Cardiomyopathies (DCM, HOCM, RCM)- definition,etiology, classification, symptoms, diagnosis, differential diagnosis treatment
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
cardiomyopathy dilated hypertrophic restrictive comparison
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Searching PubMed
"cardiomyopathy" AND "guidelines"
Loading Image
Loading Image
Cardiomyopathies: DCM, HOCM, and RCM
Overview & Classification
Cardiomyopathy refers to myocardial disorders in which the heart muscle is structurally and functionally abnormal in the absence of coronary artery disease, hypertension, valvular disease, or congenital heart disease sufficient to explain the observed abnormality. The major types are classified by ventricular morphology and pathophysiology:
| Feature | DCM | HCM/HOCM | RCM |
|---|---|---|---|
| LV cavity | Dilated | Small/reduced | Normal or slightly reduced |
| Wall thickness | Normal or thin | Markedly thickened | Normal/slightly thickened |
| LVEF | <40% | 50–80% (often hyperdynamic) | 25–50% (usually preserved) |
| Dominant dysfunction | Systolic | Diastolic | Diastolic (stiff ventricle) |
| Atria | ± enlarged | ± enlarged (LA) | Markedly dilated (biatrial) |
The comparison is well illustrated on echocardiography:
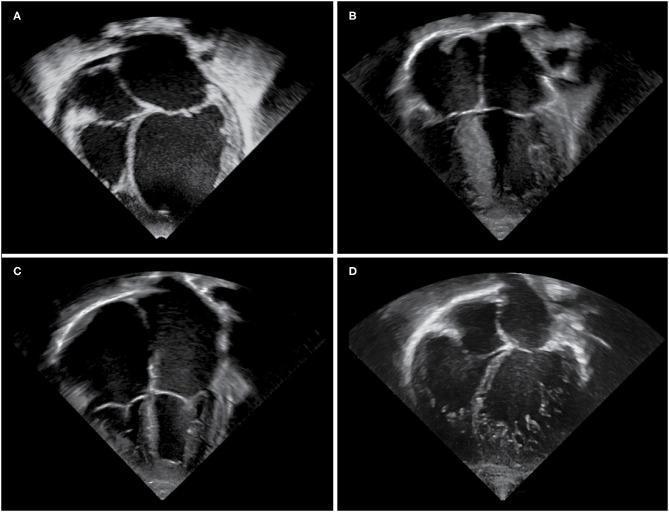
Echocardiographic phenotypes: (A) DCM—globular LV and LA enlargement with thinned walls; (B) HCM—pronounced asymmetric septal hypertrophy, reduced LV cavity; (C) RCM—massive biatrial dilation with normal-sized ventricles; (D) LVNC
I. DILATED CARDIOMYOPATHY (DCM)
Definition
DCM is characterized by progressive dilation and impaired systolic (contractile) function of the left ventricle or both ventricles, in the absence of coronary artery disease, valvular abnormalities, or pericardial disease. It is the most common form of cardiomyopathy, with an estimated adult prevalence of 1 in 250.
