I need to study gastric cancer topic in surgery
Reading File
Finding Sources
Finding Sources
Searching PubMed
"gastric cancer"[MeSH Terms] AND "surgical treatment"[MeSH Terms]
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content. Let me compile the complete study guide on gastric cancer for surgery.
🔬 Gastric Cancer — Comprehensive Surgery Study Guide
Sources: Sabiston Textbook of Surgery; Robbins & Cotran Pathologic Basis of Disease; Current Surgical Therapy 14e
1. Epidemiology
- Globally: >1 million new cases/year; >780,000 deaths/year — 5th most common cancer, 4th most common cancer death
- Geographic variation: Highest in East Asia (Japan, Korea, China), Eastern Europe, Chile, and parts of South America; lowest in North America, Northern Europe
- 5-year survival: ~80% in Japan (mass screening) vs. <50% in the USA (late detection)
- US incidence: ~26,500 cases/year; ~11,000 deaths/year; 60% in males
- Racial disparity: Higher incidence and mortality in Blacks, Asian Americans, and Hispanics vs. Whites
- Trend: Distal (noncardia) cancer declining; cardia cancer increasing (linked to GERD, obesity — similar to Barrett's/esophageal adenocarcinoma)
- Alarming: Young-onset gastric cancer (ages 25–40) has increased significantly since the 1970s
2. Risk Factors
Environmental
| Factor | Notes |
|---|---|
| H. pylori | Most important worldwide risk factor; associated with ~75% of non-cardia gastric cancers; causes chronic inflammation → atrophy → intestinal metaplasia → dysplasia → cancer |
| Epstein-Barr virus (EBV) | ~9% of gastric cancers; EBV-positive subtype (TCGA classification) |
| Diet | N-nitroso compounds, benzo[a]pyrene (smoked/salted food); decreased risk with fruits/vegetables |
| Tobacco | Increases risk ~1.5–2× |
| Obesity/GERD | Risk factors specifically for cardia gastric cancer |
| Previous gastric surgery | Partial gastrectomy → gastric stump cancer (bile reflux, hypochlorhydria) |
Premalignant Conditions
- Chronic atrophic gastritis → intestinal metaplasia → dysplasia (Correa cascade)
- Gastric polyps: Adenomatous polyps carry malignant risk; hyperplastic polyps — lower risk
- Pernicious anemia (autoimmune atrophic gastritis)
- Ménétrier disease (hypertrophic gastritis)
Genetic/Hereditary
- Hereditary Diffuse Gastric Cancer (HDGC): Germline CDH1 mutations (E-cadherin); ~40–70% lifetime risk; prophylactic total gastrectomy recommended
- Lynch syndrome (MLH1, MSH2, MSH6, PMS2) → microsatellite instability (MSI) gastric cancer
