Lung Tumors - WHO classification, etiopathogenesis, morphology
2nd yr mbbs patho refer harsh mohan
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
lung carcinoma histology adenocarcinoma squamous cell carcinoma small cell carcinoma
Loading Image
Searching Images
small cell carcinoma lung oat cell histology neuroendocrine
I have all the core content needed. Let me now compile the comprehensive answer based on Robbins Basic Pathology (the closest equivalent to Harsh Mohan for 2nd year MBBS) with the full WHO classification, etiopathogenesis, and morphology details from the textbook content I retrieved.
Lung Tumors — WHO Classification, Etiopathogenesis & Morphology
Based on Robbins & Kumar Basic Pathology, 11e and Robbins & Cotran Pathologic Basis of Disease (2021 WHO Classification)
Overview
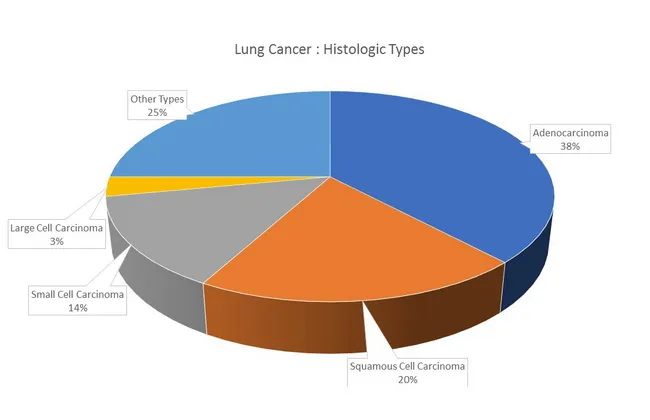
~95% of primary lung tumors are carcinomas. The remaining 5% include carcinoids, mesenchymal malignancies (fibrosarcoma, leiomyosarcoma), lymphomas, and benign lesions. The most common benign tumor is the hamartoma — a 1–4 cm "coin lesion" on CXR composed of mature cartilage, fat, fibrous tissue, and blood vessels (actually a benign neoplasm despite its name).
WHO Classification (2021) — Malignant Epithelial Lung Tumors
| Category | Subtypes |
|---|---|
| Adenocarcinoma | Acinar, papillary, micropapillary, solid, lepidic-predominant, mucinous |
| Squamous Cell Carcinoma | — |
| Large Cell Carcinoma | — |
| Neuroendocrine Carcinoma | Small cell carcinoma, Carcinoid tumor (typical & atypical), Large cell neuroendocrine carcinoma |
| Mixed Carcinomas | Adenosquamous, Small cell + other types |
| Other variants | Sarcomatoid carcinoma |
Historic Subdivision (Clinically Important)
| Group | Includes | Relevance |
|---|---|---|
| SCLC (Small Cell Lung Cancer) | Small cell carcinoma | Responds to chemo but rarely resectable |
| NSCLC (Non-Small Cell) | Adeno + Squamous + Large cell | More often resectable; targeted therapy applicable |
Epidemiology
- Leading cause of cancer-related death in men AND women (since 1987) in high-resource countries
- ~237,000 new cases / ~130,000 deaths per year (USA, 2022)
- Peak incidence: 5th–6th decade
-
50% have distant metastases at diagnosis; 25% have regional nodal involvement
- 5-year survival: ~20% overall; ~50% if localized
- Adenocarcinoma has now replaced squamous cell carcinoma as the most common type — due to decline in tobacco use and rise in peripheral adenocarcinomas in women and never-smokers
Etiopathogenesis
1. Tobacco Smoking (Most Important)
- 85% of lung carcinomas are attributable to smoking
- Squamous cell and small cell carcinomas have the strongest association
- Adenocarcinoma also associated, but less strongly