- Goldman-Cecil Medicine; Robbins & Kumar Basic Pathology
Etiology & Classification
A. Genetic / Familial DCM (20–50% of cases)
- Autosomal dominant inheritance predominates
-
50 causative genes identified, principally affecting cytoskeletal and sarcomeric proteins
- Most common: TTN (titin) mutations (~25% of familial, 18% of sporadic cases)
- Other genes: β-myosin heavy chain (MYH7), cardiac troponin T, α-myosin heavy chain, desmin (DES), lamin A/C (LMNA)
- X-linked DCM: mutations in dystrophin gene (Duchenne/Becker muscular dystrophy); also isolated X-linked DCM
- Lamin A/C mutations → arrhythmogenic form with atrial fibrillation and progressive AV conduction disease preceding heart failure
- Robbins Basic Pathology; Goldman-Cecil Medicine
B. Acquired / Toxic DCM
| Cause | Notes |
|---|---|
| Viral myocarditis | Coxsackievirus B, adenovirus, enterovirus, parvovirus B19, HHV-6, CMV, Chagas disease |
| Alcohol | Direct toxic effect + acetaldehyde metabolites; nutritional deficiencies |
| Chemotherapy | Anthracyclines (doxorubicin), cyclophosphamide |
| Peripartum | Onset in last month of pregnancy or within 5 months postpartum |
| Metabolic | Iron overload (hemochromatosis), nutritional deficiencies (thiamine) |
| Endocrinopathies | Hypothyroidism, hyperthyroidism, acromegaly, pheochromocytoma |
| Tachycardia-mediated | Reversible once tachycardia is controlled |
| Autoimmune/inflammatory | SLE, scleroderma, sarcoidosis |
| Radiation therapy | Diffuse biventricular fibrosis |
| Idiopathic | ~50% of cases; diagnosis of exclusion |
Pathology
- All four chambers dilated (LV > RV)
- Concurrent myocyte hypertrophy despite overall thinning of walls
- Mural thrombi common (especially at LV apex)
- Histology: non-specific myocyte hypertrophy, interstitial fibrosis, myocyte loss
Clinical Features / Symptoms
- Progressive heart failure: dyspnea on exertion → orthopnea → PND → dyspnea at rest
- Low cardiac output symptoms: fatigue, weakness, reduced exercise tolerance
- Arrhythmias: atrial fibrillation, ventricular tachycardia/fibrillation (risk of sudden cardiac death)
- Thromboembolic events (stroke, peripheral embolism from mural thrombi)
- Infants/children: poor feeding, irritability, sweating, failure to thrive, tachycardia, tachypnea
- Older children/adults: chest pain, palpitations, decreased exercise tolerance, syncope
Physical examination:
- Elevated JVP, displaced LV apex (laterally)
- S3 gallop (low-pitched, pathognomonic of systolic failure)
- Mitral ± tricuspid regurgitation murmurs (functional, due to annular dilatation)
- Peripheral edema, hepatomegaly, ascites in advanced disease
- Narrow pulse pressure, reduced peripheral pulses
Diagnosis
| Test | Finding |
|---|---|
| ECG | Sinus tachycardia, LBBB, non-specific ST-T changes, AF, ventricular arrhythmias |
| CXR | Cardiomegaly ("cardiac boot"), pulmonary vascular congestion, pleural effusions |
| Echocardiography | LV ± RV dilation, reduced LVEF (<40%), wall motion abnormalities, functional MR/TR, LA enlargement |
| Cardiac MRI | Gold standard for chamber volumes; LGE identifies fibrosis (mid-wall pattern) |
| Coronary angiography | Rule out ischemic cardiomyopathy (prior to labeling idiopathic) |
| BNP / NT-proBNP | Elevated, correlates with degree of HF |
| Lab | Serum electrolytes, renal/hepatic function, TSH, ferritin, CBC, troponin |
| Genetic testing | For familial DCM; also guides cascade family screening |
| Endomyocardial biopsy | Reserved for rapidly deteriorating patients or to exclude giant cell myocarditis |
Differential Diagnosis
- Ischemic cardiomyopathy (most important DDx; coronary angiography essential)
- Hypertensive heart disease
- Valvular heart disease (aortic regurgitation, mitral regurgitation)
- Myocarditis (acute presentation)
- Alcoholic cardiomyopathy (specific subset)
- Peripartum cardiomyopathy
- Tachycardia-mediated cardiomyopathy
- Takotsubo (stress) cardiomyopathy
Treatment
General / Supportive:
- Sodium (<2g/day) and fluid restriction
- Avoidance of alcohol, cardiotoxic drugs
- Submaximal exercise regimen (unless active myocarditis, peripartum, or desmosomal/lamin mutations)
Pharmacological (HFrEF guidelines):
| Drug class | Examples | Benefit |
|---|---|---|
| ACE inhibitors / ARBs / ARNi | Ramipril, perindopril; Sacubitril/valsartan | ↓ mortality, ↓ remodeling |
| Beta-blockers | Bisoprolol, carvedilol, metoprolol succinate | ↓ mortality, ↑ LVEF |
| MRA (mineralocorticoid antagonists) | Spironolactone, eplerenone | ↓ mortality in HFrEF |
| SGLT2 inhibitors | Dapagliflozin, empagliflozin | ↓ HF hospitalizations, ↓ mortality |
| Diuretics | Furosemide, torasemide | Symptom relief (volume overload) |
| Anticoagulation | DOACs or warfarin (INR 2–3) | If AF, LV/LA thrombus, ± cardiomegaly |
Device Therapy:
- ICD — primary prevention of sudden cardiac death (LVEF ≤35%, NYHA II–III on optimal medical therapy)
- CRT (cardiac resynchronization therapy) — LVEF ≤35% with LBBB and QRS ≥150ms
- Ventricular assist device (VAD) — bridge to transplant or destination therapy in advanced HF
Surgical:
- Cardiac transplantation — for refractory advanced HF
- Treatment of underlying cause (e.g., tyrosine kinase inhibitors for chemotherapy-induced; tafamidis for ATTR amyloidosis)
Prognosis: Most patients improve with treatment; 5-year mortality in idiopathic DCM ~20–30% with optimal therapy.
