Chronic eosinophil pneumonia Full details for md exam answer 15 marks
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have all the comprehensive information needed. Let me compile the full 15-mark MD exam answer.
Chronic Eosinophilic Pneumonia (CEP)
Also called Idiopathic Chronic Eosinophilic Pneumonia (ICEP)
1. Definition and Introduction
Chronic eosinophilic pneumonia (CEP) is a rare idiopathic disorder characterized by a protracted, progressive accumulation of eosinophils in the lung parenchyma — within alveolar spaces, alveolar septa, and interstitium — without an identifiable cause such as parasitic infection or drug exposure. First described in detail by Carrington et al. (1969), it is the most frequent cause of eosinophilic pneumonia in Western countries.
2. Epidemiology
| Feature | Detail |
|---|---|
| Sex | Predominantly women (F:M ratio ≈ 2:1) |
| Age | Highest incidence in the fourth decade; rare before age 20 |
| Smoking | Vast majority are non-smokers |
| Atopy | Prior history of atopy in ~50% (allergic rhinitis, drug allergy, nasal polyps, eczema) |
| Asthma | Present in ~65% at diagnosis; develops concurrently in 15%, and may appear after CEP in 13% |
— Murray & Nadel's Textbook of Respiratory Medicine
3. Etiology and Pathogenesis
The precise etiology is unknown. Current understanding implicates a Th2-skewed immune response:
- Elevated IL-5, IL-4, and IL-13 in BAL fluid drives eosinophilopoiesis and recruitment
- Eotaxin and RANTES (CCL-5) are elevated in BAL, attracting eosinophils to the lung
- Type 2 helper T (Th2) cells are central to disease perpetuation, explaining the chronic, relapsing course
- Elevated IgE levels in ~50% of patients
- Dysregulated eosinophilopoiesis — IL-5 is the key cytokine in eosinophil maturation and survival
— Fishman's Pulmonary Diseases and Disorders; Harrison's Principles of Internal Medicine 22E
4. Clinical Features
Symptoms develop progressively over weeks to months (mean interval from onset to diagnosis: several weeks to 4 months). The spectrum ranges from asymptomatic to respiratory failure.
Respiratory symptoms:
- Cough (usually dry)
- Progressive dyspnea (usually not severe initially, but respiratory failure can occur)
- Chest pain
- Wheezing (one-third of patients)
- Crackles on auscultation (38%)
- Hemoptysis (rare, <10%)
Constitutional symptoms:
- Fever, night sweats
- Weight loss (>10 kg in ~10%)
- Fatigue, malaise, anorexia, weakness
Upper respiratory tract: Chronic rhinitis or sinusitis in ~20%
— Murray & Nadel's; Goldman-Cecil Medicine
5. Laboratory Findings
| Test | Finding |
|---|---|
| Blood eosinophils | Elevated in 66–95% of cases; typically >1000/mm³ (>6–30% of differential); absent in up to 1/3 at initial evaluation |
| Mean eosinophil % | Generally >20% of differential; mean blood eosinophilia often >5×10⁹/L |
| CRP / ESR | Elevated |
| Total IgE | Elevated in ~50%; >1000 IU/mL in 15% |
| FeNO | Elevated (proposed for monitoring) |
| FBC | Normochromic normocytic anemia; thrombocytosis may be present |
| BAL eosinophils | >40% (range 12–95%); mean ~38–58%; >40% is most consistent with ICEP |
| Sputum eosinophilia | May be present |
Key point: Absence of peripheral eosinophilia does not exclude the diagnosis.
— Fishman's; Murray & Nadel's
6. Imaging
Chest Radiograph
- Peripheral, patchy, non-segmental areas of consolidation, predominantly in mid and upper lung zones
- The classic "photographic negative of pulmonary edema" (Gaensler & Carrington sign): dense peripheral opacities paralleling the chest wall, with a clear central zone — seen in only 25–50% of cases
- Infiltrates are typically bilateral, non-segmental, subsegmental, or lobar
- May be migratory in up to 25% of cases
- Pleural effusions and cavitation are rare
HRCT (more sensitive and specific)
- Bilateral peripheral opacities in up to 97.5% of cases on HRCT (vs. ~50% on CXR)
- Predominantly upper-lobe distribution
- Both ground-glass opacity and consolidation coexist
- Septal line thickening is common
- Mediastinal lymphadenopathy may be present
- With treatment: consolidation rapidly diminishes → transforms to ground-glass → then streaky/bandlike opacities parallel to chest wall → resolves
Distinguishing from organizing pneumonia (COP): CEP has peripheral opacities without nodules; nodules are seen in 32% of COP but only 5% of CEP.