- Carcinogens in tobacco: polycyclic aromatic hydrocarbons, nitrosamines → cause TP53 mutations, KRAS mutations
- Risk: 20× higher in heavy smokers; dose-dependent; risk persists even after cessation
- Passive (secondhand) smoke also increases risk
2. Industrial/Occupational Carcinogens
| Carcinogen | Tumor Type |
|---|---|
| Asbestos | Mesothelioma + lung carcinoma (synergistic with smoking — 55× increased risk) |
| Uranium, radon | Small cell carcinoma (miners) |
| Arsenic, chromium, nickel | Carcinoma |
| Bis-chloromethyl ether | Small cell carcinoma |
3. Air Pollution
- Polycyclic hydrocarbons in polluted air — modest but real risk
4. Molecular/Genetic Alterations
| Mutation | Tumor Type | Targeted Therapy |
|---|---|---|
| KRAS (30% of adenoCA) | Adenocarcinoma (smokers) | Limited targets |
| EGFR (10–15% NSCLC; >50% in never-smokers) | Adenocarcinoma | Erlotinib, Gefitinib |
| ALK rearrangement (5%) | Adenocarcinoma (young, never-smoker) | Crizotinib, Alectinib |
| ROS1 rearrangement | Adenocarcinoma | Crizotinib |
| TP53 mutations | All types (especially SCLC) | — |
| RB deletion | SCLC (near-universal) | — |
| MYC amplification | SCLC | — |
5. Precursor Lesions (Stepwise Carcinogenesis)
- Squamous cell CA: Normal epithelium → squamous metaplasia → dysplasia → carcinoma in situ → invasive carcinoma (due to repeated tobacco carcinogen exposure)
- Adenocarcinoma: Atypical adenomatous hyperplasia (AAH) → Adenocarcinoma in situ (AIS, formerly BAC) → Minimally invasive adenocarcinoma → Invasive adenocarcinoma
Morphology of Individual Types
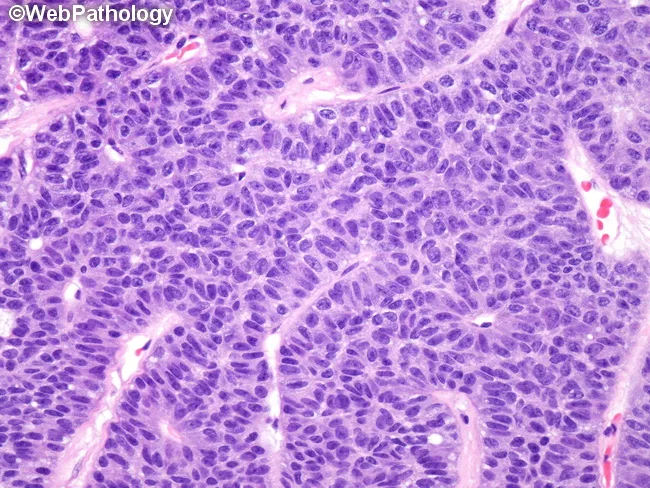
1. Adenocarcinoma
Most common overall (~38%); most common in women, never-smokers, and persons <45 years
Location: Peripheral (subpleural), often in scars
Gross: Peripheral gray-white mass; may show pleural puckering
Microscopy:
- Gland-forming, papillary, micropapillary, or solid growth patterns
- Lepidic pattern (formerly BAC): tumor cells grow along pre-existing alveolar walls without stromal invasion — best prognosis
- Mucinous variant: columnar cells with mucin, grow along alveoli, produce "pneumonia-like" consolidation
- Clara cells or type II pneumocytes as cells of origin
- Mucin secretion (PAS/Alcian blue positive)
- IHC: TTF-1 positive, Napsin A positive, CK7 positive
2. Squamous Cell Carcinoma
Second most common (~20%); strongest tobacco association
Location: Central — arises from bronchial epithelium near the hilum
Gross: Central hilar mass → may cause bronchial obstruction → obstructive pneumonitis/atelectasis; cavitation common in large tumors
Microscopy:
- Sheets and nests of polygonal cells
- Keratin pearls (concentric whorls of keratin)
- Intercellular bridges (desmosomes)
- Individual cell keratinization (pink glassy cytoplasm)
