |
| 2. Wave propagation | Sound waves travel radially outward from the catheter through blood, vessel wall layers (intima, media, adventitia), and surrounding tissue |
| 3. Echo reception | Acoustic impedance differences at tissue boundaries (e.g., blood–intima, media–adventitia) cause partial reflection of echoes back to the transducer |
| 4. Time-of-flight calculation | Distance = (speed of sound × time) / 2 — reconstructs depth from each echo |
| 5. Signal processing | Reflected signals are converted into a grayscale radial image, building a 360° cross-sectional slice |
| 6. Pullback | The catheter is withdrawn at a fixed speed (0.5–1 mm/sec) to generate a longitudinal 3D reconstruction of the vessel |
- Higher frequency → better axial resolution (~100 µm) but limited penetration (~10 mm)
- Lower frequency → deeper penetration for larger vessels (e.g., aorta)
3. Catheter-Based Transducer Design
A. Mechanical (Rotational) IVUS
- A single piezoelectric transducer element at the catheter tip rotates at ~1,800 RPM driven by a flexible driveshaft
- Produces one scan line per rotation; a full 360° image is formed per revolution
- Advantage: High image quality, uniform resolution
- Disadvantage: Mechanical artifacts — "NURD" (Non-Uniform Rotational Distortion) when the driveshaft binds
B. Solid-State (Phased Array) IVUS
- 64 or more piezoelectric elements arranged circumferentially around the catheter tip, activated electronically in sequence (no moving parts)
- Electronic beam steering generates the 360° image
- Advantage: No NURD artifact; more flexible catheter; real-time imaging
- Disadvantage: Slightly lower resolution; near-field artifacts (ring-down artifact from catheter itself)
| Feature | Mechanical (Rotational) | Solid-State (Phased Array) |
|---|---|---|
| Moving parts | Yes (rotating driveshaft) | No |
| Elements | Single | 64+ |
| Artifact | NURD | Ring-down / near-field |
| Resolution | Higher | Slightly lower |
| Flexibility | Less | More |
| Common frequency | 40–45 MHz | 20 MHz |
4. Imaging Mechanism Inside the Vessel
What is Visualized (Layer by Layer):
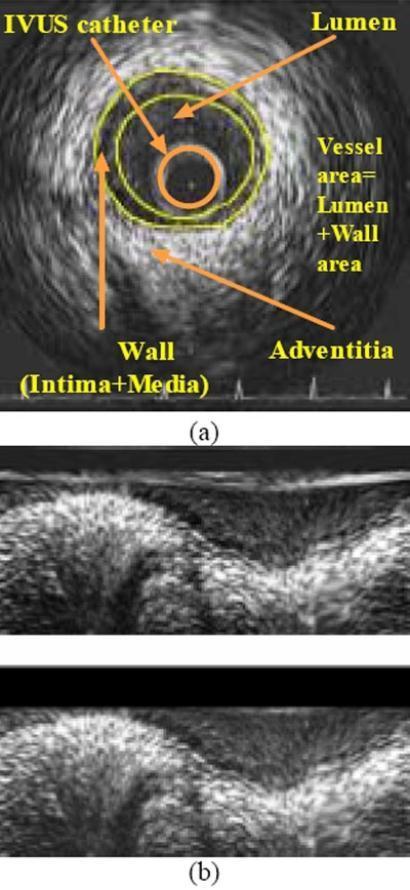
| Structure | IVUS Appearance |
|---|---|
| IVUS catheter | Central hypoechoic circular artifact (the catheter itself) |
| Lumen (blood) | Hypoechoic (dark) — blood has low acoustic impedance |
| Intima | Bright (echogenic) thin inner ring — first reflective boundary |
| Media | Hypoechoic (dark) middle band — smooth muscle is less reflective |
| Adventitia | Hyperechoic (bright) outer layer — dense collagen |
| Atherosclerotic plaque | Varies: fibrous = echogenic; lipid = hypoechoic; calcified = bright with shadowing |
| External elastic lamina | Echogenic boundary separating media from adventitia |
Key Measurements Obtained:
- Lumen Cross-Sectional Area (LCSA) — inner boundary of intima
- Vessel Cross-Sectional Area (VCSA) — outer boundary of media (external elastic lamina)
- Wall/Plaque Area (WCSA) = VCSA − LCSA
- Plaque burden (%) = (WCSA / VCSA) × 100
Clinical Applications:
- Intermediate stenosis assessment (40–70% lesions on angiography)
- Pre-PCI planning — vessel sizing, lesion characterization
- Post-stent assessment — stent apposition, expansion, edge dissection
- Plaque vulnerability — identifying lipid-rich or thin-cap fibroatheroma
- Left main coronary disease evaluation
- Detecting positive (outward) remodeling — vessel enlarges to accommodate plaque (Glagov phenomenon), invisible on angiography
Summary Diagram of the IVUS System
[Console/Processor] ←→ [Pullback Device] ←→ [IVUS Catheter (transducer at tip)]
↓
Emits 40 MHz ultrasound pulses radially
↓
Echoes reflected from vessel wall layers
↓
360° cross-sectional grayscale image
↓
Pullback at 0.5 mm/s → 3D longitudinal reconstruction
Intravascular ultrasound instrumentation
IVUS intravascular ultrasound instrumentation system components console
IVUS intravascular ultrasound instrumentation system diagram components
IVUS Instrumentation
- The IVUS Catheter
- The Transducer (ultrasound element)
- The Pullback Device
- The Imaging Console
- Signal Processing & Display
1. The IVUS Catheter
Physical Specifications
| Parameter | Typical Value |
|---|---|
| French size (shaft) | 2.6 Fr – 3.5 Fr (coronary); up to 8.2 Fr (peripheral/aorta) |
| Tip-to-transducer distance | 8–15 mm |
| Working length | 135–150 cm |
| Guidewire compatibility | 0.014" (coronary); 0.018"–0.035" (peripheral) |
| Monorail vs. over-the-wire | Both designs available |
Catheter Construction (Layers, Proximal to Distal)
Proximal Hub (connector to console)
↓
Outer Shaft (polyimide/nylon braid for torque transmission)
↓
Inner Lumen (guidewire channel)
↓
Drive Cable / Electrical Conductors (to transducer)
↓
Transducer Housing (tip)
↓
Atraumatic Distal Tip (soft, tapered)
- The outer shaft must be flexible enough to navigate tortuous coronary anatomy, yet stiff enough to transmit torque and pullback motion
- The inner guidewire lumen runs alongside or coaxially depending on design (monorail = rapid-exchange; over-the-wire = full lumen)
- Flush ports allow saline flushing to remove air (air causes acoustic shadowing)
2. Transducer — The Core Ultrasound Element
A. Piezoelectric Crystal
- Material: PZT (Lead Zirconate Titanate) or newer PVDF (polyvinylidene fluoride)
- Principle: Piezoelectric effect — mechanical deformation produces voltage; applied voltage produces mechanical vibration (ultrasound pulse)
- The crystal is excited by a short electrical pulse → vibrates at its resonant frequency → emits an ultrasound burst
B. Frequency Selection
| Vessel Type | Frequency | Axial Resolution | Penetration |
|---|---|---|---|
| Coronary arteries | 40–60 MHz | ~100 µm | ~6–10 mm |
| Peripheral arteries | 20–40 MHz | ~150–200 µm | ~10–20 mm |
| Aorta / large vessels | 10–20 MHz | ~300 µm | ~20–40 mm |
Higher frequency = finer resolution but shallower penetration. Coronary IVUS uses 40 MHz as an optimal trade-off.