- Goldman-Cecil Medicine
II. HYPERTROPHIC (OBSTRUCTIVE) CARDIOMYOPATHY — HCM / HOCM
Definition
HCM is defined as unexplained LV hypertrophy in the absence of abnormal loading conditions (valve disease, hypertension, congenital heart defects) sufficient to explain the degree of hypertrophy. HOCM refers to the obstructive subtype with dynamic LVOT gradient ≥50 mmHg at rest or with provocation.
- Prevalence: 0.2–0.5% worldwide, all racial groups
- Goldman-Cecil Medicine
Etiology & Genetics
- Usually familial with autosomal dominant inheritance (variable penetrance)
- 50–60% caused by sarcomeric contractile protein gene mutations (>1400 mutations identified)
- Most commonly affected genes:
- MYH7 (β-myosin heavy chain) — commonest
- MYBPC3 (myosin-binding protein C) — also very common
- Others: TNNT2, TNNI3, TPM1, MYL2, MYL3, ACTC1
- Non-sarcomeric causes: Friedreich's ataxia, glycogen storage diseases (Pompe), Anderson-Fabry disease, Noonan syndrome, infants of diabetic mothers
Key concept: Gain-of-function mutations in sarcomere genes cause HCM; loss-of-function mutations in the same genes cause DCM.
Classification
| Type | Definition |
|---|---|
| Obstructive (HOCM) | Resting or provokable LVOT gradient ≥50 mmHg (>3.6 m/s on Doppler) |
| Latent obstructive | Resting gradient <50 mmHg; obstruction occurs only with exercise |
| Non-obstructive | Gradient <50 mmHg at rest and with all provocation (~25% of patients) |
Pathology
- Asymmetric septal hypertrophy (ASH) — basal anterior septum most common; LV wall thickness often >15mm
- Myocyte disarray — pathognomonic; >20% of cross-sectional area
- Interstitial fibrosis
- Intramural coronary artery disease (small vessel)
- SAM (systolic anterior motion) of the mitral valve leaflet → LVOT obstruction and mitral regurgitation
- Fibrous endocardial plaque at the site of SAM-septal contact
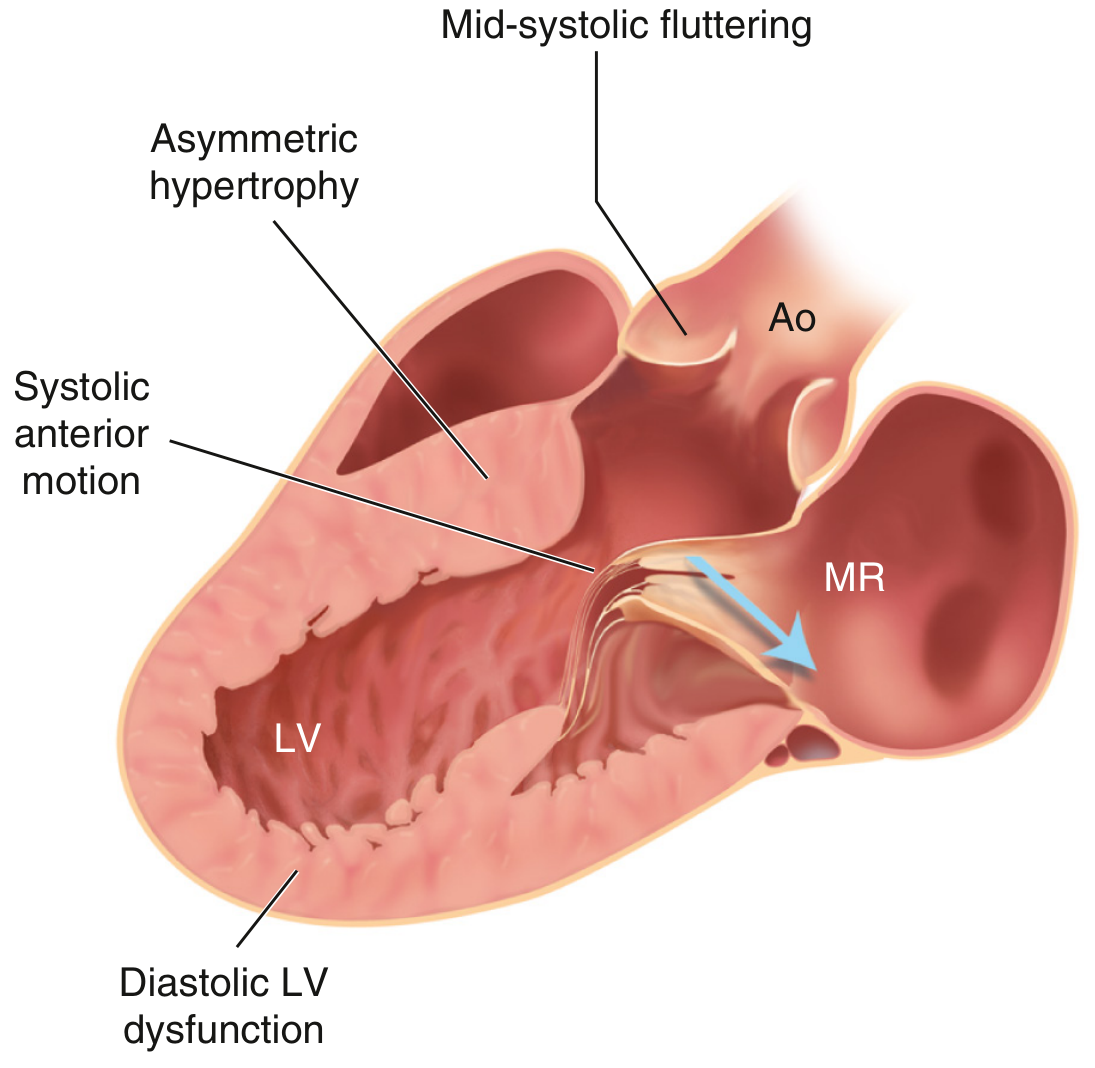
Hypertrophic cardiomyopathy: Asymmetric septal hypertrophy with systolic anterior motion (SAM) of the mitral valve, causing dynamic LVOT obstruction and mitral regurgitation (MR, blue arrow). — Textbook of Clinical Echocardiography
Clinical Features / Symptoms
