NorAdrenaline in detail it's pathway mechanisms in body and why noradrenaline used in emergency drugs and where should we used and why in detail way explain clinical importance in detail and its criteria and protocols for emergency
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching PubMed
norepinephrine vasopressor septic shock
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Searching Images
norepinephrine synthesis pathway catecholamine tyrosine
Loading Image
Loading Image
Reading File
Reading File
I now have comprehensive data from multiple authoritative textbooks. Let me compose the full detailed answer.
Noradrenaline (Norepinephrine) — A Comprehensive Reference
1. Chemistry & Classification
Noradrenaline (NA; norepinephrine, NE) is an endogenous catecholamine — a molecule bearing a catechol ring (3,4-dihydroxybenzene) plus an ethylamine side chain. It is both a neurotransmitter (postganglionic sympathetic, locus coeruleus) and a hormone (adrenal medulla). Its chemical formula is C₈H₁₁NO₃; it lacks the N-methyl group that distinguishes adrenaline (epinephrine).
2. Biosynthesis Pathway
The biosynthetic route is the classic catecholamine cascade, starting from the amino acid tyrosine:
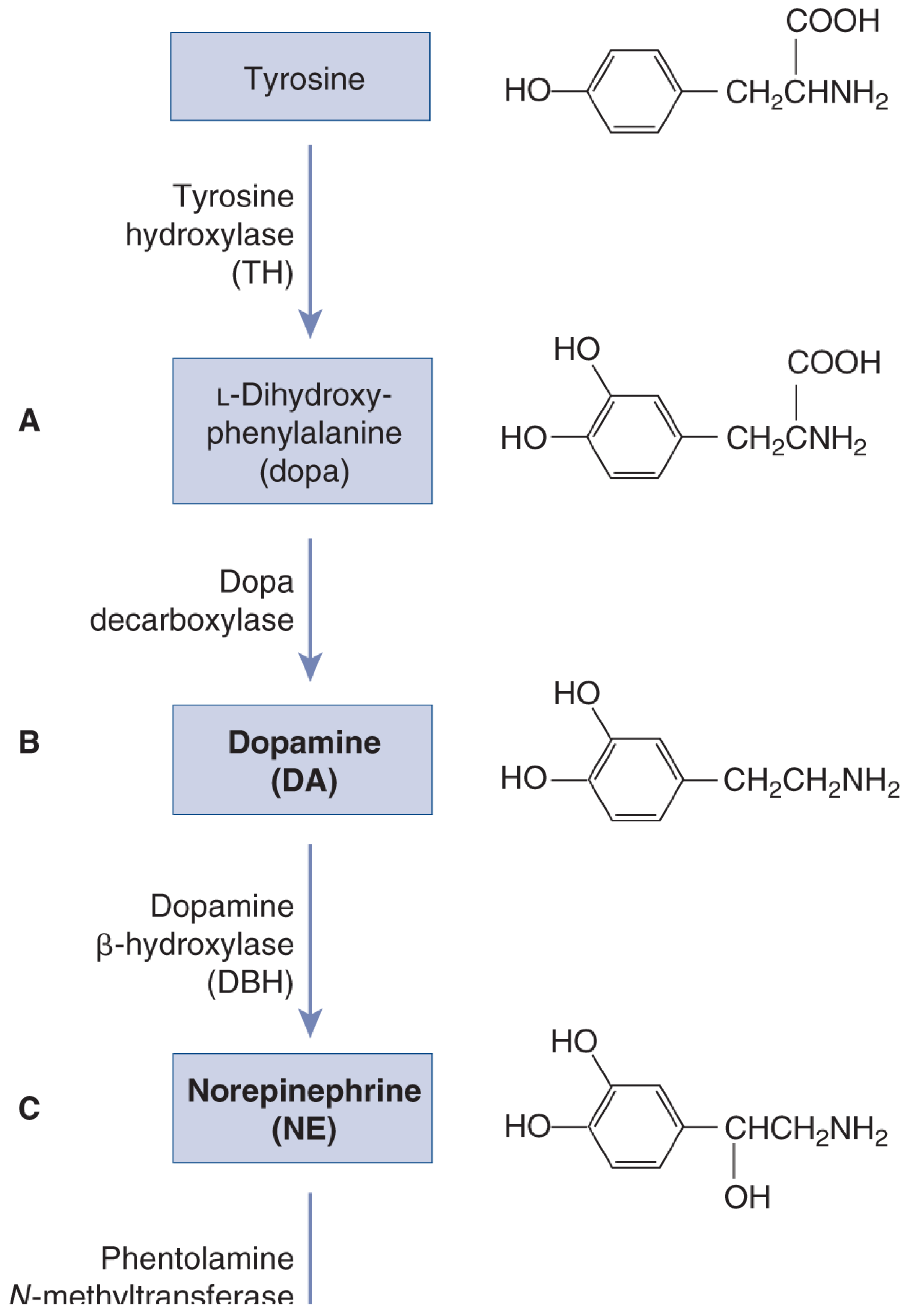
| Step | Substrate | Enzyme | Product | Key Notes |
|---|---|---|---|---|
| 1 | Tyrosine | Tyrosine hydroxylase (TH) | L-DOPA | Rate-limiting step; regulated by end-product inhibition and Ca²⁺-driven activation |
| 2 | L-DOPA | Dopa decarboxylase (AAAD) | Dopamine | Abundant enzyme; synthesis tracks DOPA availability |
| 3 | Dopamine | Dopamine β-hydroxylase (DBH) | Norepinephrine | Only enzyme bound inside synaptic vesicles; NE is the only transmitter synthesised within vesicles |
| 4 (adrenal only) | NE | Phenylethanolamine-N-methyltransferase (PNMT) | Epinephrine | Occurs in adrenal medulla chromaffin cells; requires SAM as methyl donor |
Regulation of TH:
- End-product inhibition: rising cytosolic catecholamine levels suppress TH
- Ca²⁺ activation: during high-rate firing, elevated [Ca²⁺] accelerates TH, matching supply to demand
- Long-term demand: sustained stimulation upregulates TH mRNA