C. Two Transducer Architectures
i. Mechanical (Single-Element Rotational) Transducer
Motor Drive Unit
↓
Flexible Torque Cable (driveshaft)
↓
Single Piezoelectric Element
(rotates at ~1,800 RPM)
↓
Ultrasound emitted radially as it rotates
→ 360° image assembled from sequential scan lines
- One scan line per pulse; full rotation = one cross-sectional frame
- Frame rate: ~30 frames/sec
- Acoustic mirror variant: crystal is fixed, mirror rotates (reduces size)
- Artifact — NURD (Non-Uniform Rotational Distortion): uneven rotation due to driveshaft friction in tortuous vessels causes image smearing/distortion
ii. Solid-State (Phased Array / Multi-Element) Transducer
64 piezoelectric elements arranged circumferentially
around catheter tip (no moving parts)
↓
Electronic multiplexer activates elements sequentially
↓
Synthetic aperture reconstruction → 360° image
- No mechanical rotation → no NURD artifact
- More flexible catheter profile
- Near-field artifact: ring-down artifact (reverberation near catheter surface)
- Frame rate: up to 30 frames/sec
3. Pullback Device (Motorized Pullback Unit)
Components
| Component | Function |
|---|---|
| Motor drive unit | Connects to catheter hub; rotates driveshaft (mechanical) or provides electrical connection (solid-state) |
| Motorized pullback rail | Withdraws catheter at precise speed |
| Pullback speed | 0.5 mm/sec (standard); 1.0 mm/sec (faster survey) |
| Pullback length | Up to 150 mm in one pass |
| Gating interface | ECG-gated pullback available to reduce cardiac motion artifact |
Why Controlled Pullback Matters
- Constant speed = known distance between frames → accurate volumetric measurements
- Enables 3D reconstruction of plaque volume and vessel geometry
- Manual pullback is unreliable for quantitative analysis
4. Imaging Console
Console Subsystems
┌──────────────────────────────────────┐
│ IVUS CONSOLE │
│ │
│ ┌─────────────┐ ┌───────────────┐ │
│ │ Pulser / │ │ Receiver / │ │
│ │ Transmitter│ │ Amplifier │ │
│ └─────────────┘ └───────────────┘ │
│ ↓ ↑ │
│ ┌───────────────────────────────┐ │
│ │ Time-Gain Compensation │ │
│ │ (TGC) │ │
│ └───────────────────────────────┘ │
│ ┌───────────────────────────────┐ │
│ │ Analog-to-Digital Converter │ │
│ │ (ADC) │ │
│ └───────────────────────────────┘ │
│ ┌───────────────────────────────┐ │
│ │ Digital Signal Processor │ │
│ │ (DSP) │ │
│ └───────────────────────────────┘ │
│ ┌───────────────────────────────┐ │
│ │ Scan Converter / Display │ │
│ └───────────────────────────────┘ │
└──────────────────────────────────────┘
Each Console Component Explained
a. Pulser / Transmitter
- Generates short, high-voltage electrical pulses (excitation pulses) sent to the piezoelectric crystal
- Pulse duration is very brief (~nanoseconds) to achieve short spatial pulse length → better axial resolution
b. Receiver / Pre-Amplifier
- Amplifies the weak returning echo signals before digitization
- Must handle a very wide dynamic range (strong near-field echoes vs. weak far-field echoes)
c. Time-Gain Compensation (TGC)
- Ultrasound attenuates as it travels deeper (deeper echoes are weaker)
- TGC applies progressively increasing amplification with depth to equalize image brightness across all depths
- Result: uniform grayscale appearance regardless of depth
d. Analog-to-Digital Converter (ADC)
- Converts analog echo waveforms → digital data
- Sampling rate: typically 200–500 MHz for 40 MHz IVUS
- Higher sampling rate → more precise depth resolution
e. Digital Signal Processor (DSP)
- Performs envelope detection (extracts amplitude of RF signal)
- Log compression — compresses wide dynamic range into displayable grayscale
- Scan conversion — converts polar coordinate data (radius, angle) to Cartesian (x, y) for display
- Image filtering and smoothing
f. ECG Module
- Synchronizes imaging with cardiac cycle
- Allows ECG-gated frame acquisition to reduce motion artifact from heartbeat
- Displays cardiac rhythm alongside IVUS images
5. Signal Processing & Display
Scan Conversion
Image Display Modes
| Mode | Description |
|---|---|
| 2D Cross-section | Real-time circular grayscale slice of vessel |
| Longitudinal view | Side-by-side long-axis reconstruction along pullback |
| 3D reconstruction | Volume rendering of vessel from complete pullback dataset |
| Color-flow overlay | Doppler color mapping of blood flow (some systems) |
| Virtual Histology (VH-IVUS) | Spectral analysis of RF signal backscatter to color-code plaque components |
Virtual Histology (VH-IVUS) — Advanced Signal Processing
| Color Code | Tissue Type |
|---|---|
| Green | Fibrous tissue |
| Yellow-green | Fibro-fatty tissue |
| Red | Necrotic core (lipid-rich) |
| White | Dense calcium |
Complete IVUS System — Integrated Overview
GUIDEWIRE → advanced into coronary artery
↓
IVUS CATHETER → delivered over guidewire to target lesion
↓
TRANSDUCER (40 MHz piezoelectric crystal)
→ emits ultrasound pulses radially (mechanical rotation or phased array)
↓
ECHOES return from vessel wall layers
↓
SIGNAL travels via cable to:
↓
CONSOLE:
Pulser → Receiver → TGC → ADC → DSP → Scan Converter
↓
DISPLAY: Real-time 360° cross-sectional grayscale image
↓
PULLBACK DEVICE: Withdraws catheter at 0.5 mm/sec
↓
LONGITUDINAL / 3D RECONSTRUCTION of full vessel segment
IVUS Catheter System — Clinical Illustration
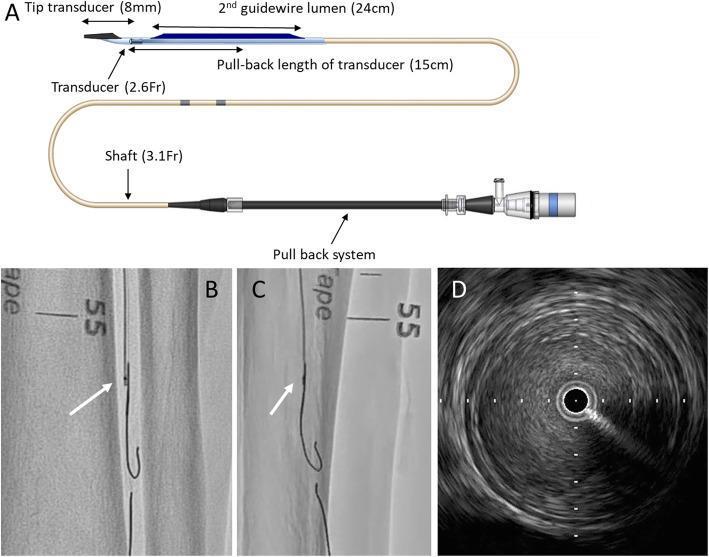
Summary Table — IVUS Instrumentation Components
| Component | Type | Key Specification |
|---|---|---|
| IVUS Catheter | Monorail / OTW | 2.6–3.5 Fr, 0.014" wire |
| Transducer | Mechanical / Solid-state | 20–60 MHz PZT crystal |
| Drive cable | Flexible torque shaft | 1,800 RPM (mechanical) |
| Pullback device | Motorized | 0.5–1.0 mm/sec |
| Pulser | High-voltage transmitter | Nanosecond pulse |
| Receiver | Pre-amplifier | Wide dynamic range |
| TGC | Depth compensation | Progressive gain |
| ADC | Digitizer | 200–500 MHz sampling |
| DSP | Signal processor | Envelope, compression, scan convert |
| Console display | Workstation | 2D, longitudinal, 3D, VH-IVUS |
Intravascular ultrasound instrumentation and explain each part in detail
Intravascular Ultrasound (IVUS) Instrumentation
Detailed explanation of each part
1) IVUS Catheter Assembly (intravascular hardware)
a) Distal soft tip
- Atraumatic flexible tip at the front
- Helps cross lesions safely and reduces vessel injury risk
b) Transducer housing (near distal tip)
- Contains the ultrasound transducer (single rotating element or array)
- Positioned close to lesion for high-quality intraluminal imaging
c) Shaft
- Thin torqueable polymer-braided shaft
- Balances pushability (to advance), trackability (through tortuous vessels), and kink resistance
d) Guidewire lumen
- Channel for guidewire compatibility (commonly 0.014" in coronary IVUS)
- May be rapid-exchange (monorail) or over-the-wire design
e) Proximal hub/connector
- Connects catheter to motor drive unit and console cable
- Provides electrical pathway and mechanical coupling
2) Ultrasound Transducer (core sensing element)
Types
A) Mechanical (rotational) IVUS
- One piezoelectric element rotates rapidly (about 1800 rpm)
- Produces 360° cross-sectional image line-by-line
- Pros: high image quality
- Limitation: rotational artifacts (NURD) if rotation becomes nonuniform
B) Solid-state (phased-array) IVUS
- Multiple fixed elements around tip, fired electronically
- No rotating shaft
- Pros: less rotational distortion, better mechanical reliability
- Limitation: different near-field artifact profile
