Placental development. The role of the chorion in the formation of the placenta. The structure and role of the hematoplacental barrier
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have all the material needed for a comprehensive, well-illustrated answer. Here is the full synthesis:
Placental Development, the Role of the Chorion, and the Hematoplacental Barrier
1. Overview: The Placenta as a Fetomaternal Organ
The placenta is a fetomaternal organ with two distinct components:
- A fetal part — derived from the chorionic sac (outermost fetal membrane)
- A maternal part — derived from the decidua basalis (endometrium of the pregnant uterus)
Its functions include: protection, nutrition, respiration, excretion of waste products, hormone production, and metabolism.
— The Developing Human: Clinically Oriented Embryology, p. 336
2. The Decidua (Maternal Contribution)
The decidua is the functional layer of the pregnant endometrium, named for its regions relative to the implantation site:
| Region | Location | Role |
|---|---|---|
| Decidua basalis | Deep to the conceptus | Forms the maternal part of the placenta |
| Decidua capsularis | Overlying the conceptus | Thin capsule; eventually fuses with decidua parietalis |
| Decidua parietalis | Remaining uterine lining | No direct placental role |
Rising progesterone levels cause endometrial connective tissue cells to differentiate into decidual cells — enlarged cells loaded with glycogen and lipids — via the decidual reaction.
3. Role of the Chorion in Placental Formation
The chorion — the outermost fetal membrane — is the primary fetal contributor to the placenta. Its role unfolds through the stepwise development of chorionic villi.
3.1 Establishment of the Uteroplacental Circulation (~Day 9)
After implantation, the syncytiotrophoblast erodes the endometrium, creating fluid-filled cavities called trophoblastic lacunae. Maternal sinusoids (derived from capillaries) anastomose with these lacunae, establishing a pressure gradient that drives directional blood flow — the primitive uteroplacental circulation. Pinocytotic vesicles in the syncytiotrophoblast begin transferring nutrients to the embryo.
3.2 Primary Chorionic Villi (Days 11–13)
The cytotrophoblast proliferates rapidly and sends cellular cords into the blood-filled trophoblastic lacunae within the syncytiotrophoblast. These finger-like projections are primary chorionic villi — composed entirely of an inner cytotrophoblast core and an outer syncytiotrophoblast layer.
3.3 Secondary Chorionic Villi (~Day 16)
Loose connective tissue (chorionic mesenchyme) invades the cytotrophoblast core of the primary villi, converting them into secondary chorionic villi. These consist of:
- Outer layer: syncytiotrophoblast
- Middle layer: cytotrophoblast
- Core: extraembryonic mesenchyme
Secondary villi cover the entire surface of the chorionic sac at this stage.
3.4 Tertiary Chorionic Villi (End of Week 3)
Blood vessels differentiate within the mesenchymal cores of the secondary villi — vascularizing them and producing tertiary chorionic villi. By ~Day 21, fetal blood begins circulating through these vessels and the embryonic cardiovascular system. This establishes the definitive fetoplacental circulation.
Simultaneously, cytotrophoblastic cells grow out through the syncytiotrophoblast to form the cytotrophoblastic shell — anchoring the conceptus to the decidua basalis.