- Dyspnea on exertion (most common; from diastolic dysfunction ± outflow obstruction)
- Angina (from increased myocardial O₂ demand + microvascular disease)
- Syncope / presyncope — exertional (from LVOT obstruction) or from arrhythmia
- Palpitations (AF, SVT, VT)
- Sudden cardiac death — especially in young athletes; can be the first manifestation
- Some patients are asymptomatic (found incidentally on echo or ECG)
Physical examination findings:
- Systolic ejection murmur at LLSB/apex — increases with Valsalva (decreased preload ↑ obstruction), standing; decreases with squatting, passive leg raise (increased preload ↓ obstruction)
- Bisferiens pulse (double-peaked carotid pulse)
- S4 gallop (from reduced LV compliance)
- Loud S1, paradoxically split S2
- Mitral regurgitation murmur (secondary to SAM)
- LV apex: forceful, double or triple impulse
Diagnosis
| Test | Findings |
|---|---|
| ECG | LVH voltage criteria, deep Q waves (inferolateral leads from septal hypertrophy), T-wave inversions, LBBB, delta waves (if WPW co-exists); AF |
| Echocardiography | ASH (septum/posterior wall ratio >1.3–1.5), SAM of MV, LVOT gradient, diastolic dysfunction; MR; LA enlargement |
| Cardiac MRI | Confirms extent of hypertrophy; LGE (patchy mid-wall fibrosis); superior for apical variant |
| Genetic testing | Recommended when diagnostic phenotype present; enables cascade family screening |
| Exercise stress echo / Holter | Assess provokable gradient; screen for VT (arrhythmia risk) |
| Cardiac catheterization | Brockenbrough-Braunwald sign (post-PVC gradient increase); "spike-and-dome" aortic pressure waveform |
Differential Diagnosis
- Hypertensive heart disease (concentric LVH — history of HTN, symmetric)
- Aortic stenosis (fixed obstruction, valvular changes on echo)
- Athlete's heart (physiological hypertrophy — regression with detraining; no SAM, no myocyte disarray)
- Cardiac amyloidosis (RCM phenotype, "granular sparkling" pattern)
- Fabry disease (globotriaosylceramide deposits)
- Glycogen storage diseases (Pompe, Danon)
- LVNC (left ventricular non-compaction)
- Friedreich's ataxia
Treatment
The 2024 AHA/ACC Guideline for HCM (PMID: 38718139) guides current management.