Frequency role
- Typical coronary IVUS around 40 MHz (as described in Harrison’s, p. 6916)
- Higher frequency gives better resolution but shallower penetration
- Lower frequency gives deeper penetration but lower resolution
3) Drive/Interface Unit
a) Motor drive (for rotational systems)
- Spins the driveshaft or rotating transducer at constant speed
- Stable rotation is critical for circular geometry and accurate measurements
b) Electrical interface
- Sends transmit pulses to transducer
- Receives tiny echo signals and routes them to console receiver
4) Pullback Device
Function
- Withdraws catheter at a known constant speed (commonly 0.5 or 1.0 mm/s)
- Converts many cross-sectional frames into longitudinal vessel mapping
Why it matters
- Enables reproducible measurements:
- lesion length
- plaque volume
- stent segment analysis
- Manual pullback is less accurate for quantitative analysis
5) IVUS Console Electronics
a) Pulser/Transmitter
- Generates very short high-voltage electrical impulses
- Excites transducer to emit ultrasound pulses into vessel wall
b) Receiver/Pre-amplifier
- Captures weak returning echo signals
- Amplifies them while preserving signal fidelity
c) Time Gain Compensation (TGC)
- Compensates for depth-dependent attenuation
- Deep echoes are amplified more so image brightness is balanced
d) Analog-to-Digital Converter (ADC)
- Converts analog echo waveforms into digital data
- High sampling needed for precise axial localization
e) Digital Signal Processor (DSP)
- filtering
- envelope detection
- dynamic range/log compression
- artifact handling
- scan conversion support
6) Scan Conversion and Image Reconstruction
Display monitors use Cartesian coordinates (x-y).
Scan converter
- Transforms polar data into familiar circular cross-sectional image
Display outputs
- Real-time 2D vessel cross section
- Longitudinal “pullback” view
- Quantitative overlays (lumen and vessel contours, area values)
7) Software Measurement and Analysis Module
Core measurements
- Lumen CSA
- Vessel (EEM) CSA
- Plaque burden = (vessel area - lumen area) / vessel area
- Lesion length and stent expansion metrics
Clinical use
- Pre-PCI vessel sizing
- Post-stent optimization (underexpansion, malapposition, edge dissection)
- Plaque characterization support
8) Display, Recording, and Data Management
Includes
- High-resolution monitor
- Cine loop playback (frame-by-frame review)
- Procedure recording and export
- Integration with cath lab systems (DICOM/PACS in many setups)
9) Ancillary Components
- Sterile flush system (reduces air artifacts)
- ECG gating input (optional motion-reduced analysis)
- Footswitch/user controls for acquisition and pullback actions
End-to-end workflow (instrumentation in action)
- Guidewire crosses lesion
- IVUS catheter advanced distal to lesion
- Console sends pulses; transducer emits ultrasound
- Echoes return from lumen-intima-media-adventitia/plaque interfaces
- Receiver + DSP process signals into grayscale image
- Pullback device withdraws catheter at constant speed
- Software quantifies lumen/vessel/plaque and supports PCI decisions
Quick system block diagram
- a labeled exam-style IVUS instrumentation diagram, or
- a comparison table of IVUS vs OCT instrumentation, which is commonly asked in viva/practical exams.
Give me notes on ivus transducer based catheters, definition, components, and working
IVUS transducer catheter types mechanical rotational solid state phased array components working
IVUS catheter transducer mechanical rotational phased array design
IVUS Transducer-Based Catheters
Definition
Harrison's Principles of Internal Medicine (21st Ed., p. 6916): "IVUS is performed using a small flexible catheter with a 40-mHz transducer at its tip that is advanced into the coronary artery over a guidewire."
Classification of IVUS Transducer Catheters
IVUS Transducer Catheters
|
_____|______
| |
Mechanical Solid-State
(Rotational) (Phased Array)
|
|_______________
| |
Single-element Acoustic mirror
rotating (fixed crystal,
crystal rotating mirror)
TYPE 1 — Mechanical (Rotational) IVUS Catheter
Definition
Components
1. Distal Atraumatic Tip
- Soft, rounded, tapered polyurethane tip
- Positioned beyond the transducer
- Purpose: guides catheter across lesions without traumatizing vessel wall; allows smooth advancement through tight stenoses
2. Transducer Housing / Window
- A thin-walled acoustic window surrounding the rotating element
- Made of acoustically transparent material (minimal signal loss)
- Purpose: allows ultrasound to pass freely in and out while protecting the rotating inner assembly
3. Single Piezoelectric Crystal (Rotating Element)
- The actual sound-generating element
- Material: Lead Zirconate Titanate (PZT) or PVDF (polyvinylidene fluoride)
- Frequency: 40–45 MHz (coronary); 20 MHz (peripheral)
- Size: extremely small (fraction of a millimeter)
- Purpose: converts electrical pulses → ultrasound pulses (transmit); converts returning echoes → electrical signals (receive)
4. Acoustic Mirror Variant (optional design)
- In some designs, the crystal is fixed and a tiny rotating mirror redirects the beam laterally
- Allows even smaller catheter profile
- Eliminates issues from rotating electrical connections
5. Flexible Drive Cable (Torque Shaft)
- A stainless-steel coaxial cable running the full catheter length (~135–150 cm)
- Connects rotating transducer to the external motor drive unit
- Purpose: transmits rotational force from external motor to spinning transducer tip; also conducts electrical signals to/from transducer
6. Outer Catheter Shaft
- Multi-layer polymer construction (inner PTFE + braided reinforcement + outer nylon/polyurethane)
- Purpose: provides structural integrity, torque response, and kink resistance during navigation
7. Guidewire Lumen
- Inner channel for 0.014" guidewire
- Rapid-exchange (monorail) or over-the-wire configurations
- Purpose: tracks over pre-placed guidewire for safe coronary delivery
8. Flush Port / Saline Sheath
- Allows saline flushing around the rotating assembly
- Critical because air causes acoustic shadowing — saline displaces air and acts as an acoustic coupling medium between transducer and vessel wall
9. Proximal Connector Hub
- Interfaces catheter with:
- Motor drive unit (mechanical coupling for rotation)
- Electrical connector (signal transmission)
- Purpose: bridges disposable catheter to reusable console hardware
Working of Mechanical IVUS Catheter
STEP 1: Preparation
→ Catheter flushed with saline to remove air bubbles
→ Connected to motor drive unit at proximal hub
STEP 2: Delivery
→ Advanced through guiding catheter over 0.014" guidewire
→ Positioned distal to target lesion
STEP 3: Rotation
→ Motor drive unit rotates drive cable at ~1,800 RPM
→ Single piezoelectric element spins inside housing
STEP 4: Pulse Emission
→ Console sends electrical pulse to spinning transducer
→ Transducer vibrates at resonant frequency (40 MHz)
→ Short ultrasound burst emitted radially into vessel wall
STEP 5: Echo Reception
→ Ultrasound hits acoustic interfaces:
blood/intima → intima/media → media/adventitia → plaque
→ Partial echoes reflected back at each boundary
→ Transducer receives echoes, converts back to electrical signals
STEP 6: Image Line Formation
→ One transmitted pulse + received echoes = ONE radial scan line
→ Depth encoded by time-of-flight of each echo
STEP 7: 360° Frame Assembly
→ As transducer rotates, new scan lines acquired at each angular position