Source: Neuroscience: Exploring the Brain, 5th Ed., pp. 503–504; Eric Kandel: Principles of Neural Science, 6th Ed., p. 409
3. Sites of Synthesis and Release
Central Nervous System
- Locus coeruleus (LC): the primary noradrenergic nucleus, located in the pons. Though containing relatively few neurons, it projects broadly to the cortex, cerebellum, hippocampus, and spinal cord, modulating arousal, attention, stress response, and autonomic tone. — Kandel, Principles of Neural Science
Peripheral Nervous System
- Postganglionic sympathetic nerve terminals: NE is the principal neurotransmitter here, released onto effector organs (heart, vasculature, kidney, gut, etc.)
- Adrenal medulla: releases ~20% NE + ~80% epinephrine directly into the bloodstream as a hormonal response to systemic stress
4. Receptor Pharmacology and Downstream Signalling
Noradrenaline acts on adrenergic receptors — all are GPCRs:
| Receptor | Coupling | Signal Cascade | Tissue Effect |
|---|---|---|---|
| α₁ | G_q/11 → PLC → IP₃/DAG → ↑Ca²⁺/PKC | Vascular smooth muscle contraction | Vasoconstriction (↑SVR, ↑MAP) |
| α₂ | G_i/o → ↓cAMP; ↑K⁺ (hyperpolarisation) | Presynaptic autoreceptor; vascular | Inhibits NE release (negative feedback); venous constriction |
| β₁ | G_s → adenylyl cyclase → ↑cAMP → PKA | Heart (atria + ventricles) | ↑Chronotropy, ↑inotropy, ↑dromotropy, ↑lusitropy |
| β₂ | G_s → ↑cAMP | Vascular smooth muscle (minor for NE) | Vasodilation — NE has minimal β₂ activity |
NE receptor selectivity: α₁ = α₂ >> β₁ >> β₂ (contrast with epinephrine which strongly activates β₂)
β₁ signalling in myocardium (Barash, Clinical Anesthesia, 9th Ed.):
- β₁ agonist → Gs → adenylyl cyclase → cAMP ↑ → PKA activation
- PKA phosphorylates L-type Ca²⁺ channels → greater Ca²⁺ entry
- PKA phosphorylates troponin I → increased TnC efficacy
- PKA phosphorylates phospholamban → faster SERCA2 activity → faster diastolic relaxation
Net cardiac result: increased contractility (inotropy) + slightly faster rate, but baroreceptor reflex-mediated bradycardia often predominates — making NE distinct from pure β₁ agonists.
