Thyroglossal fistula and Thyroglossal duct cyst in detail
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Searching PubMed
thyroglossal duct cyst Sistrunk procedure
I now have comprehensive content from multiple authoritative textbooks. Let me compile the complete answer.
Thyroglossal Duct Cyst & Thyroglossal Fistula
1. Embryological Basis
The thyroid gland originates at approximately the 3rd week of gestation as a median endodermal thickening (diverticulum) at the floor of the pharynx — specifically at the foramen cecum, located at the junction of the anterior two-thirds and posterior third of the tongue. This thyroid primordium descends in the midline of the neck, passing anterior to structures that will form the hyoid bone and laryngeal cartilages, to reach its final pretracheal position by the 7th week.
During its descent, the thyroid anlage remains connected to the foramen cecum by an epithelial-lined tube — the thyroglossal duct. The duct lumen normally begins to obliterate during the 5th week and disappears completely by the 8th week.
Key anatomical relationship: As the ventral cartilages of the 2nd and 3rd branchial arches form the hyoid bone, the thyroglossal duct passes through the central portion of the developing hyoid. This explains why the definitive surgical procedure must include resection of the hyoid body.
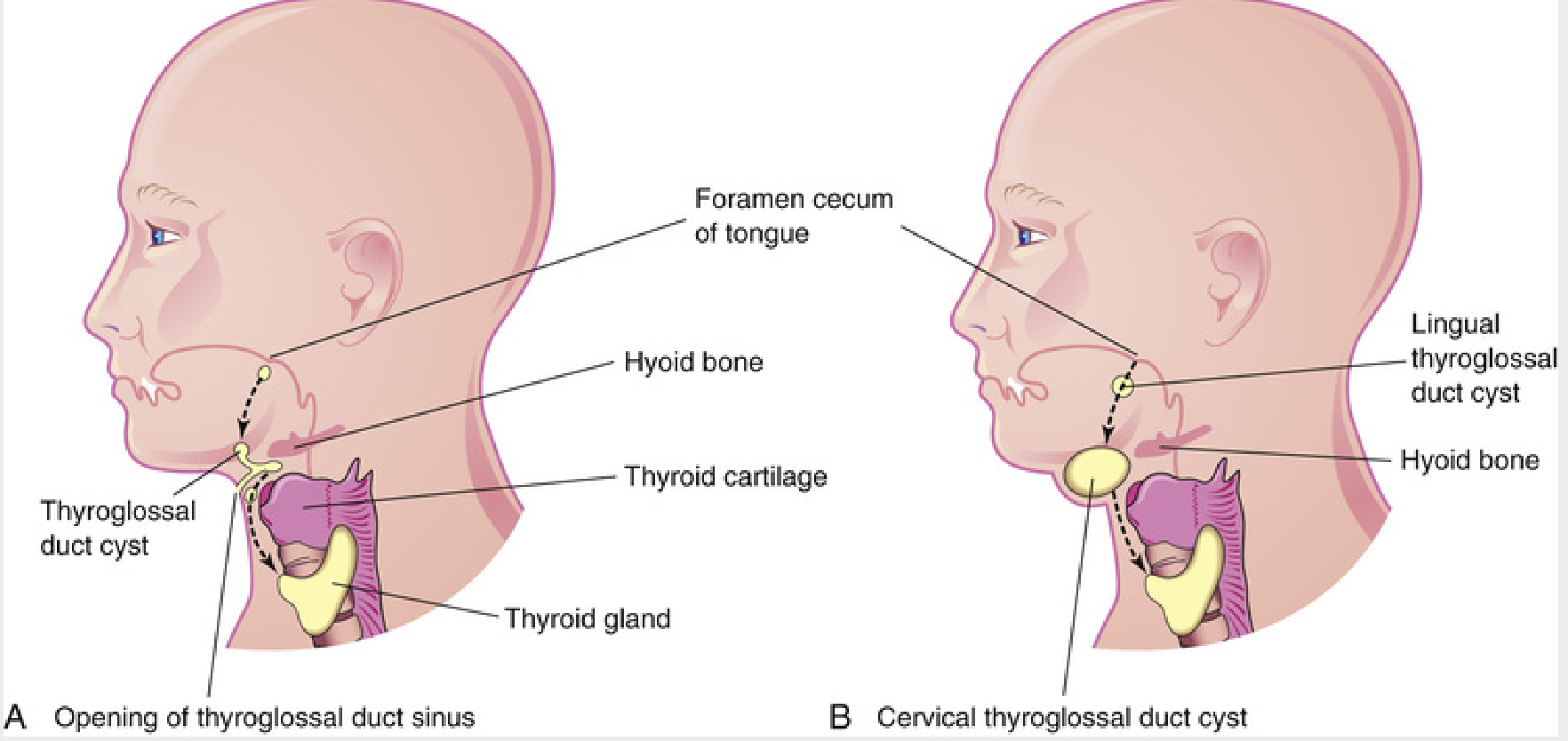
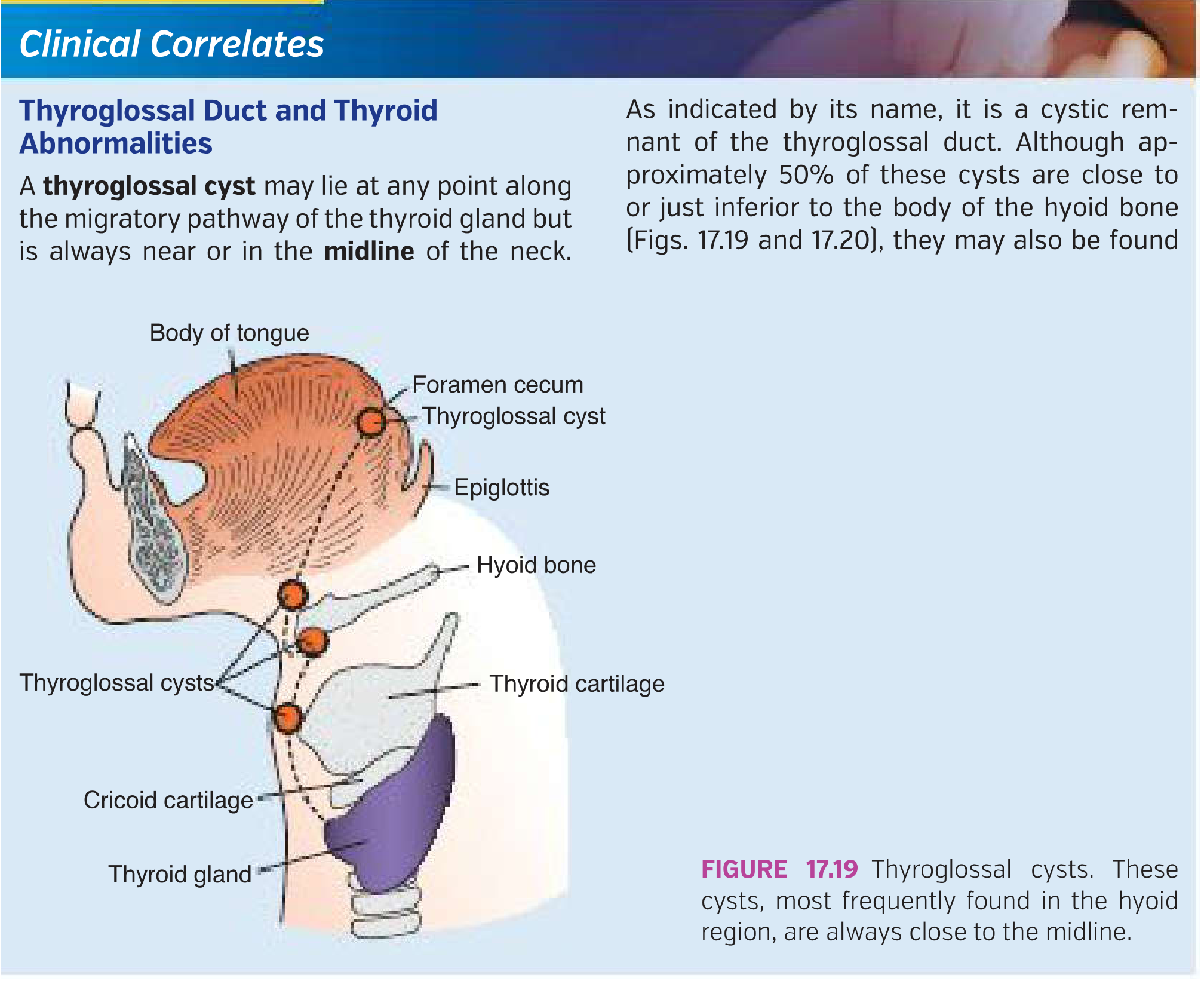
2. Thyroglossal Duct Cyst (TDC)
Definition
A thyroglossal duct cyst forms when elements of the thyroglossal duct persist despite complete thyroid descent. The persistent duct segment gradually fills with mucus/secretions, forming a cyst. TDCs are the most commonly encountered congenital cervical anomalies.
Location
- Can occur anywhere along the migratory path of the thyroid — from the base of the tongue to just above the isthmus
- ~80% are found at or just below the hyoid bone (juxtahyoid position)
- ~20% are suprahyoid; rarely, a TDC forms at the base of the tongue (lingual thyroglossal cyst) or within the thyroid itself (intrathyroidal)
Clinical Features
| Feature | Details |
|---|---|
| Age of presentation | Most common in 2–4-year-olds (when baby fat disappears); can present at any age including adults |
| Sex | Equal male:female distribution |
| Location | Midline of the neck |
| Character | Smooth, well-defined, painless, mobile cystic mass, 1–4 cm |