→ ~360 scan lines per revolution = one complete cross-sectional frame
→ Frame rate: ~30 frames/second
STEP 8: Pullback
→ Motorized pullback withdraws catheter at 0.5 mm/sec
→ Sequential frames stacked → longitudinal & 3D vessel reconstruction
Artifact — NURD (Non-Uniform Rotational Distortion)
| Feature | Detail |
|---|---|
| Cause | Uneven rotation of drive cable due to friction in tortuous vessels |
| Appearance | Smearing or compression of part of the image arc |
| Prevention | Minimize catheter bending; use appropriate guide catheter; newer low-friction designs |
TYPE 2 — Solid-State (Phased Array) IVUS Catheter
Definition
Components
1. Multi-Element Phased Array Transducer
- 64 individual piezoelectric elements arranged as a ring around the catheter circumference
- Each element is tiny (~50–100 µm wide)
- Elements fired sequentially by electronic multiplexer
- Purpose: each element contributes scan lines at different angles → full 360° coverage without any moving parts
2. Application-Specific Integrated Circuit (ASIC)
- Miniaturized integrated circuit embedded within the catheter tip
- Purpose: controls timing of element firing, multiplexing, and preliminary signal conditioning — essential because 64 signal wires cannot run the full catheter length
3. Flexible Printed Circuit Board (PCB)
- Thin flexible substrate connecting elements to ASIC and cable
- Purpose: routes electrical signals within extremely tight catheter dimensions
4. Catheter Shaft (No Drive Cable Needed)
- Simpler construction than mechanical type
- No rotating driveshaft — catheter is more flexible and trackable
- Multi-lumen design carries signal conductors
5. Guidewire Lumen
- Same as mechanical type (0.014" compatibility)
6. Proximal Electrical Connector
- Pure electrical interface (no mechanical coupling required)
- Connects to console for power supply and signal transfer
Working of Solid-State IVUS Catheter
STEP 1: Preparation & Delivery
→ No flushing required (no rotating assembly, no air trapping concern)
→ Advanced over guidewire to target site
STEP 2: Electronic Activation
→ Console sends firing sequence to ASIC in catheter tip
→ ASIC activates elements one-by-one (or in small groups)
around the 360° circumference
STEP 3: Pulse-Echo per Element
→ Each activated element emits an ultrasound pulse
→ Listens for returning echoes
→ One element = one angular scan line
STEP 4: Synthetic Aperture Processing
→ Signals from adjacent elements mathematically combined
→ Improves lateral resolution beyond what one element achieves
→ DSP in console performs this reconstruction
STEP 5: 360° Frame Assembly
→ All 64 elements complete one firing cycle = one full frame
→ Frame rate: ~30 frames/sec
STEP 6: Pullback & Reconstruction
→ Same motorized pullback process
→ Sequential frames build longitudinal/3D vessel map
Artifact — Ring-Down Artifact
| Feature | Detail |
|---|---|
| Cause | Reverberation of ultrasound within catheter body itself near elements |
| Appearance | Bright halo/ring immediately surrounding catheter in near-field |
| Effect | Creates a blind zone very close to catheter surface (~0.5–1 mm) |
| Mitigation | Software subtraction algorithms; accounted for in measurements |
Comparison Table — Mechanical vs Solid-State IVUS Catheter
| Feature | Mechanical (Rotational) | Solid-State (Phased Array) |
|---|---|---|
| Transducer elements | 1 rotating crystal | 64 fixed circumferential elements |
| Motion | Physical rotation (~1800 RPM) | No mechanical movement |
| Drive shaft | Required (flexible torque cable) | Not required |
| Frequency | 40–45 MHz | ~20 MHz |
| Axial resolution | ~100 µm (better) | ~150–200 µm |
| Catheter flexibility | Less flexible | More flexible |
| Primary artifact | NURD | Ring-down (near-field) |
| Saline flush | Required (air displacement) | Less critical |
| Image quality | Excellent near-field | Good; near-field limited |
| ASIC required | No | Yes (within catheter tip) |
| Example system | Boston Scientific iLab | Volcano/Philips Eagle Eye |
CMUT — Emerging Transducer Technology
- Fabricated using semiconductor microfabrication (MEMS technology)
- Integrated directly with ASICs on flexible PCB within catheter
- Frequency tunable by adjusting bias voltage (e.g., 8–20 MHz range from same device)
- Enables switchable modes: high-penetration vs high-resolution imaging
- Smaller profile, better bandwidth, and improved signal-to-noise ratio
Piezoelectric Transducer — Physical Principle (Core of all IVUS catheters)
TRANSMIT MODE:
Electrical pulse applied to crystal
↓
Crystal deforms mechanically (inverse piezoelectric effect)
↓
Rapid mechanical vibration at resonant frequency
↓
Ultrasound wave emitted into medium
RECEIVE MODE:
Returning echo strikes crystal
↓
Mechanical deformation of crystal
↓
Voltage generated across crystal faces (direct piezoelectric effect)
↓
Electrical signal sent to console receiver
f = v / 2t (where v = speed of sound in crystal, t = thickness)
- Thinner crystal → higher frequency → better resolution but more fragile
Key Technical Parameters of IVUS Transducer Catheters
| Parameter | Typical Value | Clinical Significance |
|---|---|---|
| Frequency | 20–60 MHz | Determines resolution vs penetration trade-off |
| Axial resolution | 100–200 µm | Minimum detectable layer thickness |
| Lateral resolution | 200–400 µm | Ability to separate adjacent structures side-by-side |
| Frame rate | 30 fps | Real-time imaging quality |
| Catheter size | 2.6–3.5 Fr | Deliverability in coronary arteries |
| Guidewire | 0.014" | Standard coronary compatibility |
| Pullback speed | 0.5–1.0 mm/sec | Determines frame spacing for 3D reconstruction |
| Field of view | ~10 mm radius | Adequate for coronary/peripheral imaging |
Summary — Key Points for Exam
- IVUS transducer catheters use the piezoelectric effect to generate and detect ultrasound from within the vessel
- Two main types: mechanical (single rotating element) and solid-state (64-element phased array)
- Mechanical type uses a rotating drive shaft → excellent image quality but NURD artifact
- Solid-state type uses electronic beam steering → no moving parts but ring-down artifact
- Coronary IVUS uses 40 MHz — optimal balance of resolution (~100 µm) and penetration (~10 mm)
- Saline flushing is essential in mechanical systems to eliminate air acoustic shadowing
- CMUT technology is the next-generation transducer using MEMS fabrication with tunable frequency
- The drive cable in mechanical systems transmits both rotation force and electrical signals simultaneously
- Pullback at 0.5 mm/sec allows volumetric plaque analysis and lesion length measurement
- All designs ultimately produce the same output: 360° grayscale cross-sectional images of the vessel wall
Give me notes on ivus transducer based catheters, definition, components, and working
IVUS transducer catheter piezoelectric crystal mechanical rotational solid state array detailed components
IVUS transducer catheter cross section components diagram rotational phased array
IVUS Transducer-Based Catheters
Complete Notes — Definition | Components | Working
SECTION 1 — DEFINITION
Harrison's Principles of Internal Medicine, 21st Ed. (p. 6916): "IVUS is performed using a small flexible catheter with a 40-mHz transducer at its tip that is advanced into the coronary artery over a guidewire... used to image atherosclerotic plaque precisely, determine luminal cross-sectional area, and measure vessel size."