- Familial Adenomatous Polyposis (FAP): APC mutations → fundic gland polyposis + increased gastric cancer risk
- Li-Fraumeni syndrome, BRCA2 mutations also increase risk
- Blood type A — historically associated with increased risk (diffuse type)
3. Pathology
Classification by Location
- Cardia (proximal/GE junction) — increasing incidence
- Body/Fundus (middle third)
- Antrum/Pylorus (distal) — most common historically
Lauren Histologic Classification
| Type | Intestinal | Diffuse |
|---|---|---|
| Morphology | Gland-forming, cohesive cells, polypoid or ulcerating mass | Poorly cohesive signet ring cells, infiltrative ("linitis plastica") |
| Spread | Hematogenous (liver) | Peritoneal, transmural |
| H. pylori | Strongly associated | Less associated |
| CDH1 (E-cadherin) | KRAS, TP53 mutations | CDH1 loss-of-function (50% of sporadic) |
| Prognosis | Better | Worse |
| Demographics | Older age, males, improving incidence | Younger patients, females; stable incidence |
Signet Ring Cell Carcinoma
- Cells distended with mucin, nucleus pushed to periphery
- Infiltrates gastric wall without forming discrete mass
- Linitis plastica: Diffuse thickening of the gastric wall — "leather bottle stomach"
Special Metastatic Sites (must memorize)
| Site | Name |
|---|---|
| Left supraclavicular node | Virchow's node |
| Periumbilical nodule | Sister Mary Joseph nodule |
| Ovarian metastasis | Krukenberg tumor (mucin-secreting signet ring cells) |
| Left axillary node | Irish node |
| Pouch of Douglas (rectal shelf) | Blumer's shelf |
4. Molecular Classification (TCGA)
Four molecular subtypes:
- EBV-positive (~9%): PIK3CA mutations, extreme CpG island methylation, PD-L1 amplification → responds to immunotherapy
- MSI (microsatellite instability) (~22%): Hypermutated; generally unresponsive to cytotoxic chemo but excellent immunotherapy responders; best prognosis
- CIN (chromosomal instability) (~50%): TP53 mutations, KRAS amplification, HER2 amplification; intestinal-type histology; cardia-predominant
- GS (genomically stable) (~20%): CDH1/RHOA mutations; diffuse-type histology; worst prognosis
Key biomarkers for treatment:
- HER2 (ERBB2): Overexpressed in ~15–20% → trastuzumab eligibility
- PD-L1: Immunotherapy eligibility
- MSI-H: Pembrolizumab eligibility
5. Diagnosis & Workup
Symptoms
- Early disease: Often asymptomatic or nonspecific (dyspepsia, nausea)
- Advanced disease: Weight loss (most common), persistent abdominal pain, dysphagia (proximal tumors), gastric outlet obstruction (distal tumors), anemia (40% have GI bleeding)