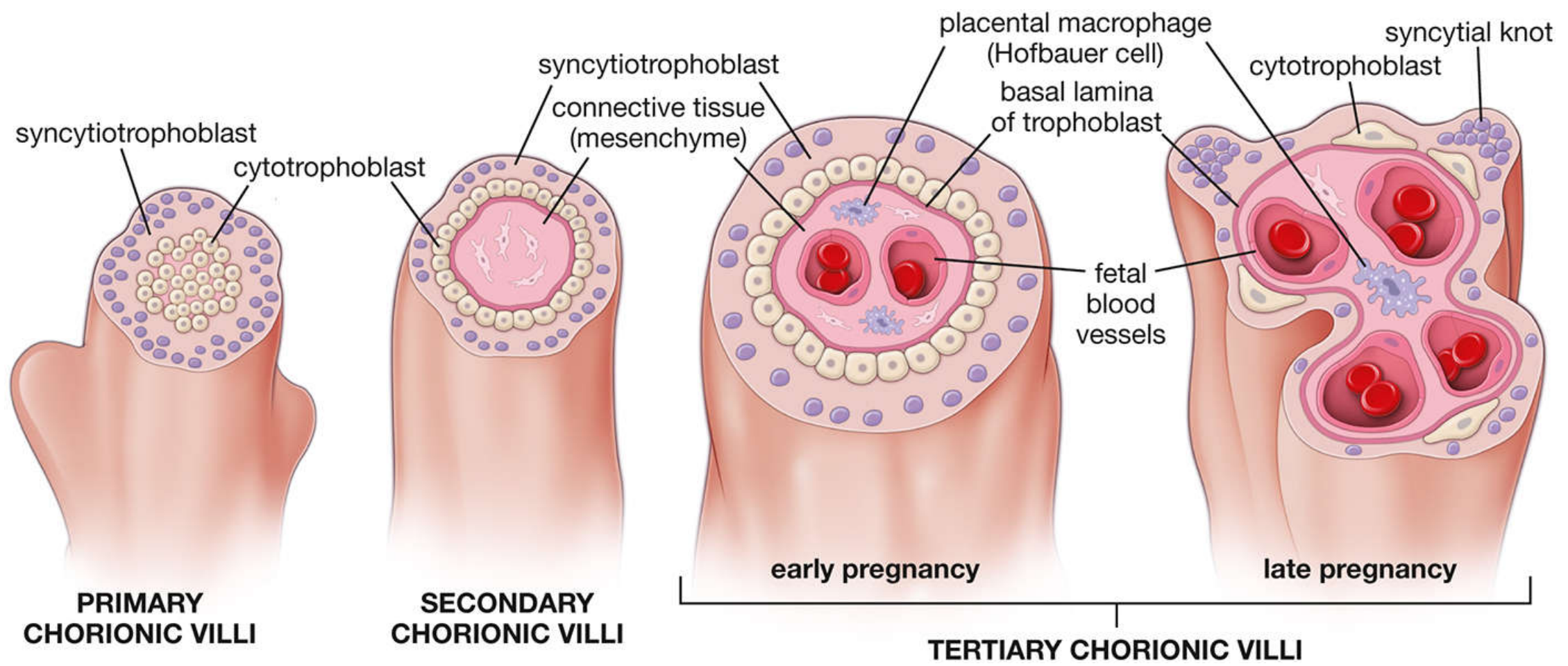
Fig. 23.26 — Histology: A Text and Atlas, p. 2262
3.5 Chorion Frondosum vs. Chorion Laeve
During the first 8 weeks, villi cover the entire chorionic surface. As growth proceeds:
- Villi adjacent to the decidua capsularis degenerate → chorion laeve (smooth chorion; avascular)
- Villi adjacent to the decidua basalis proliferate massively → chorion frondosum (villous chorion; the fetal component of the placenta)
The chorionic plate is the layer from which villi project into the intervillous space.
3.6 Cotyledon Formation (~Months 4–5)
As the chorion frondosum expands rapidly, wedge-shaped projections of the decidua basalis — placental septa — grow toward the chorionic plate, dividing the placenta into 15–25 cotyledons. Each cotyledon contains 2 or more main stem villi and many branching villi. The septa do not fuse with the chorionic plate, allowing free maternal blood circulation between cotyledons.