- Moderately to well-differentiated tumors show classic squamous features
- IHC: p40 positive, p63 positive, CK5/6 positive
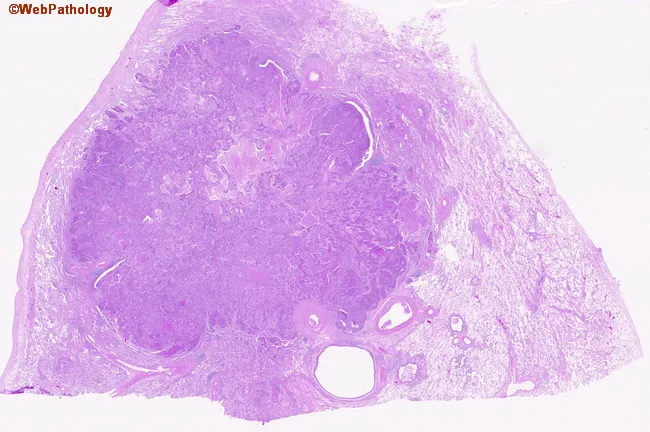
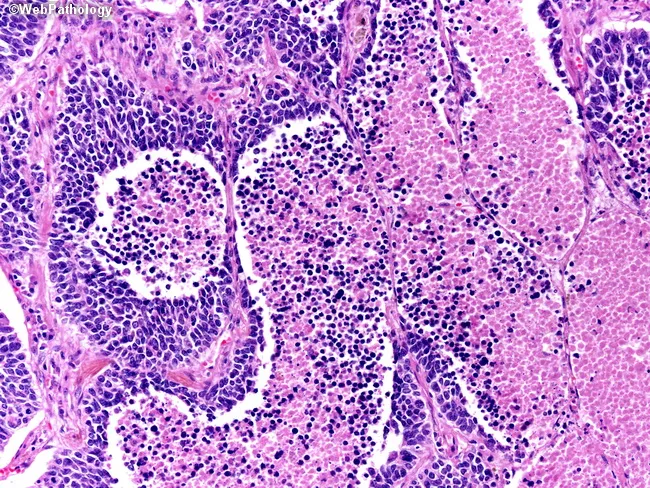
3. Small Cell Carcinoma (SCLC)
~14%; most aggressive; almost always due to smoking
Location: Central (hilar/perihilar); almost always disseminated at diagnosis
Gross: Soft, white, bulky perihilar mass; extensive necrosis; involves bronchial mucosa and submucosa diffusely
Microscopy:
- Oat cell appearance: small (2× the size of lymphocyte), round-to-fusiform cells
- Scant cytoplasm
- Salt-and-pepper chromatin (finely granular, neuroendocrine pattern)
- No visible nucleoli
- Nuclear molding (nuclei indent each other)
- Abundant mitoses and geographic necrosis
- Azzopardi effect: DNA encrustation around vessel walls from necrotic tumor
- Arranged in sheets, ribbons, and nests
- IHC: Synaptophysin+, Chromogranin A+, CD56+, TTF-1 often positive; NSE positive
- Paraneoplastic syndromes are common: SIADH (ectopic ADH), Cushing's (ectopic ACTH), Eaton-Lambert syndrome (anti-VGCC antibodies)
4. Large Cell Carcinoma
~3%; a diagnosis of exclusion — anaplastic tumor that lacks squamous, glandular, or neuroendocrine differentiation
Location: Peripheral
Gross: Large, bulky peripheral mass with necrosis
Microscopy:
- Large polygonal cells with abundant cytoplasm
- Prominent nucleoli, vesicular nuclei
- No keratin pearls, no gland formation
- IHC negative for squamous and adenocarcinoma markers
- Large cell neuroendocrine carcinoma (LCNEC): shows neuroendocrine morphology (organoid nesting, rosettes) + large cells; IHC: Synaptophysin+, Chromogranin+; very aggressive
5. Carcinoid Tumors
Low-grade neuroendocrine tumors (~5% of lung tumors); NOT strongly linked to smoking
Types:
| Typical Carcinoid | Atypical Carcinoid | |
|---|---|---|
| Mitoses | <2/2mm² | 2–10/2mm² |
| Necrosis | Absent | Focal |
| Prognosis | Excellent (>90% 5-yr) | Worse (50–70% 5-yr) |
| Mets | Rare | More frequent |
Gross: Polypoid intraluminal bronchial mass (central type common); cherry-red, well-vascularized
Microscopy:
- Uniform cells arranged in nests, cords, and trabeculae (organoid pattern)
- Round nuclei with salt-and-pepper chromatin
- Scant to moderate cytoplasm
- No significant pleomorphism in typical carcinoid
- IHC: Chromogranin A+, Synaptophysin+, CD56+
Secondary/Metastatic Lung Tumors
- The lung is the most common site of metastasis (hematogenous spread)
- Sources: breast, colon, kidney, uterus, prostate, stomach
- Pattern: multiple bilateral nodules ("cannonball metastases") vs. solitary nodule
- Lymphangitic carcinomatosis: tumor spread along lymphatics → miliary pattern, dyspnea
Summary Table: Key Distinguishing Features