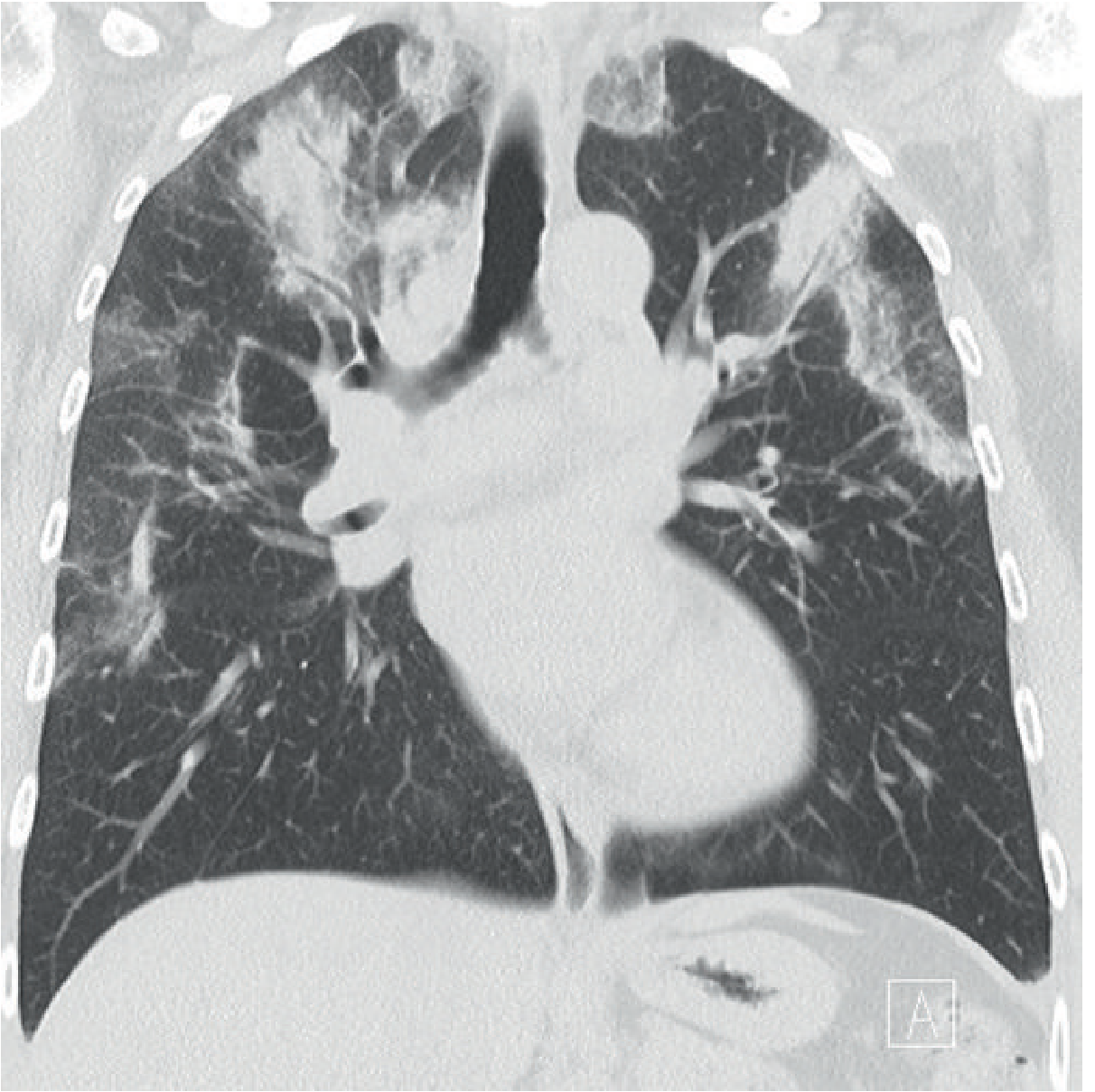
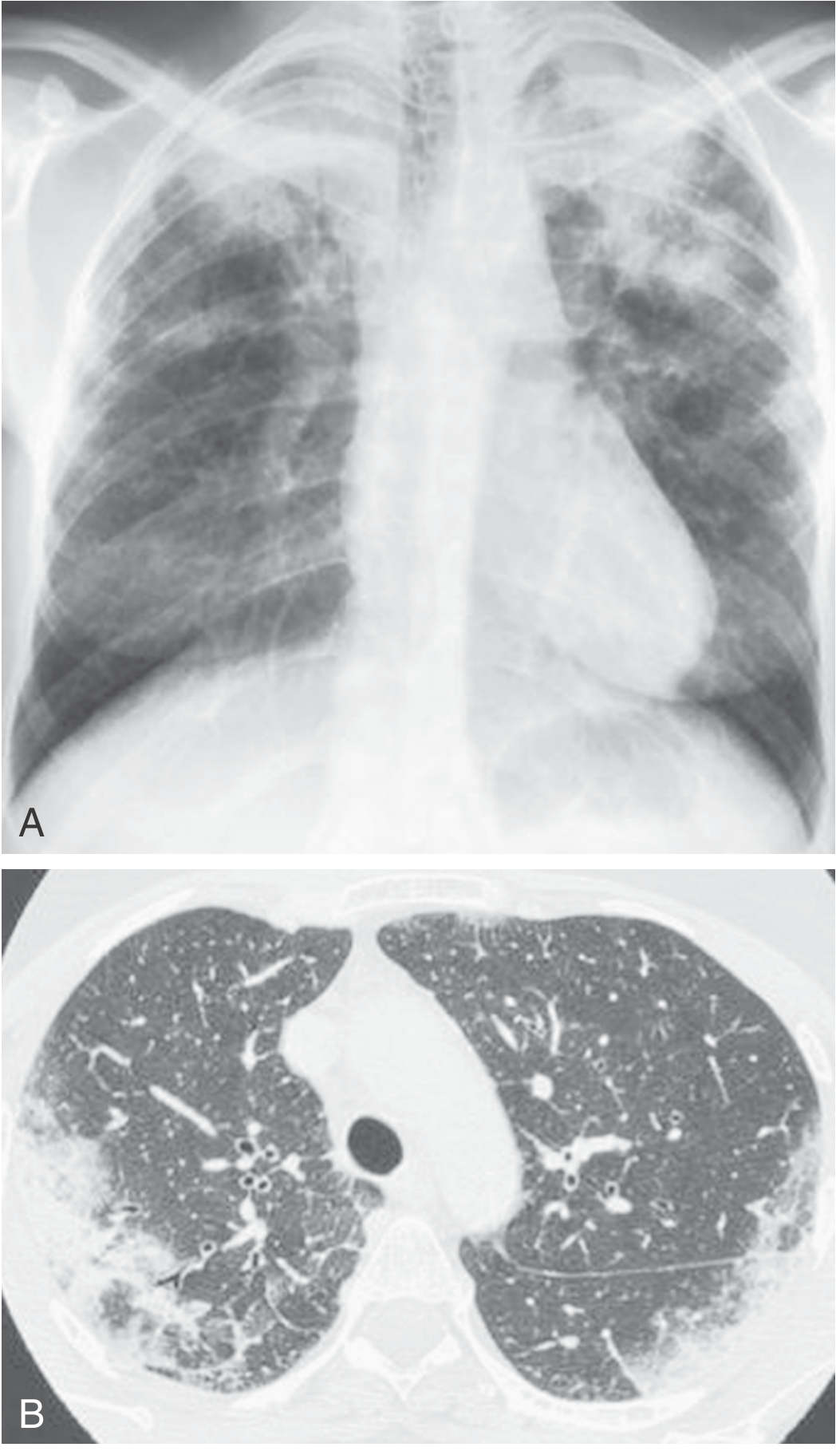
— Grainger & Allison's Diagnostic Radiology; Murray & Nadel's; Fishman's
7. Pulmonary Function Tests
- Obstructive ventilatory defect in ~50% (more common in asthmatics)
- Restrictive physiology in the other ~50% (due to parenchymal infiltration)
- Reduced DLCO (diffusing capacity) in some; transfer coefficient (DLCO/VA) impaired in only 1/4
- Elevated alveolar-arterial oxygen gradient in 90%
- PaO₂ ≤75 mmHg in 64%
- PFTs normalize rapidly after treatment in most patients
- A persistent obstructive defect may develop in 1/3 over time, especially in those with high BAL eosinophilia
8. Pathology
Lung biopsy is rarely required when BAL has been performed. When done:
- Alveolar spaces and alveolar septa: Predominantly eosinophilic infiltrates with macrophages, lymphocytes, occasional plasma cells, and multinucleated giant cells
- Fibrinous exudate within alveoli
- Eosinophilic microabscesses may be present
- Focal edema of capillary endothelium; focal type II pneumocyte hyperplasia
- Basal lamina may be disrupted but frank alveolar necrosis is absent (unlike AEP)
- Mild non-necrotizing microangiitis of small venules in some cases
- Proliferative bronchiolitis obliterans/BOOP pattern in up to 1/3 of cases
- Lymph nodes: lymphoid hyperplasia and eosinophil infiltration
— Fishman's Pulmonary Diseases and Disorders
9. Diagnostic Criteria (Carrington Criteria / Current Consensus)
Diagnosis of CEP is based on all of the following:
- Compatible clinical presentation — progressive cough, dyspnea, constitutional symptoms over weeks to months
- Peripheral opacities on chest imaging (radiograph or CT)
- BAL eosinophilia >25% (most consistent when >40%)
- Exclusion of known causes: parasitic infection, drug-induced, TB, fungal, sarcoidosis, ABPA, EGPA, organising pneumonia
- Dramatic response to corticosteroids — considered near-diagnostic
A surgical lung biopsy is rarely required. Because of profound steroid responsiveness, a therapeutic trial of steroids is itself diagnostically informative.