| Pathognomonic sign | Moves upward with protrusion of the tongue or swallowing (due to its attachment via the duct remnant to the tongue base) |
| Symptoms | Usually asymptomatic; may become tender and swollen if infected |
| Rare presentations | Dysphagia, dysphonia (if lingual), airway compromise (large lingual TDC) |
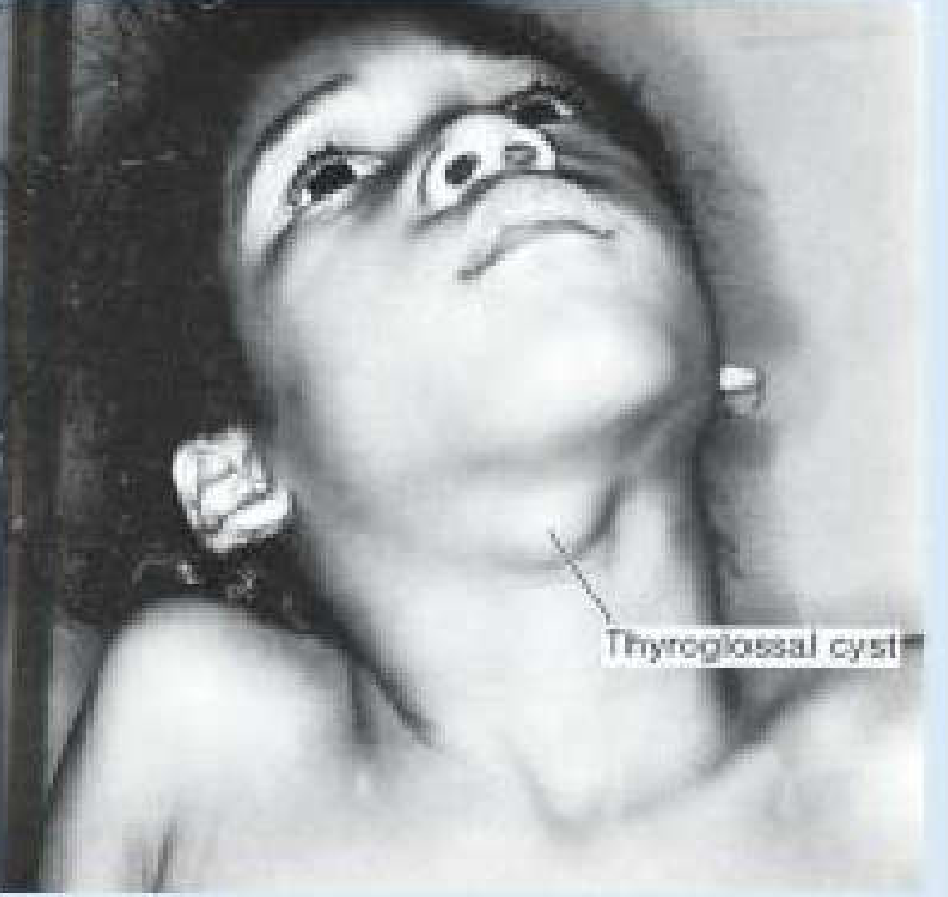
Complications
- Infection — most common; often triggered by upper respiratory tract infections via oral bacteria ascending through a patent duct. Results in abscess formation
- Fistula formation — infected cysts may spontaneously rupture through the skin, creating a thyroglossal fistula (see below)
- Malignant transformation — exceedingly rare (~1% of cases); usually papillary thyroid carcinoma (most common), followed by squamous cell carcinoma. The identification of malignancy within a TDC is associated with a 6.6% risk of microcarcinoma in the normal-appearing thyroid gland. Suspect malignancy if the cyst grows rapidly or ultrasound shows a complex anechoic pattern or calcification
- Ectopic thyroid — rarely, the TDC may represent the patient's only thyroid tissue (especially if the thyroid fails to descend — lingual thyroid). This occurs when caudal migration of the thyroid is arrested
Histopathology (Robbins Pathology)
- Wall lined by stratified squamous epithelium near the tongue base, or pseudostratified ciliated columnar (respiratory) epithelium in lower positions
- Transitional epithelial differentiation patterns also occur
- Fibrous cyst wall often includes lymphoid aggregates and thyroid follicular remnants (heterotopic thyroid tissue present in ~20% of cases)
Diagnosis
- Clinical — characteristic midline neck mass moving with tongue protrusion is usually sufficient
- Ultrasound — first-line imaging; confirms cystic nature, confirms presence of a normal orthotopic thyroid gland in the neck (critical — must rule out that the TDC is not the only thyroid tissue before surgery). Also identifies infection, septa, solid components
- CT/MRI — for large or atypical lesions; delineates extent and relationship to surrounding structures
- Thyroid scintigraphy (radionuclide scan) — used selectively when US is inconclusive about whether normal thyroid tissue is present
- FNAC — can rule out malignancy in suspicious lesions
Important: Routine thyroid function tests and nuclear imaging are not required preoperatively in all cases. However, if a normal thyroid cannot be palpated or confirmed on US, further investigation is mandatory before surgery.