Investigations
| Test | Purpose |
|---|---|
| Upper endoscopy (EGD) + biopsy | Gold standard for diagnosis |
| Endoscopic ultrasound (EUS) | T and N staging (local) |
| CT chest/abdomen/pelvis with contrast | Distant metastasis staging |
| PET/CT | If no evidence of M1 disease; identifies distant/nodal disease |
| Diagnostic laparoscopy + peritoneal washings | Mandatory for all patients with suspected locoregional disease; positive cytology = M1 disease |
| HER2/NEU testing | If metastatic disease suspected |
| MSI/MMR testing, PD-L1 | For unresectable/metastatic disease |
| Genetic counseling + CDH1 | In young patients (<50) with diffuse-type |
6. Staging
TNM Staging (AJCC 8th Edition)
T (Primary Tumor)
- T1a: Invades lamina propria or muscularis mucosae
- T1b: Invades submucosa
- T2: Invades muscularis propria
- T3: Invades subserosa (penetrates subserosal connective tissue without visceral peritoneal involvement)
- T4a: Perforates serosa (visceral peritoneum)
- T4b: Invades adjacent structures
N (Regional Lymph Nodes)
- N0: No regional nodes
- N1: 1–2 positive nodes
- N2: 3–6 positive nodes
- N3a: 7–15 positive nodes
- N3b: ≥16 positive nodes
M (Distant Metastasis)
- M0: No distant metastasis
- M1: Distant metastasis (includes positive peritoneal cytology)
Stage Groups & 5-Year Survival (SEER data)
| Stage | Description | ~5-Year Survival |
|---|---|---|
| IA | T1N0 | ~90% |
| IB | T1N1 or T2N0 | ~70% |
| IIA | T1N2, T2N1, T3N0 | ~55% |
| IIB | T1N3a, T2N2, T3N1, T4aN0 | ~40% |
| IIIA | T2N3a, T3N2, T4aN1, T4aN2, T4bN0-1 | ~25% |
| IIIB/C | More advanced nodal | ~10–20% |
| IV | M1 | <5% |
7. Surgical Treatment
Principles of Resection
- Goal: R0 resection (microscopically negative margins) — most important determinant of cure
- Proximal margin: ≥5 cm for intestinal type; ≥8 cm or total gastrectomy for diffuse type
- Frozen section of margins intraoperatively
Types of Gastrectomy
| Tumor Location | Procedure | Notes |
|---|---|---|
| Distal (antrum/pylorus) | Subtotal/distal gastrectomy | Preferred if adequate margins achievable; superior quality of life; Billroth II or Roux-en-Y reconstruction |
| Middle third (body) | Distal/total gastrectomy | Pylorus-preserving segmental gastrectomy for cT1N0 (East Asia, KLASS-04 trial) |
| Proximal (cardia, fundus) | Total gastrectomy with Roux-en-Y esophagojejunostomy OR proximal gastrectomy | Roux-en-Y preferred (less bile reflux, reflux esophagitis) |
| Entire stomach / linitis | Total gastrectomy |