5. Physiological Actions — System by System
Cardiovascular
| Parameter | Effect | Mechanism |
|---|---|---|
| SVR / Afterload | ↑↑↑ (intense) | α₁-mediated arterial vasoconstriction |
| Venous return | ↑ | α₁ constricts capacitance veins |
| MAP / DBP | ↑↑ | Combined ↑SVR + ↑venous return |
| Cardiac output | Unchanged or slight ↑ | β₁ inotropic effect balanced by ↑afterload |
| Heart rate | Often ↓ (reflex bradycardia) | Baroreceptor activation overrides direct β₁ chronotropy |
| Stroke volume | Modest ↑ | β₁ + venous return |
| Coronary perfusion | ↑ (when MAP profoundly low) | ↑Perfusion pressure dominates; but also ↑MVO₂ |
Barash, Clinical Anesthesia 9e, p. 962
Renal / Splanchnic
- At normal MAP: α₁ constriction reduces renal, hepatic, splanchnic, and skeletal muscle flow
- When MAP is profoundly reduced (shock): NE restores perfusion pressure, increasing urine output and GFR
- Prolonged high-dose NE risks sustained renal and mesenteric ischaemia — a major limitation
Pulmonary
- Stimulates α₁ in pulmonary arteries → dose-related ↑ pulmonary arterial pressure → risk of RV dysfunction at high doses in patients with pulmonary hypertension
CNS (physiological)
- Locus coeruleus firing increases arousal, attention, and "fight-or-flight" readiness
- Amygdala activates LC during acute stress → high NE impairs prefrontal cognition while sharpening reactive responses
6. Metabolism and Elimination
NE is inactivated by two pathways:
- Uptake-1 (neuronal reuptake) via the norepinephrine transporter (NET) — primary mechanism at the synapse
- Enzymatic degradation:
- MAO (monoamine oxidase, mitochondrial) → deamination
- COMT (catechol-O-methyltransferase, cytosolic) → methylation
- Final metabolite: vanillylmandelic acid (VMA) — measured in 24-h urine for pheochromocytoma diagnosis
Plasma half-life ~2 minutes; continuous infusion is required for clinical use.
7. Why Noradrenaline is the First-Line Emergency Vasopressor
The Core Rationale
NE provides the ideal combination of:
- Powerful α₁ vasoconstriction → rapidly restores MAP in vasodilated/distributive shock
- Adequate β₁ inotropism → maintains/improves cardiac output without solely increasing afterload
- No meaningful β₂ activity → avoids peripheral vasodilation that would worsen hypotension
- Lower arrhythmia risk than dopamine or epinephrine — clinically significant advantage
- Baroreceptor-mediated bradycardia prevention → heart is protected from tachyarrhythmias
"The preferred vasopressor for distributive shock is norepinephrine, because it is a strong vasoconstrictor with enough beta-adrenergic effects to increase myocardial contractility and to protect the heart against the increased afterload posed by vasoconstriction." — Goldman-Cecil Medicine, International Edition
Evidence Over Dopamine
A large prospective study comparing NE vs dopamine in shock demonstrated:
- Fewer adverse events, particularly arrhythmias, with NE
- Dopamine was associated with higher mortality in cardiogenic shock
- Meta-analysis confirmed NE superior to dopamine in in-hospital and 28-day mortality
- NE increases GFR and urine output equally to dopamine in septic patients, but without dopamine's side effects
— Rosen's Emergency Medicine, 10th Ed.