| Feature | Adenocarcinoma | Squamous Cell CA | Small Cell CA | Large Cell CA |
|---|---|---|---|---|
| Frequency | 38% (most common) | 20% | 14% | 3% |
| Location | Peripheral | Central | Central | Peripheral |
| Smoking link | Moderate | Strong | Strongest | Strong |
| Gross | Gray-white peripheral mass | Hilar mass, cavitation | Soft hilar mass, necrosis | Bulky peripheral mass |
| Histology | Glands, lepidic pattern | Keratin pearls, bridges | Oat cells, molding | Large pleomorphic cells |
| Mucin | Yes | No | No | No |
| IHC | TTF-1+, Napsin A+ | p40+, CK5/6+ | Synaptophysin+, CD56+ | Negative for all |
| Metastasis | Late | Late | Early (universal) | Early |
| Paraneoplastic | Hypertrophic osteoarthropathy | Hypercalcemia (PTHrP) | SIADH, ACTH, Eaton-Lambert | — |
| Targeted therapy | EGFR, ALK, ROS1 | — | — | — |
Source: Robbins & Kumar Basic Pathology 11e (equivalent to Harsh Mohan Textbook of Pathology for 2nd year MBBS); 2021 WHO Classification of Thoracic Tumours
Leukemia* - define*, classify*, CML CLL ALL AML- peripheral blood smear, bone marrow findings; Acute Leukemia - FAB classification, cytochemical stains, laboratory findings
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
peripheral blood smear leukemia CML CLL blast cells Auer rods
Reading File
Loading Image
Loading Image
Reading File
Searching Images
AML acute myeloid leukemia Auer rods myeloblasts bone marrow
Searching Images
ALL acute lymphoblastic leukemia lymphoblasts peripheral blood smear bone marrow
I have all the core content needed. Let me now compile the comprehensive, well-structured answer.
Leukemia — Definition, Classification, and Detailed Pathology
Based on Robbins & Kumar Basic Pathology 11e and Robbins & Cotran Pathologic Basis of Disease 10e
Definition
Leukemia is a malignant neoplasm of hematopoietic stem cells or their precursors, characterized by clonal proliferation of abnormal leukocytes that accumulate in the bone marrow and peripheral blood, suppressing normal hematopoiesis.
Key hallmark: Replacement of normal bone marrow → pancytopenia (anemia, thrombocytopenia, neutropenia) despite elevated WBC count.
Classification of Leukemia
By Maturity of Cells
| Acute | Chronic | |
|---|---|---|
| Cell type | Immature blasts (arrested differentiation) | Mature/maturing cells |
| Onset | Abrupt, aggressive | Insidious |
| Prognosis | Rapidly fatal without treatment | Indolent initially |
| Blasts in marrow | ≥20% (WHO) | <20% |
By Cell Lineage
LEUKEMIA
├── MYELOID
│ ├── Acute Myeloid Leukemia (AML)
│ └── Chronic Myeloid Leukemia (CML)
└── LYMPHOID
├── Acute Lymphoblastic Leukemia (ALL)
└── Chronic Lymphocytic Leukemia (CLL)
I. CHRONIC MYELOID LEUKEMIA (CML)
Definition & Epidemiology