Key defining features:
- Works from inside the vessel (intravascular)
- Uses high-frequency ultrasound (20–60 MHz)
- Produces tomographic (cross-sectional) images
- Both transmitter and receiver are the same element (pulse-echo mode)
- Provides anatomical truth — vessel wall layers, plaque burden, lumen geometry
SECTION 2 — CLASSIFICATION OF IVUS TRANSDUCER CATHETERS
IVUS Transducer-Based Catheters
|
_____________|_____________
| |
TYPE 1: MECHANICAL TYPE 2: SOLID-STATE
(Rotational) (Phased Array)
|
_____|_____
| |
Single Acoustic
rotating mirror
crystal design
SECTION 3 — TYPE 1: MECHANICAL (ROTATIONAL) IVUS CATHETER
3.1 Definition
3.2 Detailed Components
COMPONENT 1 — Distal Atraumatic Tip
| Feature | Detail |
|---|---|
| Material | Soft polyurethane or silicone polymer |
| Shape | Tapered, rounded, flexible |
| Length | 1–3 mm beyond transducer |
| Function | Prevents vessel wall injury during advancement; allows crossing of tight lesions; guides tracking through tortuous anatomy |
COMPONENT 2 — Acoustic Window / Transducer Housing
| Feature | Detail |
|---|---|
| Material | Acoustically transparent thin polymer membrane |
| Location | Surrounds rotating transducer element |
| Function | Protects rotating inner assembly from blood contact; allows ultrasound pulses to pass in and out with minimal attenuation; maintains sterility of inner components |
COMPONENT 3 — Piezoelectric Crystal (Single Rotating Element)
| Feature | Detail |
|---|---|
| Material | Lead Zirconate Titanate (PZT) or Polyvinylidene Fluoride (PVDF) |
| Size | Sub-millimeter (fraction of catheter tip) |
| Frequency | 40–45 MHz (coronary); 20 MHz (peripheral vessels) |
| Shape | Flat disc or curved (focused beam) |
| Thickness | Determines resonant frequency: f = v/2t |
TRANSMIT:
Electrical pulse applied to crystal
↓
Inverse piezoelectric effect
↓
Crystal mechanically vibrates at resonant frequency
↓
Ultrasound pulse emitted radially into vessel
RECEIVE:
Returning echo strikes crystal surface
↓
Direct piezoelectric effect
↓
Mechanical pressure → voltage generated
↓
Electrical signal sent to console
COMPONENT 4 — Flexible Drive Cable (Torque Shaft)
| Feature | Detail |
|---|---|
| Material | Multi-layer coaxial stainless steel cable |
| Length | Full catheter length (~135–150 cm) |
| Construction | Inner electrical conductors + outer torque-transmitting coil |
| Dual function | 1. Transmits rotational torque from motor to transducer; 2. Conducts electrical signals to and from crystal |
| Critical property | Must transmit rotation uniformly to avoid NURD artifact |
COMPONENT 5 — Outer Catheter Shaft
| Feature | Detail |
|---|---|
| Construction | Inner PTFE liner + braided polymer reinforcement + outer polyurethane |
| French size | 2.6 Fr – 3.5 Fr (coronary); up to 8.2 Fr (peripheral) |
| Properties needed | Pushability + trackability + torque response + kink resistance |
| Function | Structural backbone; allows catheter to navigate coronary anatomy while protecting drive cable inside |
COMPONENT 6 — Guidewire Lumen
| Feature | Detail |
|---|---|
| Wire size | 0.014 inch (coronary standard) |
| Designs | Rapid-exchange (monorail) — short distal rail only; Over-the-wire — lumen runs full length |
| Function | Allows catheter to track safely over pre-placed guidewire to target vessel segment |
| Monorail | Over-the-wire | |
|---|---|---|
| Guidewire lumen | Distal 20–30 cm only | Full catheter length |
| Exchange ease | Easier, faster | Requires longer wire |
| Use | Most coronary IVUS | Complex anatomy |
COMPONENT 7 — Saline Flush Port / Sheath
| Feature | Detail |
|---|---|
| Location | Near proximal hub |
| Fluid | Normal saline (0.9% NaCl) |
| Function | Displaces air from around rotating assembly; air has very high acoustic impedance difference → causes complete signal drop-out (shadowing) |
| Critical importance | Without flushing, image quality is severely degraded or lost entirely |
COMPONENT 8 — Proximal Connector Hub
| Feature | Detail |
|---|---|
| Type | Dual interface — mechanical + electrical |
| Mechanical | Couples drive cable to motor drive unit |
| Electrical | Routes crystal signals to/from console cable |
| Function | Bridges disposable single-use catheter to reusable motor drive and console hardware |
3.3 Working of Mechanical IVUS Catheter — Step by Step
STEP 1 — PREPARATION
→ Catheter purged with saline to eliminate all air
→ Proximal hub connected to motor drive unit
→ Motor drive unit connected to imaging console
STEP 2 — DELIVERY INTO VESSEL
→ Guiding catheter positioned at coronary ostium
→ Guidewire advanced past lesion
→ IVUS catheter tracked over guidewire
→ Positioned distal to target lesion/stenosis
STEP 3 — ROTATION INITIATED
→ Motor drive unit activates
→ Torque transmitted along full drive cable
→ Single piezoelectric crystal rotates at ~1,800 RPM
→ One complete rotation = one 360° sweep
STEP 4 — ULTRASOUND PULSE EMISSION
→ Console sends brief high-voltage electrical pulse to crystal
→ Crystal vibrates at 40 MHz resonant frequency
→ Very short ultrasound burst (~20 nanoseconds) emitted radially
→ Beam travels through saline → vessel wall tissue
STEP 5 — ECHO REFLECTION
→ Sound encounters acoustic impedance interfaces:
Blood / Intima boundary → partial echo reflected back
Intima / Media boundary → partial echo reflected back
Media / Adventitia boundary → partial echo reflected back
Plaque / Normal wall → partial echo reflected back
→ Each boundary produces echo with characteristic strength
STEP 6 — ECHO RECEPTION
→ Returning echoes strike crystal surface
→ Direct piezoelectric effect → voltage generated
→ Amplitude of voltage encodes echo strength (tissue type)
→ Time delay encodes depth: Distance = (Speed × Time) / 2
STEP 7 — ONE SCAN LINE FORMED
→ One pulse + received echoes = one radial scan line
→ Depth information along that angular direction captured
STEP 8 — 360° FRAME ASSEMBLY
→ Crystal continues rotating
→ New pulse fired at each angular position
→ ~360 scan lines assembled into one circular frame
→ Frame rate: approximately 30 frames per second
STEP 9 — MOTORIZED PULLBACK
→ Pullback device withdraws catheter at 0.5–1.0 mm/sec
→ Each frame represents a new axial position in vessel
→ Stacking frames → longitudinal vessel map
→ 3D reconstruction of plaque, lumen, vessel geometry
3.4 Acoustic Mirror Variant
- Crystal is fixed (does not rotate)
- A tiny rotating acoustic mirror redirects the beam laterally
- Eliminates need for rotating electrical contacts (reduces noise)
- Allows even smaller catheter tip profile
- Signal quality preserved; same 360° imaging principle
3.5 Major Artifact — NURD
| Feature | Detail |
|---|---|
| Cause | Friction on drive cable when catheter bends sharply in tortuous artery → uneven rotation speed |
| Appearance | Part of 360° image smeared, compressed, or stretched |
| Clinical impact | Can misrepresent plaque distribution and lumen geometry |
| Prevention | Minimize guide catheter angulation; ensure adequate catheter support; newer low-friction designs |
SECTION 4 — TYPE 2: SOLID-STATE (PHASED ARRAY) IVUS CATHETER
4.1 Definition
4.2 Detailed Components
COMPONENT 1 — Multi-Element Circumferential Array