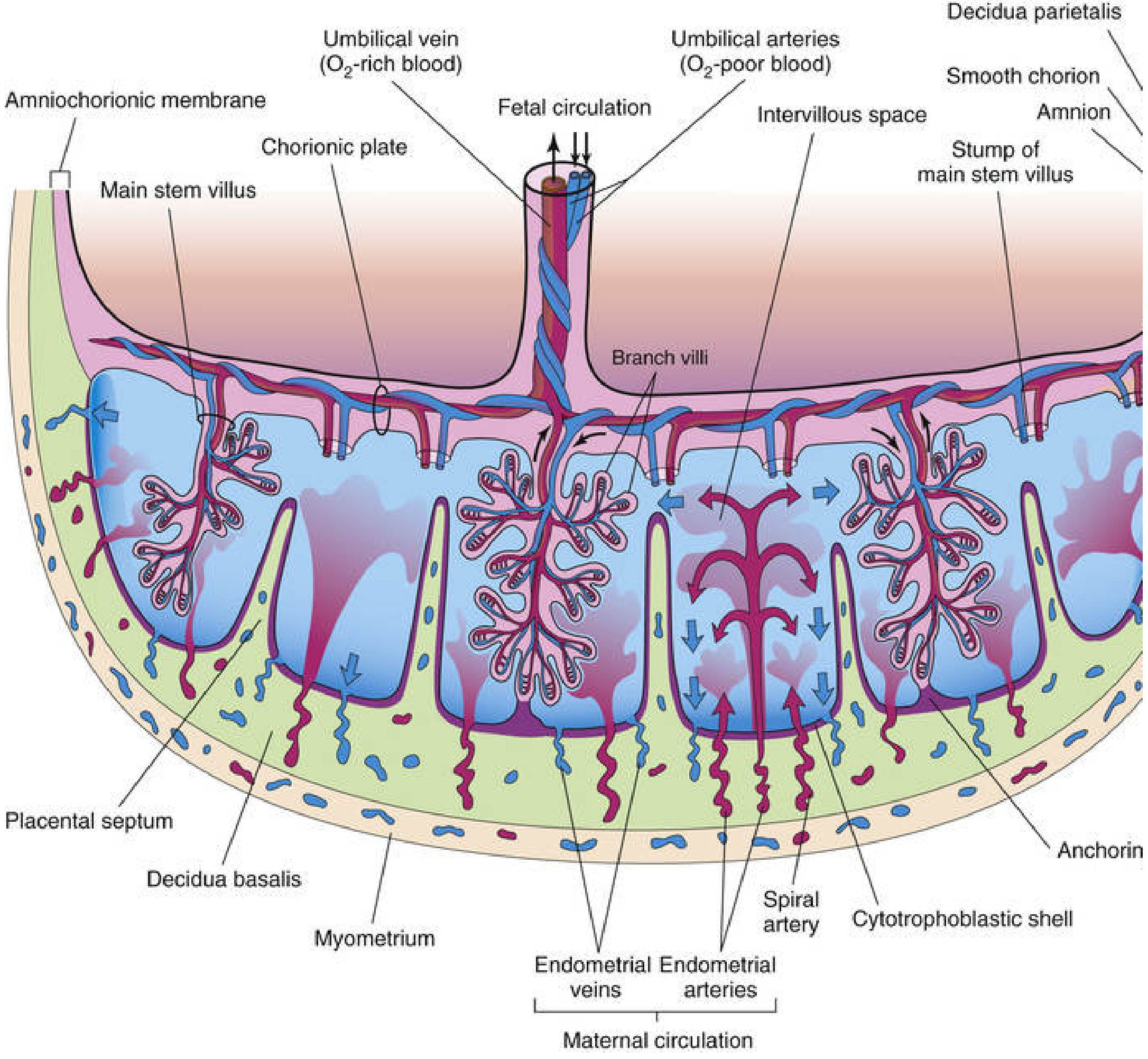
Fig. 7.5 — The Developing Human, p. 346
4. Placental Circulation
Fetal Circulation
Deoxygenated blood travels from the fetus via two umbilical arteries → chorionic arteries in the chorionic plate → capillary networks within branch villi. Oxygenated blood returns via the umbilical vein.
Maternal Circulation
80–100 spiral endometrial arteries in the decidua basalis discharge maternal blood in pulsatile spurts into the intervillous space (through gaps in the cytotrophoblastic shell). Blood flows over the branch villi (exchange occurs), then drains through endometrial veins back to the maternal circulation. The intervillous space holds ~150 mL of blood at term, replenished 3–4 times per minute.
Under normal conditions, fetal and maternal blood do not mix.