3. Thyroglossal Fistula
Definition & Pathogenesis
A thyroglossal fistula (also called a thyroglossal duct sinus) is an abnormal communication between the skin of the anterior neck and the pharynx (at the foramen cecum), tracking along the path of the former thyroglossal duct.
- It is almost always a secondary event, arising after:
- Spontaneous rupture of an infected thyroglossal duct cyst through the skin
- Incision and drainage of an abscess formed within a TDC (inadequate/incomplete primary surgery)
- Rarely, it may be present at birth as a primary congenital anomaly (Langman's Medical Embryology)
- Unlike branchial cleft fistulas (which are true fistulas with two openings from the outset), thyroglossal fistulas are typically sinuses with only one external opening — the term "fistula" is used loosely since the internal opening at the foramen cecum may not always be patent
Anatomy of the Tract
- The external opening lies in the median plane of the anterior neck, anterior to the laryngeal cartilages (typically between the hyoid bone and the thyroid notch)
- The tract runs superiorly, passing through or closely related to the body of the hyoid bone, ultimately directed toward the foramen cecum at the tongue base
- The tract is lined by pseudostratified ciliated columnar epithelium (similar to the cyst) with surrounding fibrous tissue and lymphoid infiltrate
Clinical Features
- Intermittent discharge of mucoid or mucopurulent material from a midline neck opening
- The opening may appear as a small dimple or pore with surrounding skin changes (induration, granulation tissue, erythema)
- History of a previously swollen midline neck mass that spontaneously drained or was incised
- Periodic flare-ups with discharge, especially after URTI
- The tract moves with swallowing/tongue protrusion (like the cyst)
Diagnosis
- Clinical — history + midline location
- Fistulogram/sinogram — injection of contrast to delineate the tract
- Ultrasound — may trace the tubular tract
- CT/MRI — for complex or recurrent cases
4. Surgical Treatment: The Sistrunk Procedure
This is the definitive treatment for both TDC and thyroglossal fistula, first described by Walter Sistrunk in 1920 (published 1928). He recognized the high recurrence rate from simple excision and identified the embryological reason — the intimate relationship of the duct with the hyoid bone and the tract's continuation to the tongue base.