| Feature | Detail |
|---|---|
| Number of elements | 64 (standard); up to 128 in advanced designs |
| Arrangement | Ring of elements around catheter circumference |
| Element size | ~50–100 µm each |
| Material | PZT or PVDF ceramic |
| Frequency | ~20 MHz (standard); higher in newer designs |
| Function | Each element independently transmits and receives at its angular position → collectively cover full 360° without rotation |
COMPONENT 2 — Application-Specific Integrated Circuit (ASIC)
| Feature | Detail |
|---|---|
| Location | Embedded within catheter tip (critical innovation) |
| Size | Extremely miniaturized chip |
| Function | Controls timing of element firing sequence; multiplexes 64 elements through limited conductors; performs initial signal conditioning at source |
| Why essential | Cannot run 64 individual signal wires for 150 cm catheter length — ASIC reduces wiring requirement dramatically |
COMPONENT 3 — Flexible Printed Circuit Board (PCB)
| Feature | Detail |
|---|---|
| Type | Thin-film flexible substrate |
| Function | Mechanically supports and electrically connects array elements to ASIC; routes signals within ultra-compact catheter tip dimensions |
| Significance | Enables integration of complex electronics in sub-3 Fr space |
COMPONENT 4 — Catheter Shaft (Simplified, No Drive Cable)
| Feature | Detail |
|---|---|
| Construction | Multi-lumen polymer with embedded signal conductors |
| Advantage | No rotating driveshaft → catheter is more flexible, more trackable in tortuous anatomy |
| Function | Carries power and signal lines from console to ASIC at tip |
COMPONENT 5 — Guidewire Lumen
- Same 0.014 inch compatibility as mechanical type
- Runs full length or as rapid-exchange design
COMPONENT 6 — Proximal Electrical Connector
| Feature | Detail |
|---|---|
| Type | Pure electrical interface only (no mechanical coupling) |
| Function | Connects to console for power delivery and bidirectional signal transfer |
| Simpler than mechanical | No driveshaft coupling required |
4.3 Working of Solid-State IVUS Catheter — Step by Step
STEP 1 — DELIVERY
→ No saline flush needed (no rotating assembly)
→ Catheter advanced over 0.014" guidewire to target site
STEP 2 — ELECTRONIC ACTIVATION
→ Console sends power and timing signals to ASIC in catheter tip
→ ASIC initializes firing sequence for 64 elements
STEP 3 — SEQUENTIAL ELEMENT FIRING
→ Element 1 fires: emits ultrasound pulse at 0°
→ Element 1 listens: receives echoes from 0° direction
→ Element 2 fires: emits at ~5.6° (360/64)
→ Element 2 listens: receives echoes from that direction
→ Continues through all 64 elements sequentially
STEP 4 — SYNTHETIC APERTURE PROCESSING
→ Signals from multiple adjacent elements combined mathematically
→ Virtual aperture larger than single element
→ Improves lateral resolution significantly
→ DSP in console performs this reconstruction algorithm
STEP 5 — 360° FRAME ASSEMBLY
→ All 64 elements complete one full firing cycle
→ 64 angular scan lines assembled into circular cross-section
→ Frame rate: ~30 frames per second
STEP 6 — PULLBACK AND 3D RECONSTRUCTION
→ Same motorized pullback at 0.5–1.0 mm/sec
→ Sequential frames produce longitudinal vessel map
→ 3D plaque and lumen reconstruction
4.4 Major Artifact — Ring-Down
| Feature | Detail |
|---|---|
| Cause | Reverberation of ultrasound within catheter body near elements |
| Appearance | Bright concentric ring halo immediately surrounding catheter on image |
| Effect | Creates near-field blind zone (~0.5–1 mm around catheter) |
| Mitigation | Software subtraction algorithms remove static ring-down pattern |
SECTION 5 — CMUT: NEXT-GENERATION TRANSDUCER TECHNOLOGY
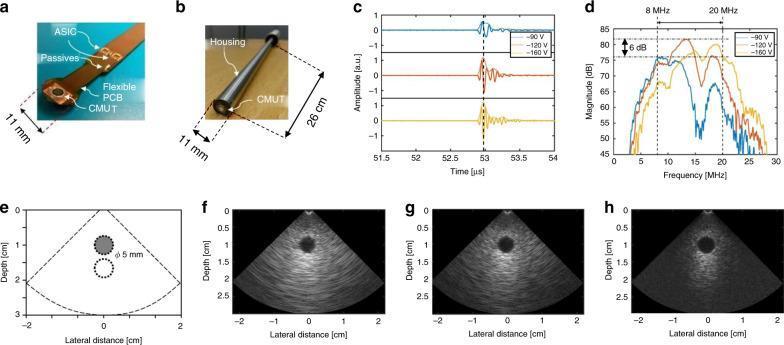
| Feature | CMUT | Conventional PZT |
|---|---|---|
| Fabrication | MEMS semiconductor microfabrication | Ceramic piezoelectric crystal |
| Operating principle | Capacitive membrane vibration | Piezoelectric deformation |
| Frequency tuning | Variable by bias voltage (8–20 MHz same device) | Fixed at manufacture |
| Integration with ASIC | Direct on-chip | Separate bonding |
| Bandwidth | Wider | Narrower |
| Sensitivity | Higher at high frequencies | Standard |
| Size | Smaller possible | Slightly larger |
SECTION 6 — COMPLETE COMPARISON TABLE
| Feature | Mechanical (Rotational) | Solid-State (Phased Array) | CMUT |
|---|---|---|---|
| Elements | 1 rotating crystal | 64 fixed elements | MEMS membrane array |
| Motion | Physical rotation 1800 RPM | None | None |
| Drive cable | Required | Not required | Not required |
| ASIC in tip | No | Yes | Yes (on-chip) |
| Frequency | 40–45 MHz | ~20 MHz | 8–20 MHz (tunable) |
| Axial resolution | ~100 µm (best) | ~150–200 µm | Comparable to PZT |
| Catheter flexibility | Less | More | Most flexible |
| Primary artifact | NURD | Ring-down | Under investigation |
| Saline flush | Mandatory | Not critical | Not critical |
| Image near-field | Excellent | Blind zone present | Improved |
SECTION 7 — KEY TECHNICAL PARAMETERS
| Parameter | Value | Clinical Importance |
|---|---|---|
| Frequency | 20–60 MHz | Resolution vs penetration trade-off |
| Axial resolution | 100–200 µm | Minimum detectable tissue layer thickness |
| Lateral resolution | 200–400 µm | Separating adjacent structures |
| Frame rate | 30 fps | Real-time imaging smoothness |
| Catheter size | 2.6–3.5 Fr | Deliverability without vessel trauma |
| Pullback speed | 0.5–1.0 mm/sec | Frame spacing for 3D accuracy |
| Field of view | ~10 mm radius | Adequate for coronary/peripheral imaging |
| Guidewire | 0.014 inch | Standard coronary compatibility |
SECTION 8 — SUMMARY (EXAM POINTS)
| # | Key Point |
|---|---|
| 1 | IVUS catheter carries a piezoelectric transducer inside the vessel lumen |
| 2 | Piezoelectric effect — mechanical ↔ electrical energy conversion — is the core principle |
| 3 | Two main designs: mechanical (single rotating element) and solid-state (64-element array) |
| 4 | Mechanical design needs a torque drive cable; solid-state needs an ASIC chip at tip |
| 5 | Coronary IVUS uses 40 MHz for ~100 µm axial resolution with ~10 mm penetration |
| 6 | Mechanical systems require mandatory saline flush to remove air artifacts |
| 7 | NURD artifact is specific to mechanical; ring-down artifact is specific to solid-state |
| 8 | Motorized pullback at 0.5 mm/sec converts 2D frames into 3D vessel reconstruction |
| 9 | CMUT technology uses MEMS fabrication with bias-voltage-tunable frequency |
| 10 | Both designs ultimately produce the same clinical output — 360° grayscale vessel cross-sections |
Give me notes on ivus transducer based catheters, definition, components, and working
IVUS transducer based catheter definition components working principle piezoelectric
IVUS catheter transducer components working diagram
IVUS Transducer-Based Catheters
Definition | Components | Working
1. DEFINITION
Harrison's Principles of Internal Medicine, 21st Ed. (p. 6916): "IVUS is performed using a small flexible catheter with a 40-mHz transducer at its tip that is advanced into the coronary artery over a guidewire... used to image atherosclerotic plaque precisely, determine luminal cross-sectional area, and measure vessel size."