- Myeloproliferative neoplasm arising from a pluripotent hematopoietic stem cell
- Adults aged 25–60 years; peak: 4th–5th decade
- ~4,500 new cases/year (USA)
Etiopathogenesis
- Hallmark: Philadelphia chromosome (Ph¹) — present in >90% of cases
- Reciprocal translocation t(9;22)(q34;q11)
- Creates BCR::ABL1 fusion gene → encodes BCR-ABL tyrosine kinase (210 kDa)
- BCR-ABL constitutively activates RAS and other pro-survival signals, making cells growth factor–independent
- Does NOT block differentiation → cells proliferate AND mature (explains high WBC with mature cells)
- Remaining 5%: complex/cryptic rearrangements — detected by FISH or PCR
Peripheral Blood Smear (CML)
- Markedly elevated WBC — often >100,000/μL
- Entire spectrum of granulocytic maturation: myeloblasts, promyelocytes, myelocytes, metamyelocytes, band forms, mature neutrophils
- Basophilia and eosinophilia — characteristic; distinguishes from leukemoid reaction
- Thrombocytosis (platelets elevated)
- Anemia (normocytic normochromic)
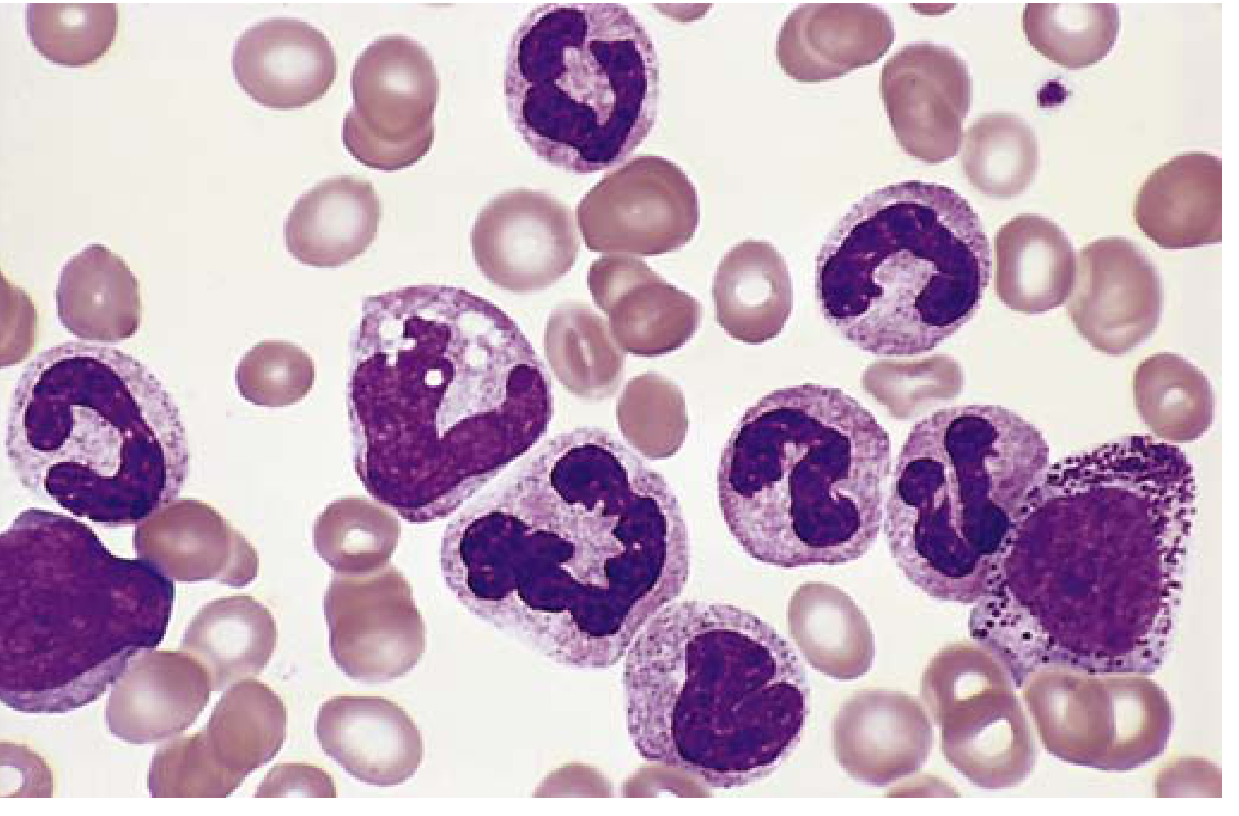
Bone Marrow (CML)
- Hypercellular (markedly); virtual disappearance of fat spaces
- Increased granulocytic and megakaryocytic precursors
- Blasts <10% in chronic phase
- Leukocyte alkaline phosphatase (LAP) score: LOW (vs. high in leukemoid reaction — key differentiator)
Key Distinguishing Feature vs. Leukemoid Reaction
| Feature | CML | Leukemoid Reaction |
|---|---|---|
| LAP score | Low | High |
| Ph chromosome | Present | Absent |
| Basophilia | Present | Absent |
| Cause | Neoplasm | Infection/stress |
Phases of CML
- Chronic phase (3–5 years): Mature granulocytes, <10% blasts
- Accelerated phase: 10–19% blasts, increasing basophilia
- Blast crisis: ≥20% blasts → transforms to AML (70%) or ALL (30%)
Treatment
- Imatinib (Gleevec) — BCR-ABL tyrosine kinase inhibitor; revolutionary targeted therapy
II. CHRONIC LYMPHOCYTIC LEUKEMIA (CLL)
Definition & Epidemiology
- Neoplasm of mature CD5+ B cells (arrested at a stage of naive B-cell development)
- Most common leukemia of adults in the Western world
- Predominantly older adults; more common in men
- CLL and SLL (Small Lymphocytic Lymphoma) are the same disease:
- CLL: peripheral blood lymphocytes >5,000/μL
- SLL: predominantly lymph node involvement
Etiopathogenesis
- BCL-2 overexpression (from 13q deletion → loss of miRNA negative regulators) → inhibits apoptosis → tumor survival > proliferation
- BTK (Bruton Tyrosine Kinase) signaling via surface immunoglobulin (BCR) promotes growth/survival
- Immune dysregulation: hypogammaglobulinemia → susceptibility to infections
- 15% develop autoimmune hemolytic anemia (warm autoantibodies — made by non-malignant bystander B cells)
Peripheral Blood Smear (CLL)