Reconstruction options:
- Billroth I (gastroduodenostomy) — rarely used now
- Billroth II (gastrojejunostomy)
- Roux-en-Y — preferred: less reflux, better QOL at 1 year
- Double-tract reconstruction — after proximal gastrectomy (connects remnant stomach and esophagus to jejunum)
Siewert Classification (GE Junction Tumors)
- Type I (1–5 cm above GE junction): True esophageal cancer → esophagectomy protocol
- Type II (1 cm above to 2 cm below): Cardiac cancer — controversial; gastrectomy via transhiatal approach generally preferred over thoracoabdominal (same survival, less morbidity)
- Type III (2–5 cm below GE junction): True gastric cancer → total gastrectomy
Extent of Lymphadenectomy (Japanese Gastric Cancer Association)
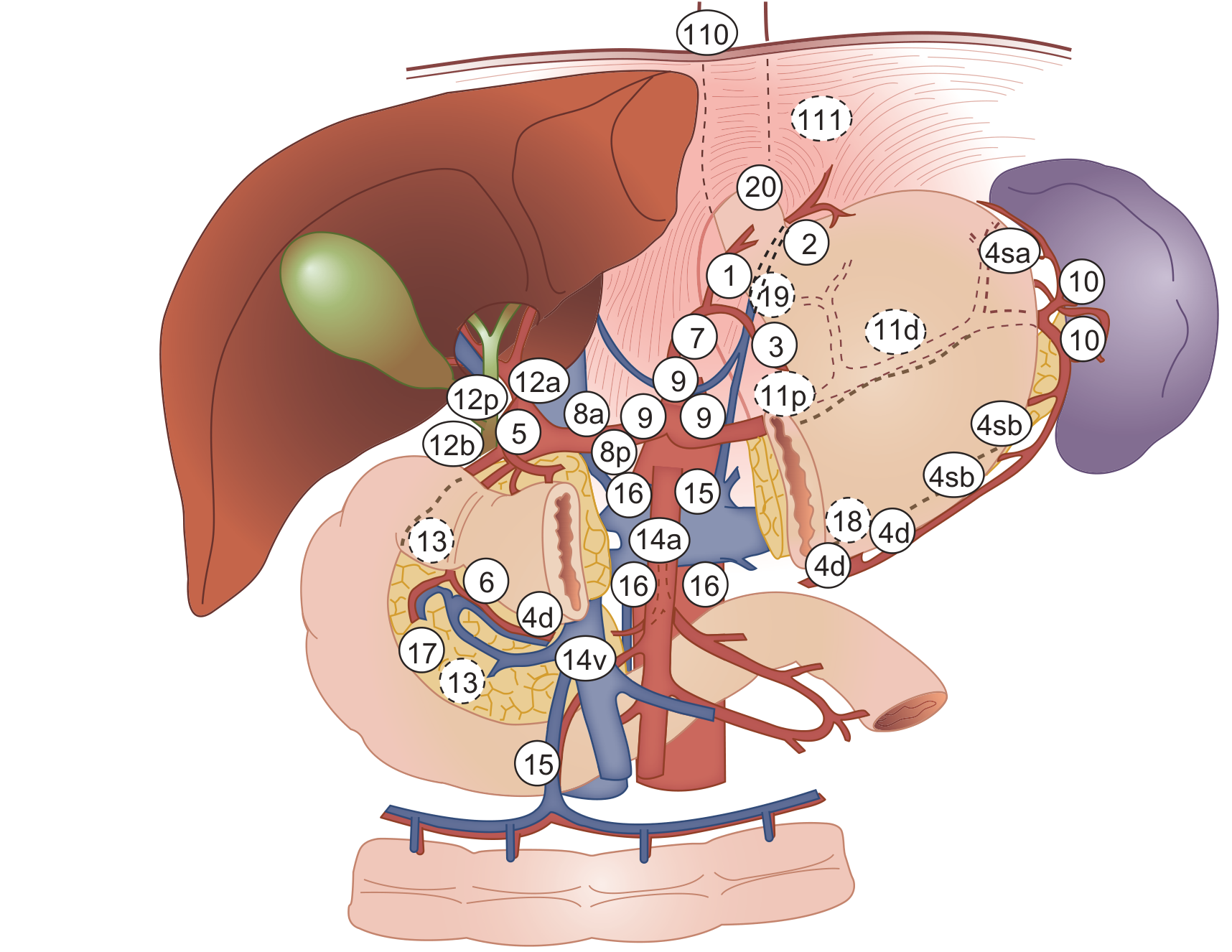
| Dissection | Nodes Removed | Indication |
|---|---|---|
| D1 | Perigastric nodes (stations 1–6) | Early gastric cancer (T1) not amenable to endoscopic resection |
| D1+ | D1 + stations 7–8a, 9, 11p (for distal) | Selected early cancers |
| D2 | D1 + stations 7–12a perigastric + celiac axis nodes (hepatic, splenic, left gastric arteries) | Standard for potentially curative resection of locally advanced disease |
Key evidence on D2:
- Dutch Gastric Cancer Trial: D2 vs D1 — initially no OS benefit, but 15-year follow-up showed D2 superior in OS, local recurrence, and cancer-specific mortality; routine splenopancreatectomy not recommended (increases morbidity without benefit)
- Minimum 16 lymph nodes should be retrieved and examined for adequate staging (AJCC recommendation)
- D2 without splenopancreatectomy is the current standard of care in Western countries
Minimally Invasive Surgery
- Laparoscopic distal gastrectomy: Equivalent oncologic outcomes to open; favored for early gastric cancer in experienced centers
- Robotic gastrectomy: Promising outcomes, especially for D2 lymphadenectomy
- KLASS-01 RCT (Korean): Laparoscopic vs. open distal gastrectomy for early gastric cancer — equivalent 5-year OS (94.2% vs 93.3%)
8. Endoscopic Treatment (Early Gastric Cancer)
Endoscopic Mucosal Resection (EMR) vs. Endoscopic Submucosal Dissection (ESD)
ESD preferred for larger lesions — allows en bloc resection regardless of size.
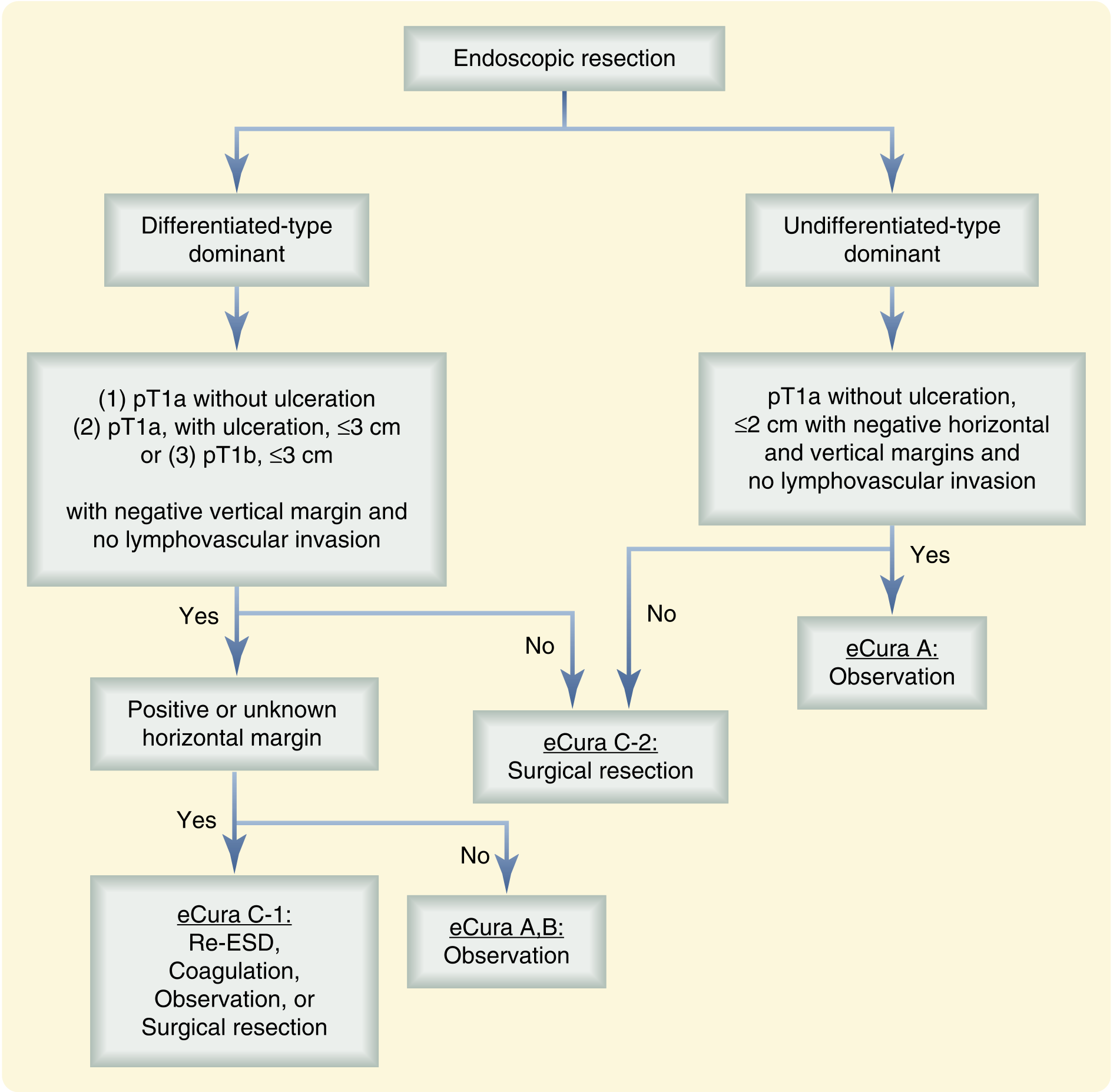
Standard Criteria for Endoscopic Resection (Japanese Guidelines)
Absolute indications (ESD):
- Differentiated-type adenocarcinoma
- No ulceration
- ≤2 cm (EMR) or any size (ESD)
- Clinical T1a (mucosa only)
Expanded indications (ESD):