The Procedure Includes:
- Complete excision of the cyst (or fistulous tract)
- Resection of the central portion (body) of the hyoid bone — this is essential because the tract is intimately associated with or passes through the hyoid, and residual hyoid periosteum leads to recurrence
- Excision of the tract from below the hyoid continuing superiorly as a core of muscle (genioglossus) up to the foramen cecum at the base of the tongue — ensuring no duct epithelium is left behind
- Ligation at the foramen cecum — a pharyngotomy may be required; a modification of the procedure omits formal pharyngotomy
Outcomes
| Procedure | Recurrence Rate |
|---|---|
| Simple cyst excision | 40–50% |
| Modified Sistrunk procedure | ~5.8% |
| Full Sistrunk with pharyngotomy | <5% |
Factors predictive of recurrence:
- More than 2 episodes of infection prior to surgery
- Age under 2 years at time of surgery
- Inadequate initial operation (especially failure to include hyoid body)
- Prior incision and drainage
Surgical Illustration
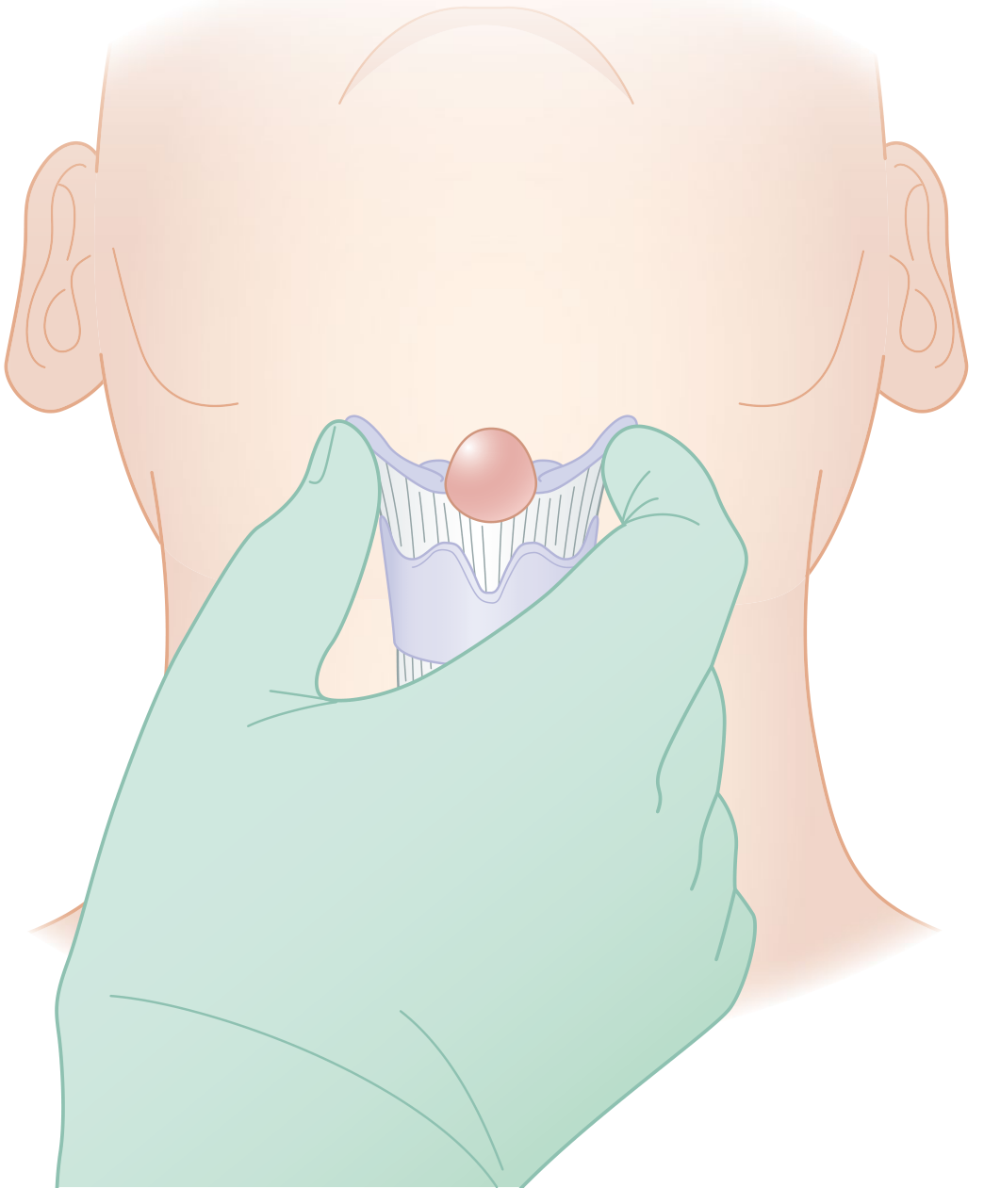
(The illustration shows the cyst held within the U-shaped central hyoid, demonstrating the en-bloc excision principle of the Sistrunk procedure)
Management of Infected TDC/Fistula
- Acute phase: IV/oral antibiotics; if fluctuant → incision and drainage (not definitive excision)
- Definitive surgery: Defer until full resolution of acute inflammation (usually 4–6 weeks). Operating through inflamed tissue dramatically increases recurrence risk and wound complications
5. Important Differentials (Midline Neck Masses)
| Condition | Features distinguishing from TDC |
|---|---|
| Dermoid cyst | Also midline; does NOT move with tongue protrusion; doughy consistency; contains keratin |
| Submental lymph node | Firm, may be bilateral; no movement with tongue |
| Lingual thyroid | Pinkish-red submucosal mass at tongue base; no normal thyroid elsewhere |
| Ranula | Floor of mouth, not neck |
| Lipoma | Soft, lobulated, does not move with tongue |
| Midline ectopic thyroid | No movement sign; radionuclide scan confirms thyroid tissue |
6. Malignancy in TDC
- Incidence: ~1% of all TDCs
- Most common histotype: papillary thyroid carcinoma (>90%); rarely squamous cell carcinoma, Hurthle cell carcinoma
- Suspect if: solid or mixed echogenicity on US, rapid growth, hard consistency, associated cervical lymphadenopathy, calcifications on imaging
- Management: Sistrunk procedure is sufficient for most intrinsic TDC carcinomas. Thyroidectomy is not automatically required but the thyroid should be closely evaluated and monitored lifelong given the 6.6% risk of concurrent microcarcinoma in the thyroid gland (Cummings Otolaryngology, 2024)
Key Summary Points
| TDC | Thyroglossal Fistula | |
|---|---|---|
| Cause | Persistence of thyroglossal duct remnant | Secondary to rupture/drainage of infected TDC |
| Position | Midline, subhyoid (80%) | Median plane, anterior neck |
| Key sign | Moves up with tongue protrusion | Midline discharge; tract moves with swallowing |
| Histology | Pseudostratified columnar/stratified squamous epithelium | Same; lined by duct remnant epithelium |
| Treatment | Sistrunk procedure | Sistrunk procedure (tract excision in continuity with hyoid) |