Core concept in one line:
The catheter brings the ultrasound source inside the vessel — imaging outward through the wall, rather than inward from the body surface.
2. TYPES OF IVUS TRANSDUCER-BASED CATHETERS
IVUS Transducer-Based Catheters
|
____________|____________
| |
TYPE 1 TYPE 2
MECHANICAL SOLID-STATE
(Rotational) (Phased Array)
|
___|___
| |
Single Acoustic
crystal mirror
rotating variant
3. TYPE 1 — MECHANICAL (ROTATIONAL) IVUS CATHETER
Definition
Components
A. Distal Atraumatic Tip
[ soft tapered polymer tip ]──► guides catheter across lesions
| Feature | Detail |
|---|---|
| Material | Soft polyurethane / silicone |
| Shape | Tapered, rounded, flexible |
| Length | 1–3 mm beyond transducer |
| Purpose | Prevents vessel wall trauma; allows crossing of tight stenoses; enables smooth tracking through tortuous coronary anatomy |
B. Acoustic Window / Transducer Housing
| Feature | Detail |
|---|---|
| Material | Acoustically transparent thin polymer membrane |
| Location | Surrounds the rotating transducer element |
| Purpose | Protects rotating assembly from blood; allows ultrasound pulses to pass freely in and out with minimal signal loss |
C. Piezoelectric Crystal — THE CORE ELEMENT
| Feature | Detail |
|---|---|
| Material | Lead Zirconate Titanate (PZT) or Polyvinylidene Fluoride (PVDF) |
| Size | Sub-millimeter |
| Frequency | 40–45 MHz (coronary); 20 MHz (peripheral) |
| Shape | Flat disc or curved (focused beam design) |
| Thickness | Determines resonant frequency — f = v / 2t (thinner = higher frequency) |
TRANSMIT MODE
─────────────
Electrical pulse → applied to crystal faces
↓
Inverse piezoelectric effect
↓
Crystal mechanically vibrates at resonant frequency
↓
Ultrasound pulse emitted radially into vessel wall
RECEIVE MODE
────────────
Returning echo strikes crystal surface
↓
Direct piezoelectric effect
↓
Mechanical pressure → voltage generated across crystal
↓
Electrical signal sent back to console receiver
D. Flexible Drive Cable (Torque Shaft)
[Motor drive unit] ──torque──► [Drive cable running 150 cm] ──► [Rotating crystal at tip]
◄──signal── ◄──
| Feature | Detail |
|---|---|
| Material | Multi-layer coaxial stainless steel cable |
| Length | Full catheter length (~135–150 cm) |
| Construction | Inner electrical conductors + outer torque-transmitting coil layers |
| Dual function | 1. Transmits rotational torque from motor to spinning crystal; 2. Conducts electrical signals to and from the transducer |
| Critical property | Must rotate uniformly — any uneven rotation causes NURD artifact |
E. Outer Catheter Shaft
| Feature | Detail |
|---|---|
| Construction | Inner PTFE liner + braided polymer reinforcement + outer nylon/polyurethane |
| French size | 2.6–3.5 Fr (coronary); up to 8.2 Fr (peripheral) |
| Required properties | Pushability + Trackability + Torque response + Kink resistance |
| Function | Structural backbone that protects the drive cable inside while navigating coronary anatomy |
F. Guidewire Lumen
| Feature | Detail |
|---|---|
| Wire compatibility | 0.014 inch (coronary standard) |
| Design options | Rapid-exchange (monorail) or Over-the-wire |
| Function | Tracks catheter safely over a pre-placed guidewire to target vessel segment |
| Rapid-Exchange | Over-the-Wire | |
|---|---|---|
| Lumen extent | Distal 20–30 cm only | Full catheter length |
| Exchange | Fast, single operator | Needs longer wire |
| Common use | Most coronary IVUS | Complex anatomy |
G. Saline Flush Port
| Feature | Detail |
|---|---|
| Fluid | Normal saline (0.9% NaCl) |
| Location | Near proximal hub |
| Function | Displaces air from around rotating transducer assembly |
| Why critical | Air has extremely high acoustic impedance → causes complete signal dropout and shadowing artifacts. Saline acts as acoustic coupling medium between crystal and vessel wall |
H. Proximal Connector Hub
| Feature | Detail |
|---|---|
| Type | Dual — mechanical + electrical interface |
| Mechanical side | Couples drive cable to motor unit |
| Electrical side | Routes crystal signals to console |
| Function | Bridges the single-use disposable catheter to the reusable motor drive and imaging console |
Working — Mechanical IVUS Catheter (Step by Step)
STEP 1 ── PREPARATION
Catheter flushed with saline → all air removed
Proximal hub connected to motor drive unit + console
STEP 2 ── DELIVERY
Guiding catheter seated at coronary ostium
Guidewire advanced past the target lesion
IVUS catheter tracked over guidewire
Tip positioned distal to lesion
STEP 3 ── ROTATION BEGINS
Motor drive unit activates
Torque transmitted along full drive cable (~150 cm)
Single piezoelectric crystal spins at ~1,800 RPM
STEP 4 ── PULSE EMISSION
Console sends brief high-voltage electrical pulse to crystal
Crystal vibrates at 40 MHz resonant frequency
Short ultrasound burst (~20 nanoseconds) emitted radially
Beam travels: saline → vessel wall tissue layers
STEP 5 ── ECHO REFLECTION
Sound hits acoustic impedance boundaries:
Blood / Intima → partial echo reflected
Intima / Media → partial echo reflected
Media / Adventitia → partial echo reflected
Plaque / Normal wall → partial echo reflected
Each boundary returns echo of characteristic amplitude
STEP 6 ── ECHO RECEPTION
Returning echoes strike crystal surface
Direct piezoelectric effect → voltage proportional to echo amplitude
Time delay of each echo encodes depth:
Distance = (Speed of sound × Time) / 2
STEP 7 ── ONE SCAN LINE FORMED
One transmitted pulse + all received echoes
= one radial scan line at that angular position
STEP 8 ── 360° FRAME ASSEMBLY
Crystal continues rotating
New pulse fired at each angular step
~360 scan lines assembled → one complete circular frame
Frame rate: ~30 frames per second
STEP 9 ── MOTORIZED PULLBACK
Pullback device withdraws catheter at 0.5–1.0 mm/sec
Each frame represents a new axial slice of vessel
Stacked frames → longitudinal map + 3D reconstruction
NURD Artifact — Mechanical Catheter
| Feature | Detail |
|---|---|
| Full name | Non-Uniform Rotational Distortion |
| Cause | Friction on drive cable when catheter bends sharply in tortuous artery → uneven rotation speed |
| Appearance | Part of 360° image smeared, compressed, or geometrically distorted |
| Prevention | Minimize sharp bends; use proper guide catheter support; newer low-friction drive cable designs |
Acoustic Mirror Variant