- Differentiated-type, T1a, any size, with ulceration
- Differentiated-type, T1a, no ulceration, >2 cm (ESD)
- Differentiated-type, T1b (SM1 <500 μm), ≤3 cm, no ulceration
- Undifferentiated-type, T1a, no ulceration, ≤2 cm
9. Perioperative & Systemic Therapy
Perioperative Chemotherapy (Resectable Disease)
- FLOT (5-FU, leucovorin, oxaliplatin, docetaxel) — standard of care in Europe; superior to ECF/ECX in the FLOT4 trial (OS 50 months vs 35 months)
- FOLFOX / XELOX (CAPOX) — also used perioperatively
- ECF / ECX (epirubicin, cisplatin, 5-FU/capecitabine) — historical standard (MAGIC trial)
- Standard approach: 3–6 months neoadjuvant → surgery → adjuvant chemotherapy
Adjuvant Therapy (Post-resection)
- Japan/Korea: Adjuvant S-1 monotherapy (ACTS-GC trial) or XELOX (CLASSIC trial) after D2 resection
- US (SWOG S0116): Adjuvant chemoradiation (5-FU/leucovorin + 45 Gy) — INT-0116 trial showed survival benefit, but patients had inadequate lymphadenectomy (D0/D1), raising debate about whether radiation compensates for inadequate surgery
Metastatic/Unresectable Disease
- First-line: Platinum + fluoropyrimidine backbone
- + Trastuzumab if HER2-positive (ToGA trial — OS benefit)
- + Pembrolizumab if MSI-H or PD-L1 CPS ≥10 (KEYNOTE-590/811)
- Nivolumab + chemotherapy (CheckMate-649): OS benefit in PD-L1 CPS ≥5
10. Management of Peritoneal Disease
- Positive peritoneal cytology (washing) = M1 disease → systemic therapy
- HIPEC + CRS (cytoreductive surgery):
- Consider for limited peritoneal disease (PCI <6–10) after good response to systemic therapy
- PHOENIX-GC trial: IP paclitaxel trend toward survival benefit (17.7 vs 15.2 months)
- GYMSSA trial: CRS+HIPEC+systemic → 11.3 months vs 4.3 months (systemic alone)
- GASTRIPEC trial: CRS+HIPEC did not improve OS but doubled PFS (7.1 vs 3.5 months)
- Prophylactic HIPEC: Under investigation (GASTRICHIP trial)
- Palliative gastrectomy (REGATTA trial): No survival benefit over systemic therapy alone → not recommended routinely in metastatic setting
11. Outcomes & Surveillance
Recurrence Patterns
- Locoregional recurrence: ~40% after curative resection; sites — gastric remnant, anastomosis, nodal basins; reduced with D2 lymphadenectomy
- Distant recurrence: Liver (most common), peritoneum
- Median OS after curative resection in Western countries: ~2 years; in Asia: better
Surveillance Protocol (NCCN)
- H&P every 3–6 months × 2 years, then every 6–12 months × 3–5 years
- CT chest/abdomen/pelvis: Every 6 months × 2 years, then annually × 5 years (stage II+)