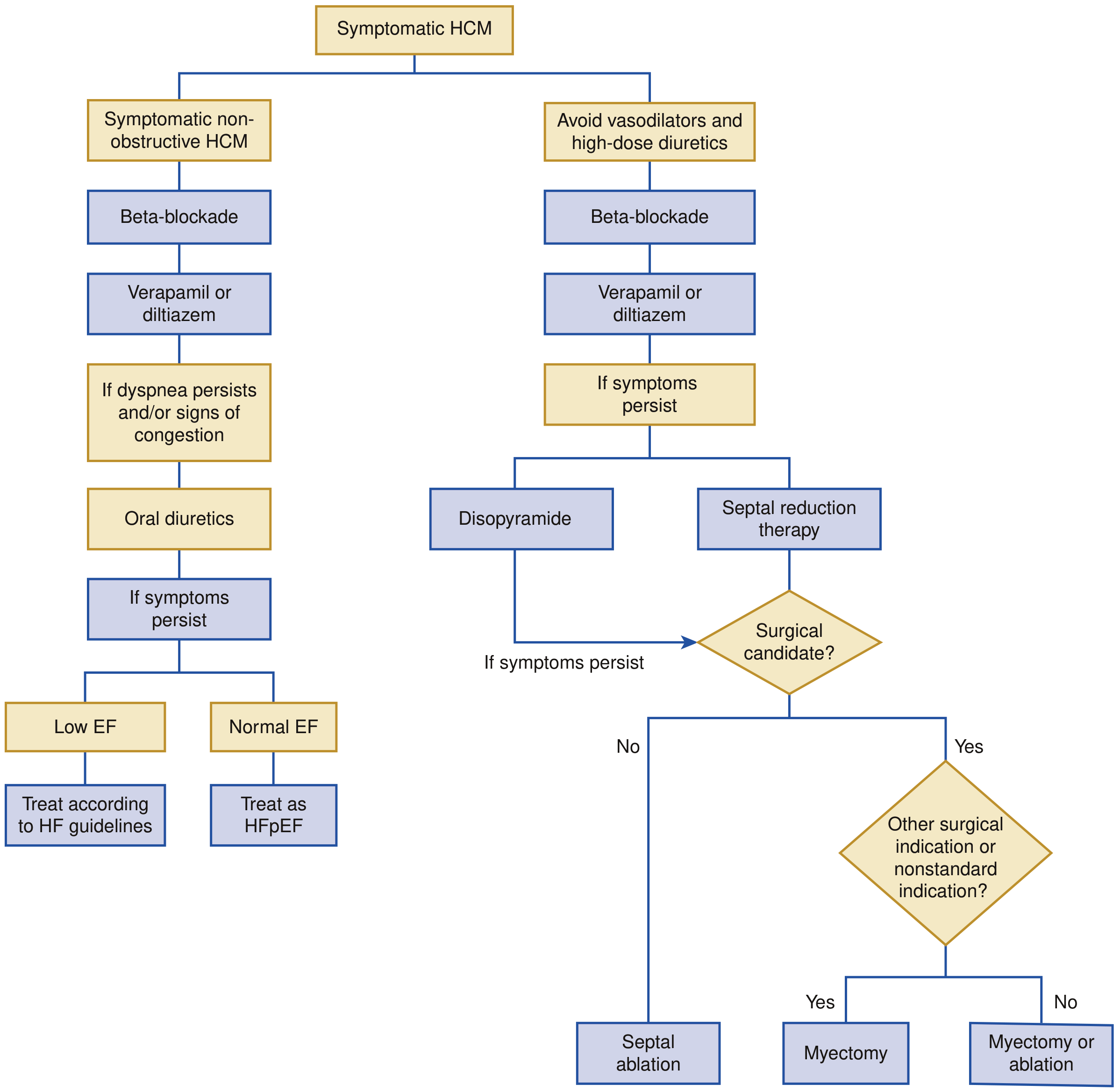
HCM management algorithm. Left: Non-obstructive HCM — beta-blocker → verapamil/diltiazem → diuretics if congestion; if EF low treat per HFrEF, if EF normal treat as HFpEF. Right: Obstructive HOCM — avoid vasodilators and high-dose diuretics; beta-blocker → verapamil/diltiazem; if persistent: disopyramide or septal reduction therapy (myectomy vs. ablation). — Braunwald's Heart Disease
General:
- Avoid vasodilators (nitrates, phosphodiesterase inhibitors), high-dose diuretics, digoxin, dehydration, extreme exertion — all worsen obstruction
- Genetic counseling and first-degree family screening (ECG + echo at baseline; repeat every 1–5 years)
Pharmacological:
| Drug | Indication | Mechanism |
|---|---|---|
| Beta-blockers (1st line) | Obstructive and non-obstructive HCM | ↓ heart rate → ↑ diastolic filling time; ↓ contractility → ↓ gradient |
| Verapamil / Diltiazem (2nd line) | If beta-blocker intolerant | ↓ LVOT gradient; improve diastolic function |
| Disopyramide | Obstructive HCM (add-on) | Class Ia antiarrhythmic; ↓ contractility → ↓ LVOT gradient |
| Mavacamten (novel, FDA-approved 2022) | Obstructive HCM | Selective cardiac myosin ATPase inhibitor; ↓ actin-myosin cross-bridges → ↓ gradient and symptoms (EXPLORER-HCM trial) |
| Anticoagulation | AF (any onset) | DOACs preferred |
Interventional / Surgical (for drug-refractory obstructive HCM):
- Surgical septal myectomy (Morrow procedure) — gold standard; resects hypertrophied basal septum; preferred if surgical candidate or other cardiac surgery needed
- Alcohol septal ablation (ASA) — catheter-based; injects ethanol into septal perforator → controlled septal infarct; for non-surgical candidates