- Crystal is fixed — does not rotate
- A tiny rotating acoustic mirror redirects the beam radially
- Eliminates rotating electrical contacts → reduces electrical noise
- Allows even smaller catheter tip profile
- Same 360° imaging principle achieved
4. TYPE 2 — SOLID-STATE (PHASED ARRAY) IVUS CATHETER
Definition
Components
A. Multi-Element Circumferential Array
| Feature | Detail |
|---|---|
| Number | 64 elements (up to 128 in advanced designs) |
| Arrangement | Ring of elements around catheter circumference |
| Individual element size | ~50–100 µm |
| Material | PZT or PVDF ceramic |
| Frequency | ~20 MHz standard |
| Function | Each element fires at its angular position → collectively cover full 360° without any rotation |
B. Application-Specific Integrated Circuit (ASIC)
| Feature | Detail |
|---|---|
| Location | Embedded within the catheter tip (critical design innovation) |
| Function | Controls timing of element firing sequence; multiplexes 64 elements through limited wire conductors; performs initial signal conditioning at source |
| Why essential | Cannot physically run 64 individual signal wires for 150 cm of catheter — ASIC drastically reduces wiring requirement at source |
C. Flexible Printed Circuit Board (PCB)
| Feature | Detail |
|---|---|
| Type | Thin-film flexible substrate |
| Function | Mechanically supports array elements; electrically connects elements to ASIC; routes all signals within ultra-compact sub-3 Fr space |
D. Simplified Catheter Shaft (No Drive Cable)
| Feature | Detail |
|---|---|
| Construction | Multi-lumen polymer with embedded signal conductors |
| Advantage over mechanical | No rotating driveshaft → catheter is more flexible and trackable in tortuous vessels |
| Function | Carries power and bidirectional signals between console and ASIC at tip |
E. Proximal Electrical Connector
| Feature | Detail |
|---|---|
| Type | Pure electrical interface — no mechanical coupling needed |
| Function | Connects to console for power delivery and signal transfer |
| Simpler | No driveshaft coupling required compared to mechanical type |
Working — Solid-State IVUS Catheter (Step by Step)
STEP 1 ── DELIVERY
No saline flush required (no rotating assembly)
Catheter advanced over 0.014" guidewire to target site
STEP 2 ── ELECTRONIC ACTIVATION
Console sends power + timing signals to ASIC in catheter tip
ASIC initializes firing sequence for all 64 elements
STEP 3 ── SEQUENTIAL ELEMENT FIRING
Element 1 fires → emits pulse at 0°
Element 1 listens → receives echoes from 0° direction
Element 2 fires → emits pulse at ~5.6° (360°/64)
Element 2 listens → receives echoes
Continues sequentially through all 64 elements
STEP 4 ── SYNTHETIC APERTURE PROCESSING
Signals from multiple adjacent elements combined mathematically
Creates virtual aperture larger than any single element
Significantly improves lateral resolution
DSP in console performs reconstruction algorithm
STEP 5 ── 360° FRAME ASSEMBLY
All 64 elements complete one full firing cycle
64 angular scan lines assembled into one circular frame
Frame rate: ~30 frames per second
STEP 6 ── PULLBACK AND RECONSTRUCTION
Same motorized pullback at 0.5–1.0 mm/sec
Sequential cross-sectional frames build longitudinal vessel map
3D reconstruction of plaque, lumen, and vessel geometry
Ring-Down Artifact — Solid-State Catheter
| Feature | Detail |
|---|---|
| Cause | Reverberation of ultrasound energy within catheter body near elements |
| Appearance | Bright concentric halo ring immediately surrounding catheter in image |
| Effect | Creates near-field blind zone (~0.5–1 mm immediately around catheter) |
| Solution | Software subtraction algorithms remove the static ring-down pattern |
5. HEAD-TO-HEAD COMPARISON
| Feature | Mechanical | Solid-State |
|---|---|---|
| Transducer elements | 1 rotating crystal | 64 fixed elements |
| Physical motion | Yes (~1,800 RPM) | None |
| Drive cable | Required | Not required |
| ASIC in tip | No | Yes |
| Frequency | 40–45 MHz | ~20 MHz |
| Axial resolution | ~100 µm (better) | ~150–200 µm |
| Catheter flexibility | Less flexible | More flexible |
| Primary artifact | NURD | Ring-down |
| Saline flush | Mandatory | Not critical |
| Near-field imaging | Excellent | Blind zone present |
| Example system | Boston Scientific iLab | Volcano Eagle Eye |
6. TECHNICAL PARAMETERS — QUICK REFERENCE
| Parameter | Value | Significance |
|---|---|---|
| Frequency | 20–60 MHz | Resolution vs penetration balance |
| Axial resolution | 100–200 µm | Minimum detectable layer thickness |
| Lateral resolution | 200–400 µm | Separating adjacent structures |
| Frame rate | ~30 fps | Real-time image smoothness |
| Catheter size | 2.6–3.5 Fr | Safe coronary deliverability |
| Guidewire | 0.014 inch | Standard coronary compatibility |
| Pullback speed | 0.5–1.0 mm/sec | Frame spacing for 3D accuracy |
| Field of view | ~10 mm radius | Covers coronary/peripheral vessel wall |
7. VESSEL LAYERS VISUALIZED BY IVUS CATHETER
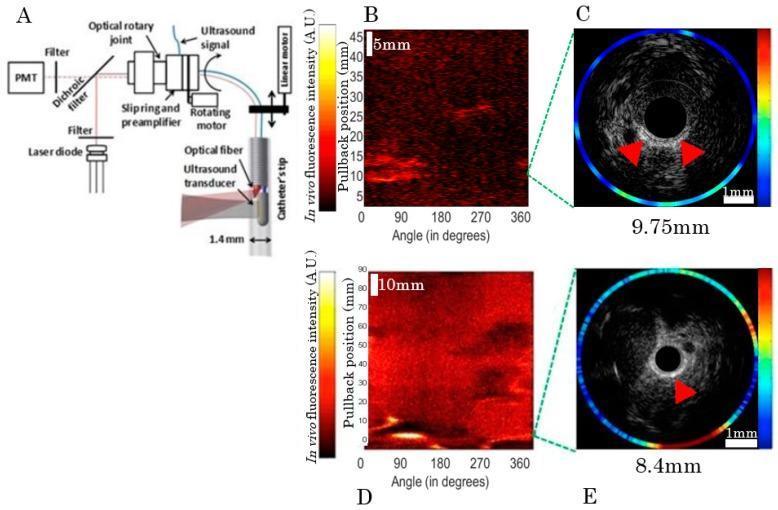
| Layer | IVUS Appearance |
|---|---|
| Catheter itself | Central dark circle |
| Blood / Lumen | Hypoechoic (dark) |
| Intima | Bright (echogenic) thin inner ring |
| Media | Hypoechoic (dark) middle band |
| Adventitia | Bright (hyperechoic) outer layer |
| Fibrous plaque | Echogenic (bright) |
| Lipid plaque | Hypoechoic (dark) |
| Calcified plaque | Very bright with acoustic shadow behind |
8. SUMMARY — EXAM POINTS
| # | Key Point |
|---|---|
| 1 | IVUS catheter carries piezoelectric transducer inside the vessel |
| 2 | Piezoelectric effect converts electrical ↔ mechanical energy — core operating principle |
| 3 | Two main types: mechanical (single rotating element) and solid-state (64-element array) |
| 4 | Mechanical needs torque drive cable; solid-state needs ASIC chip at tip |
| 5 | Coronary IVUS operates at 40 MHz → axial resolution ~100 µm, penetration ~10 mm |
| 6 | Mechanical systems require mandatory saline flush to eliminate air shadowing |
| 7 | NURD = artifact of mechanical; Ring-down = artifact of solid-state |
| 8 | Motorized pullback at 0.5 mm/sec converts 2D cross-sections into 3D vessel reconstruction |
| 9 | Both designs produce the same output — 360° grayscale cross-sectional vessel images |
| 10 | Frequency formula: f = v/2t — thinner crystal = higher frequency = better resolution |