- Upper endoscopy: First at 1 year post-resection, then every 2 years (monitor for second primary)
- Lab tests: CBC, chemistry, vitamin B12, iron studies, vitamin D, bone density (post-gastrectomy nutritional sequelae)
12. Postgastrectomy Syndromes (Surgical Complications)
| Syndrome | Timing | Mechanism | Treatment |
|---|---|---|---|
| Dumping syndrome (early) | 15–30 min post-meal | Rapid gastric emptying → osmotic fluid shift → vasomotor symptoms | Dietary modification, octreotide |
| Dumping syndrome (late) | 1–3 hrs post-meal | Reactive hypoglycemia | Small frequent meals |
| Bile reflux gastritis | Chronic | Loss of pylorus | Roux-en-Y reduces this |
| B12 deficiency | Months–years | Loss of intrinsic factor (total gastrectomy) | Lifelong B12 supplementation |
| Iron deficiency anemia | Months | Reduced acid for Fe absorption | Oral/IV iron |
| Bone disease | Long-term | Ca/vitamin D malabsorption | Supplementation |
| Afferent loop syndrome | Variable | Biliopancreatic secretions obstruct afferent limb | Reoperation |
Key Summary — "High-Yield" Points for Surgery Exams
- Most common type: Adenocarcinoma (>90%), followed by lymphoma, GIST, carcinoid
- Most important risk factor globally: H. pylori
- Hereditary diffuse gastric cancer: CDH1 mutation → E-cadherin loss → prophylactic total gastrectomy
- Virchow's node (left supraclavicular) = distant metastasis = M1 disease
- Linitis plastica = diffuse-type infiltration, signet ring cells, poor prognosis
- Krukenberg tumor = bilateral ovarian metastases from signet ring cell gastric cancer
- D2 lymphadenectomy = standard for curative resection (without routine splenopancreatectomy)
- Minimum 16 lymph nodes needed for adequate staging
- Perioperative FLOT = current standard of care in Europe for resectable disease
- R0 resection = the single most important prognostic factor
- Positive peritoneal cytology = M1 disease (even without gross metastasis)
- Roux-en-Y reconstruction preferred over Billroth (less bile reflux, better QOL)
- ESD = preferred endoscopic technique for early gastric cancer
- HER2 positivity (~15–20%) → add trastuzumab in metastatic setting
- MSI-H → excellent immunotherapy response, better prognosis with checkpoint inhibitors
Sabiston Textbook of Surgery (9780443124341), pp. 1798–1812; Robbins & Cotran Pathologic Basis of Disease (9780443264528), pp. 2354–2365; Current Surgical Therapy 14e (9780323796835), pp. 312–316
Is this are enough for me to crack every question in neet pg nd incit for this topic ??