- ICD implantation — for sudden cardiac death prevention (high-risk features: massive LVH ≥30mm, NSVT, family history of SCD, unexplained syncope, abnormal blood pressure response to exercise, extensive LGE on MRI)
- Cardiac transplantation — end-stage HCM with systolic failure
III. RESTRICTIVE CARDIOMYOPATHY (RCM)
Definition
RCM is characterized by stiffness, impaired diastolic filling, elevated ventricular diastolic pressures, and reduced diastolic volume of the LV or RV, despite normal or near-normal systolic function and wall thickness. Both ventricles fail to fill adequately due to rigid ventricular walls — the fundamental defect is diastolic, not systolic.
- Goldman-Cecil Medicine
Etiology & Classification
Primary (idiopathic) RCM — rare; ~30% familial (mutations in TNNI3, MYH7, DES)
Secondary RCM:
| Category | Causes |
|---|---|
| Infiltrative | Amyloidosis (most prevalent — especially ATTR wild-type in elderly; also AL amyloidosis), Sarcoidosis |
| Storage diseases | Hemochromatosis, Fabry disease, Glycogen storage diseases, Gaucher disease |
| Fibrotic | Radiation fibrosis, Scleroderma, Drug-induced (doxorubicin, serotonin, ergotamine) |
| Endomyocardial | Endomyocardial fibrosis (tropical; Africa; hypereosinophilia/parasitic infections) — most common RCM worldwide; Löffler endocarditis (HES with eosinophilic myocardial infiltration) |
| Metabolic | Carnitine deficiency, fatty acid oxidation defects |
| Miscellaneous | Carcinoid syndrome, metastatic tumors, radiation |
Key point: Wild-type transthyretin (ATTR) cardiac amyloidosis is increasingly recognized as the most prevalent form of RCM in older patients.