| Recurrence without proper surgery | 40–50% | High without hyoid excision |
Sources: — The Developing Human: Clinically Oriented Embryology | — Langman's Medical Embryology | — Sabiston Textbook of Surgery (21e) | — Cummings Otolaryngology Head and Neck Surgery | — Schwartz's Principles of Surgery (11e) | — Robbins, Cotran & Kumar Pathologic Basis of Disease
Recent evidence: Two 2025 systematic reviews are available:
- Thyroglossal duct cyst carcinomas — systematic review (PMID 40347268) — confirms Sistrunk as definitive treatment; thyroidectomy not mandated by TDC carcinoma alone
- Diagnostic utility of thyroid scan and ultrasound in TDC management (PMID 40757114) — supports routine ultrasound; nuclear scan reserved for equivocal cases
Ménière’s disease in Detail
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching PubMed
"Meniere disease" AND treatment
Reading File
Loading Image
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material. Let me compile the full, detailed answer.
Ménière Disease — A Detailed Review
1. Historical Background
Prosper Ménière first described this symptom complex in 1861, correctly proposing the inner ear (labyrinth) as the pathologic site — a landmark departure from the then-prevailing belief that all vertigo was of central origin. He initially attributed the pathophysiology to intralabyrinthine hemorrhage, which has since been disproven. Prior to Ménière's description, "Ménière disease" was used loosely as a generic term for any peripheral vertigo. The histologic hallmark — endolymphatic hydrops — was not demonstrated until 1938 by Hallpike and Cairns. — Cummings Otolaryngology Head and Neck Surgery
2. Definition
Ménière disease (MD) is a disorder of the inner ear defined by recurrent, spontaneous episodes of vertigo associated with fluctuating sensorineural hearing loss (SNHL), tinnitus, and aural fullness. It is considered the clinical expression of idiopathic endolymphatic hydrops — the accumulation of excess endolymph within the membranous labyrinth.
3. Epidemiology
| Parameter | Data |
|---|---|
| Incidence (USA) | ~1 in 500 persons |
| Incidence (UK) | ~13.1 per 100,000 person-years |
| Gender | Approximately equal; slight female preponderance in some series |
| Race | More prevalent in Caucasians |
| Age of peak onset | 4th and 5th decades (but can occur at any age) |
| Bilaterality | True incidence ~19–24%; up to 50% within 20 years in some series |
| Familial occurrence | 10–20% of cases; autosomal dominant pattern suggested |
Note: Wide published variation (17–513/100,000) largely reflects inconsistent diagnostic criteria. — Cummings Otolaryngology
4. Pathogenesis & Pathophysiology
4a. Anatomy of Endolymph
The inner ear membranous labyrinth contains two fluid compartments:
- Endolymph — high K⁺, low Na⁺; produced by the stria vascularis (cochlea) and dark cells (vestibular labyrinth)
- Perilymph — high Na⁺, low K⁺; resembles extracellular fluid
Endolymph circulates in a radial and longitudinal fashion and is absorbed by the endolymphatic sac in the posterior fossa.
4b. Endolymphatic Hydrops
Endolymphatic hydrops = overaccumulation of endolymph → distension of the membranous labyrinth at the expense of the perilymphatic space.