Searching the Web
NEET PG INI CET surgery gastric cancer frequently asked questions high yield topics 2024 2025
Finding Sources
Honest answer: Almost, but not quite. Here's a gap analysis:
What the Guide Above Covers Well ✅
Everything in that guide maps directly to NEET PG / INI-CET high-yield zones:
- Epidemiology, H. pylori, risk factors
- Lauren classification (intestinal vs diffuse)
- Virchow's / Sister Mary Joseph / Krukenberg / Blumer's shelf
- TNM staging, surgical principles
- D1 vs D2 lymphadenectomy
- Reconstruction types
- Perioperative chemotherapy (FLOT, MAGIC trial)
- Peritoneal metastasis / HIPEC
- Postgastrectomy syndromes
- HER2, MSI, molecular subtypes
What's Missing for Full PYQ Coverage ❌
These topics appear repeatedly in NEET PG / INI-CET previous year questions (PYQs) but weren't in the guide:
1. Gastric Lymphoma / MALT Lymphoma (frequently asked)
- Most gastric lymphomas = B-cell Non-Hodgkin lymphoma
- MALT (Mucosa-Associated Lymphoid Tissue) lymphoma: directly caused by H. pylori; first-line treatment = H. pylori eradication (not surgery) — classic exam favourite
- High-grade MALT → chemotherapy (R-CHOP); surgery rarely needed now
- Ann Arbor staging used
2. GIST (Gastrointestinal Stromal Tumor) (very high yield)
- Most common mesenchymal tumor of GI tract; most common site = stomach
- CD117 (c-KIT) and DOG1 positive on immunohistochemistry
- KIT mutations in 75–85%; PDGFRA mutations in 10%
- Treatment: Imatinib (Gleevec) — targeted therapy for unresectable/metastatic; also adjuvant for high-risk
- Surgery: wedge resection with clear margins; no lymphadenectomy needed (GISTs rarely spread to nodes)
- Malignant potential based on size + mitotic rate (Fletcher/NIH criteria)
3. Carcinoid / Neuroendocrine Tumors of Stomach (occasionally asked)
- Type I (most common, fundic gland polyps, pernicious anemia, low malignant potential) vs Type II (MEN1/ZES) vs Type III (sporadic, high malignant potential)
4. Specific Exam-Favourite One-Liners (PYQ staples)
| Fact | Answer |
|---|---|
| Correa's cascade steps | Normal mucosa → Chronic superficial gastritis → Atrophic gastritis → Intestinal metaplasia → Dysplasia → Carcinoma |
| Most common site of gastric cancer | Pyloric antrum (distal) |
| Best prognosis in gastric cancer | EBV-positive subtype (molecular) |
| Macroscopic classification | Borrmann classification (I–IV): I = polypoid, II = ulcerating, III = ulcero-infiltrative, IV = diffuse infiltrating (linitis plastica) |
| Radiological sign of linitis plastica | "Leather bottle stomach" on barium meal |
| Surgical margin for diffuse type | ≥8 cm (vs 5 cm for intestinal type) |
| Minimum nodes for adequate staging | 16 lymph nodes |
| Trial that proved perioperative chemo benefit (UK) | MAGIC trial (ECF regimen) |
| Current best perioperative regimen | FLOT (FLOT4 trial) |
| Adjuvant therapy after D2 in Asia | S-1 (ACTS-GC trial) or XELOX (CLASSIC trial) |
| "Pulled-up" mucosa in barium = | Malignant ulcer (vs benign ulcer: punched-out, regular folds radiating to edge) |
| Most important prognostic factor | R0 resection status |
5. Borrmann Classification (diagrams appear in image-based Qs)
- Type I: Polypoid/fungating — best prognosis
- Type II: Ulcerating with raised edges — well-defined
- Type III: Ulcero-infiltrative — most common
- Type IV: Diffuse infiltrating (linitis plastica) — worst prognosis
6. Early vs Advanced Gastric Cancer
- Early gastric cancer: Limited to mucosa ± submucosa (T1), regardless of nodal status — 5-year survival >90% with surgery
- Advanced: Beyond submucosa
- Japan detects 35% as early (mass screening) vs <20% in the West
7. Benign vs Malignant Gastric Ulcer Differentiation (classic exam Q)
| Feature | Benign | Malignant |
|---|---|---|
| Margins | Regular, punched-out | Irregular, raised, heaped-up |
| Floor | Smooth | Necrotic/shaggy |
| Rugae | Radiate to ulcer edge | Stopped/fused before edge |
| Location | Lesser curve | Anywhere, often greater curve |
| Barium appearance | Hampton's line | Meniscus sign / Carman's sign |
Verdict
The original guide covers ~75–80% of what NEET PG / INI-CET asks on gastric cancer. Add these gaps — especially Borrmann classification, MALT lymphoma, GIST, Correa cascade, benign vs malignant ulcer signs, and the key trial names — and you'll be equipped to answer >95% of PYQs on this topic.
The questions in these exams are heavily MCQ-based and love:
- Eponymous signs (Virchow, Krukenberg, Blumer, Irish node)
- Classification systems (Lauren, Borrmann, Siewert, D1/D2)
- Trial names (MAGIC, FLOT4, ACTS-GC, Dutch trial, KLASS-01)
- Single best-treatment questions (MALT → H. pylori eradication; GIST → imatinib)
- Image-based (Borrmann types on barium, endoscopy findings, histology)
This is a shared conversation. Sign in to Orris to start your own chat.