- Absolute lymphocytosis (sustained >5,000/μL)
- Small, mature-appearing lymphocytes with:
- Dark round nuclei
- Condensed/clumped ("soccer ball") chromatin
- Scant cytoplasm
- No prominent nucleoli
- Smudge cells (basket cells) — pathognomonic: fragile leukemic lymphocytes crushed during smear preparation
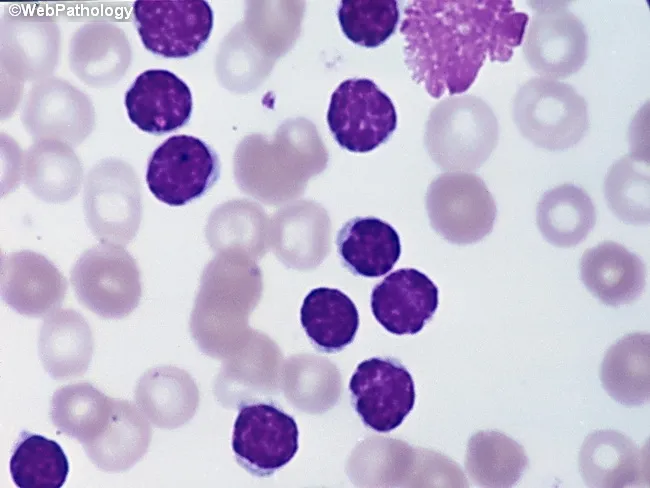
Bone Marrow (CLL)
- Interstitial or nodular lymphocytic infiltration initially; later diffuse replacement
- Sheets of small lymphocytes identical to peripheral blood
- Residual hematopoietic elements reduced
Lymph Node (CLL/SLL)
- Diffuse effacement by small lymphocytes
- Proliferation centers (pseudofollicles) — pale foci of larger, actively dividing cells — pathognomonic for CLL/SLL
IHC: CD5+, CD19+, CD23+, CD20 (dim), surface Ig (dim)
Prognosis Markers
| Marker | Poor Prognosis |
|---|---|
| 17p deletion (TP53) | Yes — resistant to chemotherapy |
| 11q deletion | Yes |
| Unmutated IgVH | Yes |
| ZAP-70 expression | Yes |
| 13q deletion | Favorable (if sole abnormality) |
III. ACUTE LYMPHOBLASTIC LEUKEMIA (ALL)
Definition & Epidemiology
- Neoplasm of immature lymphoid precursors (lymphoblasts) — pre-B or pre-T cells
- Most common cancer in children (peak: 3 years for B-ALL; adolescence for T-ALL)
- ~2,500 new cases/year in USA
- 85% are B-ALL (bone marrow/blood); 15% T-ALL (often thymic mass in adolescent males)
Etiopathogenesis
- Mutations in transcription factors essential for lymphoid development:
- B-ALL: PAX5, TCF3, ETV6, RUNX1, BCR::ABL1 (poor prognosis), KMT2A (11q23)
- T-ALL: NOTCH1 mutations (most common)
- Leads to maturation arrest + increased self-renewal
- Down syndrome: 10–20× increased risk of ALL (often B-ALL)
- Cytogenetic subtypes (prognostically important):
| Abnormality | Prognosis |
|---|---|
| Hyperdiploidy (>50 chromosomes) | Favorable |
| t(12;21) ETV6::RUNX1 | Favorable (most common in children) |
| t(9;22) BCR::ABL1 ("Ph+ ALL") | Poor (adult ALL, ~25%) |
| t(4;11) KMT2A::AFF1 | Poor (infant ALL) |
| Hypodiploidy | Very poor |
Peripheral Blood Smear (ALL)
- Variable WBC — may be low, normal, or high
- Lymphoblasts: medium-sized cells with:
- High N:C ratio
- Scant agranular pale-blue cytoplasm
- Finely stippled/homogeneous chromatin ("immature" pattern)
- Nucleoli present (1–2, inconspicuous to prominent)
- Round/oval or convoluted nuclei (T-ALL)
- Anemia (normocytic), thrombocytopenia
Bone Marrow (ALL)
- Hypercellular with diffuse replacement by lymphoblasts
- ≥20% lymphoblasts (often 80–100%)
- Suppression of all normal hematopoietic lineages
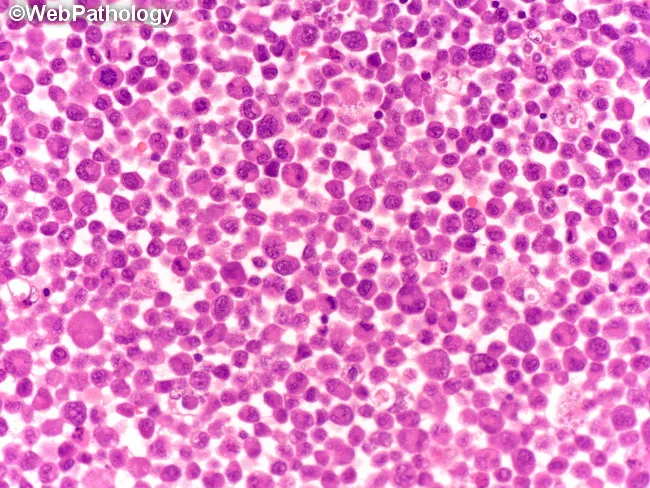
IHC/Immunophenotype
- TdT (Terminal deoxynucleotidyl Transferase) positive — hallmark of ALL (marks lymphoid immaturity); negative in AML
- B-ALL: CD10+, CD19+, CD20 (variable)