8. Clinical Indications — Where and When to Use
Primary Indications
| Indication | Evidence Level | Details |
|---|---|---|
| Septic shock | IA (highest) | First-line vasopressor per Surviving Sepsis Campaign; preferred over all others |
| Distributive shock (all causes) | I | Vasodilatory states: anaphylaxis (after epinephrine), neurogenic shock |
| Refractory hypotension post-fluid resuscitation | I | Any shock unresponsive to 30 mL/kg crystalloid |
| Vasoplegic syndrome (post-cardiopulmonary bypass) | II | Combined with methylene blue or hydroxocobalamin |
| Cardiogenic shock (limited role) | II | May be required temporarily; all vasopressors worsen afterload |
| Neurogenic shock | II | With fluid resuscitation |
Specific Contexts
Septic shock (most common emergency use):
- First-line when MAP <65 mmHg despite fluid resuscitation (≥30 mL/kg crystalloid)
- Goal MAP 65 mmHg (equivalent outcomes to 80–85 mmHg target; higher target does NOT improve mortality and increases adverse events)
- NE preferred over: dopamine (more arrhythmias), phenylephrine (no β₁ support), vasopressin alone (not a first-line sole agent)
Vasoplegic syndrome (post-cardiac surgery):
- NE + methylene blue (nitric oxide scavenger) or hydroxocobalamin for severe cases refractory to NE alone
Where NE is NOT the primary agent:
- Anaphylaxis: epinephrine remains first-line (β₂ bronchodilation is needed)
- Cardiac arrest: epinephrine first-line (α₁ + β₁)
- Isolated bradycardia: atropine / pacing
9. Dosing and Administration Protocols
Dosing (Adult)
| Source | Starting Dose | Titration Range | Target |
|---|---|---|---|
| Rosen's Emergency Medicine | 3–5 μg/min | Up to 30–50 μg/min | MAP ≥65 mmHg |
| Tintinalli's Emergency Medicine | Per weight-based protocol | — | MAP ≥65 mmHg |
| Goldman-Cecil Medicine | 1 μg/min | Up to 50 μg/min | MAP 65–70 mmHg |
Paediatric (Harriet Lane Handbook):
- Start 0.05–0.1 μg/kg/min; titrate up
- Fluid-refractory shock → add NE or epinephrine
Route of Administration
- Central venous catheter (CVC) preferred — to prevent tissue necrosis from extravasation
- Peripheral IV is acceptable for short durations in dilute solutions (≤20 μg/mL) via antecubital or larger peripheral veins with close monitoring — a large retrospective observational study supports this when CVC insertion would cause unacceptable delay (Barash, Clinical Anesthesia 9e, p. 963)
Preparation
- Standard concentration: 4 mg in 250 mL D5W or NS = 16 μg/mL
- High concentration (fluid-restricted): 16 mg in 250 mL = 64 μg/mL
10. Emergency Protocol — Step-by-Step (Septic Shock)
SEPTIC SHOCK VASOPRESSOR PROTOCOL
════════════════════════════════════════════════════
STEP 1: Fluid Resuscitation
• 30 mL/kg IV crystalloid (lactated Ringer preferred) over ≤3 hours
• Reassess MAP, lactate, urine output, mental status
STEP 2: If MAP <65 mmHg despite fluids
→ START Norepinephrine
• Dose: 3–5 μg/min IV (central line preferred)
• Titrate every 5–15 min to MAP ≥65 mmHg
• Maximum: ~50 μg/min before adding second agent
STEP 3: If MAP target not achieved on NE alone
→ ADD Vasopressin 0.03 units/min (fixed dose, do not titrate)
Purpose: NE-sparing, may reduce catecholamine dose
OR
→ ADD Epinephrine 1–15 μg/min if cardiac output depressed
STEP 4: If myocardial dysfunction present (echo evidence)
→ ADD Dobutamine 2–15 μg/kg/min
(do NOT substitute for NE — add on top)
STEP 5: Refractory vasodilatory shock
→ Consider Angiotensin II 20 ng/kg/min (titrate to 200 ng/kg/min)
Adrenergic-sparing effect; does not reduce mortality but reduces NE requirements
STEP 6: Ongoing monitoring
• MAP target: 65 mmHg (unless prior hypertension → target 75 mmHg)
• CVP target: 8–12 mmHg (on mechanical ventilation: 12–15 mmHg)
• Lactate clearance: target >10% per 2 hours
• Urine output: >0.5 mL/kg/hr
• Wean NE as haemodynamics and source control improve
════════════════════════════════════════════════════
Compiled from: Rosen's Emergency Medicine; Goldman-Cecil Medicine; Surviving Sepsis Campaign 2021
11. Adverse Effects and Contraindications