5. The Hematoplacental Barrier (Placental Membrane)
The hematoplacental barrier (also called the placental membrane) is the composite of fetal tissues that separates maternal blood in the intervillous space from fetal blood in the villous capillaries.
5.1 Structure: Early Pregnancy (up to ~20 weeks)
The barrier consists of 4 layers (from maternal to fetal side):
| Layer | Description |
|---|---|
| 1. Syncytiotrophoblast | Multinucleated, continuous outer layer; covered by microvilli on its maternal surface to maximize exchange area |
| 2. Cytotrophoblast | Inner, continuous cellular layer |
| 3. Villous connective tissue | Mesenchymal stroma containing Hofbauer cells (fetal macrophages) |
| 4. Fetal capillary endothelium | Endothelium of fetal placental capillaries |
5.2 Structure: After 20 Weeks — Thinning of the Barrier
The cytotrophoblast layer becomes progressively attenuated and discontinuous, leaving only thin patches of syncytiotrophoblast. The barrier is reduced to 3 effective layers in most regions:
- Syncytiotrophoblast
- Connective tissue of the villus
- Fetal capillary endothelium
In the thinnest areas, the syncytiotrophoblast comes into direct contact with fetal capillary endothelium, forming the vasosyncytial placental membrane — maximally efficient for gas exchange.
The thinnest barrier at full term consists of:
- Syncytiotrophoblast (+ trophoblast basal lamina)
- Thin/discontinuous cytotrophoblast
- Connective tissue of the villus
- Endothelial basal lamina
- Fetal capillary endothelium
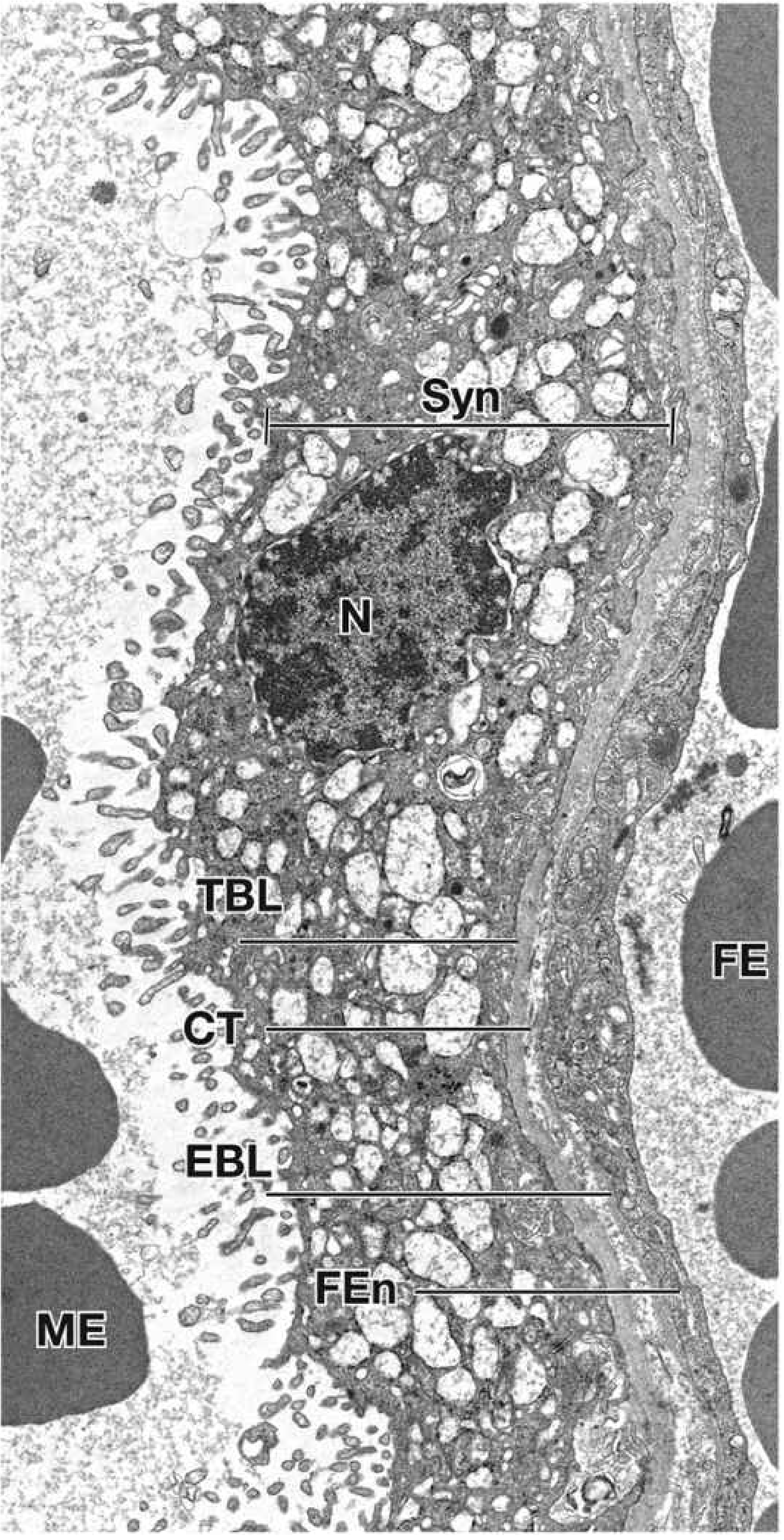
Fig. 23.28 — Histology: A Text and Atlas, p. 2267–2268
5.3 Hofbauer Cells
Resident fetal placental macrophages (Hofbauer cells) in the villous stroma are antigen-presenting cells of fetal origin. They proliferate in response to antigens, upregulate MHC II receptors, and are involved in placental innate immunity. They are more numerous early in pregnancy. HIV is primarily localized within these cells and the syncytiotrophoblast in infected placentas.
5.4 Syncytial Knots