The prevailing theory is inadequate reabsorption by the endolymphatic sac. Supporting evidence includes:
- Surgical disruption of the endolymphatic sac in animals consistently produces hydrops
- Perisaccular fibrosis and decreased endolymphatic duct size found in affected temporal bones
- CT and MRI show hypoplasia of the endolymphatic sac/duct and reduced vestibular aqueduct visualization in MD patients
- MRI after gadolinium shows enhancement of the endolymphatic sac and perilymphatic space
- These anatomic variations develop by age 3 and may represent a predisposing factor
Important caveat: Endolymphatic hydrops is a necessary but not sufficient finding — hydrops is also found post-labyrinthitis, otitis media, head trauma, mumps, and meningitis. Not all subjects with hydrops on temporal bone studies had clinical Ménière disease. — Cummings Otolaryngology
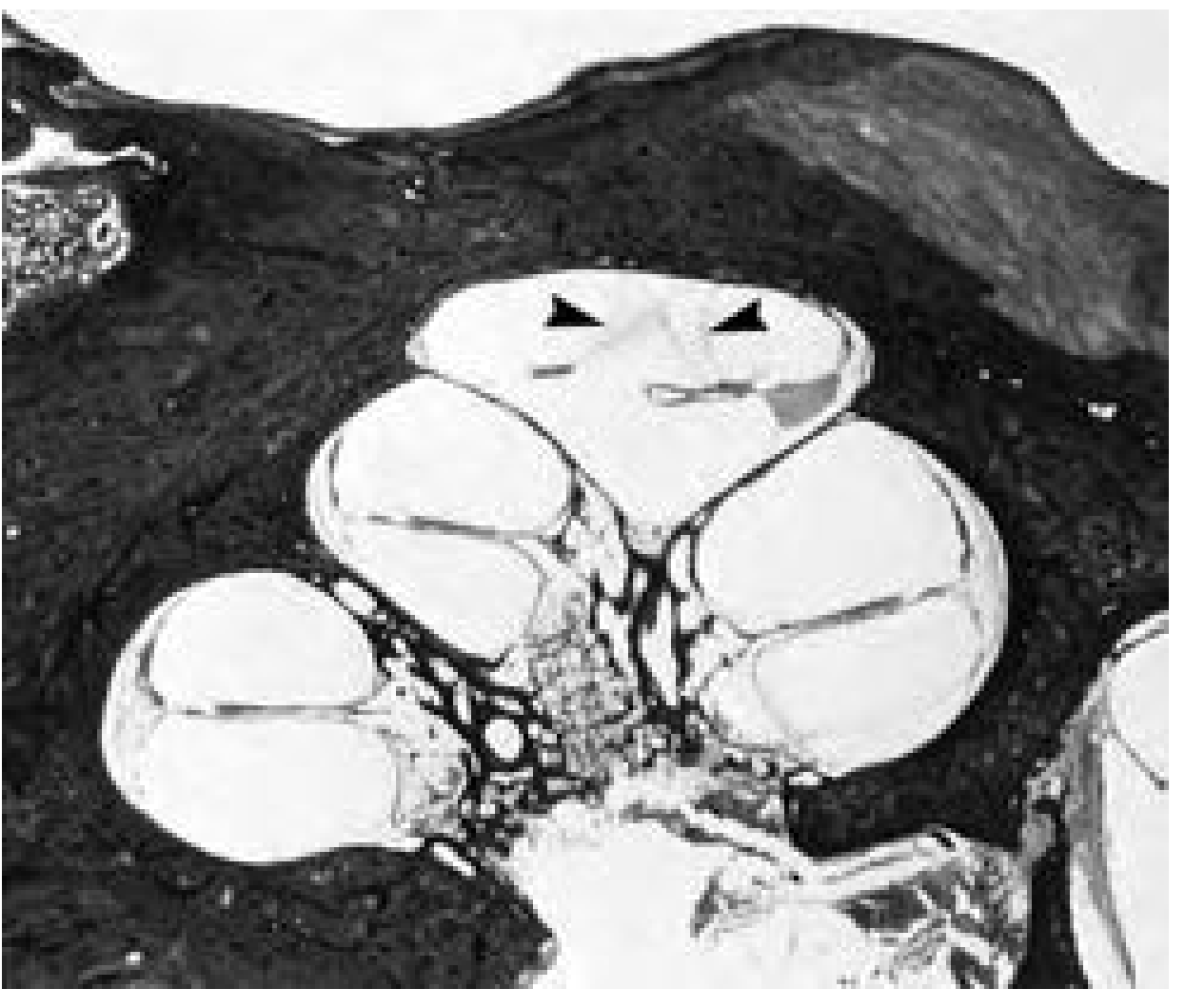
4c. How Hydrops Causes Symptoms
Two competing theories explain the episodic nature of attacks:
- Membrane rupture theory — Increased endolymphatic pressure causes rupture of the membranous labyrinth (Reissner membrane), allowing K⁺-rich endolymph to flood into the perilymph → depolarization block of cochlear and vestibular neurons → sudden deafness and vertigo. As the rupture heals and ionic gradients restore, symptoms resolve
- Pressure/distortion theory — Mechanical distortion of the sensory epithelium and hair cells by pressure fluctuations alters their firing pattern
4d. Etiology of Idiopathic Endolymphatic Hydrops
The underlying cause remains elusive despite 70+ years of research. Proposed contributing mechanisms include:
- Impaired endolymph absorption (dysfunctional endolymphatic sac)
- Autoimmune mechanisms — association with HLA-B8/DR3 and Cw7; familial cases linked to autoimmune disease
- Viral/infective — prior herpes virus infection postulated in some cases
- Migraine — strong association, especially in familial MD
- Gluten sensitivity — recently implicated
- Anatomic predisposition — small vestibular aqueduct
- Genetic factors — autosomal dominant, recessive, and mitochondrial inheritance described; no specific gene identified yet
When a known disease causing hydrops is identified (syphilis, trauma, autoimmune disease, labyrinthitis, etc.), the diagnosis is secondary endolymphatic hydrops — not Ménière disease, which is strictly idiopathic.