| Adverse Effect | Mechanism | Management |
|---|---|---|
| Peripheral ischaemia (fingers, toes, gut) | α₁ vasoconstriction | Lowest effective dose; phentolamine infiltration for extravasation |
| Renal ischaemia (prolonged high dose) | ↓ renal blood flow | Add low-dose vasopressin to spare NE dose |
| Pulmonary hypertension / RV dysfunction | α₁ pulmonary vasoconstriction | Add inhaled NO or prostacyclin if PHT is present |
| Arrhythmias | β₁ + ischaemia | Less than dopamine/epinephrine; substitute NE for epinephrine when AF occurs |
| Bradycardia | Baroreceptor reflex | Usually benign; reassess if haemodynamically significant |
| Hypertension | Excessive vasoconstriction | Titrate dose down |
| Tissue necrosis | Extravasation of NE | Use CVC; treat with phentolamine 5–10 mg in 10 mL NS infiltrated locally |
| Metabolic acidosis | Sustained visceral ischaemia at high doses | Address source control, wean NE |
Relative contraindications:
- Hypovolaemia — must correct volume first; NE on empty tank worsens microvascular ischaemia
- Coronary artery disease with flow-limiting stenoses — NE increases MVO₂; may precipitate myocardial ischaemia or graft spasm (internal mammary / radial artery grafts)
- Severe pulmonary hypertension — use with inhaled pulmonary vasodilator
- Halothane anaesthesia — risk of sensitised arrhythmias (historical, largely obsolete)
12. Noradrenaline vs Other Vasopressors — At a Glance
| Agent | α₁ | β₁ | β₂ | DA | Role |
|---|---|---|---|---|---|
| Norepinephrine | +++ | ++ | + | 0 | First-line vasopressor |
| Epinephrine | +++ | +++ | +++ | 0 | Anaphylaxis, cardiac arrest, second-line shock |
| Dopamine | ++ | +++ | + | +++ | Largely replaced; positive inotropy if NE unavailable |
| Phenylephrine | +++ | 0 | 0 | 0 | Spinal hypotension, AF with fast rate; NOT in septic shock |
| Vasopressin | V₁ only | 0 | 0 | 0 | Second-line add-on; NE-sparing; renal-protective |
| Dobutamine | 0 | +++ | ++ | 0 | Inotrope, NOT vasopressor; add-on for LV dysfunction |
13. Recent Evidence (2024–2026)
Three high-quality meta-analyses confirm and refine NE use in septic shock:
-
Ahn et al., Chest 2024 [PMID 38972348] — Systematic review/meta-analysis comparing early vs late NE initiation in septic shock: early initiation reduces time to MAP target and may improve outcomes.
-
Kotani et al., Crit Care 2024 [PMID 39736782] — Meta-analysis of non-adrenergic vasopressors (vasopressin, angiotensin II) vs NE in vasodilatory shock: non-adrenergic agents are useful adjuncts but do not replace NE as the anchor agent.
-
Shi et al., Crit Care 2025 [PMID 40329359] — Updated meta-analysis with trial sequential analysis: early NE significantly lowers 28-day mortality and time to shock reversal in septic shock patients.
⚠️ Current evidence update: The 2025 meta-analysis by Shi et al. supports initiating NE early — concurrently with fluid resuscitation rather than waiting for full 30 mL/kg fluid challenge — in patients with obvious vasodilatory shock, potentially reducing mortality.
Summary
Noradrenaline is the cornerstone emergency vasopressor because its receptor profile (strong α₁, moderate β₁, minimal β₂) precisely matches the haemodynamic deficit in distributive/vasodilatory shock: profound vasoplegia requiring both vasoconstriction and preserved cardiac output. Its superiority over dopamine in safety, arrhythmia profile, and mortality is established across multiple trials. Protocol-driven use — titrated to MAP 65 mmHg, layered with vasopressin and inotropes when needed, and initiated early — represents current best practice in septic shock and other vasodilatory emergencies.
Sources: Barash Clinical Anesthesia 9e; Rosen's Emergency Medicine 10e; Goldman-Cecil Medicine International Ed.; Tintinalli's Emergency Medicine; Goodman & Gilman's Pharmacological Basis of Therapeutics; Neuroscience: Exploring the Brain 5e; Kandel: Principles of Neural Science 6e; Surviving Sepsis Campaign Guidelines; PMIDs 38972348, 39736782, 40329359
This is a shared conversation. Sign in to Orris to start your own chat.