- T-ALL: CD2+, CD3+, CD5+, CD7+, CD4/CD8 double positive (thymic stage)
IV. ACUTE MYELOID LEUKEMIA (AML)
Definition & Epidemiology
- Neoplasm of immature myeloid progenitors (myeloblasts) — impaired differentiation → accumulation of blasts in marrow
- Incidence rises with age; peak after 60 years; also occurs in children
- ~13,000 new cases/year in USA
Etiopathogenesis — Driver Mutations (4 categories)
- Transcription factor mutations (most important):
- t(8;21) → RUNX1::RUNX1T1 — blocks myeloid differentiation
- inv(16) → CBFB::MYH11 — same pathway
- t(15;17) → PML::RARA — Acute Promyelocytic Leukemia (APL/AML-M3); ATRA therapy blocks this fusion protein → differentiation induced
- Growth factor/RAS signaling mutations: FLT3 (ITD — poor prognosis), RAS, KIT
- Epigenetic regulator mutations: DNMT3A, TET2, IDH1/2
- Tumor suppressor mutations: TP53, NPM1
Risk factors: Prior radiation/chemotherapy (especially alkylating agents), myelodysplastic syndrome, Down syndrome, Fanconi anemia, benzene exposure
WHO Classification of AML (2022) — Simplified
| Class | Example/Prognosis |
|---|---|
| AML with t(8;21) RUNX1::RUNX1T1 | Favorable |
| AML with inv(16) CBFB::MYH11 | Favorable |
| APL with t(15;17) PML::RARA | Very favorable (ATRA-responsive) |
| AML with KMT2A rearrangement t(11q23) | Poor |
| AML with mutated NPM1 | Favorable |
| AML with myelodysplasia-related changes | Poor |
| Therapy-related AML | Poor |
| AML with FLT3 mutations | Intermediate→Poor |
FAB Classification of AML (French-American-British, 1976)
| FAB Subtype | Name | Key Morphology |
|---|---|---|
| M0 | Minimally differentiated AML | Blasts with no granules; MPO negative by light microscopy; positive by IHC/EM |
| M1 | AML without maturation | >90% myeloblasts; few granules; Auer rods occasionally |
| M2 | AML with maturation | Myeloblasts + some maturation; Auer rods frequent; t(8;21) |
| M3 | Acute Promyelocytic Leukemia (APL) | Hypergranular promyelocytes; Faggot cells (bundles of Auer rods); t(15;17); DIC common |
| M4 | Acute Myelomonocytic Leukemia | Both myeloid + monocytic differentiation; serum/urine lysozyme ↑ |
| M4Eo | AML M4 with eosinophilia | Like M4 + abnormal eosinophils; inv(16) |
| M5 | Acute Monocytic Leukemia | ≥80% monocytic cells; gum infiltration; skin involvement |
| M6 | Erythroleukemia (Di Guglielmo) | ≥50% erythroid + ≥20% myeloblasts; bizarre erythroblasts |
| M7 | Acute Megakaryoblastic Leukemia | Megakaryoblasts; marrow fibrosis; Down syndrome association |
Auer Rods — pathognomonic of AML (never in ALL). Pink needle-like crystalline inclusions in the cytoplasm of blasts. Composed of fused azurophilic granules (condensed MPO). Especially prominent and numerous in APL (M3) — "faggot cells."