5. Clinical Features
5a. The Classic Tetrad (AAO-HNS Criteria)
| Symptom | Details |
|---|---|
| 1. Episodic vertigo | Spontaneous, rotational; lasts 20 min to 24 hours (usually 1–6 hrs); associated nausea, vomiting, diaphoresis; no loss of consciousness |
| 2. Sensorineural hearing loss | Fluctuating, initially affects low frequencies; may progress over years to moderate–severe pan-frequency loss; profound loss is rare |
| 3. Tinnitus | Usually low-pitched, roaring or hissing; unilateral; worsens during attacks |
| 4. Aural fullness | Pressure sensation in the affected ear; often a premonitory symptom heralding an impending attack |
5b. Symptom Pattern and Natural History
- Hallmark: Dramatic variability and unpredictability
- Attacks may cluster and then remit for months to years
- Between attacks, patients may be entirely asymptomatic, or describe chronic disequilibrium/lightheadedness
- ~60% achieve spontaneous remission over time
- ~85% unilateral at onset; bilateral disease develops in ~20–30%, often within 36 months of second-ear involvement; up to 50% over 20 years
- Hearing loss: fluctuates early; tends to progressively stabilize at a moderate flat loss over years
5c. Important Variants
1. Lermoyez Syndrome
- Reversed Ménière: tinnitus, hearing loss, and aural fullness progressively worsen → then suddenly resolve when vertigo occurs
- The vertiginous episode paradoxically relieves the auditory symptoms
- Temporal bone studies showed hydrops and membrane ruptures isolated to basal cochlear turns and saccule
2. Cochlear Hydrops
- Fluctuating SNHL and tinnitus without episodic vertigo
- Considered a forme fruste
3. Vestibular Hydrops
- Episodic vertigo + aural fullness without documented hearing loss
4. Otolithic Catastrophe of Tumarkin (Drop Attacks)
- Sudden, violent fall without warning, loss of consciousness, or associated vertigo
- Thought to represent acute utriculosaccular dysfunction → erroneous vertical gravity reference → inappropriate vestibulospinal reflex → sudden fall
- Patients describe being "pushed" or "thrown" to the ground
- Occur in 2–6% of MD patients; tend to cluster then spontaneously remit
- Injury is common due to the abrupt, unprotected fall
5. Delayed Endolymphatic Hydrops
- Profound SNHL occurs in one ear (from previous infection, etc.)
- Years later, symptoms of endolymphatic hydrops develop either in the same ear (ipsilateral) or opposite ear (contralateral)
6. Diagnostic Criteria (AAO-HNS 1995)
The American Academy of Otolaryngology – Head and Neck Surgery (AAO-HNS) criteria stratify certainty of diagnosis:
| Category | Criteria |
|---|---|
| Possible | Episodic vertigo without documented hearing loss, OR SNHL (fluctuating/fixed) with dysequilibrium but no definite episodes. Other causes excluded |
| Probable | One definitive episode of vertigo + audiometrically documented hearing loss on ≥1 occasion + tinnitus or aural fullness. Other causes excluded |
| Definite | ≥2 definitive spontaneous episodes of vertigo each lasting ≥20 min + audiometrically documented hearing loss on ≥1 occasion + tinnitus or aural fullness in the suspected ear. Other causes excluded |
| Certain | Definite Ménière disease + histopathologic confirmation (temporal bone) |
Definitive vertigo episode = spontaneous rotational vertigo lasting ≥20 min, not attributable to another cause
7. Investigations
7a. Audiometry (Pure Tone Audiogram)
- Low-frequency SNHL (250–1000 Hz) — characteristic early finding
- Carhart notch at 2000 Hz occasionally
- Fluctuating pattern — repeat audiograms over time are essential
- No tone decay (distinguishes from retrocochlear pathology)
- Recruitment present (loudness recruitment on SISI test)
7b. Electrocochleography (ECoChG)
- Measures cochlear electrical potentials in response to sound
- Elevated summating potential/action potential (SP/AP) ratio (>0.4) suggests endolymphatic hydrops
- Useful for confirming cochlear hydrops but not universally abnormal
7c. Electronystagmography (ENG) / Videonystagmography (VNG)
- Between attacks: may show reduced caloric response on the affected side (reflects vestibular hypofunction)
- During attacks: spontaneous horizontal nystagmus — direction changes over the course of the attack (not useful for lateralization)
- Head thrust test (video head impulse test): usually normal between attacks
7d. Glycerol Test (Dehydration Test)
- Oral glycerol 1.2 mL/kg + equal volume saline (osmotic diuretic)
- Positive test: ≥10 dB improvement in PTA or ≥12% improvement in speech discrimination score within 1–3 hours
- A positive result supports the diagnosis of endolymphatic hydrops and implies reversibility
- Useful as a diagnostic adjunct; not universally performed
7e. VEMP (Vestibular-Evoked Myogenic Potentials)
- Cervical VEMP (cVEMP) tests the saccule and inferior vestibular nerve
- Ocular VEMP (oVEMP) tests the utricle and superior vestibular nerve
- Abnormal in advanced MD; can help lateralize the lesion
7f. Imaging
- MRI with gadolinium — primary role is to exclude a vestibular schwannoma or endolymphatic sac tumor
- Delayed gadolinium MRI (intravenous or intratympanic gadolinium with high-Tesla MRI): can directly visualize endolymphatic hydrops, but not yet in standard diagnostic criteria
- CT temporal bone: reduced vestibular aqueduct width/pneumatization supports diagnosis
8. Treatment
Treatment is directed at: (1) aborting acute attacks, (2) reducing attack frequency and severity, and (3) managing progressive hearing loss. No treatment has been proven to arrest hearing loss.