In the third trimester, syncytiotrophoblast nuclei aggregate to form multinucleated protrusions — syncytial knots — which regularly break off into the maternal circulation and are destroyed by pulmonary enzymes. Their number increases with gestational age and is elevated in pathological conditions such as uteroplacental malperfusion.
6. Functions of the Hematoplacental Barrier
The barrier is not a true barrier in the restrictive sense — most substances cross it. Its functional selectivity depends on the size, configuration, and charge of molecules.
| Substance | Mechanism of Transfer |
|---|---|
| O₂, CO₂, CO | Simple diffusion |
| Water | Simple diffusion (increasing with advancing pregnancy) |
| Glucose | Facilitated diffusion (GLUT-1 transporter) |
| Amino acids | Active transport (fetal concentration > maternal) |
| Free fatty acids | Passive transfer (long-chain PUFAs predominate) |
| Vitamins | Crossing (water-soluble faster than fat-soluble) |
| IgG | Transcytosis (begins ~16 weeks; peaks ~26 weeks; confers passive immunity) |
| Steroid hormones | Freely cross (testosterone → fetal masculinization risk) |
| Protein hormones (insulin, pituitary) | Do not cross in significant amounts |
| Heparin | Does not cross (size/charge restriction) |
| Drugs and toxins | Most cross (clinical significance: teratogenesis) |
Eosinophilic fibrinoid material accumulates on villus surfaces toward the end of pregnancy, progressively reducing placental transfer capacity.
Summary
The chorion is the structural engine of placental formation. Through successive villous generations — primary → secondary → tertiary — and the differential growth of the chorion frondosum over the decidua basalis, the fetal component of the placenta is built. The hematoplacental barrier, formed within the walls of the tertiary villi, separates maternal and fetal circulations while enabling selective exchange. It begins as a 4-layer structure and thins progressively to maximize efficiency at term, functioning analogously to the air-blood barrier of the lung.
— The Developing Human: Clinically Oriented Embryology, pp. 335–357; Histology: A Text and Atlas with Correlated Cell and Molecular Biology, pp. 2261–2270
This is a shared conversation. Sign in to Orris to start your own chat.