Pathology
- Biatrial dilation with small, non-dilated ventricular cavities
- Thrombus in atrial appendages
- Patchy endocardial fibrosis
- Histology: varying from minimal to extensive patchy interstitial fibrosis; cause-specific deposits (amyloid, iron, eosinophilic infiltrate)
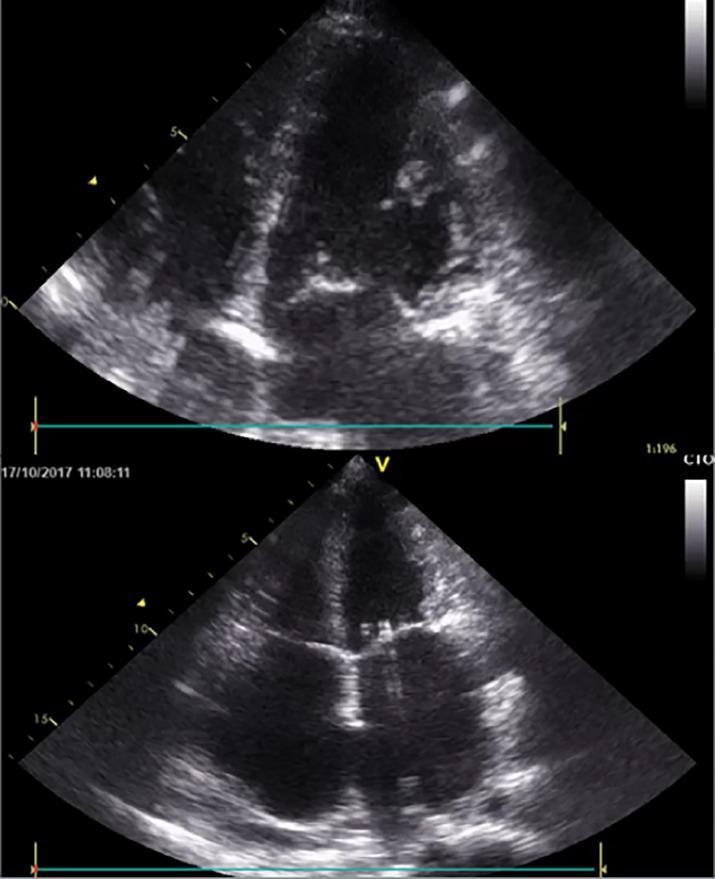
Apical 4-chamber echocardiogram: RCM with severe biatrial enlargement dwarfing the normal-sized ventricles — hallmark of impaired ventricular filling and elevated filling pressures
Clinical Features / Symptoms
- Dyspnea on exertion → rest dyspnea, orthopnea, PND
- Fatigue and weakness
- Right heart failure symptoms: peripheral edema, ascites, hepatomegaly, abdominal discomfort
- Palpitations, AF, heart block
- Rarely, sudden cardiac death as the initial manifestation
- Chest pain
Physical examination:
- Elevated JVP with prominent y descent (rapid early diastolic filling)
- Kussmaul sign — JVP fails to fall (or rises) on inspiration (shared with constrictive pericarditis)
- Loud P2 (if pulmonary hypertension)
- S3 and/or S4 gallop
- Peripheral edema, hepatomegaly, ascites
Diagnosis
| Test | Findings |
|---|---|
| ECG | P mitrale/pulmonale, nonspecific ST-T changes, conduction abnormalities, abnormal Q waves; low voltage QRS in amyloidosis |
| Echocardiography | Biatrial dilation >> ventricular dilation; normal/near-normal LVEF; "sparkling granular" myocardium (amyloid); diastolic dysfunction: ↑E/A ratio, ↓E deceleration time, ↓IVRT; restrictive filling pattern |
| Doppler/Tissue Doppler | Increased E/e' ratio; diastolic annular velocities reduced |
| Cardiac catheterization | "Dip-and-plateau" (square root sign) on ventricular pressure trace; LVEDP usually >5 mmHg above RVEDP (distinguishes from constrictive pericarditis where pressures equalize) |
| Cardiac MRI | LGE pattern (amyloidosis: diffuse subendocardial; sarcoidosis: patchy); T1 mapping (elevated in amyloid) |
| Endomyocardial biopsy | Congo red staining for amyloid; Prussian blue for hemochromatosis; specific deposits |
| Nuclear imaging (Tc-99m PYP) | Highly sensitive/specific for ATTR amyloidosis |
| Lab | Serum free light chains, SPEP/UPEP (AL amyloid); ferritin/transferrin saturation (hemochromatosis); ACE level (sarcoidosis) |
Differential Diagnosis — RCM vs. Constrictive Pericarditis
This is the most critical differential (and clinically the most challenging):
| Feature | RCM | Constrictive Pericarditis |
|---|---|---|
| Etiology | Myocardial infiltration/fibrosis | Pericardial fibrosis/calcification |
| JVP | Elevated, prominent y descent | Elevated, prominent x and y descents |
| Kussmaul sign | Present | Present |
| Pulsus paradoxus | Absent | Present |
| Cardiac catheterization | LVEDP > RVEDP by >5 mmHg | LVEDP = RVEDP (within 5 mmHg); equalization of diastolic pressures |
| Tissue Doppler e' velocity | Reduced (<8 cm/s) | Normal or elevated (>8 cm/s, "annulus reversus") |