8a. Acute Attack Management
- Vestibular suppressants: Meclizine, dimenhydrinate, promethazine
- Antiemetics: Prochlorperazine, ondansetron
- Benzodiazepines: Lorazepam (acute sedation and vestibular suppression)
- Rest in a quiet, dark environment
- IV hydration if vomiting is severe
8b. Medical Prophylaxis (First-Line)
Low-salt diet + diuretics — most widely accepted medical regimen:
- Sodium restriction: <2 g/day (reduces endolymph production by altering body fluid homeostasis)
- Diuretics: Hydrochlorothiazide/triamterene (Dyazide) most common; acetazolamide also used
- First proposed by Furstenberg; widely accepted despite limited level 1 evidence for hearing preservation
Additional medical measures:
- Betahistine (histamine H₃ antagonist/weak H₁ agonist): improves cochlear microcirculation, enhances endolymph absorption; widely used in Europe (16–48 mg TID); evidence remains mixed
- Vasodilators (cinnarizine, nimodipine)
- Caffeine and alcohol restriction
- Stress reduction
8c. Intratympanic Therapies
Intratympanic (IT) Steroids (hearing-sparing):
- Dexamethasone 4 mg/mL or methylprednisolone most commonly used
- Delivered through a tympanostomy tube or by injection across the tympanic membrane
- Vertigo control achieved in ~70–80% of patients in some series
- No proven effect on hearing loss — multiple RCTs have failed to show hearing benefit
- Mechanism: anti-inflammatory + possible mineralocorticoid effects on Na⁺/K⁺ channel activity in the stria vascularis
- 2025 systematic review (PMID 40421807) confirms role of IT gentamicin for refractory cases
Intratympanic Gentamicin (selective chemical vestibular ablation):
- Low-dose gentamicin selectively destroys vestibular hair cells (type I > type II) while relatively sparing cochlear function
- Protocols: single injection, titration (inject until vertigo is controlled or caloric hypofunction confirmed), or multiple fixed-dose injections
- Vertigo control: 80–90% in most series
- Risk of SNHL: 10–30% (dose-dependent)
- Preferred when vertigo is disabling and hearing is already significantly reduced
- 2025 meta-analysis (PMID 40421807) confirms effectiveness for vertigo control
8d. Surgical Treatment
Reserved for patients with refractory vertigo despite ≥6 months of adequate medical therapy. Surgical options stratified by residual hearing:
Hearing-Preserving Procedures
| Procedure | Details |
|---|---|
| Endolymphatic sac surgery | Decompression ± shunting into mastoid or subarachnoid space; theoretically corrects sac physiology. Blinded RCTs comparing it to mastoidectomy alone showed no significant difference in vertigo control or hearing. Placebo effect may account for 50–70% of benefit |
| Vestibular nerve section (selective) | Cuts the vestibular nerve via retrolabyrinthine, retrosigmoid, or middle cranial fossa approach; most effective hearing-sparing procedure for vertigo; ~90–95% control. Major intracranial surgery with associated risks |
Hearing-Destructive Procedures
| Procedure | Details |
|---|---|
| Labyrinthectomy (surgical) | Complete destruction of labyrinthine end organ; reserved for non-serviceable hearing (<30 dB SRT); near-complete vertigo control (~95%) |
| Cochleosacculotomy | Indicated in elderly patients with disabling vertigo, poor hearing, and preserved vestibular function; performed under local anesthesia |
8e. Staging and Outcome Reporting (AAO-HNS 1996)
Vertigo control is graded by the AAO-HNS numeric value formula:
Ratio = (mean definitive spells/month post-therapy) ÷ (mean spells/month pre-therapy) × 100
| Class | Numeric Value | Meaning |
|---|---|---|
| A | 0 | Complete control |
| B | 1–40 | Substantial control |
| C | 41–80 | Limited control |
| D | 81–120 | Insignificant control |
| E | >120 | Worse / Secondary treatment initiated |
Important note (2024 meta-analysis, PMID 38101129): The large placebo effect in Ménière disease RCTs (up to 50–70% improvement in placebo arms) makes it extremely difficult to interpret therapeutic efficacy. The unpredictable, fluctuating nature of the disease further confounds study design.
9. Differential Diagnosis
| Condition | Key Distinguishing Features |
|---|---|
| BPPV | Positional vertigo, lasts seconds (<1 min), no hearing loss, positive Dix-Hallpike |
| Vestibular migraine | Migraine headache, photophobia; vertigo may last minutes to days; no progressive hearing loss |
| Vestibular neuritis | Single prolonged attack (days); no hearing loss; severe canal paresis on ENG |
| Acoustic neuroma | Gradual unilateral SNHL, tinnitus; rarely episodic vertigo; confirmed on MRI |
| Autoimmune inner ear disease | Rapidly progressive bilateral SNHL; responds to steroids; serum autoantibody screen |
| Syphilitic labyrinthitis | FTA-ABS positive; bilateral involvement; hydrops on temporal bone |
| Superior canal dehiscence | Pressure-induced vertigo; Tullio phenomenon; CT shows dehiscence |
| Perilymphatic fistula | History of barotrauma or straining; pressure-induced vertigo |
10. Prognosis
- Spontaneous remission of vertigo attacks occurs in ~60% of patients over time
- Hearing loss progresses over the years in most, though at variable rates; profound loss is rare
- Eventually, the attacks often "burn out" as vestibular function is destroyed — but the patient is left with chronic imbalance and significant hearing loss
- Bilateral disease worsens the prognosis considerably
11. Associated Conditions
- Migraine — strong bidirectional association, especially in familial cases
- Autoimmune disease — association with HLA-B8/DR3; autoimmune inner ear disease may represent a spectrum
- Thyroid disease — 2025 meta-analysis (PMID 40134376) confirms a significant association between Ménière disease and thyroid pathology
- Anxiety and depression — common comorbidities given the unpredictable disabling nature of attacks
Sources:
— Cummings Otolaryngology Head and Neck Surgery (primary source, chapters 158 & 167)
— Bradley and Daroff's Neurology in Clinical Practice
— K.J. Lee's Essential Otolaryngology
— Goldman-Cecil Medicine
Recent evidence:
- IT gentamicin meta-analysis 2025 (PMID 40421807) — confirms gentamicin effectiveness for refractory vertigo
- Placebo effect in Ménière RCTs (PMID 38101129) — highlights the significant placebo response confounding trial interpretation
- Ménière disease & thyroid disease association (PMID 40134376) — 2025 meta-analysis confirming association
This is a shared conversation. Sign in to Orris to start your own chat.