Peripheral Blood Smear (AML)
- Myeloblasts with:
- Large cells, moderate to abundant cytoplasm
- Fine/open chromatin
- Prominent nucleoli (2–5) — key feature
- Auer rods (M1, M2, M3) — pathognomonic
- Cytoplasmic azurophilic granules
- Anemia, thrombocytopenia, neutropenia
- WBC variable (may be high, normal, or low — "aleukemic leukemia")
Bone Marrow (AML)
- Hypercellular; virtual replacement by myeloblasts
- ≥20% blasts required for AML diagnosis (WHO)
- Auer rods in blasts
- Suppression of normal erythroid, megakaryocytic, and lymphoid elements
Cytochemical Stains (Acute Leukemia Diagnosis)
| Stain | AML (Myeloid) | ALL (Lymphoid) |
|---|---|---|
| MPO (Myeloperoxidase) | POSITIVE ✓ (M1–M3 strongly +) | Negative |
| Sudan Black B (SBB) | POSITIVE ✓ (parallels MPO) | Negative |
| NSE (Non-Specific Esterase / α-naphthyl acetate esterase) | Positive in monocytic (M4, M5) | Negative |
| Specific esterase (Chloroacetate esterase / CAE) | Positive in granulocytic (M1–M3) | Negative |
| PAS (Periodic Acid-Schiff) | Diffuse/granular in erythroid (M6) | Block-positive in ALL (coarse chunky blocks) |
| TdT (Terminal deoxynucleotidyl Transferase) | Negative (except M0 occasionally) | POSITIVE ✓ (hallmark of ALL) |
| Acid Phosphatase | Variable | Positive in T-ALL (focal dot-like) |
Key mnemonic: "MPO/SBB = Myeloid; PAS blocks + TdT = Lymphoid"
Laboratory Findings Summary Table
| Finding | CML | CLL | AML | ALL |
|---|---|---|---|---|
| WBC | Very high (>100K) | High (lymphocytes) | Variable | Variable |
| Predominant cell | Mature granulocytes (all stages) | Mature lymphocytes | Myeloblasts | Lymphoblasts |
| Auer rods | Absent | Absent | Present | Absent |
| Smudge cells | Absent | Present | Absent | Absent |
| Basophilia | Present | Absent | Absent | Absent |
| Bone marrow | Hypercellular, granulocytic | Lymphocytic infiltration | ≥20% myeloblasts | ≥20% lymphoblasts |
| TdT | Negative | Negative | Negative | Positive |
| MPO | Negative (blast phase+) | Negative | Positive | Negative |
| LAP score | Low | Normal | Variable | Variable |
| Philadelphia chr. | Present (>90%) | Absent | Absent (except Ph+ ALL) | Present in 25% adults |
| Key genetic marker | BCR::ABL t(9;22) | del 13q, BTK | FLT3, NPM1, t(15;17) | ETV6::RUNX1, NOTCH1 |
| CNS involvement | Rare | Rare | Occasional | Common (prophylaxis needed) |
| Age | 25–60 years | >50 years | >60 years | Children <15 years |
| Prognosis | Good (imatinib) | Indolent | Variable | Good in children (80–90% cure) |
Sources: Robbins & Kumar Basic Pathology 11e; Robbins & Cotran Pathologic Basis of Disease 10e — equivalent content to Harsh Mohan Textbook of Pathology for 2nd year MBBS pathology syllabus
This is a shared conversation. Sign in to Orris to start your own chat.