| CT/MRI | No pericardial thickening | Pericardial thickening/calcification |
| LGE | Disease-specific pattern | Normal myocardium |
| Treatment | Medical (cause-specific) | Pericardiectomy (curative) |
Other DDx for RCM:
- HCM (especially overlap phenotype)
- HFpEF (hypertensive heart disease)
- Tricuspid valve disease
Treatment
General:
- Treat the underlying cause when possible:
- ATTR amyloidosis — tafamidis (TTR stabilizer; reduces cardiovascular mortality); patisiran/inotersen (TTR silencers)
- AL amyloidosis — chemotherapy/stem cell transplant
- Hemochromatosis — phlebotomy, iron chelation
- Sarcoidosis — corticosteroids
- Fabry disease — enzyme replacement therapy (agalsidase-α/β)
- Löffler endocarditis — corticosteroids ± tyrosine kinase inhibitors (if associated myeloid neoplasm)
- Endomyocardial fibrosis — surgical endocardiectomy in selected cases
Symptomatic treatment:
- Diuretics — cautiously for congestion (preload-dependent state; aggressive diuresis worsens output)
- Beta-blockers / rate-limiting agents — slow HR to maximize diastolic filling time
- Anticoagulation — for AF, atrial thrombus (high risk due to stasis in dilated atria)
- Antiarrhythmics — for AF, arrhythmia control
- Pacemaker / ICD — for complete heart block or high-risk arrhythmias (especially sarcoidosis, desminopathy)
Avoid: ACE inhibitors and vasodilators should be used cautiously (fixed cardiac output; vasodilation → hypotension without benefit)
Cardiac transplantation — for end-stage, refractory disease (but contraindicated in systemic amyloidosis without simultaneous liver/stem cell transplant for AL type)
Comparison Summary
| Feature | DCM | HOCM | RCM |
|---|---|---|---|
| Definition | Dilated chambers + systolic dysfunction | LVH without loading condition; dynamic LVOT obstruction | Stiff ventricle + diastolic dysfunction; normal systole |
| Key gene | TTN, LMNA, dystrophin | MYH7, MYBPC3 | TNNI3, DES |
| LVEF | <40% | 50–80% (hyperdynamic) | 25–50% (preserved) |
| Classic murmur | Functional MR/TR (pan) | LLSB ejection murmur (↑ Valsalva) | None or S3/S4 |
| Key echo | Global dilation, ↓ EF | ASH, SAM, LVOT gradient | Biatrial dilation, restrictive filling |
| Key cath finding | Elevated LVEDP | Spike-and-dome, Brockenbrough sign | Dip-and-plateau (square root sign) |
| DDx | Ischemic CM | HTN, aortic stenosis, athlete's heart | Constrictive pericarditis |
| Mainstay treatment | ACEi/ARNi + BB + MRA + SGLT2i; ICD if EF ≤35% | BB, verapamil, disopyramide, mavacamten; myectomy/ablation | Treat cause; cautious diuretics; avoid vasodilators |
Sources: Robbins & Kumar Basic Pathology 10e | Robbins, Cotran & Kumar Pathologic Basis of Disease | Goldman-Cecil Medicine International Edition | Braunwald's Heart Disease 12e | Textbook of Clinical Echocardiography | Tintinalli's Emergency Medicine | 2024 AHA/ACC HCM Guideline (PMID 38718139)
This is a shared conversation. Sign in to Orris to start your own chat.