10. Differential Diagnosis
| Condition | Distinguishing Feature |
|---|---|
| Acute eosinophilic pneumonia (AEP) | Acute onset, respiratory failure, BAL eos >25% but blood eosinophilia absent at onset, no relapse after treatment |
| Eosinophilic granulomatosis with polyangiitis (EGPA/Churg-Strauss) | Systemic vasculitis, ANCA positive in 30–40%, multiorgan involvement |
| Organizing pneumonia (COP) | Nodules more common; less peripheral eosinophilia |
| ABPA | Asthma/CF, Aspergillus sensitization, central bronchiectasis |
| Drug-induced eosinophilic pneumonia | Drug exposure history |
| Parasitic infections / Loeffler syndrome | Parasitic exposure, migratory eosinophilia |
| Sarcoidosis | Bilateral hilar adenopathy, non-caseating granulomas, less eosinophilia |
11. Treatment
Corticosteroids — Mainstay of Therapy
Initial regimen:
- Oral prednisone 0.5 mg/kg/day (typically 40–60 mg/day)
- Continue until 2 weeks after resolution of symptoms and radiographic abnormalities (~4–6 weeks)
- Then taper slowly by 0.25 mg/kg/d over 8 weeks
- Total treatment duration: optimally 6–9 months (minimum 3 months)
Response timeline:
| Timeframe | Response |
|---|---|
| 6 hours | Fever resolves |
| 24–48 hours | Dyspnea, cough, blood eosinophilia improve |
| 2–3 days | Hypoxemia resolves |
| 1–2 weeks | Radiographic improvement |
| 2–3 weeks | Complete symptom resolution |
| ~2 months | Chest radiograph normalizes |
This rapid, dramatic response is considered near-diagnostic — failure to respond should prompt reconsideration of diagnosis.
Steroid-Sparing / Biologic Agents
Used when steroids are not tolerated, in relapsing disease, or to reduce steroid dose:
- Mepolizumab (anti-IL-5): associated with fewer relapses, improved imaging, reduced oral steroid dose
- Benralizumab (anti-IL-5 receptor antagonist): reports of success in relapsed/refractory disease
- Omalizumab (anti-IgE): steroid-sparing in cases with elevated IgE/allergic sensitization
- Inhaled corticosteroids: not adequate as monotherapy; may have a role in reducing relapses, especially in asthmatics
— Fishman's; Murray & Nadel's
12. Prognosis and Relapse
- Prognosis is generally favorable; mortality directly from CEP is very rare
- Relapse rate is high: 50–80% when steroids are tapered or discontinued
- Relapses may occur in the same or different anatomic distribution
- Up to 50% require long-term maintenance prednisone (2.5–10 mg/day)
- Some patients require 1–3 years of initial steroid treatment
- Relapses respond well to resuming prednisone ≥40 mg/day
- Asthma often worsens after diagnosis of ICEP; may require long-term oral steroids
- Persistent obstructive ventilatory defect develops in 1/3 over time
- If left untreated, may result in pulmonary fibrosis
Key contrast with AEP: AEP does NOT relapse after treatment is stopped; CEP has a high relapse rate.
Summary Table: CEP vs AEP
| Feature | CEP (ICEP) | AEP (IAEP) |
|---|---|---|
| Onset | Weeks to months | Days (<1 month) |
| Sex | Women (2:1) | Young men (20–40 yr) |
| Smoking | Non-smokers | Often recent smokers |
| Fever | Mild to moderate | High fever |
| Respiratory failure | Rare initially | Common, may need ventilation |
| Blood eosinophilia | Present in 66–95% | Usually absent at onset |
| BAL eosinophils | >40% | >25% |
| CXR/CT | Peripheral upper-zone consolidation | Bilateral diffuse ground-glass ± effusions |
| Pleural effusion | Rare | Common (up to 65–70%) |
| Response to steroids | Dramatic, but high relapse | Dramatic, no relapse |
| Prognosis | Good; relapses common | Excellent; no relapse |
Sources: Goldman-Cecil Medicine, International Edition; Murray & Nadel's Textbook of Respiratory Medicine; Fishman's Pulmonary Diseases and Disorders; Grainger & Allison's Diagnostic Radiology; Harrison's Principles of Internal Medicine 22E (2025)
This is a shared conversation. Sign in to Orris to start your own chat.